Abstract
Objectives
The aim of this study was to describe the pharmacokinetics and bioavailability of itraconazole (ITR) oral solution in healthy cats.
Methods
The pharmacokinetics of ITR were studied in eight healthy, fasted cats after a single intravenous (IV) and oral (PO) administration at a dose of 5 mg/kg, in a two-period crossover design study. Blood was obtained at predetermined intervals for the determination of ITR concentrations with high-performance liquid chromatography. Pharmacokinetic characterisation was performed by a non-compartmental method using WinNonlin 5.2.1.
Results
After IV administration, the major pharmacokinetic parameters were as follows (mean ± SD): terminal elimination half-life (T1/2λz ) 15.8 ± 1.88 h; area under the curve from time zero to infinity (AUC0–∞) 13.9 ± 3.17 h·μg/ml; total body clearance 0.37 ± 0.08 l/h/kg; apparent volume of distribution 8.51 ± 1.92 l/kg; mean residence time 20.6 ± 3.95 h. After PO administration, the principal pharmacokinetic parameters were as follows (mean ± SD): T1/2λz 15.6 ± 3.20 h; AUC0–∞ 7.94 ± 2.83 h·μg/ml; peak concentration 0.70 ± 0.14 μg/ml; time of peak 1.43 ± 0.53 h. The absolute bioavailability of ITR oral solution after oral administration was 52.1 ± 11.6%.
Conclusions and relevance
The disposition of ITR oral solution in cats is characterised by a long terminal half-life, a short peak time and moderate bioavailability.
Introduction
In recent years, an increasing number of fungal infections have been spread by animals. Almost 20% of fungal infections of human skin can be the result of close contact with cats and dogs. The infections not only endanger the health of cats and dogs, but also threaten human health.1,2
Itraconazole (ITR) is a first-generation synthetic triazole antifungal agent that is commonly used for a number of indications, including systemic and superficial mycosis.3,4 It has a wide antifungal spectrum, including moulds, dimorphic fungi, yeasts and dermatophytes. 5 Similar to the mode of action of ketoconazole, ITR can selectively destroy cytochrome P-450-mediated ergosterol synthesis in fungal membranes, which leads to death of the fungal cells. 6 Although a limited number of ITR products are licensed for use in animals, many of those available for the treatment of mycoses in humans are used by veterinary practitioners.
Currently, the pharmacokinetics of ITR in rats, humans, dogs and horseshoe crabs have been reported.7–12 Although several studies on the determination of ITR in cat plasma have been evaluated, 13 a literature search revealed that pharmacokinetic evaluations after ITR oral solution to cats are scarce.
The aim of this study was to assess the pharmacokinetics of ITR in eight cats after intravenous (IV) and oral (PO) administration, and to calculate the oral bioavailability of ITR oral solution in cats.
Materials and methods
Animals
Eight healthy adult family cats (four females, four males) weighing (mean ± SD) 2.39 ± 0.18 kg were studied. They were kept individually in cages under the same standard laboratory conditions. The cats were fed the same regular volume of dry cat food without salt restriction twice a day. Water was freely available. Food was withheld before the experiment and until 10 h after drug delivery.
The study was performed with the approval of the Institutional Animal Care and Use Committee of South China Agricultural University.
Chemicals and reagents
A standard of ITR (99.3% purity) was purchased from the National Institute for the Control of Pharmaceutical and Biological Products. The bulk of ITR (99.9% purity) and ITR oral solution (10 mg/ml) were provided by Shanghai Hanwei Bio-Pharmaceutical Technology Co. The ITR intravenous solution (10 mg/ml) was prepared by the College of Veterinary Medicine (South China Agricultural University). Analytical grade trifluoroacetic acid (TFA), n-hexane, acetic acid and methyl tert-butyl ether (MTBE) were purchased from Guangzhou Reagent Factory, and the other solvents were high-performance liquid chromatography (HPLC) grade and purchased from CNW Technologies GmbH.
