Abstract
Objectives
The aim of the study was to determine if methimazole applied in a transdermal formulation to the internal pinna will cross to the external pinna in an in vitro Franz cell model.
Methods
The ears from six cats were harvested soon after death. Whole ears were mounted onto Franz-type diffusion cells with the stratum corneum of the inner pinnae uppermost. A commercial transdermal preparation containing methimazole (0.1 ml/10 mg) was applied to the inner pinnae. At 1, 2, 4, 6, 8, 12, 18, 24 and 30 h, a 200 µl sample of reservoir solution was removed to determine the methimazole concentration by high-performance liquid chromatography. The ears were then dissected, separating the internal pinna from the cartilage and the external pinna, before the methimazole concentration was measured at each site. The thickness of the different regions of the ear was measured on paraffin histology sections.
Results
Mean ± SD methimazole concentrations at 30 h for the right and left ear, respectively, were: inner ear, 1.25 ± 0.53 mg/g, 0.39 ± 0.26 mg/g; cartilage, 1.36 ± 0.47 mg/g, 0.33 ± 0.20 mg/g; and outer ear, 1.0 ± 0.32 mg/g, 0.33 ± 0.14 mg/g. There was a difference between the left and right ears (P <0.001). Minimal methimazole concentrations were detected in the receptor fluid. The mean methimazole concentration absorbed by the skin after application of 10 mg was, for the right ear, 3.65 ± 1.27 mg/g and, for the left, 1.08 ± 0.27 mg/g. There was no correlation between methimazole concentrations and thickness of each region of the ear.
Conclusions and relevance
Methimazole in a lipophilic vehicle applied to the inner pinna will penetrate to the outer pinna of cats in an in vitro model, which may have safety implications for humans associated with cats treated with transdermal methimazole. Substantial inter-individual variation was found. Further research is required in the area of transdermal penetration of drugs in cats.
Introduction
Transdermal drug therapy has become popular in cats since the early 2000s, with many compounding pharmacies formulating drugs into gels for systemic absorption.1,2 Despite a recent survey in the UK reporting that 62% of cat owners could orally administer a pill to their cat twice a day without a problem, many cat owners find it difficult to medicate their cats and thus transdermal drug therapy for cats is proving a popular alternative to oral medication.1–3 In cats the inner pinna is the most common site for transdermal drug therapy for ointments and gels as opposed to liquid spot-ons and patches. The inner pinna is relatively hairless, has a thin stratum corneum and has limited exposure to self-grooming.4,5
To date, the drug that has been most extensively studied in cats for transdermal absorption is methimazole (United States Adopted Name [USAN]; thiamazole International Nonproprietary Name [INN]).4,6–10 The transdermal route is a good alternative to the oral administration of methimazole for cats with hyperthyroidism, showing both short- and long-term efficacy.4,6–10 Methimazole is also suitable for topical therapy, as its chemical structure facilitates transdermal penetration (low molecular weight [144 Da], logP of 0.75 and a melting point of 144–147 °C, dose rate <20 mg/day) and efficacy can be assessed easily, as a biological endpoint can be measured (serum thyroxine concentrations and clinical signs). 11 Currently, there are no studies in cats that describe the kinetics of methimazole permeation across the skin.
One concern when administering methimazole is that the drug is teratogenic to the human fetus.12–14 Precautions, such as wearing gloves or finger cots, are indicated when administering transdermal formulations, as the drug can also cross human skin. 15 Washing hands with soap is recommended after use of both the oral and transdermal formulation. Recommendations when administering methimazole in any form are that pregnant women need to wear gloves when handling methimazole, and women of childbearing age should wear gloves when cleaning the litterbox of cats treated with methimazole.12,13 While it is obvious that the transdermal formulation of methimazole that remains on the inner pinna should be treated with the same caution as the initial administration, it has not been considered whether the methimazole applied to the inner pinna can pass through the ear and be within the skin of the outer pinna.
Transdermal penetration kinetics can be studied using in vitro models, such as the Franz cell (Figure 1), and in vitro studies correlate well with the in vivo situation.16,17 The aim of the current study was to determine if the percutaneous application of a commercial transdermal methimazole (currently available in New Zealand) in a lipophilic vehicle, applied to the internal pinna (non-haired region) will cross to the external (haired) pinna of the ear in an in vitro Franz-type cell model. Throughout this article, the term methimazole is used, the most common term used in the veterinary literature, and as stated by the USAN, the Recommended INN is thiamazole.

