Abstract
Objectives
Feline allergic asthma is a chronic inflammatory disorder of the lower airways that may manifest with acute, life-threatening clinical signs. Tachykinins released from sensory nerves and immune cells binding neurokinin (NK)-1, NK-2 and NK-3 receptors have been implicated in asthma pathogenesis. Maropitant, an NK-1 receptor antagonist, blocks neuroimmune pathways and may be a viable treatment option for cats in asthmatic crisis. Using an experimental chronic allergic feline asthma model, we hypothesized that a single dose of maropitant given immediately after allergen challenge would blunt clinical signs, airway hyperresponsiveness (AHR) and airway eosinophilia.
Methods
Cats (n = 7) induced to have an asthmatic phenotype using Bermuda grass allergen (BGA) were enrolled in a prospective, placebo-controlled crossover design study. Cats randomly received maropitant (2 mg/kg SC) or placebo (saline SC) immediately post-BGA challenge, followed 12 h later by pulmonary mechanics testing and measurement of airway eosinophils. After a 2 week washout, cats were crossed-over to the alternate treatment. Study endpoints included subjective clinical scoring systems post-BGA challenge, ventilator-acquired pulmonary mechanics to assess AHR after bronchoprovocation with methacholine and collection of bronchoalveolar lavage fluid to quantify airway eosinophilia. Data were analyzed using a Mann–Whitney rank sum test with P <0.05 considered significant.
Results
A single injection of maropitant failed to diminish clinical composite score (P = 0.902), visual analogue scale scoring (P = 0.710), AHR (P = 0.456) or airway eosinophilia (P = 0.165) compared with placebo.
Conclusions and relevance
A single injection of maropitant given immediately post-allergen challenge was ineffective at blunting clinical signs, AHR and airway eosinophilia, and cannot be recommended as treatment for feline status asthmaticus.
Introduction
Feline asthma is thought to be due to allergen-specific airway responses, leading to a constellation of acute and chronic clinical signs. Acute exposure to clinically relevant allergens can trigger a potentially life threatening crisis called ‘status asthmaticus’. Airflow limitation causing respiratory distress can either be associated with the early phase of asthma that occurs minutes after allergen exposure or the late phase of asthma that can appear hours later. Early events are associated with allergen-specific immunoglobulin E (IgE) cross-linking and local mast cell degranulation; late-phase events occur secondary to eosinophil and T-cell influx with elaboration of other cytokines and eicosanoids. The releases of tachykinins from sensitized neurons have also been implicated in both the early and late stages phases of asthma. The result of tachykinin release is enhanced lymphocyte proliferation, immunoglobulin production, alterations in vascular permeability and cytokine secretion from airway leukocytes.1–6 Substance P (SP) is a neuropeptide released from sensory neurons and immune cells within the respiratory tract that exerts its effects by binding neurokinin 1 (NK-1) receptors. SP and NK-1 activity have been found to be particularly important in the development of neurogenic inflammation.
SP immunoreactive nerves are stimulated by local irritants and nerve growth factors, and have been shown to increase in number in severely affected human asthmatics. 7 NK-1 receptors have been localized to respiratory leukocytes, the vascular endothelium and goblet cells, and contribute to cellular influx, edema and mucous production.1,8,9 Additionally, increased NK-1 receptor mRNA has been identified by alveolar macrophages in a rodent model. 10
Bronchodilators and corticosteroids represent the gold standard of therapy for asthma, with bronchodilators representing the first line of therapy for an acute asthmatic crisis.11–13 Unfortunately, comorbidities such as cardiac disease often make these treatments relatively or absolutely contraindicated in many asthmatic cats. Studies examining rodent models of asthma have shown that NK-1 receptor antagonism may have promise as a means of reducing inflammatory cell influx and airway hyperresponsiveness (AHR).9,14,15 Additionally, studies in human asthmatics utilizing combined NK-1 and NK-2 antagonists have shown mild effects on AHR.3,16 However, studies evaluating the effect of specific NK-1 receptor antagonists have not been reported.
Maropitant citrate (Cerenia; Zoetis) is an NK-1 receptor antagonist marketed as an antiemetic in cats. 17 It has been used anecdotally for the treatment of naturally occurring feline asthma, including during acute asthmatic episodes, without the benefit of supporting scientific evidence. However, if effective in modulating the SP-mediated neuroimmune axis by disrupting airway inflammation and/or AHR, maropitant may represent an alternative treatment for asthmatic cats. Experimental models of feline allergic asthma can be useful to test therapeutics that may be beneficial in spontaneous feline asthma. This study hypothesized that in experimentally asthmatic cats, a single dose of maropitant administered immediately after allergen challenge would reduce clinical signs, AHR measured by ventilator-acquired pulmonary mechanics and airway eosinophilia.