Experimental design
On the day before an experiment, IV catheters (24 G, 4.8 cm) were placed in the right elbow vein of the cats and protected with an adhesive bandage. The cats were allowed to recover for 12 h.
According to a single dose, two period crossover design with a washout of 15 days, the eight fasted cats were randomly divided into two groups for IV and PO administrations followed by a single dose of 5 mg/kg. After the 15 day washout period, the treatment was changed repeatedly. The first administration of ITR was delivered as an IV solution to the left cephalic vein using a plastic syringe fitted with an infusion set. The second administration was by instillation using a plastic syringe at the corners of the mouth within 10 s. Each cat was observed daily and abnormal findings were recorded during the experiments.
Blood sampling
After IV and PO administration, blood samples were collected into heparin tubes at 0.083 h, 0.25 h, 0.5 h, 1 h, 2 h, 3 h, 4 h, 6 h, 9 h, 12 h, 24 h, 36 h, 48 h, 60 h, 72 h and 0.167 h, 0.25 h, 0.5 h, 1 h, 2 h, 3 h, 4 h, 6 h, 9 h, 12 h, 24 h, 36 h, 48 h, 60 h, 72 h after treatment, respectively. All samples were immediately centrifuged at 2254 g/min for 10 mins and stored at −20ºC until analysis.
Sample preparation
A 500 μl blood sample was placed into a 15 ml polypropylene centrifuge tube with 200 μl sodium hydroxide (1 mol/l) and extracted with 5 ml of MTBE. The mixture was vortexed for 5 mins and then centrifuged at 9016g/min for 10 mins at 4ºC. The supernatants were transferred to another tube. The extract was concentrated up to dryness under nitrogen, and the residues were dissolved in 500 μl acetonitrile/aqueous solution of 6% acetic acid (2:3, v/v) and directly injected into the HPLC system.
Instrumentation and analytical method
ITR concentrations in serum were determined by using a slightly modified method adapted from Redmann and Charles. 14 Briefly, the chromatographical system was a Waters 2475 Series HPLC and the column used was a reversed-phase Hypersil BDS C18 (5 μm, 250 mm × 4.6 mm). The mobile phase consisted of 0.2% TFA aqueous solution/acetonitrile (45:55, v/v). The elution was carried out on a C18 column with a flow rate of 1.0 ml/min. Excitation and emission wavelengths were 260 nm and 365 nm, respectively. The temperature was controlled at 25ºC and the injection volume was 20 μl. The calibration curve was linear over the concentration range of 0.02– 2.00 μg/ml, with a correlation coefficient (r) >0.999. The limit of quantification for ITR in plasma was 0.02 μg/ml. Both the inter- (n = 5) and intra-assay (n = 15) precision values were <9.16%, and the accuracy values ranged from 86.94–99.4% for the quality control samples.
Pharmacokinetic analysis
The pharmacokinetic parameters of ITR were calculated by non-compartmental analysis using WinNonlin 5.2.1 (Pharsight). The area under the curve from time zero to infinity (AUC0–∞) was calculated by the trapezoidal rule with extrapolation to infinity. The absolute oral bioavailability (F) of ITR oral solution was estimated according to the calculation formula: (AUC0-∞ )oral/(AUC0–∞ )iv × 100.
Results
Clinical reflection
There were no side effects such as nausea, swelling or severe pains associated with IV or PO administration of ITR in cats.