Two-chambered Franz-type diffusion cell used in the study of percutaneous absorption of methimazole in the cat. The skin is clamped between the upper donor compartment and lower receptor compartment. The receptor compartment solution simulates the physical conditions surrounding the subcutaneous tissues. All cells were mounted in a diffusion apparatus, and placed in a water bath set to maintain the temperature of the skin in the diffusion cell at approximately 32°C. The receptor compartment solution is magnetically stirred. Samples of the receptor solution are removed via the sampling port at set time points, with equal volumes of fresh solution being replaced into the port
Materials and methods
Animals
Six domestic shorthair cats (four female, one male neutered, one intact male) at a local pound were euthanased by an intravenous injection of sodium pentobarbital and the pinnae harvested soon after death. The ears were stored at −20°C for 3 days. This study protocol was approved by the animal ethics committee of the University of Queensland (approval number SVS/494/12).
Whole feline ears were used for the study. The ear was rinsed with tap water to remove any adherent blood, and dried gently with a gauze swab. One circular section measuring 2 cm2 was cut from the same site in each ear using a round biopsy punch.
Histology
Samples of the ear skin surrounding the hole left from the biopsy punch were trimmed to determine thickness of the epidermis, dermis and cartilage, and fixed in 10% buffered formalin for 24–48 h, processed and embedded into paraffin blocks. Haematoxylin and eosin staining was performed on 3 μm sections of tissue cut from the formalin-fixed, paraffin-embedded blocks for light microscopy.
Histological analysis was performed using a Nikon Eclipse Ni microscope. Histological images were captured using NIS Elements software (Nikon Instruments) and analysed using Image J software. 18 Briefly, for each sample, the inner and outer stratum corneum, inner and outer epidermis, number of nucleated epidermal cell layers, inner and outer dermis and pinnal cartilage were measured or counted at three randomly selected locations and averaged as previously described. 5 Both left and right ears from each cat were examined.
In vitro skin penetration studies
Whole ears were mounted onto Franz-type diffusion cells with the ventral inner pinnae of the stratum corneum uppermost as previously described (Figure 1). 19 The surface area of skin exposed to drug in the diffusion cells was 1.13 cm2. The dermal chamber was filled with 3.5 ml of a reservoir solution suitable for a lipophilic drug of phosphate-buffered isotonic saline (PBS; sodium chloride 8.0 g/l, potassium chloride 0.2 g/l, disodium hydrogen phosphate 1.15 g/l, potassium dihydrogen phosphate 0.2 g/l [MP Biomedicals]) with 4% bovine serum albumin (BSA), 20 pH 7.4 ± 0.1 and the donor compartment left open to ambient laboratory conditions. All cells were mounted in a diffusion apparatus with the dermal bathing solution being magnetically stirred. The temperature of the skin in the diffusion cell was approximately 32°C. After mounting, the upper surface (inner pinna) of the skin was hydrated for 1 h with sodium phosphate buffer (0.1 M) and the system was allowed to equilibrate. The buffer solution was aspirated from the surface of the skin and the commercial formulation of methimazole (0.1 ml/10 mg) was applied to the inner pinnae using 1 ml syringes, pre-weighed to ensure accurate application of 0.1 ml. Each application of methimazole was spread across the ear with a glass rod. The donor compartment was left open to ambient laboratory conditions, to simulate the natural absorption of the drug.
The commercial formulation of methimazole was applied as a finite dose, where a limited amount of the each formulation is applied to the skin surface, simulating conditions for the patient in vivo. The Organisation for Economic Co-operation and Development (OECD) Guideline 428 and Guidance Document 28 define finite dose absorption experiments as the application of ⩽10 μl/cm2 of a liquid formulation to the skin.21,22 For semisolid and solid substances, values range between 1 and 10 mg/cm2. In this study, 10 mg of methimazole was applied to the skin, with a skin surface area of 1.13 cm2 (8.8 mg/cm2), criteria that would meet conditions for a finite dose under OECD guidelines.
Sampling time and sample collection
A 200 µl sample of reservoir solution was removed at 1, 2, 4, 6, 8, 12, 18, 24 and 30 h, and was replaced by 200 µl of fresh solution into the sampling port.
