Abstract
Anaesthetic and analgesic effects of three different injectable anaesthetic combinations for prepubertal gonadectomy (PPG) in cats were studied. One anaesthetic protocol was compared with a similar one for gonadectomy at traditional age (TAG). Kittens were randomly assigned to PPG or TAG. For PPG, three different protocols were compared: (1) intramuscular (IM) administration of 60 μg/kg dexmedetomidine plus 20 μg/kg buprenorphine followed by an IM injection of the anaesthetic agent (20 mg/kg ketamine) (DB-IM protocol); (2) oral transmucosal (OTM) administration of 80 μg/kg dexmedetomidine plus 20 μg/kg buprenorphine followed by an IM injection of 20 mg/kg ketamine combined with 20 µg/kg dexmedetomidine (DB-OTM protocol); (3) IM injection of a 40 μg/kg medetomidine–20 μg/kg buprenorphine–20 mg/kg ketamine combination (MBK-IM protocol). For TAG, a DB-IM protocol was used, but with different doses for dexmedetomidine (40 μg/kg) and ketamine (5 mg/kg). All cats (PPG and TAG) received a non-steroidal anti-inflammatory before surgery. Anaesthetic and analgesic effects were assessed pre- and postoperatively (until 6 h). Cumulative logit, linear and logistic regression models were used for statistical analysis. Compared with the DB-OTM protocol, the DB-IM and MBK-IM protocols provided better anaesthesia with fewer adverse effects in PPG cats. Postoperative pain was not significantly different between anaesthetic protocols. PPG and TAG cats anaesthetised with the two DB-IM protocols differed significantly only for sedation and pain scores, but sedation and pain scores were generally low. Although there were no anaesthesia-related mortalities in the present study and all anaesthetic protocols for PPG in cats provided a surgical plane of anaesthesia and analgesia up to 6 h postoperatively, our findings were in favour of the intramuscular (DB-IM and MBK-IM) protocols.
Introduction
Prepubertal gonadectomy (PPG) is promoted for population control in cats1–6 and is gradually becoming an acceptable technique in veterinary practice. Nevertheless, some veterinarians are still hesitant to perform PPG, partially because of the lack of experience with paediatric anaesthesia.7,8 To promote PPG to both the veterinary and lay public, the Belgian government supported a large-scale project on early age gonadectomy in cats. In this project, different anaesthetic and surgical protocols for PPG were investigated. The evaluation of surgical technique is the topic of a subsequent publication. 9 The present study, as part of the project, describes the characteristics of anaesthesia and postoperative pain in cats following PPG and gonadectomy at traditional age (TAG).
Previous studies described anaesthetic protocols for PPG in cats, but often with a limited number of animals, without being case-controlled10,11 or with cats and dogs evaluated together. 3 Moreover, postoperative pain was often not assessed. Injectable anaesthetic drug combinations have been used to anaesthetise cats undergoing neutering procedures.1,12,13 A more recent focus has been the evaluation of pain in kittens following PPG.4,14
The purpose of the present study was two-fold: (1) to compare the characteristics of anaesthesia and postoperative pain between different anaesthetic protocols for PPG in cats (while taking into account the surgical technique), and (2) to compare anaesthetic effects and postoperative pain in kittens (PPG) and young adult cats (TAG) with a comparable anaesthetic protocol and surgical technique in a large prospective randomised clinical trial.
Materials and methods
The study protocol was approved by the local Ethical Committee (Faculty of Veterinary Medicine, Ghent University, Belgium; licence number EC 2010/019 and 2011/077) and Deontological Committee (Federal Public Service Health, Food Chain Safety and Environment, Brussels, Belgium).
