Abstract
Objectives
Many cats with gastrointestinal signs have coexisting abnormalities in the intestine, liver and pancreas. Investigations typically involve clinicopathological tests, diagnostic imaging and biopsy, either at coeliotomy or by non-invasive means. While exploratory coeliotomy enables all organs to be sampled simultaneously, it is invasive and might not be necessary. The aim of the current study was to assess the performance of preliminary clinical information in predicting the histopathological presence of abnormalities in alimentary tract organs in cats.
Methods
The records of 38 cats with alimentary tract signs, which had ultimately undergone exploratory coeliotomy and surgical biopsy, were reviewed. The clinical signs, clinicopathological findings, diagnostic imaging findings and histopathology results were reviewed.
Results
On histopathological analysis, lesions were detected in 29/37 (78%) liver biopsies, in 29/35 (83%) gastrointestinal biopsies and in 17/37 (46%) pancreatic samples, the majority of which were inflammatory in nature. Clinical signs were generally poor markers of the presence of lesions in the alimentary tract. Further, while liver enzyme activity was relatively specific (88–100%) for detecting histopathological abnormalities in the liver, sensitivity was poor (11–50%). Pancreatic histopathological abnormalities were present in 1/3 of the cats with a positive pancreas-specific lipase result, and in 6/8 cats with a negative result. While relatively specific (57–100%) for both intestinal (57–100%) and hepatic (71–80%) histopathological abnormalities, abdominal ultrasonography lacked sensitivity for both organs (intestine 50–80%; liver 20–25%). In contrast, ultrasonography was relatively sensitive (50–80%), but not specific (17–22%) for detecting pancreatic lesions.
Conclusions and relevance
Clinical signs, and clinicopathological and ultrasonographic abnormalities lack precision for hepatic and pancreatic histopathological lesions in cats with alimentary tract signs, and cannot reliably predict from which organs biopsies should be collected. Arguably, therefore, exploratory coeliotomy is necessary to determine the site of histopathological abnormalities in feline alimentary tract disorders.
Introduction
Diagnosis of alimentary disorders can be challenging in cats as there can be concurrence of lesions in the gastrointestinal tract, the liver and the pancreas. This association was first described in 1984, 1 and the term feline inflammatory disease (FID) was coined to describe such cases. Since then, this syndrome has been extensively characterised in the veterinary literature.2–7 Although the precise pathogenesis is not understood, the proximity between the liver, pancreas and duodenum might predispose, 8 and it has been hypothesised that inflammatory lesions arise secondary to passage of enteric bacteria along the pancreatic and common bile ducts.
Histopathological examination of tissue samples is said to be the gold standard for diagnosis of FID.2,9–12 Given the fact that lesions can be present in any or all of the alimentary organs, exploratory coeliotomy is arguably the most appropriate method for biopsy collection, although its invasiveness makes it difficult to justify. As a result, some clinicians use data from preliminary clinical investigations, namely history, physical examination, clinicopathological testing and diagnostic imaging, to decide which organs might be involved and to what extent. For instance, when vomiting or diarrhoea are observed, or when thickened intestinal loops are identified on abdominal palpation, these would prompt further investigation of and biopsy sampling from the gastrointestinal tract. Similarly, the identification of icterus, increased liver enzyme activity, hyperbilirubinaemia and bilirubinuria might prompt a clinician to investigate and biopsy the liver, while the identification of abdominal pain and increased serum pancreas-specific lipase concentration might suggest the need for pancreatic biopsy. The findings of abdominal ultrasonography can also be used to identify organ-specific changes. In this way, preliminary clinical data are used to help the clinician decide which organs to biopsy, whether coeliotomy is needed or whether a less invasive method of biopsy collection would suffice (ie, ultrasound-guided biopsy or gastrointestinal endoscopy). However, the reliability of this approach has not been critically assessed. The aim of the current study was to compare the results of preliminary clinical data with the results of histopathological examination in a cohort of suspected FID cases that ultimately had gastrointestinal, pancreatic and hepatic biopsies collected at exploratory coeliotomy.
