Abstract
Objectives
In this study, we evaluated the potential association between the habitat types of feral cats and the prevalence of selected infectious pathogens and health status based on a set of blood parameters.
Methods
We live-trapped 72 feral cats from two different habitat types: an urban area (n = 48) and a rural agricultural area (n = 24). We compared blood values and the prevalence of feline immunodeficiency virus (FIV), feline leukaemia virus (FeLV) and haemotropic Mycoplasma infection in feral cats from the two contrasting habitats.
Results
Significant differences were observed in several blood values (haematocrit, red blood cells, blood urea nitrogen, creatinine) depending on the habitat type and/or sex of the cat. Two individuals from the urban area were seropositive for FIV (3.0%), and eight (12.1%) were positive for FeLV infection (five from an urban habitat and three from a rural habitat). Haemoplasma infection was more common. Based on molecular analysis, 38 cats (54.3%) were positive for haemoplasma, with a significantly higher infection rate in cats from rural habitats (70.8%) compared with urban cats (47.8%).
Conclusions and relevance
Our study recorded haematological and serum biochemical values, and prevalence of selected pathogens in feral cat populations from two different habitat types. A subset of important laboratory parameters from rural cats showed values under or above the corresponding reference intervals for healthy domestic cats, suggesting potential differences in the health status of feral cats depending on the habitat type. Our findings provide information about the association between 1) blood values (haematological and serum biochemistry parameters) and 2) prevalence of selected pathogen infections and different habitat types; this may be important for veterinarians who work with feral and/or stray cats and for overall cat welfare management.
Introduction
With their extreme adaptability and ecological flexibility, feral cats (Felis catus) are found in diverse habitat types, from deserted islands to the core of metropolitan areas. 1 Feral cats in urbanised habitats typically have high population densities and there are increasing concerns regarding both the potential role of feral cats in the transmission of zoonotic pathogens, and their welfare, as well as conflict with the public due to the wide range of human attitudes towards feral cats. 2 So far, few studies have evaluated the potential risk factors that may be related to pathogen infection and feral cats, such as habitat type (eg, urban, agricultural) and/or lifestyle (pet, stray, feral).3,4 In contrast, many studies about pathogen infections in feral cats have focused on describing the prevalence of zoonotic diseases.5,6 In this study, we compared blood values and the prevalence of selected pathogens of feral cats inhabiting two different habitats: urban and rural. For the purposes of this study, feral cats were defined as unowned, untamed or unconfined cats. 7 Blood values composed of haematological and serum biochemistry parameters are considered to reflect the general health status of animals, including physiological condition and the function of important organs and are commonly applied to evaluate the trend of physiological status in natural feline populations, such as feral cats and the Iberian lynx.8 –10 Although urbanised areas are generally not considered to be ideal habitats for wild mammals, feral cats in urban environments commonly use abundant food sources provided indirectly through human refuse or directly by cat caretakers. 11 In addition, feral cats that rely on cat caretakers are known to have a smaller home range and altered breeding patterns compared with their conspecifics living on farmland or in rural areas where they are relatively independent from humans. 12 We hypothesised that the blood parameters of feral cats would differ depending on their habitats (urban vs rural) as a result of differences in their environments and resources, which would affect the physiological status of individual cats.