Animals
Healthy, dewormed and vaccinated kittens were recruited from animal shelters in Flanders (Belgium). Female and male kittens were enrolled between the estimated age of 8 and 12 weeks on the basis of their body weight (between 0.7 and 1.4 kg). 15 A complete physical examination was performed before the kittens were transported to the Faculty of Veterinary Medicine, Ghent University. During transport to and their stay at the Faculty, kittens from the same litter were housed together to minimise stress and discomfort.16,17
Between May 2011 and August 2012, standardised anaesthetic protocols as well as standardised surgical techniques were used. Kittens were randomly assigned by a stratified randomisation scheme (using different anaesthetic and surgical protocols as stratification factors to ensure treatment assignment balance within each stratum) to one of the two treatment age groups, using unequal group sizes (2/3 PPG; 1/3 TAG). Kittens belonging to the PPG group were gonadectomised upon assignment. In the TAG group, gonadectomy was postponed until kittens were 6–8 months of age. Only cats (PPG and TAG) with normal macroscopic anatomy of the uterus or normal descent of both testicles were included in the present study. All kittens (PPG and TAG) were microchipped and remained hospitalised at the Faculty of Veterinary Medicine and monitored until at least 6 h postoperatively. Kittens were returned to the shelter and offered for adoption. TAG cats returned to the Faculty around the age of 6–8 months for neutering and 6 h postoperative follow-up. At 3 and 6 h after surgery (PPG and TAG), vital functions (heart/respiratory rate, capillary refill time, femoral pulse) and body temperature were recorded, and sedation and pain were assessed. Environmental conditions were kept as constant as possible throughout all procedures and recovery. Caretakers from the shelters (PPG) and pet owners (TAG) received written instructions for postoperative care beyond 6 h.
Anaesthesia
Prior to treatment, food, but not water, was withheld for 2–4 h (PPG) or 10–12 h (TAG), the health of the cats was assessed by a general physical examination and body weight was measured (accurate to 0.1 kg). All cats (PPG and TAG) received general anaesthesia according to their assigned protocol (see below). An intramuscular (IM) injection was given in the quadriceps muscle, a subcutaneous (SC) injection in the lumbar region. Oral transmucosal (OTM) administration was performed by inserting the nozzle of a 1 ml syringe into the buccal cavity of the cat’s mouth and gently squirting the content into the buccal area.18,19 For IM administration, cats were gently handled and restrained if necessary with a scruffing technique (ie, holding on the skin of the cat’s neck) by a veterinary student or nurse, while for OTM administration cats were gently handled and restrained by the main researcher (NP), and no additional handler was involved.
For PPG, three different anaesthetic protocols were compared:
Dexmedetomidine–buprenorphine intramuscular group (DB-IM): IM injection of 60 μg/kg (0.12 ml/kg) dexmedetomidine combined with 20 μg/kg (0.07 ml/kg) buprenorphine IM (Dexdomitor, Orion Corporate and Vetergesic Multidose; Alstoe Animal Health), followed by an IM injection of 20 mg/kg (0.2 ml/kg) ketamine (Anesketin; Eurovet) and a SC injection of 4 mg/kg (0.08 ml/kg) carprofen (Rimadyl; Zoetis).
Dexmedetomidine–buprenorphine oral transmucosal group (DB-OTM): OTM administration of 80 μg/kg (0.16 ml/kg) dexmedetomidine combined with 20 μg/kg (0.07 ml/kg) buprenorphine OTM, followed by an IM injection of 20 mg/kg (0.2 ml/kg) ketamine combined with 20 μg/kg (0.04 ml/kg) dexmedetomidine IM and a SC injection of 4 mg/kg (0.08 ml/kg) carprofen.
Medetomidine–buprenorphine–ketamine intramuscular group (MBK-IM): a single IM injection of 40 μg/kg (0.04 ml/kg) medetomidine (Sedator; Eurovet) plus 20 μg/kg (0.07 ml/kg) buprenorphine plus 20 mg/kg (0.2 mg/kg) ketamine IM and a SC injection of 0.3 mg/kg (0.06 ml/kg) meloxicam (Acticam; Ecuphar). The latter, based on a protocol previously used in kittens, 10 was only implemented in the third study year (2012) at the request of shelter veterinarians already involved in PPG but with specific concerns about protocol efficiency and potential side effects.
TAG cats were anaesthetised similarly to the PPG DB-IM group but using different drug doses: 40 μg/kg (0.08 ml/kg) dexmedetomidine combined with 20 μg/kg (0.07 ml/kg) buprenorphine IM, followed by 5 mg/kg (0.05 ml/kg) ketamine IM and 4 mg/kg (0.08 ml/kg) carprofen SC.