Materials and methods
Study recruitment and eligibility criteria
The study was a retrospective, cross-sectional, cohort study involving cats seen at a single centre. To identify eligible cases, the medical records of cats referred to the Clinique Alliance Digestive Diseases Consultation, Bordeaux, France, between January 2006 and February 2013 were reviewed. Eligibility criteria included the presence of signs compatible with FID (eg, anorexia, lethargy, vomiting, diarrhoea, icterus and weight loss), and having a clinical history available for review, which included clinical signs, routine clinicopathological examination, abdominal ultrasonographic examination and collection of hepatic, pancreatic and small intestinal biopsies at exploratory coeliotomy. All cats must have had either liver enzyme activity or abdominal ultrasonography, or both performed, in addition to histopathological assessment of at least two alimentary tract organs. Additional eligibility criteria included the absence of complete biliary tract obstruction, signs of encephalopathy, marked abdominal lymphadenopathy, or histopathological findings compatible with idiopathic hepatic lipidosis. Either primary or referral cases were eligible for inclusion. All diagnostic investigations were performed for the direct clinical benefit of the case, and owners gave written consent.
Data recording
Preliminary clinical data
Information was recorded on signalment, clinical signs (eg, what signs were present, as well as duration and severity), dietary and medication history, and physical examination findings (eg, body weight, body condition score, temperature, thoracic auscultation findings and abdominal palpation, as well as the presence of icterus, peripheral lymphadenopathy and dehydration).
Clinicopathological investigations
The clinicopathological investigations used in cases included haematological and serum biochemical analyses, total thyroxine measurement and pancreas-specific lipase assay. Haematological and serum biochemical analyses were not repeated if the referring veterinarian had recently performed them. Total bilirubin concentration was only measured if the serum was visibly icteric, or if the clinician requested it specifically. During the course of the study, haematology was performed with one of two in-house haematology analysers (ProCyte Dx Hematology Analyzer [IDEXX] and LaserCyte Dx Hematology Analyzer [IDEXX]), both calibrated for feline cell morphology. Similarly, two in-house serum biochemistry analysers (IDEXX VetLab Station [IDEXX] and Kbio 4 biochemistry analyser [KITVIA]) were used. In 21 cats, total thyroxine concentration was measured with one of two in-house biochemistry analysers (SNAP Reader [IDEXX] and SNAPshot Dx Analyzer [IDEXX]), while pancreatic lipase was measured using a feline-specific immunoassay (feline pancreas-specific lipase, fPL Test; IDEXX) in 11 cats. Basic urinalysis (eg, dipstick analysis and measurement of specific gravity by refractometer) was not required for a case to be eligible for inclusion, but was performed in 15 cats. Faecal analysis was performed in only two cats, with negative results in all, and these data are therefore not presented.
Abdominal ultrasonography
The senior study author (VF) performed all abdominal ultrasonographic examinations using a MyLab Twice ultrasound system (Esaote Medical SAS). Although this author is not a board-certified diagnostic imager, she has >15 years of experience in the field, and has received extensive training in ultrasonography from board-certified diagnostic imagers. Data were recorded regarding the presence of hepatic (eg, hepatomegaly, altered hepatic echogenicity, biliary tract dilation, biliary sludge and gall bladder wall thickening), pancreatic (eg, altered pancreatic echogenicity and homogeneity), gastrointestinal (eg, increased small intestinal wall thickness, increased muscularis layer thickness [defined as being more than twice the width of the submucosa], altered intestinal wall layering and retention of contents within the stomach) and abdominal lymph node (eg, enlargement of mesenteric lymph nodes and lymphadenopathy [>5 mm thickness]) abnormalities.
Exploratory coeliotomy
In all cases, exploratory coeliotomy was performed by one of three European-Board certified surgeons, and the name of the surgeon involved was recorded. The reason for surgical intervention was also recorded in each case and included the presence of lymph node thickening, distal small intestinal lesions, suspected cholangitis and suspected pancreatic lesions. In all cases, the decision to perform surgery was based upon preliminary clinical data and, most notably, the findings of ultrasonographical examination.
Histological evaluation of biopsy samples
Biopsy samples were placed in 10% neutral buffered formalin (pH 7.4), and submitted to a single diagnostic pathology laboratory (Laboratoire d’Anatomie Pathologique Vétérinaire du Sud Ouest [LAPVSO], Toulouse, France). Samples were processed by routine methods for histological examination and examined by histopathologists working at the same diagnostic pathology laboratory (LAPVSO). A number of pathologists were involved in assessing the samples, although most (23/39; 59%) were conducted by one of three certified pathologists (Frédérique Degorce-Rubiales, Claire Dally and Mélanie Fine). All liver samples were scored according to World Small Animal Veterinary Association (WSAVA) Liver Standardization Group criteria, 13 while all intestinal samples were scored according to the WSAVA Gastrointestinal Standardization Group criteria. 14
Data handling and analysis
Data recorded from cases included signalment data, presenting findings, the results of clinicopathological investigations, abdominal ultrasonography and histopathological examination. Data for duration of clinical signs were not normally distributed (Shapiro–Wilk test) and are expressed as median (range); although age data were normally distributed, for consistency, median (range) is again used. Categorical data (eg, clinical signs and abnormalities identified with the diagnostic investigations) are reported as numbers, proportions and percentages. A clinical sign was defined as being present when its presence was recorded in the medical record. Given that different haematological and serum biochemical analysers were used throughout the study, results are recorded as normal or abnormal, based upon the appropriate reference interval for the analyser. These included leukocytosis (based upon an increased white blood cell count), anaemia (based upon decreased haemoglobin concentration), increased liver enzyme activity, hypoalbuminaemia, hyperbilirubinaemia and increased pancreas-specific lipase activity.