The 200 µl aliquots were frozen at −20°C until subsequent analysis, conducted within 2 months of sampling.
In vitro retention
At the end of the experiment, the Franz cells were dismantled; the skin removed and patted dry with gauze swabs. The skin exposed to the formulation containing methimazole was excised. Ears were dissected, removing the internal pinna from the cartilage and the external pinna. These three sections were individually macerated, placed in pre-weighed vials and then re-weighed. All vials were labelled and stored at −20°C until required for further analysis, also within 2 months of sampling.
Chemicals
A commercially available methimazole in a lipophilic formulation was supplied by Bayer NZ (Hyper-T Earspot, batch number NZ05784; date of manufacture: December 2012; date of expiration: December 2013; date of study: 6 June 2013).
BSA was purchased from Trace Biosciences and PBS from MP Biomedicals.
Analytical methods
The methods for high-performance liquid chromatography (HPLC) analysis of methimazole from serum have been previously described. 7 Samples were analysed for methimazole concentration by a Waters HPLC (600 Model controller with 717 plus auto sampler and a 2998 Model photodiode array detector). For each run, 10 µl was injected at a flow rate of 0.6 ml/min onto the column (Phenomenox Luna C18, 150 × 4.6 mm, 5 µm, with a guard column) at 30°C. The detection wavelength was 252 nm, run time 15 mins. The data were processed and integrated with Waters software (Empower 2). Methimazole standards were run before all samples were analysed. The limit of detection was 20 ng/ml; the limit of quantification was 70 ng/ml and the precision was 1%. Frozen samples were thawed to room temperature and 90 µl was removed and mixed with 10 µl protein extraction solution.
The samples were vortexed and centrifuged at 14,000 g for 20 mins. An additional 100 µl of dimethyl sulfoxide was added to neutralize the acid. An aliquot of 100 µl was taken for analysis.
For validation of the procedure, aliquots of (0.1 ml) of the receiver medium (PBS and BSA) were spiked with standard solutions of the commercial transdermal methimazole.
Skin extraction
Frozen macerated skin samples were thawed to room temperature and 1 ml of water was added. The sample was left for 24 h at 4°C for methimazole desorption.
The sample liquid was taken (90 µl) and mixed with 10 µl protein extraction solution. The protein extraction solution was made of 0.1 g/ml of 5-sulfosalicylic acid in 60% water and 40% acetonitrile.
The samples were vortexed and centrifuged at 14,000 g for 20 mins. An additional 100 µl of dimethyl sulfoxide was added to neutralise the acid. An aliquot of the supernatant (100 µl) was collected and analysed by HPLC as described above.
Extraction efficiency validation
The extraction efficiency (drug recovery) of methimazole from the ear skin sections was calculated by spiking blank skin samples with known amounts of methimazole. Ten skin samples were spiked with methimazole in the lipophilic vehicle at concentrations of 0.01 mg, 0.1 mg, 0.5 mg, 1 mg and 5 mg. Samples were left at room temperature (25°C) for 24 h while being gently roller mixed to permit absorption of methimazole. The methimazole was then extracted as described above and a percentage recovery determined, which was then used to adjust methimazole concentrations extracted from the skin at the conclusion of the study.
Data analysis
A linear regression model was created to determine the effect of the left and right ear, and the ear regions (inner, cartilage and outer ear), on the concentration of methimazole; both ear and region were added to the model as fixed effects. A Student’s t-test was used to compare the mean total concentration in the right and left ear, and to compare the thickness of the ears. Scatter plots and correlations were used to look at the relationship between methimazole concentration and the thickness of each ear region. Analyses were conducted in Stata version 12 (Statacorp LP) and P <0.05 was considered significant.
One cat had an ear tattoo that penetrated the cartilage (Figure 2), and it was decided that this cat should be excluded from the analysis of the right ear owing to higher methimazole concentrations compared with the other cats.