In addition to blood values, we examined the prevalence of selected feline pathogens – haemotropic Mycoplasma species, feline immunodeficiency virus (FIV) and feline leukaemia virus (FeLV) – in rural and urban habitat types. Owing to their potential to cause clinical signs that may be lethal to infected animals or negatively impact their health, haemoplasmas and retroviruses are pathogens that have frequently been studied in domestic cats.13 –15 Haemotropic Mycoplasma species are bacteria that parasitise red blood cells that may cause anaemia to varying degrees in cats, 16 especially when co-infected with retroviruses. 17 In cats, Mycoplasma haemofelis, Candidatus Mycoplasma haemominutum and Candidatus Mycoplasma turicensis have been reported to cause infection. 16 FIV and FeLV are both retroviruses that have a significant impact on feline health. 18 Although feline retroviruses have been widely studied, current knowledge about feline retroviruses is mostly from domestic cats rather than feral cats. Therefore, it should be noted that feral cats may respond differently physiologically and/or immunologically to retroviruses than domestic cats owing to different environmental factors such as diet and stress.19,20 In pet cats, FIV can cause AIDS-like clinical signs through immunosuppression and immunological dysregulation, leading to increased risk of opportunistic infections, onset of tumours, such as leukaemia and lymphoma, and pathological clinical signs related to immune complex deposition disorders. 18 FeLV is often more pathogenic than FIV, causing up to 80% mortality after infection. 21 Clinical signs of FeLV are most commonly related to tumours, immunosuppression, haematological disorders and immune-mediated diseases. 18
The aim of this study was to identify any differences in haematological and serum biochemical values, and in the prevalence of infection for the selected pathogens – FIV, FeLV and haemoplasma – between feral cat populations from two contrasting environments.
Materials and methods
Field site
We collected blood samples from feral cats in seven different sites: five urbanised habitats and two rural areas. The urban sites were selected within Seoul (37°33′59.53″N 126°58′40.69″E), Korea, which is an intensively developed urban area, with a population density of 17,000/km2. 22 The landscape of Seoul is highly fragmented and densely occupied by buildings and roads. 23 Feral cats are mostly observed in residential areas within the city, where they engage in both positive (food provisioning) and negative (animal abuse incidents) interactions with humans. Thus, all urban trapping sites were selected among similar areas that represented densely packed multi-storey residential buildings, one of the most typical housing structures in the city. 24
Rural sites were selected among typical agricultural landscape composed of extensive rice paddies and sparsely dispersed houses. Two rural sites were selected among areas surrounding Seoul, Yangpyeong County (37°46′15.262″N 127°42′10.875″E) and Gwangju city (37°42′83.21″N 127°33′48.157″E), located at a distance of approximately 70 km from Seoul. The vegetation of the area is typically dominated by rice, with shrubs between the rice paddies. The farming areas are surrounded by low, hilly, forest-covered landscapes. The population densities of the rural sites are approximately 136–189/km2. In order to collect independent samples with respect to geographical locations, trapping sites within both urban and rural areas were sufficiently dispersed from each other (mininum 10 km, maximum 18 km) to avoid the possibility of trapping the same cat from different sites.
Live trapping and sample collection
Feral cats were live-trapped with a total of 480 trap nights in the summer of 2013 using Tomahawk traps (Tomhawk Live Trap). All traps were baited with wet cat food. In urban neighbourhoods, traps were set in relatively hidden and confined places, where feral cats were commonly observed. Such spots also reduced the chance of trapped cats being exposed to pedestrians before we checked the traps. In rural neighbourhoods, most traps were placed within the farmland area with the permission of the farm owners. All traps were set between 6 and 8 pm in all areas, and sample collection started around 2–3 am the next morning in close proximity (<5 min drive) to the trapping sites. To reduce the visual stress of the trapped cats until sample collection, every trap with a cat was covered with a plastic panel. Trapped cats were anaesthetised with a mixture of Zoletil (zolazepam and tiletamine 2 mg/kg; Virbac) and Domitor (medetomidine 50 mg/kg; Zoetis). Before sample collection a physical examination was performed for each cat, which included measurement of body weight, and a check of teeth condition and ear-tipping (marking for neutering).
Approximately 3–4 ml of blood were collected from the jugular vein. Half of the whole-blood samples were collected in EDTA tubes and the other half were collected in serum separation tubes containing clot activator. An aliquot of whole-blood sample in an EDTA tube was sent for haematology analysis within 1–2 days of collection; the remaining blood samples were stored at −70°C for future DNA analysis. Serum separation tubes were maintained in an upright position until centrifugation. Centrifugation was performed within 3–4 h of blood collection. Separated serum samples were stored at −70°C for serum biochemistry analysis and serology pathogen diagnostic assays.