All data were entered into a spreadsheet (Microsoft Excel for Mac 2011 version 14.4.4; Microsoft Corporation), and statistical software (StatsDirect version 2.7.9; StatsDirect Ltd) was used to assist with data manipulation and analysis. The aim was to determine the relationship between key clinical findings (from history, physical examination, clinicopathological tests and diagnostic imaging) and the results of histopathological examination. The different clinical signs were categorised according to the organ most likely to be affected when that sign was present. Therefore, vomiting, diarrhoea and the presence of thickened intestinal loops on abdominal palpation were classified as gastrointestinal indicators, icterus was classed as a liver indicator and abdominal pain was classed as a pancreatic indicator. The different clinicopathological abnormalities were also classified in a similar manner, such that increased liver enzyme activity (alanine aminotransferase [ALT] and alkaline phosphatase [ALP]), hyperbilirubinaemia, and bilirubinuria were assumed to be liver indicators, and increased pancreas-specific lipase activity was assumed to be a pancreatic indicator. Finally, abnormal findings on abdominal ultrasonography were also used as indicators of disease in the organ affected.
We were interested in determining to what extent the organ indicators identified could rule disease of that organ either in or out. Therefore, sensitivity, specificity, positive predictive value (PPV) and negative predictive value (NPV) were calculated for each indicator for the presence or absence of histopathological abnormalities in their specific organ. For descriptive purposes, results were classified arbitrarily as very poor (<50%), poor (50–65%), moderate (66–80%) or good (>80%). Given our specific interest in FID, sensitivity, specificity, PPV and NPV were also calculated for the presence of histopathological changes of an inflammatory nature in the organ of interest. There were two exceptions: first, for thickened intestinal loops, sensitivity, specificity, PPV and NPV were also calculated for the presence of gastrointestinal neoplasia; second, for feline pancreas-specific lipase, the limited number of test results meant that it was inappropriate to calculate sensitivity, specificity, PPV and NPV. Instead, the proportions of positive and negative results with and without pancreatic abnormalities were reported.
In addition to this, we wished to determine whether the presence of an organ-specific indicator could rule out disease in another alimentary organ, such that targeted biopsy of the specific organ could then be justified. Therefore, for each organ indicator, we also recorded the proportion (and percentage where appropriate) of cases with or without that indicator that had histopathological changes in the other organs.
Results
Study population and signalment
During the study period, a total of 258 cats were investigated for possible alimentary tract disease. Of these, 38 met the eligibility criteria for the study, including 19 primary care cases and 19 referral cases. The domestic shorthair breed comprised 28 (74%) of the cats in the study, with the remaining breeds including domestic longhair (n = 1; 3%), Persian (n = 3; 85%), oriental (n = 5; 13%) and Chartreux (n = 1; 3%). A total of 22 (58%) were neutered males and 16 (42%) were neutered females. The median age of the cats was 112 months (range 15–188 months). Median duration of clinical signs before examination was 75 days (range 1–1095 days), and duration was classed as chronic (ie, >21 days) in 28 (73%).
Preliminary clinical findings
History and physical examination findings
From the history, vomiting was present in 30 (79%) cats, and diarrhoea was present in 16 (42%), with 24 (63%), 24 (63%) and 18 (47%), respectively, demonstrating anorexia, weight loss and lethargy. On physical examination, thickened intestinal loops were noted in four (11%) cats, with abdominal pain and icterus seen in three (8%) cats each. Other physical examination findings included dehydration (13 cats; 34%), abdominal mass lesion (three cats; 8%), peripheral lymphadenopathy (one cat; 3%), poor body condition (10 cats; 26%), and hyperthermia (⩾39.1 °C) (eight cats; 21%).
Clinicopathological investigations
Increased ALT was noted in 5/35 (14%) cats, and increased ALP was seen in 4/35 (11%) cats. Total serum bilirubin concentration was measured in 15 cats, and was increased in two (13%), while serum fPL was measured in 11 cats, and was increased in three (27%). Urine bilirubin was measured by dipstick in 15 cats, and was present in seven (47%). Other clinicopathological findings included leucocytosis (5/30; 17%), anaemia (8/31; 26%), hypoalbuminaemia (4/24; 17%) and proteinuria (4/15; 27%).