Materials and methods
Cats
Seven cats were enrolled in a prospective, placebo-controlled, crossover study using an established experimental model of chronic feline asthma. Asthma was induced using Bermuda grass allergen (BGA) as previously described.18,19 The asthmatic phenotype was confirmed with positive skin reactivity to BGA and a bronchoalveolar lavage fluid (BALF) eosinophil percentage of >17%. Cats received bimonthly-to-weekly BGA aerosol challenges to maintain the asthmatic phenotype until the study commenced; weekly BGA challenges were then continued for the duration of this study. The study was approved by the University of Missouri Care and Use Committee (Institutional Animal Care and Use Committee protocol #7891). Cats were cared for in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
Treatment
Each cat was randomly assigned to either the maropitant group (2 mg/kg SC) or the placebo group. A placebo (saline) was administered using the equivalent volume to the maropitant group using the same protocol in order to reduce introduction of variables between groups.
Twelve hours prior to bronchoprovocation testing and BALF collection, cats received a BGA aerosol challenge, followed immediately by one of the aforementioned treatments. After a 2 week washout period, the cats were crossed over to the alternate treatment.
Subjective clinical scoring systems
A composite clinical score was assigned to each cat 10 mins after BGA challenge using a nine-point system (Table 1). A visual analog scale (VAS) was also used to score clinical signs 10 mins after airway challenge with BGA and subsequent administration of maropitant or saline. The VAS used a 10 cm scale to assess signs observed ranging from no clinical signs (0 cm) to extreme respiratory distress (10 cm). The distance from 0 to the patient score was measured to provide a subjective, yet quantitative VAS score. The subjective clinical scores were assigned by a single investigator (MG).
A composite clinical scoring system was used to evaluate all seven cats following aerosol challenge with Bermuda grass allergen. The clinical score and visual analog scale score were assessed by the same individual (MG). For the clinical score, cats were given a score between 0 and 9 20

RR = respiratory rate
Measurement of AHR
Ventilator-acquired pulmonary mechanics was performed on anesthetized and mechanically ventilated cats as previously described. 19 Briefly, ketamine (30 mg IV) was used as a premedicant, propofol was used for induction (6 mg/kg) and maintenance (0.3 mg/kg/min), and cisatracurium (0.1mg/kg IV, with additional doses of 0.01–0.02 mg/kg titrated to effect) was administered for neuromuscular blockade. Neuromuscular blockade was monitored with a peripheral nerve stimulator. Edrophonium (0.5 mg/kg IV) was administered to reverse neuromuscular blockade after completion of data collection as needed. Pulse oximetry was used to monitor oxygen saturation (SpO2). Initial ventilator settings were as follows: tidal volume of 10 ml/kg, respiratory rate of 10 breaths/min, inspired oxygen fraction 0.4, inspiratory to expiratory ratio of 1:3 and a positive end expiratory pressure of 2 cm H2O. Parameters recorded on a breath-by-breath basis on the ventilator included airway resistance (Raw), compliance and peak pressure. After inspiratory and expiratory breath holds, plateau pressure and endogenous positive end expiratory pressure were measured. Methacholine (MCh) challenge was terminated when airway resistance reached 200% of baseline (EC200Raw). Prior to data collection, cats were ventilated for 5 mins to allow for steady-state conditions to be reached.
Saline was nebulized for 30 s followed by 4 mins of data collection to determine baseline data. MCh was aerosolized for 30 s with 4 mins of data collection between each dose. Doubling doses of MCh from 0.0625–32.0000 mg/ml were administered until intended endpoints were reached. The intended endpoint of the study was the effective concentration of MCh to increase airway resistance by 200% over baseline (EC200Raw) or oxygen desaturation <75%. The EC200Raw was calculated by linear interpolation of the log plot of the dose response curve with the maximum airway resistance obtained over each 4 min period of data collection for each dose of MCh being used for this calculation.
Collection and analysis of BALF
Using a 20 ml aliquot of sterile saline, BALF was collected in a blind fashion through the endotracheal tube after completion of ventilator acquired pulmonary mechanics. 21 A differential count was performed on a Wright’s stained cytospin to document the percentage of the total cell count that was eosinophils.