Ethics statement
Ethical approval for the use of animals in this study was obtained from the Institutional Animal Care and Use Committee of the University of Georgia (UGA IACUC: A2013 04-009-Y1-A0) and Seoul National University (SNU) (SNUIBC-R131118-1). The fieldwork for this study was performed in areas where no specific permissions were required, and none of the field studies involved endangered or protected species.
Laboratory analysis
Both haematology and serum biochemistry analyses were performed at SNU Veterinary Medical Teaching Hospital (VMTH) using automatic analysers (haematological analysis: Siemens ADVIA 2120i haematology system; 25 serum biochemistry analysis: Hitachi 7180 clinical analyser). The haematology analytes included haematocrit (HCT), red blood cell (RBC) count, white blood cell (WBC) count, haemoglobin (HGB), mean cell volume and mean cell haemoglobin. Serum samples were tested for blood urea nitrogen (BUN), creatinine, albumin, total protein (TP) and three major liver enzymes: alanine transaminase (ALT), aspartate aminotransferase (AST) and alkaline phosphatase (ALP).
Using commercialised serum diagnostic kits (SensPERT FeLV Ag/FIV Ab kit; VetAll), serum samples were tested for the identification of FIV antibodies and FeLV p27 antigen. The manufacturer reported test sensitivities for FeLV antigen and FIV antibody assays were 98.5% and 99.7%, respectively, while the specificities were reported as 97.0% and 99.0%, respectively (VetAll). All positive samples were re-tested with another commercialised ELISA kit from a different manufacturer (SNAP FIV Antibody/FeLV Antigen Combo; IDEXX).
DNA was extracted from whole-blood samples with the QIAamp DNA mini kit (Qiagen). The extracted DNA was used to identify haemoplasma infection through a subsequent PCR assay. We amplified a partial segment of the 16S rRNA gene of Mycoplasma species using the universal primer set HBT-F (ATACGGCCCATATTCCTACG) and HBT-R (TGCTCCACCACTTGTTCA) designed by Criado-Fornelio et al. 26 The protocol for PCR was performed as previously described. 26 Every PCR run included a positive and negative control to detect bacterial contamination. Amplified products were separated by electrophoresis on a 1.2% agarose gel. PCR amplicons from the positive samples were purified and prepared for direct sequencing. The sequence homology of the sequenced 16S rRNA gene from the positive samples was examined using the program BLAST (National Center for Biotechnology Information; available at: http://www.ncbi.nlm.nih.gov).
Statistical analysis
All statistical analyses were performed using the statistical software R (http://cran.r-project.org). The association of sample numbers between habitat type and sex was analysed using the χ2 test. All continuous data, such as haematology and serum biochemistry parameters, were evaluated by the Shapiro–Wilk test before use in further tests. The means and 95% confidence intervals (CIs) of continuous parameters (haematology and serum biochemistry) were calculated with Bonferroni correction. Descriptive statistics are shown in Tables 1 and 2. Sample sizes slightly varied according to analyses because of missing data and/or specimens (whole blood or serum) of several individuals. The differences in haematology or serum biochemistry parameters between habitat types and sex were tested using two-way ANOVA. The difference in these parameters among sampling sites within each habitat type (five urban and two rural), was tested using ANOVA and Tukey’s honest significant difference post-hoc test for pairwise comparison. Post-hoc power analysis (1-b error probability) was performed using G*Power in order to evaluate the ability of the analysis to detect a significant association between blood parameters and/or pathogen infection and habitat types at an α error of 0.05. 27 The logistic regression model included two binary independent variables and their interaction (habitat type, sex and habitat type–sex interaction) to test its association with the three dependent variables (FIV, FeLV and haemoplasma infection). The odds ratio (OR) of two independent variables and the 95% CI for haemoplasma infection was calculated. Lastly, a likelihood ratio test for the logistic regression model was performed comparing the tested model (sex, habitat, habitat × sex) and the model with intercept only.