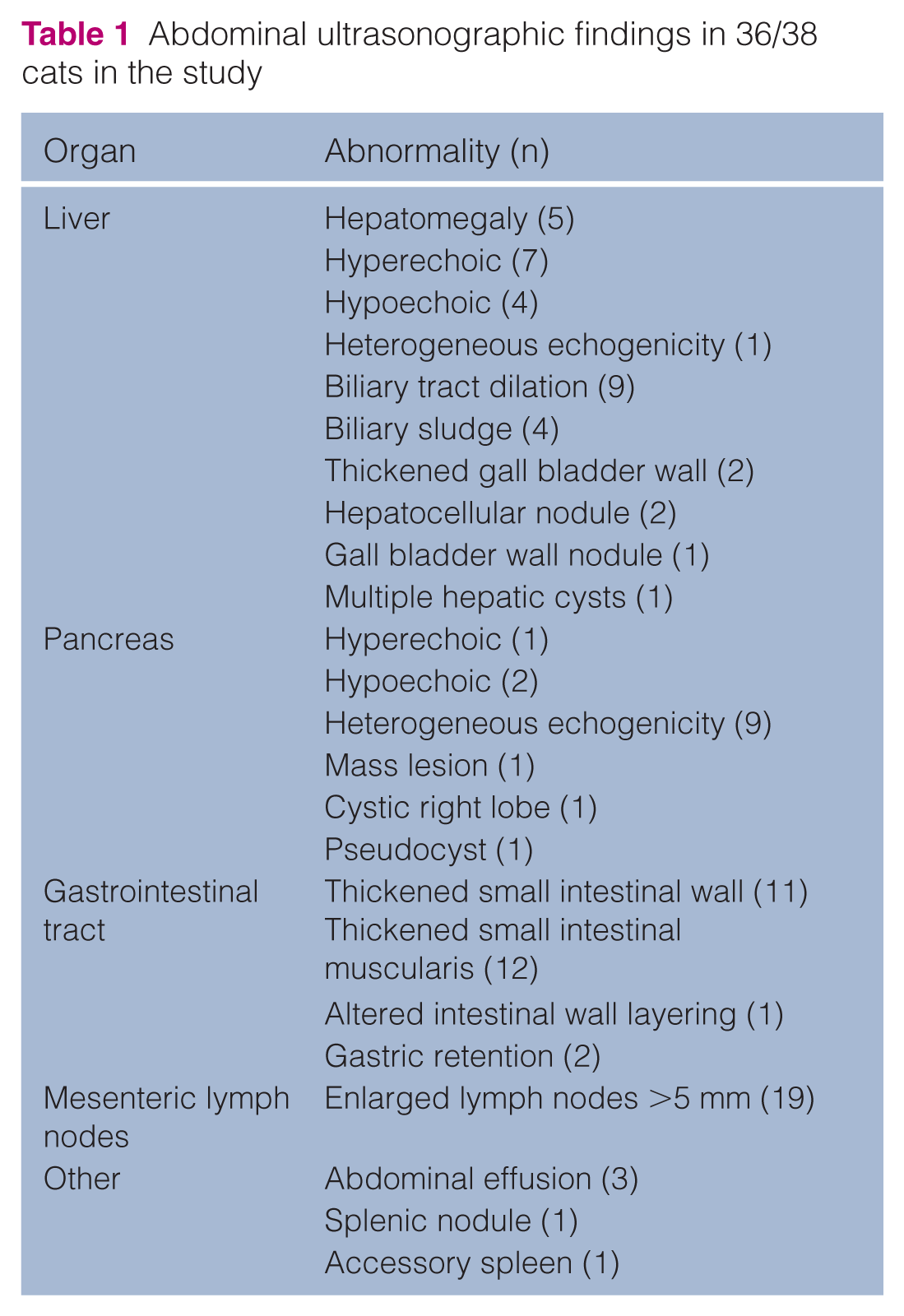
Ultrasonographic examination
Ultrasonographic examination was performed in 37/38 cats, but one of the reports was inadvertently erased and data were not available for analysis. Thus, results were available for 36 cats, and a range of hepatic, pancreatic, gastrointestinal and abdominal lymph node abnormalities were seen (Table 1).