In the DB-IM and the DB-OTM groups, ketamine was injected when profound sedation (ie, sedation score of at least 3; Table 1) was reached 15 mins after administration of dexmedetomidine plus buprenorphine. Otherwise, sedation was reassessed every 5 mins until profound sedation was reached. If a score of 3 was still not observed at 30 mins after premedication, ketamine was administered. Immediately after the ketamine injection (DB-OTM and DB-IM group), carprofen was administered. Thereafter, 5 mins elapsed before starting surgical preparation (clipping, scrubbing). In the MBK-IM group, after kittens were profoundly sedated (as soon as a sedation score 3 was reached; Table 1) and anaesthetised, meloxicam was administered and surgical preparation was started. Cats were considered to be in a surgical plane of anaesthesia when they were not responsive to a toe pinch and surgical preparation (clipping, scrubbing). In event of an inadequate plane of anaesthesia (movement and/or vocalisation), systemic or local anaesthetic drugs were given: a second injection of ketamine was administered IM (PPG: 10 mg/kg; TAG: 5 mg/kg) or, during skin closure, 2 mg/kg lidocaine was gently squirted over the incision site (Xylocaine 2%; Astrazeneca). Surgery was resumed 5 mins (systemic) or 1 min (local) after supplemental drug administration. Atipamezole (Antisedan; Orion Corporate) at half the volume of (dex)medetomidine given was injected IM at 3 h postoperatively if the cat still had a sedation score >1. Postoperative rescue analgesia (buprenorphine 20 μg/kg) was administered if a DIVAS score >50 mm or a 4AVet score >6 was recorded, or when the cat looked uncomfortable based on the opinion of the primary investigator.
Surgical procedures
Kittens (PPG) were randomly assigned to groups with a different surgical technique. The surgical techniques and the results are described in detail elsewhere.9 For prepubertal ovariectomy in female kittens, a limited ventral coeliotomy was performed more caudally than for TAG cats, and the ovariectomy hook was exclusively used to identify the right uterine horn/ovary. Kittens were randomly assigned to one of four surgical techniques for ovarian pedicle haemostasis: placement of ligatures, application of vascular clips, use of a bipolar forceps (electrocautery), or creation of a knot in the ovarian pedicle. Male PPG kittens were randomly assigned to two groups of prepubertal orchidectomy: creation of a knot by a single throw in the spermatic cord or placement of a single ligature around the spermatic cord. Both were closed techniques. In TAG cats, the traditional approaches and techniques (ligatures in females, knot in males) were used. One single surgeon (NP) performed all surgeries. Briefly, all surgical PPG techniques provided easy and reliable haemostasis in kittens and could be used for early age gonadectomy, although some required less time than others. Clips and electrocautery were the fastest techniques for female PPG; knot placement was fastest for male PPG. For both sexes, few intraoperative complications were observed, irrespective of the surgical technique used. 9
Data recording
Sedation
The extent of sedation was assessed subjectively with a Numerical Rating Scale (NRS; Table 1) modified from scales used previously in cats.20–22
Pain
Pain was assessed using three scoring methods: a Dynamic and Interactive Visual Analogue Scale (DIVAS), a multidimensional pain scoring system (4AVet) and a mechanical nociceptive threshold (MNT) testing device (ProD-Plus – pressure rate onset device; Topcat Metrology). Frightened kittens and those hiding from, trying to escape from and showing aggressive behaviour towards the investigator (NP) were excluded from pain assessment.
DIVAS – The 100 mm DIVAS lines were anchored so that 0 mm represented ‘no pain’ and 100 mm represented ‘worst imaginable pain’. 23 The DIVAS score of each cat was based on an observation, interaction and wound palpation.23,24 The wound was palpated four times using the flat part of two or three fingers of the observer’s right hand. 25
4AVet 26 – In this multidimensional composite pain scale, the score of each cat was not only based on observation, interaction and wound palpation, but also on monitoring of a physiological parameter (heart rate) and general attitude (eg, activity, appetite).