Haematological values of studied feral cats by habitat and sex, including the results of two-way ANOVA

Data are presented as mean (95% Bonferroni confidence interval) unless otherwise indicated
HCT = haematocrit; RBCs = red blood cells; HGB = haemoglobin; MCV = mean cell volume; WBCs = white blood cells
Serum biochemistry values of studied feral cats by habitat and sex, including the results of two-way ANOVA

Data are presented as mean (95% Bonferroni confidence interval) unless otherwise indicated
AST = aspartate aminotransferase; ALT = alanine transaminase; TP = total protein; BUN = blood urea nitrogen
Results
In total, biological samples were collected from 72 trapped feral cats. Among the 48 cats from the urban site, 16 were female and 32 were male, while from the rural area, 12 cats were female and 12 were male. Although the samples from the urban area had more male cats, there was no significant association between sex and habitat type (χ2 test P = 0.20). However, it should be noted that the power to detect the association between habitat and sex was only 0.29. Therefore, careful interpretation is required when analysing data combined by habitat type or sex (urban vs rural, male vs female) and the significance of interaction between habitat type and sex.
Physical examination
Based on physical examinations, two females showing round and swollen abdomens with large, well-developed nipples were determined as pregnant. Three male individuals weighing <2.0 kg and showing clean teeth, completely devoid of yellowing of tartar, 28 were categorised as juveniles. In addition, five male cats from the urban habitat had a tipped ear, which is an indication of neutering. The remaining 67 animals were assumed to be intact.
Haematology
When haematological parameters were tested for significant difference among sites (five urban sites, two rural sites) within each habitat type, none of the analytes showed a significant difference. Therefore, haematological parameters from different sites of the same habitat type were pooled for further analysis. There was a significant habitat-type difference in HCT and RBC values, with urban cats having a higher mean value (Table 1). HCT, RBC and HGB values were also different between sexes, with male cats showing higher mean values. In all haematological values, habitat–sex interaction was not significant. The WBC values were not significantly associated with habitat type or sex (Table 1). However, in general, the WBC values of all the groups were high relative to the reference interval provided by SNU VMTH, with only the WBC values of the urban males falling within the reference interval. There were three outlier samples with WBC values >150 (one urban female, one rural male and one rural female). Therefore, the impact of these three samples will need to be considered when interpreting the mean values of WBCs in each group. Despite the limited sample size, the statistical power (the probability of rejecting a false null hypothesis) of the tests comparing haematological parameters (RBCs, HCT, HGB and WBCs) between two habitat types was >0.8, with the exception of WBC count (0.59). Therefore, the lack of significance of the WBC result should be interpreted with caution.
Serum biochemistry
Serum analytes, including BUN, creatinine, ALT, AST, TP and albumin, were tested for significant differences among sites (five urban sites, two rural sites) within each habitat type. None of the analytes were significantly different among sites in urban or rural habitat types, with the exception of albumin and TP (for both protein analytes, samples from one urban site showed a significantly higher value compared with the other urban and rural sites [TP = 8.2 g/dl, albumin = 3.5 g/dl]). Therefore, serum parameters from different sites of the same habitat types were pooled for further analysis. When the serum analytes were compared between the two habitat types, a significant difference was observed in the subset of parameters, with higher ALT values in rural cats and higher BUN and creatinine values in urban cats. Nevertheless, the values were within the reference interval both provided by SNU VMTH and a previous study of free-roaming cats in New York. 8 In addition, sex was associated with TP and creatinine, with female cats having lower values (Table 2). A significant habitat–sex interaction was observed in the AST values (Figure 1). Power analysis comparing serum parameters between the two habitat types revealed the statistical power of TP and albumin analysis to be low (<0.8), which warrants cautious interpretation.