Pharmacokinetic analysis
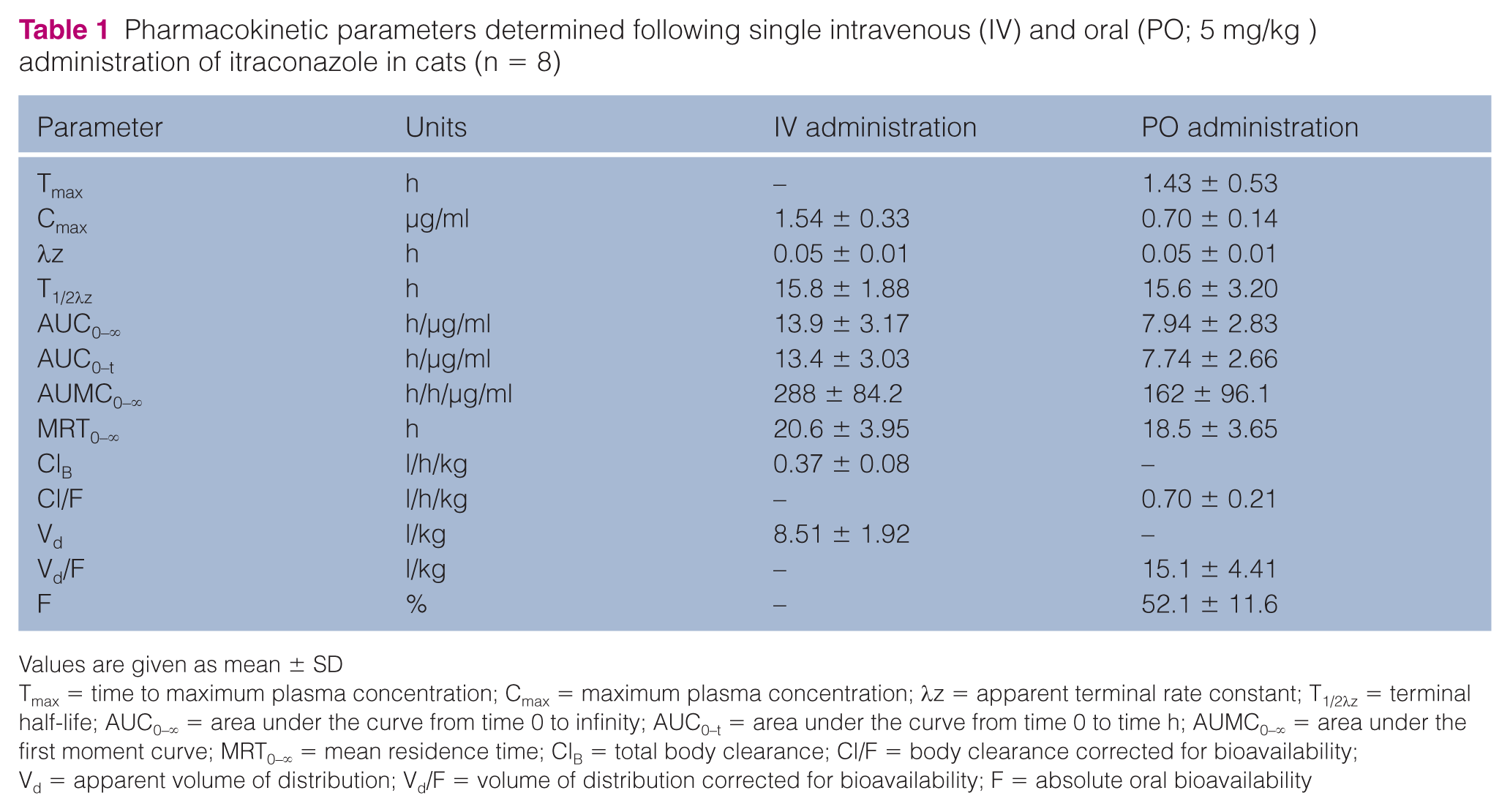
Pharmacokinetic parameters are presented as mean ± SD. The mean plasma concentration vs time curve after IV and PO administration, and their pharmacokinetic parameters, are shown in Figure 1 and Table 1, respectively.