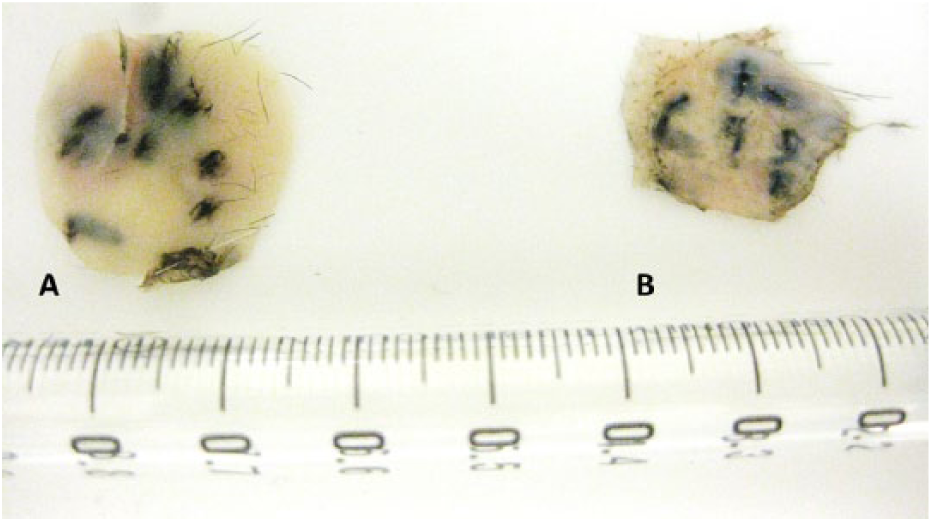
In an in vitro study showing that methimazole can cross from the inner to outer pinna of cats, one cat had a neuter tattoo that penetrated the cartilage. After application of 10 mg of methimazole to the inner pinna and the whole ear placed for 30 h in an in vitro Franz cell, the right ear of this cat had 3.3 mg/g of methimazole in the cartilage, the highest of all the cats in the study. A = cartilage; B = inner ear
Results
A significant difference was found in the concentrations of methimazole between the left and right ear (P <0.001) (Figure 3); therefore, data from each side were considered separately. Mean ± SD methimazole concentrations for the three regions of the right and left ears, respectively, were as follows: inner ear, 1.25 ± 0.53 mg/g, 0.39 ± 0.26 mg/g; cartilage, 1.36 ± 0.47 mg/g, 0.33 ± 0.20 mg/g; and outer ear, 1.0 ± 0.32 mg/g, 0.33 ± 0.14 mg/g. Minimal methimazole concentrations were detected in the receptor fluid (right ear 0.04 ± 0.07 mg/g, left ear 0.02 ± 0.03 mg/g). The mean total amount of absorbed methimazole after application of 10 mg was 3.65 ± 1.27 mg/g for the right ear and 1.08 ± 0.27 mg/g for the left ear.
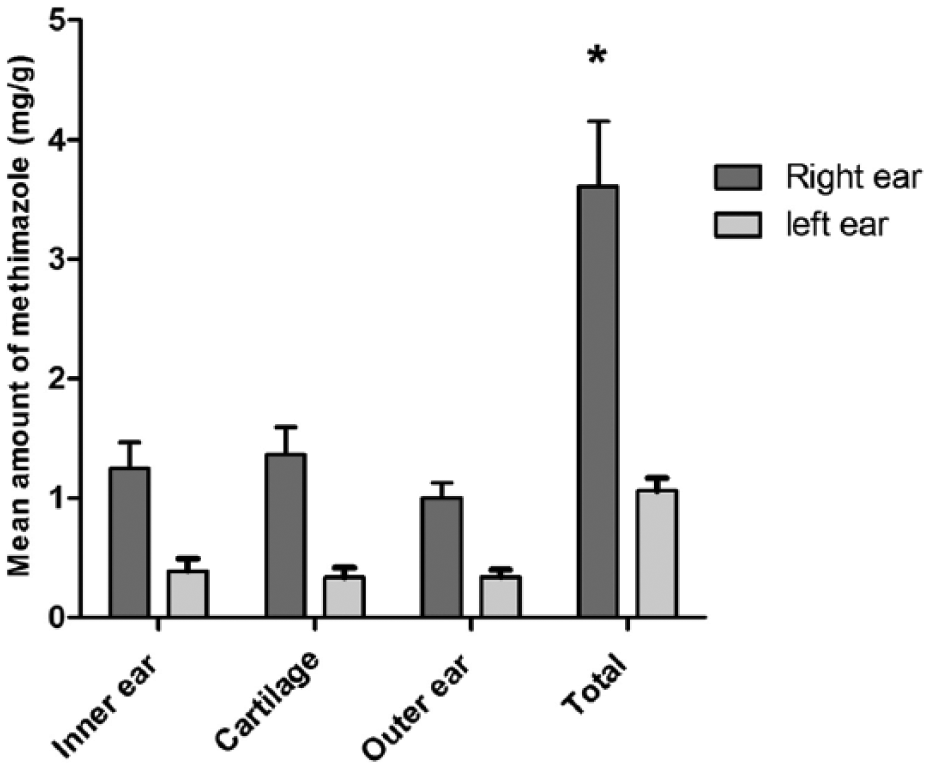
Mean amount (mg/g) of methimazole recovered from six cat (11 ears) whole-ear samples after application of 10 mg of methimazole in a lipophilic vehicle to the inner pinna and the whole ear placed for 30 h in an in vitro Franz cell, stratified by site (the right ear of one cat was excluded owing to abnormally high concentrations of methimazole). The y-axis shows the amount of methimazole (mg/g) (the SD is shown with bars). After adjusting for the effect of ear (left or right), no significant difference was found in methimazole concentration between the different sites of the ear (inner ear, cartilage, outer ear) (P = 0.47). A significant difference was found between the total methimazole concentration of the left and right ear. *P <0.001
Cat 6 was excluded from the analysis for the right ear owing to the extremely high concentrations of methimazole in the right ear compared with the left (see Figure 2): inner ear, 2.8 vs 0.6 mg/g; cartilage, 3.3 vs 0.5 mg/g; outer ear, 2.1 vs 0.4 mg/g; and total, 8.3 vs 1.5 mg/g.