Abdominal ultrasonographic findings in 36/38 cats in the study
Exploratory coeliotomy
There were various indications for performing exploratory coeliotomy in the 38 cats, with 19 (50%) having a single indication, 15 (39%) having two indications and four (10%) having three indications. Specific indications included mesenteric lymphadenopathy (20 cats; 53%), suspected distal small intestinal lesion (19 cats; 50%), suspected liver disease (13 cats; 34%), a suspected pancreatic lesion (five cats; 13%) and other reasons in four (11%) cats (focal peritonitis [one cat], collection of full-thickness biopsies to differentiate alimentary inflammatory bowel disease from lymphoma [one cat], suspected chronic pancreatitis [one cat] and reason not clear [one cat]).
The gross findings at coeliotomy are summarised in Table 2. Biopsy material was collected from the liver in 37 cases, from the gastrointestinal tract in 35 cases and from the pancreas in 37 cases. Thus, all three alimentary organs were sampled concurrently in 31 (82%) cases, with two organs sampled in the remaining seven (18%) cats. Mesenteric lymph node was also sampled in 22 (58%) cats, spleen in three cats and kidney in one cat (Table 2). Median time of hospitalisation after surgery was 2 days (range 1–6 days). One cat died 15 days after the surgery, and its final diagnosis was pancreatic neoplasia, while the remainder survived >15 days. There were transient complications in a further nine cases, including postoperative anorexia (n = 3), postoperative haemorrhage (n =1), wound dehiscence (n = 1), hyperthermia (n = 1), hypothermia (n = 2) and hypokalaemia (n = 1).
Gross and histological findings after exploratory coeliotomy in 38 cats in the study

IEL = Intra-epithelial lymphocyte; LP = lamina propria
Histopathological examination
Histopathological abnormalities are summarised in Table 2. Abnormalities were detected in 29/37 (78%) liver biopsies, in 29/35 (83%) gastrointestinal biopsies and in 17/37 (46%) pancreatic samples, the majority of which were inflammatory in nature (liver 20/29 [69%], intestine 28/29 [96%], pancreas 11/19 [58%]). Abnormalities were also noted in the mesenteric lymph node in 17/22 cases (77%), 3/4 of which 13 were inflammatory in nature.
Association between preliminary clinical information and histopathology
History and physical examination
The sensitivities, specificities, PPV and NPV of a range of clinical signs for the presence of histopathological abnormalities within liver, gastrointestinal tract and pancreas are shown in Table 3. Vomiting was both sensitive (81–84%) and specific (83–100%) for the presence of gastrointestinal abnormalities, although the NPV was very poor (29%), suggesting absence of vomiting would not exclude gastrointestinal disease. Further, 21/28 (75%) and 17/28 (61%) cats with vomiting also had hepatic and pancreatic histopathological abnormalities, respectively. The presence of diarrhoea was both very poorly sensitive (35–45%) and very poorly specific (11–20%) for gastrointestinal abnormalities, with a correspondingly very poor NPV (6%). Further, 14/17 (82%) and 10/17 (59%) cats with diarrhoea also had hepatic and pancreatic histopathological abnormalities, respectively. When the presence of thickened intestinal loops were identified, sensitivity was very poor (0–11%) but specificity was moderate-to-good (50–87%) for gastrointestinal histopathological abnormalities. Further, NPV was also very poor (3–14%). When the presence of thickened intestinal loops was used to determine the presence of gastrointestinal neoplasia, sensitivity and PPV were very poor (both 0%), and specificity and NPV were good (both 87%). Further, 4/4 and 1/4 cats with thickened intestinal loops also had hepatic and pancreatic histopathological abnormalities, respectively.
Performance of clinical signs for identifying abnormalities in liver, intestine and pancreas

NPV = negative predictive value; PPV = positive predictive value
Icterus had good specificity (94–100%) but very poor sensitivity (10%) for hepatic histopathological abnormalities, and NPV was very poor (21–45%). Further, 3/3 and 2/3 cats with icterus also had intestinal and pancreatic disease, respectively. The findings were similar for abdominal pain (sensitivity 9–11%, specificity 92–94%, NPV 48–69%). Further, all three cats with abdominal pain also had both intestinal and hepatic histopathological abnormalities.
Clinical pathology
The sensitivities, specificities, PPV and NPV of various clinicopathological tests for the presence of histopathological abnormalities within liver, gastrointestinal tract and pancreas are shown in Table 4. ALT was specific for hepatic disease (88–100%), but sensitivity (17–19%) and NPV (24–48%) were very poor. Further, 4/4 and 3/5 cats with increased ALT also had intestinal and pancreatic histopathological abnormalities, respectively. The results for ALP were similar with good specificity (88–100%), but very poor sensitivity (11–36%) and moderate NPV (47–77%) for hepatic histopathological abnormalities. Further, 3/3 and 2/4 cats with increased ALP also had intestinal and pancreatic histopathological abnormalities, respectively. When the results of ALT and ALP were combined (either ALT or ALP increased), sensitivity was good (88–100%) but specificity was poor (22–50%), with a moderate-to-poor NPV (50–75%). Once again, 5/5 and 4/6 cats with increased ALT ± ALP also had intestinal and pancreatic histopathological abnormalities, respectively.
