Abstract
Objectives
Feline allergic asthma is a common chronic lower airway disease characterized by clinical signs attributed to eosinophilic inflammation, airway hyper-responsiveness (AHR) and airway remodeling. Tachykinins released from sensory nerves and immune cells bind neurokinin-1 (NK-1) receptors in the lung. The resultant neurogenic airway inflammation has been implicated in asthma pathogenesis. In mouse models and spontaneous human asthma, NK receptor antagonists reduce bronchospasm and inflammation. We hypothesized that chronic administration of maropitant, an NK-1 receptor antagonist, would decrease clinical signs of asthma, AHR and eosinophilic inflammation in experimentally asthmatic cats.
Methods
Cats (n = 6) induced to have asthma using Bermuda grass allergen (BGA) were enrolled in a randomized, prospective, placebo-controlled crossover design study. Cats received either oral maropitant (2 mg/kg) or placebo q48h for 4 weeks; following a 2 week washout, cats were crossed-over to the alternate treatment. Study endpoints included subjective clinical scoring systems after BGA challenge, ventilator-acquired pulmonary mechanics to assess AHR after bronchoprovocation with methacholine, and collection of bronchoalveolar lavage fluid to quantify airway eosinophilia. Statistical analysis was performed using a Mann–Whitney rank sum test with P <0.05 considered significant.
Results
Administration of maropitant for 1 month in experimentally asthmatic cats produced no significant difference in clinical scoring scheme (P = 0.589 and P = 1.0), AHR (P = 0.818) or airway eosinophilia (P = 0.669) compared with placebo.
Conclusions and relevance
Chronic administration of maropitant was ineffective at blunting clinical signs, AHR and airway eosinophilia in experimental feline asthma and thus cannot be recommended as a novel treatment for this disorder.
Introduction
Feline asthma is a serious chronic respiratory condition resulting in clinical signs of cough, wheeze and episodic respiratory distress attributed to airway hyper-responsiveness (AHR), eosinophilic airway inflammation, and airway remodeling. 1 The immunologic driving force behind allergic asthma is aeroallergen-induced activated T helper 2 cells, which result in chronic inflammation and subsequently orchestrate a variety of pathologic changes. 2 Homeostasis in the lung is maintained, in part, by the neuroimmune axis. Crosstalk between these two systems is mediated via cytokines, neuropeptides and their receptors.3–5 In asthmatic individuals allergens and other airborne irritants induce neuropeptide release locally. Tachykinins are released both from sensory nerves and immune cells, and there is evidence that these neurogenic mediators play an important role in asthma pathogenesis both by facilitating acute bronchospasm and maintaining inflammation during periods of relative clinical quiescence.2–4
Tachykinin release from sensitized neurons sustains and amplifies the ongoing inflammatory response in rodent models of asthma and in humans with spontaneous asthma.6–10 This is accomplished by enhanced lymphocyte proliferation, immunoglobulin production, cytokine secretion and alterations in vascular permeability.3–5,11–15 Substance P (SP) is a neuropeptide released from sensory neurons and immune cells within the respiratory tract and exerts its effects by binding neurokinin-1 (NK-1) receptors. 3 SP and NK-1 activity have been found to be particularly important in the development of neurogenic inflammation. SP immunoreactive nerves stimulated by local irritants and nerve growth factors are increased in humans severely affected by asthma. 3 NK-1 receptors have been localized to respiratory leukocytes, the vascular endothelium and goblet cells, and are believed to contribute to cellular influx, edema and mucous production.16,17 Further, SP acts as a mitogen for smooth muscle cells, endothelial cells and fibroblasts that may contribute to the architectural changes seen the airways of asthmatic patients. 3
Corticosteroids represent the gold standard of therapy for asthma. The efficacy of corticosteroids may include attenuation of SP-induced inflammation because the NK-1 receptor gene has been found to have glucocorticoid responsive elements.2,18 However, comorbidities including diabetes mellitus, cardiac disease and recurrent upper respiratory infections make corticosteroid therapy relatively or absolutely contraindicated in asthmatic cats. Studies examining rodent models of asthma have shown the NK-1 receptor antagonism may have promise as a means of reducing inflammatory cell influx and AHR.7,17,19 Studies in human asthmatics utilizing mono-antagonism of the NK-1 receptor are lacking; however, examination of these studies in addition to those examining combined NK-1 and NK-2 antagonists have shown mild effects on AHR.10,20,21 Maropitant citrate (Cerenia) is an NK-1 receptor antagonist marketed as an antiemetic in cats with an excellent safety profile. 22 It has been used anecdotally for the treatment of naturally occurring feline asthma, without the benefit of objective scientific evaluation. However, if effective in modulating the SP-mediated neuroimmune axis to disrupt airway inflammation and/or AHR, maropitant may represent an effective treatment for asthmatic cats. This study hypothesized that in experimentally asthmatic cats, chronic every-other-day dosing of maropitant would reduce clinical signs after allergen challenge, AHR measured by ventilator-acquired pulmonary mechanics and airway eosinophilia.