After adjusting for the effect of ear (left or right), no significant difference was found in methimazole concentration between the different regions of the ear (inner ear, cartilage, outer ear) (P = 0.47) (see Figure 3).
Histology of the ears showed minimal evidence of significant freezing artefact such as cell disruption or tissue vacuolation. The mean thickness of the inner ear, cartilage and outer ear is shown in Table 1.
Mean thickness (µm) and SD of the inner and outer ear epidermis, stratum corneum, dermis and number of epidermal cell layers from the left and right ear of six cats as determined by paraffin histology sections. The mean ± SD of three replicates is provided. There was no difference in the mean thickness of the right ear compared with the left ear (P = 0.39), the inner ear thickness (P = 0.37), the cartilage thickness (P = 0.46) or the outer ear thickness (P = 0.48)
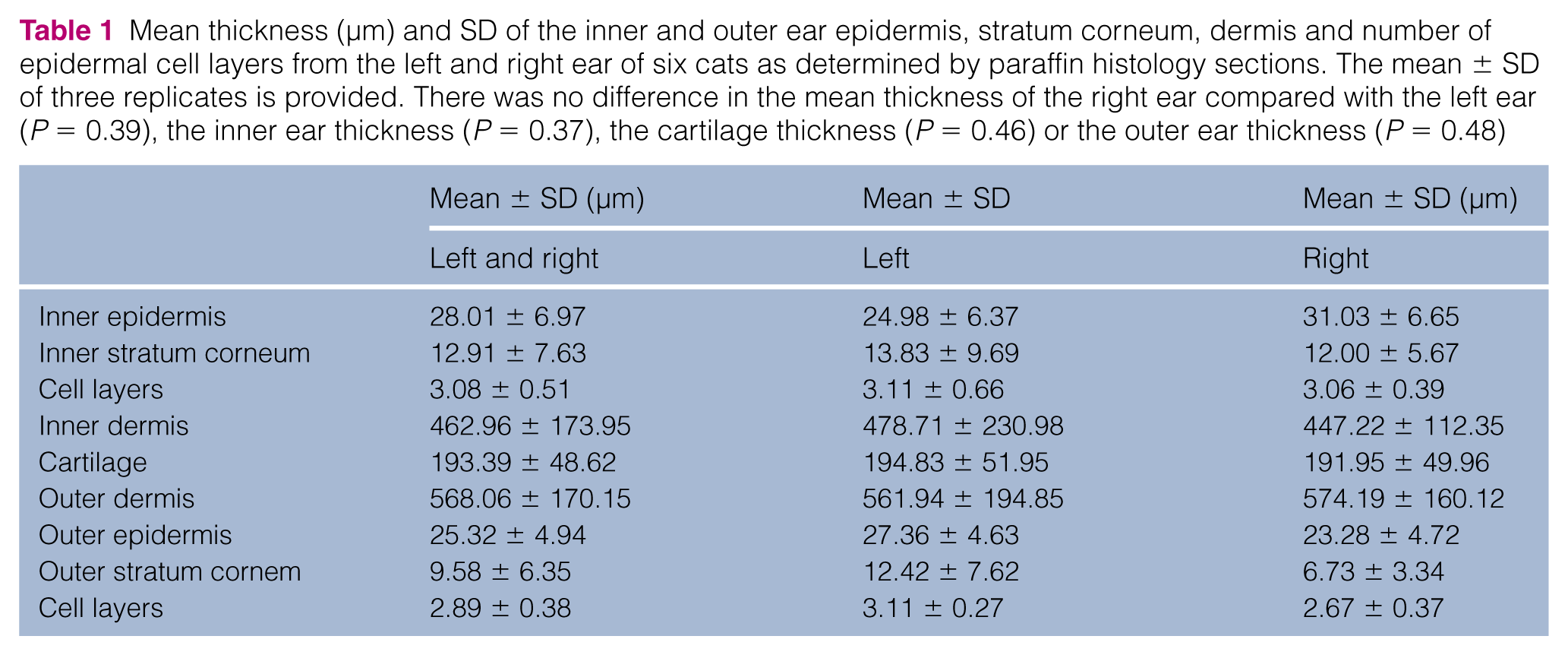
There was no difference in the mean thickness of the right ear compared with the left ear (P = 0.39), the inner ear skin thickness (P = 0.37), the cartilage thickness (P = 0.46) or the outer ear skin thickness (P = 0.48). No correlation was found with measured methimazole concentrations and the thickness of each region of the ear.
Discussion
The use of transdermal methimazole to treat cats with hyperthyroidism has increased over the past 10–15 years.1,7,10,23 The use of transdermal methimazole has the potential to increase the exposure of the veterinary client to methimazole, and, despite this risk, research into environmental exposure after the use of transdermal methimazole in cats has not been published. The outcome from the current study has shown that, in vitro, transdermal methimazole in a lipophilic vehicle, in small doses, can penetrate from the inner ear, across the cartilage to the outer portion of the ear and may therefore pose a risk to people handling cats undergoing transdermal methimazole treatment. It is not surprising that some methimazole was absorbed into the cartilage and outer ear skin in the in vitro conditions. The sink conditions in the reservoir solution (ie, low concentrations of drug in the receptor fluid) would allow for passive drug diffusion.19,24,25 Over time, methimazole concentrations remained low in the receptor fluid, maintaining sink conditions; therefore, the results of this study can potentially overestimate in vivo absorption. However, blood circulation in the skin and cartilage of the ear, such as would occur in vivo, would also be likely to remove drug from these areas, rather than allow drug to concentrate in the in vitro conditions of our study.