Performance of liver enzyme activity and abdominal ultrasonography for identifying abnormalities in liver, intestine and pancreas

ALP = alkaline phosphatase; ALT = alanine aminotransferase; NPV = negative predictive value; PPV = positive predictive value; n/a = result could not be calculated because none of the cats without bilirubinuria had normal hepatic histopathology
Increased serum bilirubin concentration was moderately specific for hepatic histopathological abnormalities (67–86%), but sensitivity (7–13%) and NPV (13–46%) were very poor. Further, both cats with increased serum bilirubin concentration also had intestinal and pancreatic histopathological abnormalities, respectively. The presence of bilirubinuria was poorly sensitive (50–55%) and moderately specific (67%) for hepatic histopathological abnormalities, and NPV was also very poor (0–29%). Further, 6/6 and 4/7 cats with increased bilirubinuria also had intestinal and pancreatic histopathological abnormalities, respectively.
Serum fPL concentration
Pancreatic histopathological abnormalities were present in 3/3 cats with a positive pancreas-specific lipase concentration and, in two of these cats, the changes were inflammatory in nature. However, pancreatic histopathological abnormalities were also present in 3/8 cats with a negative fPL concentration, and the changes were of an inflammatory type in two cases. Further, all three of the cats with increased serum fPL concentration also had intestinal and hepatic histopathological abnormalities.
Ultrasonography
The sensitivity and specificity of ultrasonography for the presence of histopathological abnormalities of liver, gastrointestinal tract and pancreas are shown in Table 5. Both sensitivity (50–80%) and specificity (57–100%) were moderate for the presence of gastrointestinal histopathological abnormalities. For lesions in the liver, ultrasonographic examination was moderately specific (71–80%), but sensitivity was very poor (20–25%). When the association between ultrasonographic findings and the presence of inflammatory pancreatic lesions was assessed, the sensitivity was moderate (80%), but specificity was very poor (22%). Results were worse for pancreatic neoplasia with poor sensitivity (50%) and very poor specificity (17%).
Performance of abdominal ultrasonography for identifying abnormalities in liver, intestine and pancreas

NPV = negative predictive value; PPV = positive predictive value
Discussion
The current study has assessed the ability of preliminary clinical data to identify alimentary tract disease, based on the presence of histopathological abnormalities on biopsy samples collected at coeliotomy. As previously reported,2,15,16 a range of histopathological abnormalities was identified in all alimentary tract organs, but the association with the presence of clinical signs, clinicopathological abnormalities and abdominal ultrasonographic findings was poor. These results call into question the use of preliminary clinical data in decision-making, namely in determining the most appropriate sites of the alimentary tract to sample. They further suggest that, unless all alimentary tract organs are sampled at exploratory coeliotomy, the full extent of pathological abnormalities might be missed.
The cats in the current study presented with a range of clinical signs that gave little clue as to the ultimate site of abnormalities. Vomiting was the most common clinical sign, as previously described for feline inflammatory bowel disease, 7 and while both sensitive and specific for histopathological abnormalities in the gastrointestinal tract, the NPV was poor, suggesting the absence of vomiting could not be used to eliminate the possibility of gastrointestinal disease. This clinical sign was assigned to the gastrointestinal category; it was recognised that many other non-gastrointestinal diseases can cause vomiting, including those of the pancreas and liver. 7 Diarrhoea is a second sign that is often used to indicate intestinal disease, and was again used in the current study as a gastrointestinal indicator. However, sensitivity, specificity and NPV were all poor, suggesting that veterinarians should not use the presence or absence of diarrhoea to decide whether or not to biopsy the gastrointestinal tract. Further, given that a significant number of cats with diarrhoea also had histopathological abnormalities in the liver or pancreas, the presence of diarrhoea cannot be used to exclude disease in other alimentary organs. Apparent thickening of intestinal loops was an uncommon abdominal palpation finding in the current study, being present in only 4/38 cats. Although it might be assumed that this sign would correlate with infiltrative disease, none of these cases had gastrointestinal neoplasia. Therefore, this is a non-specific finding, and should be interpreted cautiously.