Materials and Methods
Cats
Six cats were enrolled in a prospective, placebo-controlled, crossover study using an established experimental model of feline asthma. Asthma was induced using Bermuda grass allergen (BGA) as previously described. 23 The asthmatic phenotype was confirmed with positive skin reactivity to BGA and bronchoalveolar lavage fluid (BALF) eosinophil percentage of >17%. This study was approved by the University of Missouri Care and Use Committee (Animal Care and Use Committee protocol #7891). Cats were cared for in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
Treatment
Each cat was randomly assigned to either the maropitant group (2 mg/kg PO q48h in a food treat for 28 days) or the placebo group (food treat alone). The cats received weekly aerosol challenges, including an aerosol challenge 24 h prior to bronchoprovocation testing and BALF collection. After a 2 week washout period the cats were crossed over to the alternate treatment.
Subjective clinical scoring system
A visual analog scale (VAS) was used to score clinical signs 10 mins after airway challenge with BGA. The VAS used a 10 cm scale to assess signs observed ranging from no clinical signs (0 cm) to extreme respiratory distress (10 cm). The distance from 0 to the patient score was then measured to provide a quantitative VAS score. The VAS was performed by a single investigator (MG). A clinical score was also assigned to each cat 10 mins after BGA challenge using a nine-point system (Table 1).
A composite clinical scoring system was used to evaluate the cats following aerosol challenge with Bermuda grass allergen. The clinical visual analog scores were assessed by the same individual (MG). For the clinical score, cats were given a score between 0 and 9 24

RR = respiratory rate
Measurement of AHR
Ventilator-acquired pulmonary mechanics was performed on anesthetized and mechanically ventilated cats as previously described, with minor modification. 23 Briefly, ketamine (30 mg IV) was used as a premedicant, propofol was used for induction (6 mg/kg) and maintenance (0.3 mg/kg/min), and cisatracurium (0.1 mg/kg IV of with additional doses of 0.01–0.02mg/kg titrated to effect) was administered for neuromuscular blockade. Neuromuscular blockade was monitored with a peripheral nerve stimulator. Edrophonium (0.5 mg/kg IV) was administered to reverse neuromuscular blockade after completion of data collection when indicated. Pulse oximetry was used to monitor oxygen saturation (SpO2). Initial ventilator settings were as follows: tidal volume was 10 ml/kg, respiratory rate was 10 breaths/min, inspired oxygen fraction was 0.4, inspiratory to expiratory ratio was 1:3, and the positive end expiratory pressure was 2 cm H2O. Parameters recorded on a breath-by-breath basis on the ventilator included airway resistance, compliance and peak pressure; after inspiratory and expiratory breath hold plateau pressure and endogenous positive end expiratory pressure were measured. Methacholine (MCh) challenge was terminated when airway resistance reached 200% of baseline (EC200raw). Prior to data collection, cats were ventilated for 5 mins to allow for steady-state conditions.
Saline was nebulized for 30 s followed by 4 mins of data collection to determine baseline data. MCh was aerosolized for 30 s with 4 mins of data collection between each dose. Doubling doses of MCh from 0.0625 −32.0000 mg/ml were administered serially until the intended study endpoints were reached. The intended endpoint of the study was the effective concentration of MCh to increase airway resistance by 200% over baseline (EC200Raw) or oxygen desaturation <75%. The EC200Raw was calculated by linear interpolation of the log plot of the dose–response curve; the maximum airway resistance obtained over each 4 min period of data collection for each dose of MCh was used for this calculation.