A technique that may be useful to confirm if methimazole is present in the outer ear stratum corneum in vivo is tape stripping, where corneocytes are collected on adhesive tape, extracted and then analysed for drug content. 26 Tape stripping is frequently used to determine drug concentrations in outer epidermal layers in human transdermal research and, importantly, would be analogous to stroking or scratching the outer ear of cats. As our study has identified a possible risk, the potential for inadvertent contamination to humans owning cats treated with transdermal methimazole warrants further research.
According to manufacturers, methimazole is a suspected teratogen. 12 However, controversy exists, and previously published data were insufficient to draw a definitive conclusion as to the teratogenic potential of methimazole and carbimazole, with no prospective controlled studies supporting the teratogenicity of methimazole. 27 A recent study determined that there was an association between exposure in the first trimester to methimazole/carbimazole and birth defects such as omphalocele and choanal atresia. 14 The link of exposure to methimazole/carbimazole in the first trimester and these birth malformations definitely suggests that these malformations could be part of a specific, even if rare, embryopathy. 14 Follow-up recommendations for the therapy of hyperthyroidism in women who are pregnant have suggested that methimazole/carbimazole be avoided in the first trimester of pregnancy; however, the drug is safe for use by women in the second and third trimester of pregnancy. 28 Considering these recent recommendations, 28 the risk to pregnant women of rubbing the ear of a cat that has been treated with transdermal methimazole is likely to be small, and should be further reduced by washing hands after handling the cat. However, until further research is performed and considering the results of the current trial, recommendations should be made that women of childbearing age or pregnant women in the first trimester of pregnancy should wear gloves at all times when handling a cat that has been treated with transdermal methimazole.