Statistical analysis
Mann–Whitney rank sum tests were used to evaluate the difference between clinical composite score, VAS score, EC200Raw and BALF eosinophil percentage between the maropitant and placebo groups. A P value <0.05 was considered significant.
Results
A single dose of maropitant failed to diminish the composite clinical score or VAS score (Figure 1; P = 0.902 and P = 0.710, respectively). Maropitant did not significantly dampen AHR after MCh bronchoprovocation vs placebo (Figure 2; P = 0.456). No statistically significant decrease in airway eosinophilia was observed in the maropitant vs placebo treatment groups (Figure 3; P = 0.165). No differences in total nucleated cell counts between treatments were noted (P = 0.937). Oxygen desaturation <75% did not occur in any cat during pulmonary mechanics testing. No statistically significant alteration in any other pulmonary mechanics parameter was noted (Table 2).

(a,b) Seven experimentally asthmatic cats receiving maropitant and placebo in random order were evaluated for clinical signs after allergen challenge using (a) a subjective clinical composite score and (b) a visual analog scale (VAS) score. The boxes represent the 25th and 75th quartiles, with a horizontal line at the median. The whiskers represent the range of the data. The black squares represent the mean. No significant difference was observed between the maropitant or placebo groups based on subjective clinical composite scores or VAS (P = 0.902 and P = 0.710, respectively).

Seven experimentally asthmatic cats receiving maropitant and placebo as part of a randomized, placebo-controlled, prospective, crossover study were evaluated for airway hyperresponsiveness (AHR) following methacholine (MCh) challenge. The boxes represent the 25th and 75th quartiles, with a horizontal line at the median. The whiskers represent the range of the data. The black squares represent the mean. Maropitant did not significantly alter AHR as measured by when the airway resistance exceeded 200% of baseline values (EC200Raw) in response to MCh (P = 0.456). Baseline airway resistance was not significantly different between treatments (P = 0.535)
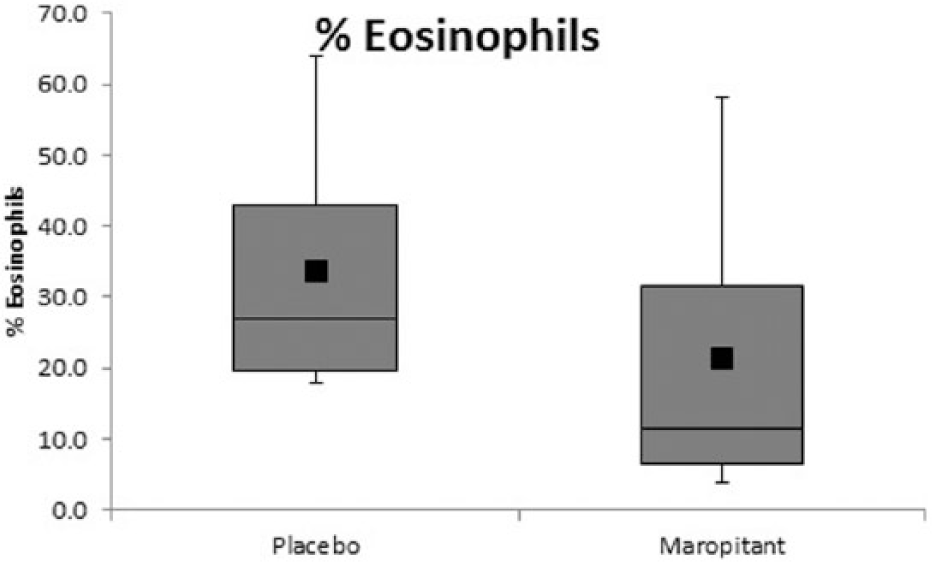
Seven experimentally induced asthmatic cats receiving maropitant and placebo in random order were evaluated for airway eosinophilia. Bronchoalveolar lavage samples were collected in a blind fashion. A differential count was performed on a Wright’s stained cytospin preparation to evaluate airway eosinophilia (expressed as a percentage of the total cell count identified as eosinophils). The boxes represent the 25th and 75th quartiles, with a horizontal line at the median. The whiskers represent the range of the data. The black squares represent the mean. Maropitant did not significantly decrease airway eosinophilia compared with placebo (P = 0.165)
Other ventilator-acquired parameters relevant to lung function were determined on experimentally asthmatic cats undergoing methacholine bronchoprovocation, including plateau pressure – positive end expiratory pressure, peak pressure – plateau pressure and change in plateau pressure. Increased plateau pressure – positive end expiratory pressure values indicate decreased respiratory compliance. Increases in peak pressure – plateau pressure are proportional to airway resistance. Increased plateau pressure (change in plateau pressure) implies enhanced airflow limitation. No significant changes in any parameter were observed between the maropitant and placebo groups

IQR = interquartile range
Discussion
To our knowledge, this is the first study to evaluate, using an experimental feline asthma model, the utility of a single dose of maropitant as a treatment for early and late phases of asthma. In contrast to our hypothesis, a single subcutaneous dose of maropitant did not diminish clinical signs after allergen challenge (indicated by lack of change in composite clinical score and VAS score), AHR or airway eosinophilia vs placebo. This is in contrast to rodent studies that demonstrated that NK-1 receptor antagonists decreased airway eosinophilia and late-phase AHR.9,15 Clinical assessment of a NK-1 receptor antagonist during acute asthmatic crisis has not been previously performed, and studies examining sole blockade of the NK-1 receptor in humans are lacking. 14 The experimental model of feline asthma is intended to be used as a preclinical model for spontaneous asthma in pet cats and in humans. While models may have differences from spontaneous disease, they are useful to test promising therapeutics prior to trials in clinically affected patients.