Collection and analysis of BALF
Using a 20 ml aliquot of sterile saline, BALF was collected in a blind fashion through the endotracheal tube after completion of ventilator-acquired pulmonary mechanics. 25 A differential count was performed on a Wright’s stained cytospin to evaluate the percentage of eosinophils.
Statistical analysis
Mann–Whitney rank sum tests were used to evaluate the differences in VAS score, EC200Raw and BALF eosinophil percentage between the maropitant and placebo groups. A P value <0.05 was considered significant.
Results
Chronically administered maropitant failed to decrease the VAS score or the composite clinical score after allergen challenge compared with placebo (P = 0.589 and P = 1.00, respectively; Figure 1a,b). Maropitant did not significantly dampen AHR, as indicated by EC200Raw in response to MCh challenge vs placebo (P = 0.818; Figure 2). No biologically significant alteration in any other measured pulmonary mechanics parameter was noted (Table 2). Oxygen desaturation <75% after MCh challenge did not occur in any cat with any treatment; the mean SpO2 in all cats over all time points was 98 ± 1%. Blockade of the NK-1 receptor by maropitant did not diminish airway eosinophilia as there was no significant difference in this parameter between maropitant and placebo treatment groups (P = 0.699; Figure 3).

(a,b) Six experimentally induced asthmatic cats receiving maropitant and placebo in random order were evaluated for clinical signs after allergen challenge using (a) a visual analog scale (VAS) score and (b) a subjective clinical composite score. The boxes represent the 25th and 75th quartiles, with a horizontal line at the median. The whiskers represent the range of the data. The black squares represent the mean. No significant difference was observed between the maropitant or placebo groups based on subjective VAS or clinical composite score (P = 0.589 and P = 1.00, respectively)
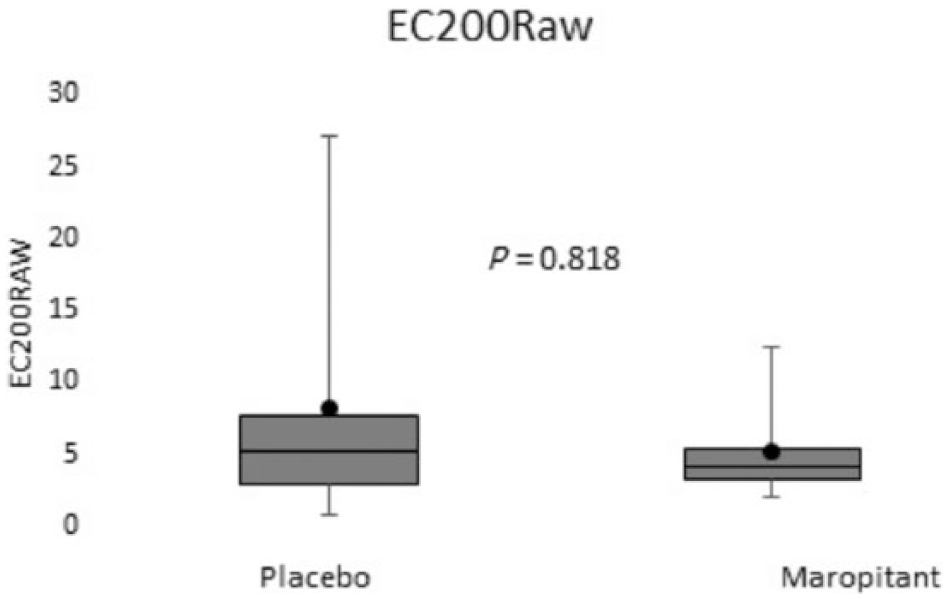
Six experimentally induced asthmatic cats receiving placebo and maropitant as part of a randomized, placebo-controlled, prospective, crossover study were evaluated for airway hyper-responsiveness (AHR) following methacholine (MCh) challenge. The boxes represent the 25th and 75th quartiles, with a horizontal line at the median. The whiskers represent the range of the data. The black squares represent the mean. Maropitant did not significantly dampen AHR as measured by the EC200Raw in response to MCh vs placebo (P = 0.818). Baseline airway resistance was not significantly different between treatments (P = 0.535); data not shown
A variety of other parameters relevant to lung function were determined on experimentally asthmatic cats undergoing methacholine bronchoprovocation, including peak pressure – plateau pressure; plateau pressure – positive end expiratory pressure; and change in plateau pressure. Increases in peak pressure – plateau pressure are proportional to airway resistance. Increased plateau pressure – positive end expiratory pressure values indicate decreased respiratory compliance. Increases in plateau pressure (change in plateau pressure) imply enhanced airflow limitation. No significant changes in any parameter were observed between the maropitant and placebo groups

Values are given as mean ± SD

Six experimentally induced asthmatic cats receiving placebo and maropitant in random order were evaluated for airway eosinophilia. Bronchoalveolar lavage samples were collected in a blind fashion. A differential count was performed on a Wright’s stained cytospin to evaluate the percentage of eosinophils. The boxes represent the 25th and 75th quartiles, with a horizontal line at the median. The whiskers represent the range of the data. The black squares represent the mean. In our cats, maropitant did not dampen airway eosinophilia compared with placebo (P = 0.699)
Discussion