In the current study, there was substantial inter-individual variability. High intra- and inter-individual variability is expected in in vitro transdermal trials. 29 In the current study, each cat received the same dose to the right and left ear, and the left and right ears were compared to determine inter-individual differences. A difference in methimazole absorption was found between the left and right ears, and there was also considerable variability between subjects for the methimazole concentrations remaining in the ear (Figure 3). The individual variability in methimazole absorption could be important in the clinical conditions, where variability in absorption between the left or the right ear may alter thyroxine concentrations and affect the resolution of clinical signs of hyperthyroidism.
Histological findings could not account for the variability in methimazole absorption in the current trial. There was no difference in the thickness of the ear regions between the left and the right ear, and the thickness of the ear regions did not correlate with methimazole concentrations absorbed and measured in the different ear regions. Considerable variability was found between subjects for thickness of all the skin regions, including the inner stratum corneum (Table 1). The major area of skin that provides resistance to drug penetration through the skin is the stratum corneum;30–32 however, at least in this in vitro study, the thickness of stratum corneum is not related to drug penetration. In the current study, blood flow could also not account for the differences in methimazole concentrations absorbed in the regions of skin. In the current study, experimental variation may explain the difference in methimazole concentrations between the left and the right side as there was no obvious biological reason. Other skin factors that may affect the penetration of transdermal drugs include the number and density of collagen fibres in the dermis or skin appendages in the epidermis (hair and sweat follicles), and further research in this area is recommended.
Another variation found in our study was the increased concentrations of methimazole in the right ear regions of the cat with a right ear tattoo indicating neuter status (Figure 2). This tattoo penetrated the cartilage of the ear, and the methimazole concentrations in the cartilage and outer ear were significantly higher than those measured in the other cats. This cat was excluded from the overall analysis of the right ear for this reason. However, in clinical conditions, the increased absorption of methimazole after the transdermal application to the ear of cats with a tattoo could be significant. Ear tattooing of cats is common in many countries to indicate whether a cat is neutered, and sometimes for identification. 33 Cats with ear tattoos may have increased systemic absorption of drugs applied to the ear, and this exception also requires further research.
There were limitations to this study. Only two replicates were possible per cat in this current trial (only one 1.13 cm2 skin disc per ear) and the number of skin replicates recommended in permeation studies is 12, as high intra- and inter-subject variability is expected. 29 A further limitation of the current study included the use of frozen, not fresh, ear skin. Freezing canine skin has been shown to increase the penetration of hydrocortisone by a constant rate over time; however, no studies have been undertaken in the cat. 34 However, while repeating the study with freshly harvested ears may be interesting, the ear skin was frozen for only 3 days, and this permitted consistent experimental conditions.
Total absorption of methimazole and subsequent recovery after application of 10 mg to the inner pinna was low (mean 3.65 mg/g for the right ear [36%] and 1.08 mg/g for the left ear [10%]), much lower than the expected 90–110% for finite dose experiments. 21 Low recovery rates in this experiment are likely owing to the fact that at the completion of the experiment, the residual amount of methimazole on the inner pinna was washed off but a methimazole concentration was not measured to account for total drug disposition. Future experiments using finite doses of drug would ensure that all equipment and skin disc washings are included in the final absorption calculations.
Conclusions
This study has shown that, in vitro, methimazole in a lipophilic vehicle will penetrate from the inner pinna to the cartilage and outer pinna. Care should be taken when handling ears (both inner pinna and outer pinna) of cats that are treated methimazole in a transdermal vehicle, as drug residues may be present in all layers of the skin. Women of childbearing age or pregnant women in the first trimester of pregnancy should wear gloves at all times when handling a cat treated with transdermal methimazole. Further research is required in this area to determine the safety of all transdermal methimazole in other vehicles and other transdermal drugs applied to the pinna of cats.
Footnotes
Acknowledgements
Thanks to Alon Meizler for technical assistance and Bayer NZ for supplying the HyperT EarSpot.
Conflict of interest
KH and PC receive some royalties for the sales of Hyper-T Earspot in New Zealand.
Funding
This study was funded by the Building Research Capability in Strategically Relevant Areas (BRCSRA) Fund.