Interaction plot of aspartate aminotransferase (AST), showing significant interaction between habitat type and sex
Identification of haemoplasma infection and its association with sex and habitat types
A total of 70 individuals (46 urban, 24 rural) were tested for haemotropic Mycoplasma infection by molecular diagnostics; two samples were excluded owing to failure of blood sample collection. Twenty-two (47.8%) of 46 urban individuals and 16 (66.7%) of 24 rural individuals were identified as haemotropic Mycoplasma PCR-positive, giving 38/70 (54.3%) haemoplasma cases in the studied cats (Table 3; Figure 2). Positive associations of haemotropic Mycoplasma infection with rural habitat type (P = 0.041, OR 7.00, 95% CI 1.22–58.56) and male sex (P = 0.002, OR 14.00, 95% CI 3.13–101.49) were shown by logistic regression analysis (likelihood ratio χ2 = 18.83, degrees of freedom = 3, P = 0.0003) (Table 4). The goodness of fit of the model, assessed by pseudo R2, was 0.32. Post-hoc power analysis showed that the power of detecting a significant difference in haemoplasma infection between the two habitat types and two sexes was 0.32 and 0.92, respectively (α = 0.05).
Occurrence of haemoplasma and retrovirus infection in feral cats from two habitat types

Data are presented as n (%) unless otherwise indicated
FIV = feline immunodeficiency virus; FeLV = feline leukaemia virus

Occurrence of haemoplasma infection in feral cats by habitat type and sex
Association between haemoplasma infection and external factors in the sampled feral cats (n = 70)
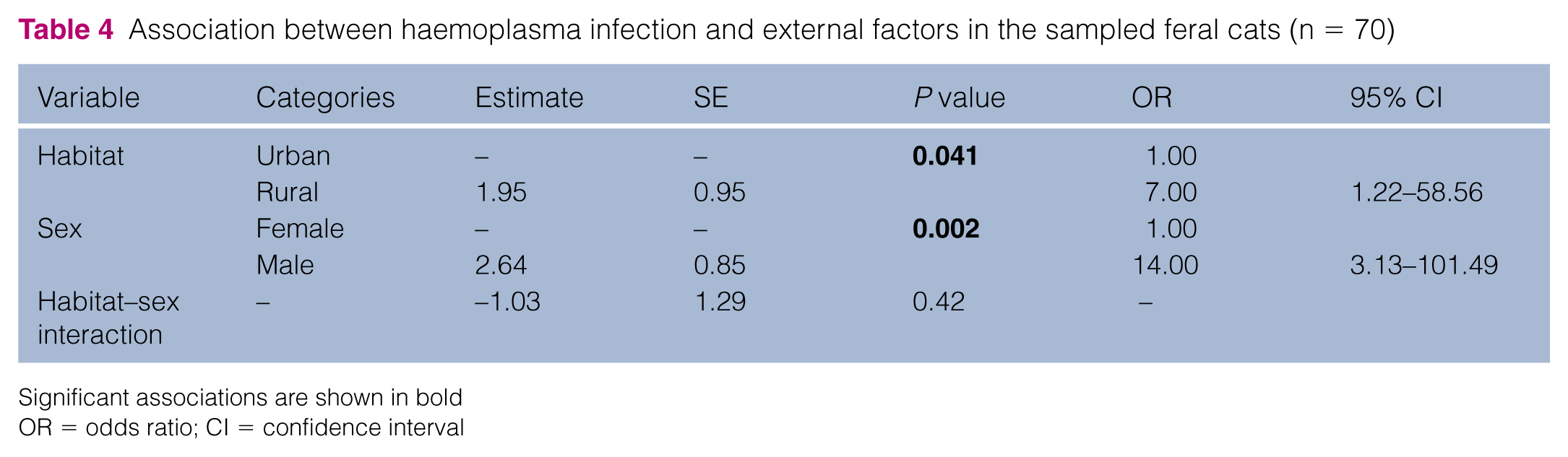
Significant associations are shown in bold
OR = odds ratio; CI = confidence interval
From the urban habitats, 5/22 positive cats (22.7%) were infected with M haemofelis and the remaining 17 (77.3%) were infected with M haemominutum. In the cats from the rural habitats, one (6.3%) was identified as M haemofelis-positive and the other 15 (93.7%) as M haemominutum-positive.