To our knowledge, this is the first study, using an experimental feline asthma model, to evaluate the utility of chronic, 1 month use of maropitant as a treatment for asthma. In contrast to our hypothesis, chronically administered maropitant did not diminish clinical signs after allergen challenge (indicated by the lack of change in VAS score or composite clinical score), AHR or airway eosinophilia vs placebo. This is in contrast to rodent studies, which showed a more favorable response to NK-1-receptor blockade by demonstrating decreased eosinophil influx and AHR, although the effects on AHR were limited to the late phase.17,19 Human studies evaluating NK-1 receptor blockade have shown mild effects on AHR, although studies are limited. 7
Maropitant has been shown to be effective as an antiemetic, with excellent affinity for the NK-1 receptor. The dose of maropitant selected in this study exceeded the recommended dose by 200%, making insufficient dosing and poor receptor affinity unlikely as the cause of the lack of a statistically significant response. 22 Chronic administration allowed us to reach steady-state concentrations, also making inadequate dosing unlikely. Maropitant was administered in a food treat to minimize the stress associated with oral administration of medication. The same food treat and timing was used in the placebo group to reduce introduction of variables between groups.
Neurogenic inflammation is complex, involving multiple receptors (NK-1, NK-2 and NK-3) and mediators (SP, neurokinin A [NKA], neurkokinin B). The NK-1 receptor is located predominantly on the bronchial vessels, epithelial cells, submucosal glands and vascular endothelium. 3 The tachykinin receptor localized to airway smooth muscle is predominantly NK-2. 3 While SP is capable of binding to NK-2 receptors, the major mediator for this receptor type is NKA.3,26,27 Human asthmatic patients are hypersensitive to both SP and NKA, with increased NKA and SP concentrations within BALF.12,13,28 As maropitant is highly specific for the NK-1 receptor, this may provide an explanation for the lack of response in clinical signs and AHR in the cats of this study.
There are several additional reasons why maropitant was not effective in this study. First, tachyphylaxis, the diminished response to a drug/compound after multiple doses, secondary to chronic inflammation and multiple allergen challenges may result in a subsequent minimal response to NK-1 receptor antagonism. The release of SP and its binding to the NK-1 receptor results in rapid internalization of the NK-1 receptor, leading to desensitization of cells to SP signaling. 29 Second, the presence of chronic inflammation may result in depletion of SP (as has been shown for vasoactive intestinal polypeptide in asthmatic compared with healthy humans,17,30–32) making the effect of NK-1 receptor blockade less effective in the management of chronic asthma. Finally, the perpetuation of airway inflammation is dependent on multiple redundant pathways and it has been speculated that following the establishment of a persistent cell-mediated inflammatory response, blockade of tissue NK-1 receptors is insufficient to prevent further eosinophil influx and airway remodeling.3,33,34
Conclusions
Maropitant did not reduce airway AHR or airway eosinophilia in experimentally induced feline asthma. Thus, this study did not support the use of maropitant in feline asthma. It is possible that dual receptor blockade (NK-1 and NK-2) may be more efficacious at blunting the hallmark features of allergic asthma. Further study is needed to determine if chronic airway allergic inflammation depletes neurogenic inflammatory mediators and therefore limits the utility of downstream tachykinin receptor antagonism to treat allergic airway disease. 35
Footnotes
Acknowledgements
We would like to thank Dr Hans Rindt for his technical assistance.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
Funding
Funding for this study was provided by the Winn Feline Foundation. Maropitant was generously donated by Zoetis.