Arguably, icterus is a more specific clinical sign as, once haemolysis is eliminated as a cause, the abnormality is likely to reflect either hepatic or posthepatic disease. As a result, this clinical sign was chosen as a liver indicator in the current study. However, although all cases with icterus were ultimately found to have histopathological abnormalities within their liver, this finding was uncommon (three cats), and the majority (90%) of those with liver disease were not icteric. Therefore, unfortunately, this clinical sign cannot be relied upon to identify all cases where a liver biopsy would be fruitful. Further, the fact that cats with icterus also had disease in other alimentary organs suggests that it cannot be used as a basis for not sampling other alimentary tract organs. However, while this sign performed poorly as a liver indicator, it is partly explained by the fact that cases with biliary tract obstruction and idiopathic hepatic lipidosis were excluded. Further studies would be required to assess performance of icterus in identifying such disorders. Abdominal pain was used as a pancreatic indicator but, as with icterus, it was only observed in three cases. In fact, only two of these cases actually had pancreatic histopathological abnormalities, while there were a further 17 cats with pancreatic disease that did not have abdominal pain. Thus, as noted in previous studies, 17 abdominal pain is uncommon in cats with pancreatic disease, and should not be used in decision-making for cases with suspected alimentary tract disease.
Liver enzymes are specific markers of hepatic disease in cats and, given the short half lives relative to other species, any abnormality is thought to be significant. 18 However, in a recent study, liver enzyme activity in cats was a poor predictor of the severity of inflammation in cats with cholangitis. 5 Similarly, in the current study, despite the fact that approximately three-quarters of cats had histopathological evidence of liver disease, increased liver enzyme activity was seen in only a minority. While specificity was good, sensitivity and NPV were both poor. Findings were equivalent for hyperbilirubinaemia, and worse for bilirubinuria. This suggests that, even when clinicopathological indicators of liver disease are normal, hepatic pathological abnormalities cannot be ruled out. Therefore, it would not be advisable for a clinician to use such results to decide against either performing exploratory coeliotomy or hepatic biopsy.
Measurement of pancreas-specific lipase is now commonplace in cats with alimentary tract signs, and previous studies have suggested reasonable sensitivity and specificity.18,19 The results of the current study did not indicate that this assay performed any better than other preliminary clinical data. However, one limitation was the fact that, given that most cases were chronic, pancreas-specific lipase activity was only assessed in 11 cats. Thus, firm conclusions on test performance should be made with caution, and future studies should be considered to address this issue.
Like liver enzyme activity, hepatic ultrasonography is also commonly used to identify the presence of liver disease, and to determine the need for biopsy. Indeed, sensitivity (87%) and specificity (90%) are reportedly good for identifying cats with cholangitis, 20 with increased echogenicity being a common feature. 21 In contrast, although the current study suggested that ultrasonography was relatively specific (71–80%), it was poorly sensitive (20–25%) for detecting hepatic abnormalities. Operator effect might be one explanation for the difference in results between studies and one potential limitation of the current work was the fact that board-certified diagnostic imagers did not perform the investigations. Any adverse effect on performance would be partly offset by the fact that only one operator performed all examinations, who had received extensive training in abdominal diagnostic imaging and used one machine. A second possible reason for the difference in results is the type of cases studied, with the current study including a wide population of cats with alimentary tract signs, rather than those that just had hepatic disease. Whatever the reason, the authors would recommend caution with using abdominal ultrasonography to determine whether or not to perform hepatic biopsy. Caution is also advised when using ultrasonography to detect gastrointestinal lesions, where performance was even worse than with hepatic abnormalities. Further studies assessing the use of ultrasonography for alimentary tract disease would help to clarify the circumstances where this modality is of greatest benefit.
Previous studies have examined the use of ultrasonography to detect pancreatic disease in cats,22,23 and both sensitivity and specificity are reported to be moderately good. 23 In contrast, the findings of the current study suggest that for ultrasonography, while sensitivity was moderately good (78%) for detecting pancreatic inflammation, specificity was poor (22%). The reasons why these results contrast with those of previous studies are not known but might again be explained by operator effect and differences in the case population examined. More concerning was the fact that, in four cats, a pancreatic tumour was identified after pancreatic biopsy, three of which were malignant, but these had been missed on ultrasonography. Therefore, once again, caution is advisable when using ultrasonography as a screening test in feline alimentary tract disease, and the authors suggest that pancreatic biopsy is a necessary step when investigating feline alimentary tract disorders.