Identification of FIV/FeLV infection
Sixty-six samples were tested for FIV antibodies and FeLV antigen using a serology assay kit. Two samples from the urban habitats showed positive exposure to FIV infection. Overall, the occurrence of the FeLV antigen was 12.1% (8/66), and the proportion of FeLV-infected cats was similar in both habitat types (urban, 5/43 [11.9%]; rural, 3/23 [12.5%]) (Table 3). When the occurrence of FeLV infection was compared between the four groups (rural males, rural females, urban males and urban females), urban females had the highest infection rate (28.6%; n = 14).
Discussion
In this study, we compared haematological and serum chemistry parameters, and the prevalence of selected pathogens of feral cats from urban and rural habitats. Subsets of important laboratory parameters, such as HCT, BUN and creatinine, of rural cats showed values under or above the corresponding reference intervals of healthy domestic cats, which suggests the potential influence of habitat type on the differing health statuses of feral cats. In addition, the prevalence of haemoplasma infection was significantly higher in rural cats.
Among the haematological parameters, HCT and RBC count were significantly higher in male or urban cats compared with female or rural cats, respectively. In wild carnivores, low HCT values may indicate lower nutritional status, in some cases owing to reduced prey availability and/or prey biomass. 29 Similarly, owing to resource limitations, rural cats may be under stronger nutritional stress than urban cats. However, testing additional variables, such as dehydration, toxin exposure and pathogen infection status (eg, hookworm infection), would be required to further clarify the cause of the lower HCT values in rural cats. Meanwhile, the HCT, RBC and HGB values for rural females were consistently below the reference interval. Two of 11 (18%) rural female cats were pregnant, and both cats showed lower-than-average haematology values, which may have caused the general decrease of values in rural female cats. However, it should be noted that the RBC and HCT values of rural female cats were below the reference interval, even when pregnant individuals were excluded from the dataset. The WBC values from the three groups, both sexes of cats from the rural areas and urban females, were above the reference interval. Although various mechanisms may cause an increase in WBCs, higher WBC values are commonly considered to be associated with pathogen infection and/or inflammation. 30
Among biochemical analytes, serum BUN and creatinine values are known to reflect protein intake and muscle mass or kidney diseases in carnivorous mammals.31 –33 Although the BUN and creatinine values of the studied cats were within the reference interval, constantly lower BUN and creatinine values of the rural cats in both sexes could potentially suggest a lower protein intake and/or muscle mass compared with cats from urban habitats.
In the case of liver enzymes, rural cats showed higher levels of AST and ALT compared with urban cats, with the difference being larger for AST in males. Although it is possible that stress and tissue damage occurred during the trapping procedure, all cats were trapped via identical protocols, and it is likely that other factors caused the difference in liver enzyme levels between the two habitat types, such as chronic stress (eg, malnutrition) and/or poor liver function.33 –35 Alternatively, the liver enzyme levels obtained in our study may be a characteristic feature of cats from each studied colony, and the observed ALT and AST values may represent the normal range of healthy cats from each colony. TP and albumin for all cats were within the reference intervals, which makes it unlikely that the cats were suffering from serious dehydration.
A potentially important factor to consider when interpreting serum analytes is the individuality of each parameter. A recent study reported the high individuality of some serum analytes, including ALP, ALT, creatinine and globulin. 36 For parameters with high individuality, population-based reference intervals are likely to have limited significance, and, when possible, repetitive sample collection of identical cats at different time intervals should be encouraged for a clear examination of the studied cat.
It should also be noted that the reference intervals used for the haematology and serology values in this study were provided by SNU VMTH and thus are based on pet domestic cats rather than feral cats, a lack of studies that report on the reference intervals of blood values of stray/feral cats. However, a recent biochemical survey of free-roaming cats in New York reported a range of serum biochemistry values that was similar to the reference interval used in our study, with the exception of creatinine (0.6–1.4 mg/dl), which was lower than the range provided by SNU VMTH (1.0–2.2 mg/dl). 8 This may suggest that based on the creatinine value, stray/feral cats have a lower muscle mass than pet domestic cats, or show a low-to-normal range for creatinine.