Semi-logarithmic plot of itraconazole concentration vs time in plasma after an intravenous and oral dose (5 mg/kg) in cats (n = 8). Values are mean ± SD
Pharmacokinetic parameters determined following single intravenous (IV) and oral (PO; 5 mg/kg ) administration of itraconazole in cats (n = 8)
Values are given as mean ± SD
Tmax = time to maximum plasma concentration; Cmax = maximum plasma concentration; λz = apparent terminal rate constant; T1/2λz = terminal half-life; AUC0–∞ = area under the curve from time 0 to infinity; AUC0–t = area under the curve from time 0 to time h; AUMC0–∞ = area under the first moment curve; MRT0–∞ = mean residence time; ClB = total body clearance; Cl/F = body clearance corrected for bioavailability; Vd = apparent volume of distribution; Vd/F = volume of distribution corrected for bioavailability; F = absolute oral bioavailability
After the IV administration, ITR distributed widely with an apparent Vd of 8.51 ± 1.92 l/kg. The antifungal elimination from the blood was slow, with a total body clearance of 0.37 ± 0.08 l/h/kg, a terminal elimination half-life of 15.8 ± 1.88 h and a mean residence time (MRT) of 20.6 ± 3.95 h. After PO administration, ITR oral solution was absorbed rapidly, with a time to maximum plasma concentration (Tmax) of 1.43 ± 0.53 h, and the mean maximum concentration was 0.70 ± 0.14 μg/ml. ITR could be readily detected from plasma samples at 10 mins after administration. The terminal elimination half-life and MRT were 15.6 ± 3.20 h and 18.5 ± 3.65 h, respectively. Moreover, the AUC0-∞ of IV and PO administration were 13.4 ± 3.03 h/μg/ml and 7.94 ± 2.83 h/μg/ml, respectively. The absolute bioavailability of ITR oral solution was 52.1 ± 11.6%.
Discussion
The pharmacokinetics of ITR have been reported in a number of animal species. However, limited pharmacokinetic data exist for ITR in cats,13,15 especially with regard to oral solutions. This study examined the pharmacokinetics and bioavailability of ITR oral solution in healthy cats, which have not previously been assessed.
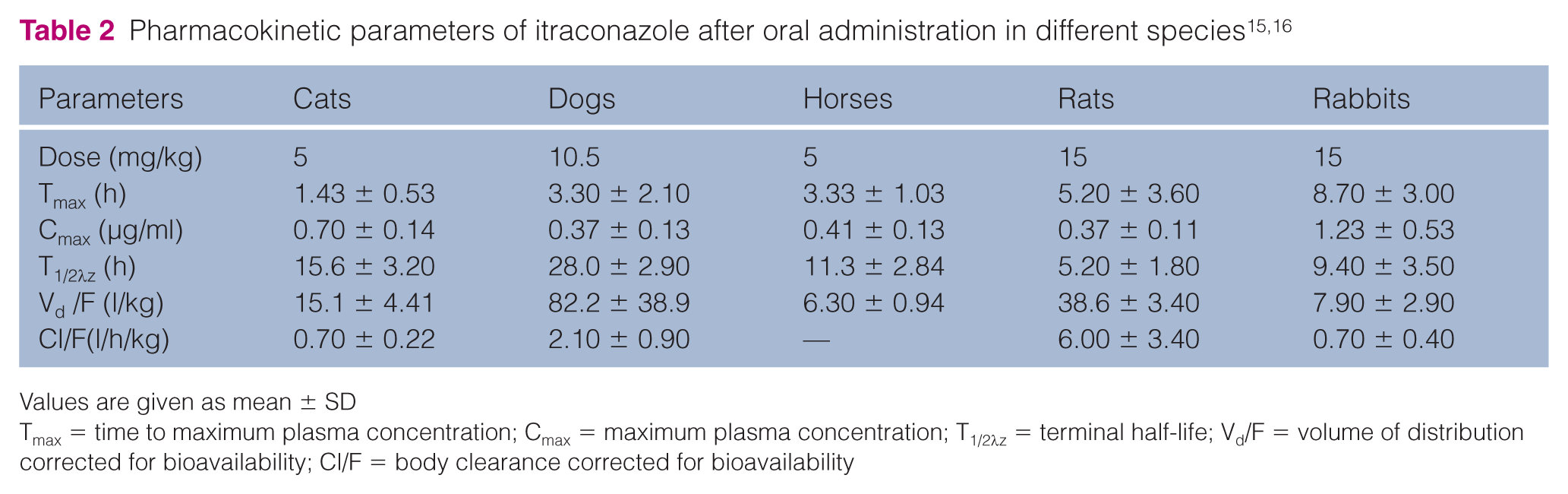
The primary pharmacokinetic parameters of ITR after PO administration in cats compared with other animal species are shown in Table 2. Based on Tmax, the absorption of ITR from the gastrointestinal tract was more rapid in cats (1.43 ± 0.53 h) than in horses (3.33 ± 1.03 h), dogs (3.30 ± 2.10 h), rats (5.20 ± 3.60 h) and rabbits (8.70 ± 3.00 h). With comparable oral doses, the maximum concentrations in cats were greater than those reported in horses (0.70 ± 0.14 vs 0.41 ± 0.13 μg/ml). The terminal elimination half-life of ITR after PO administration (15.6 ± 3.20 h) in cats was similar (P >0.05) to that of IV administration (15.8 ± 1.88 h), which is significantly shorter than in dogs (28.0 ± 2.90 h), and longer than in horses (11.3 ± 2.84 h), rabbits (9.40 ± 3.50 