Exploratory coeliotomy is an invasive procedure compared with other biopsy techniques, and many clinicians and owners are reluctant to undertake it. In the current study, the most common reasons for performing an exploratory coeliotomy, in about half of all cats, were the identification of enlarged mesenteric lymph nodes and suspicion of a distal gastrointestinal lesion on abdominal ultrasonographic examination. In other studies, lymph node enlargement is an important finding,24,25 and can suggest the presence of lymphoma. 7 In contrast, the main reason for using coeliotomy when a distal small intestinal lesion was suspected was the relative inaccessibility of this region for sampling by a less invasive method such as endoscopy. Unlike lymph node enlargement and distal small intestinal lesions, suspected hepatic and pancreatic lesions were less commonly used as the reason for performing coeliotomy. Despite its perceived invasiveness, mean duration of hospitalisation after surgery was brief (ie, <2 days) and, where complications were seen, they were typically mild and transient. One serious complication previously described is leakage of luminal contents from the site of surgical incision, 26 but this was not seen in the current study. Further, no cases of postoperative pancreatitis were reported, despite the frequent collection of biopsies from this organ. Thus, the findings of the current study would suggest that exploratory coeliotomy is relatively safe and well tolerated. Nonetheless, given the small group size and the fact that the procedures were conducted at a specialist centre, the findings might not be fully representative of cats from primary care practice. Therefore, clinicians should discuss possible complications with the owner before the procedure, so that this can form the basis for deciding how to proceed in any specific case.
The study has a number of limitations, many of which are inherent in its retrospective design. First, there was no control group for comparison, for instance cats without confirmed gastrointestinal disease, in order to compare test performance in cats with and without alimentary disease. Second, given that the study was conducted over 7 years, different analysers were used for the clinicopathological analyses. As methods and reference intervals were different, it was not possible to look at data in a quantitative manner. Instead, results were interpreted on the basis of whether or not they were normal or abnormal. Third, γ glutamyl transferase was not measured, and this might have improved the performance of liver enzyme activity in identifying hepatic histopathological abnormalities. Fourth, because exploratory coeliotomy is considered to be invasive, only a limited number (ie, up to three) full-thickness small intestinal biopsies were collected and thus gastrointestinal histopathological findings might not have been completely representative. In a similar manner, when sampling the pancreas and liver, only one biopsy sample was taken from each organ. Thus, the relatively poor associations between the clinicopathological and ultrasonographical findings and histopathological lesions might be related to the fact that genuine lesions were missed if patchy disease was present. For instance, a previous necropsy study revealed that chronic inflammation is more common in the left pancreatic lobe than in the right lobe. 27 Although biopsy samples were usually taken from the left lobe of the pancreas in the current study, the fact that the right lobe was rarely sampled might still have produced some false negative results and, once again, only one full-thickness hepatic biopsy was collected. Thus, the findings might not have been totally representative of the global degree of inflammation involving those organs.
A fifth limitation was the fact that histopathological assessments were performed by a number of different pathologists, which might have resulted in inconsistencies in interpretations. Finally, perhaps the most important issue was the fact that only a small cohort of cats was examined from a referral clinic. Cats were only included if an exploratory coeliotomy had been performed, which would likely have skewed case selection. It is possible that the very reason that the clinician selected coeliotomy, and not a non-invasive procedure, was the fact that the case was more complicated, and this might have inadvertently selected for cases where preliminary clinical data were equivocal. Finally, while we have reported positive and predictive values, these are highly dependent on the true prevalence of disease in the population assessed. Thus, our results are only meaningful relative to the population sampled, and might not be representative of other populations. Most notably, the study population was from a referral clinic, and thus affected by selection bias (ie, because they are more likely to be severe, difficult or complex cases); therefore, the predictive values might not be representative of what would be expected in primary care practice. Thus, it would be sensible to consider further studies to confirm or refute the current study findings.
Conclusions
As previously reported, it is common for cats presenting with gastrointestinal signs to have histopathological abnormalities in a number of organs concurrently. Preliminary clinical data, including signs, clinicopathological results and ultrasonography, lack precision and cannot reliably predict from which organs biopsy material should be collected. Given the fact that exploratory coeliotomy is generally well tolerated, the authors would recommend this as the procedure of choice for diagnosis of feline gastrointestinal diseases.
Footnotes
Acknowledgements
The authors would like to thank Dr P Maître and S Bureau, of the Clinique Alliance, Bordeaux, France, who performed the surgical procedures. The authors would also like to thank the pathologists at Laboratoire d’Anatomie Pathologique Vétérinaire du Sud Ouest (LAPVSO), and especially F Degorce-Rubiales for performing histopathological examination.
Conflicts of interest
AJG’s readership is financially supported by Royal Canin.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