In our study, the haemoplasma infection rate was higher in cats from rural habitats and in males. Two haemoplasma species were identified: M haemominutum was the most common species, followed by M haemofelis. The larger number of infected cats in rural areas could be related to the mode of transmission of the haemoplasma species, which is suspected to be through fighting between males and through blood-sucking arthropod vectors.14,37,38 It is possible that cats from rural habitats have daily contact with vegetation or soil, where ectoparasites live and quest for passing host animals. 39 Meanwhile, many urban feral cats are suspected to spend the majority of their time using artificial structures, such as parking lots, empty buildings and basement areas, 40 limiting their chances of being exposed to ectoparasites. 41 Further studies simultaneously comparing the occurrence of ectoparasites and vector-borne pathogen infection in relation to different habitat type are essential for revealing the pattern between sex, habitat type and the occurrence of vectors and vector-borne diseases in feral cats.
Alternatively, higher frequency of aggressive behaviours in rural males compared with urban males may be the cause for higher haemoplasma infection. In previous studies, male cats have been frequently identified to be at a higher risk of haemoplasma infection compared with female cats.42,43 As fighting is likely to be one of the major transmission routes of haemoplasma infection in males, the reduced aggressive behaviour of neutered urban male cats may have contributed to the lower prevalence of haemoplasma in the urban habitat. 44 Out of five neutered male cats, all from urban habitats, four were negative for saliva-borne pathogens, haemoplasma and FIV, and one neutered cat was co-infected with M haemofelis and M haemominutum.
The only two cats that were positive for FIV in this study were females from an urban habitat, which made it difficult to observe the potential association between FIV infection and neutering or aggressiveness. For FeLV infection, the occurrence in our study was relatively high compared with previous reports on retrovirus prevalence in feral or stray cats, which ranged from 0–12.2%.23 –25,45 The prevalence of FeLV in our study will have implications for cat practitioners in Korea who treat both pet and stray/feral cats in their clinics. Nevertheless, considering that the prevalence of retroviruses is generally low when tested at a population level, studies with a sufficient number of samples that fall into different categories of the tested variables would be required in order to explain the risk factors for retrovirus prevalence. 46
Lastly, it should be noted that insufficient sample size might have caused the lack of statistical significance in some of the analyses, such as comparison of serum biochemical parameters by sex, as evidenced by a power <0.8. Thus, the number of samples from each sex or habitat type should be considered when interpreting the results in relation to the tested factors (habitat type, sex and pathogen infection).
Conclusions
Although there is controversy regarding the role of feral cats in an ecosystem, 47 there is no doubt that they are interacting ecologically with the various components of their environment, including wildlife, domestic animals and humans, with the potential for pathogen transmission. Individual behaviours and/or the population structure of domestic cats with different lifestyles (eg, pets, feral cats, shelter cats) vary in several aspects, such as population density, allogrooming and nurturing behaviour among individuals, all of which may influence pathogen transmission. 48 Therefore, the prevalence of disease and possible interactions between pathogens and cats should be monitored separately in relation to different environmental settings and habitat types. Our study reveals possible habitat-related differences in feral cat health status, such as blood parameters and infection with selected pathogens. Findings from this study can provide important information for domestic cat practitioners and those who are involved in managing feral cat populations (eg, trap–neuter–return programmes). In addition, the pathogen infection status of feral cats in rural habitats may aid conservation efforts for wildlife species that can share pathogens with feral cats, such as leopard cats (Prionailurus bengalensis euptilurus) in Korea.
Footnotes
Acknowledgements
We are sincerely grateful to the laboratory of clinical pathology, Veterinary Medical Teaching Hospital, Seoul National University, for technical assistance and expertise on blood analysis.
Funding
This work was supported by the National Research Foundation of Korea Grant funded by the Korean Government (NRF-2012S1A5B6034265).
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