Maropitant has been shown to be effective as an antiemetic with strong affinity for the NK-1 receptor. 17 The dose of maropitant selected in this study exceeded the labeled dose by 200%, making insufficient dosing and poor receptor affinity unlikely as the cause for the lack of a statistically significant response. Bronchoprovocation was performed 12 h after a single dose of maropitant in this investigation. This time point was selected because previous studies have demonstrated therapeutic blood concentration of the drug 12 h after injection, and this time point encompasses the late-phase asthmatic response after allergen challenge. 17
Neurogenic inflammation is complex, involving multiple mediators (SP, neurokinin A [NKA], neurokinin B [NKB]), which bind to multiple receptors (NK-1, NK-2 and NK-3). 1 Each mediator preferentially activates a particular receptor, although at high concentrations each ligand can activate all receptors. The NK-1 receptor is located predominantly on bronchial vessels, epithelial cells, submucosal glands and vascular endothelium. The tachykinin receptor localized to airway smooth muscle is predominantly NK-2. While SP is capable of binding to NK-2 receptors, the major mediator for this receptor type is NKA. 1 NKA has been shown to be co-localized with SP and is released in response to similar stimuli. 22 Human asthmatic patients are hypersensitive to both SP and NKA, with increased NKA and SP concentrations within BALF, implying that both SP and NKA play a role in the pathogenesis of disease.3,23,24 As maropitant is highly specific for the NK-1 receptor, this may provide an explanation for the lack of response in clinical signs and AHR in the cats of this study.
There are additional reasons why maropitant may not have been effective in this study. First, we utilized a model that mimicked a clinical scenario in that allergen challenge preceded maropitant administration and where clinical response was assessed shortly after (10 mins) treatment. The time to maximum plasma concentration of maropitant in cats is 0.5–2.0 h following subcutaneous administration. 17 However, to be used successfully clinically, an ideal medication would be capable of blunting airflow limitation relatively quickly, which is why we elected to use this time point for evaluation. Second, the study was performed using a small sample size, which may have increased the risk of type 2 error.
The presence of chronic inflammation may result in depletion of SP (as has been shown for vasoactive intestinal polypeptide in asthmatic compared with healthy human subjects) making the effect of NK-1 receptor blockade less effective in the management of chronic asthma.9,25–27 This may represent a form of tachyphylaxis, whereby chronic inflammation and release of SP results in a diminished response over time and a minimal response to NK-1 receptor antagonism. The release of SP and binding of the NK-1 receptor also results in rapid internalization of the NK-1 receptor, potentially leading to desensitization of cells to SP signaling. 1 Finally, while inflammation in the lungs may, in part, be mediated by the influence of tachykinins, there are multiple redundant and overlapping pathways that may also be responsible for the pathogenesis of this disease and blockade of the NK-1 receptors may be insufficient to prevent further eosinophil influx.
Conclusions
The present study showed that a single dose of maropitant is ineffective at mitigating the clinical signs, AHR and airway eosinophilia in experimentally induced feline asthma.
Footnotes
Acknowledgements
We thank Dr Hans Rindt and Amber Graham for their technical assistance.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
Funding
Funding for this study was provided by the Winn Feline Foundation. Cerenia was graciously provided by Zoetis.