h) and rats (5.20 ± 1.80 h) following PO administration. The volume of distribution (corrected for bioavailability) after PO administration (15.1 ± 4.41 l/kg) was larger than that of IV administration (8.51 ± 1.92 l/kg) in cats and PO administration in horses (6.30 ± 0.94 l/kg). The large volume of distribution (corrected for bioavailability) observed after PO administration was likely due to ITR’s extensive distribution to nail, skin and tissue, 17 and its slow release from peripheral tissue to the blood. In the present study, the oral solution bioavailability in fasted cats was only 52.1 ± 11.6%, which is lower than in horses (65.0 ± 26.3%) 16 and in non-fasted cats (78.8 ± 28.0%). 13 This may be owing to different species and its first-pass metabolism, which has been reported in humans, rats and dogs.18–20 Furthermore, it is very difficult to compare the bioavailability of oral solution in cats with that of other animals as no data regarding IV administration in other animals or humans have, to our knowledge, been published.
Values are given as mean ± SD
Tmax = time to maximum plasma concentration; Cmax = maximum plasma concentration; T1/2λz = terminal half-life; Vd/F = volume of distribution corrected for bioavailability; Cl/F = body clearance corrected for bioavailability
Although the concentrations and bioavailability of oral solution in cats were low, the concentrations at the infection site were almost 10-fold higher than those in plasma concentrations.21,22 In addition, the geometric mean of minimum inhibitory concentration (MIC) values for 39 dermatophytic isolates was 0.078 μg/ml. 23 In our study, the concentrations of the target side above the MIC were likely maintained over 24.
Generally, common oral formulations of ITR are tablets and capsules. However, it is difficult to use these formulations to administer medicine to cats and dogs in veterinary clinics. ITR oral solution offers a significant advantage over capsules or tablets for veterinary use because cats may have difficulty in swallowing tablets or capsules, and it is very difficult to adjust the dosages of capsules or tablets for cats and dogs. Therefore, ITR oral solution is likely to have wide application in veterinary clinics, and there is a need to improve the absorption and bioavailability of the formulation continuously.
Conclusions
This study describes the pharmacokinetic properties of ITR oral solution, and determines the absolute bioavailability of the formulation in cats. The results contribute to the data on the kinetic disposition of ITR in cats.
Footnotes
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
Funding
This study was supported financially by the Program for Changjiang Scholars and Innovative Research Team in University of China (No. IRT13063) and the Natural Science Foundation of Guangdong Province, China (No. S2012030006590).