MNT – The antinociceptive effect (MNT) and wound sensitivity (MNT wound; only females) were measured by the cat’s response to the handheld MNT testing device. The small probe (PPG: 2.5 mm diameter; TAG: 4 mm diameter) at the angled head of the ProD-Plus was held against the pectoral muscles at the level of the shoulder joint to measure MNT. In female cats, the small probe was also applied 1 cm lateral from the wound edge (MNT wound). The lights of the device guided the user to alter the force manually at a controlled rate (2 N/s). A MNT was recorded when the cat jumped, withdrew or raised a limb, turned the head or vocalised.27–29 Moving the ears back, tail twitching or any other escape behaviour were also considered as a response to the nociceptive stimulus. 22 Whenever a response was observed, the ProD-Plus was withdrawn and the displayed peak reading of force in newtons was subsequently recorded. Since analgesics were used, the cut-off value was set at 20 N to avoid tissue trauma. 27 At every time point, the mean of three MNT measurements, obtained with an interval of 3–5 mins, was used for statistical analysis.
Adverse and physiological effects
Adverse effects such as salivation, excitation and vomiting were recorded after administration of the premedicant drugs and during recovery. The salivary pH was recorded before drug administration by a pH strip, placed in the cat’s mouth until it was moist. 18 The colour change was compared with the standard pH colours on the pH indicator paper (Carl Roth GmbH).
Before, during and after anaesthesia, respiration rate (RR) in breaths per minute was counted by observing the thoracic excursions, heart rate (HR) in beats per minute by thoracic auscultation and rectal temperature (T) using a digital thermometer. During surgery, all cats were placed on a circulating warm water blanket and pulse oximetry (Mindray PM-60 Veterinary Pulse Oximeter) using a lingual probe was performed to evaluate peripheral arterial haemoglobin oxygen saturation (SpO2) and pulse rate. During recovery, heat sources were provided and body temperature was monitored every 15 mins until the kittens were awake.
Recovery
In PPG kittens, time from induction of anaesthesia (unresponsiveness to toe pinch and surgical preparation) to sternal recumbency and eating was recorded in minutes.
Data collection
Baseline values for pain scores and physiological parameters (HR, RR and T) were recorded for each cat before drug administration. Cats were observed in their cage, then approached and handled. The ventral abdomen (female) and scrotal area (male) were palpated, and MNT (wound) scores were measured, followed by a clinical examination for physiological parameters.
Sedation in the DB-IM and DB-OTM groups was assessed 15 mins after IM or OTM premedication (dexmedetomidine combined with buprenorphine), and if sedation was not yet sufficient, the assessment was repeated every 5 mins until at least a sedation score of 3 was reached or until 30 mins after premedication. In the MBK-IM group, time to reach a sedation score of 3 was recorded in seconds.
Physiological parameters (HR, RR, SpO2) were recorded every 10 mins during surgery. Body temperature was measured after administration of the premedication and immediately after surgery.
At 3 and 6 h postoperatively, sedation and pain were reassessed and physiological parameters measured in the same way as described above for baseline values. If a cat received atipamezole at 3 h postoperatively, pain assessment was postponed another 15 mins. All assessments were performed by a single observer (NP), and occasionally some data could not be recorded.
Statistical analysis
To compare the different anaesthetic protocols for PPG in female and in male cats, a cumulative logit model for the ordinal variables (NRS, 4AVet), a linear model for the continuous variables (DIVAS, MNT, HR, RR and SpO2) and an (exact) logistic regression model for dichotomous variables (adverse effects, need for supplementary anaesthesia and rescue analgesia – data for which were sparse) were fitted with anaesthetic protocol, surgical technique, period (2011 and 2012) and two-way interactions between anaesthetic protocol, surgical technique and period as fixed effects at the 5% global significance level. Non-significant interactions were removed and will not be discussed. Pairwise multiple comparisons are based on the Tukey–Kramer method and adjusted P values are reported.
To compare PPG and TAG only, data of cats with a similar anaesthetic protocol (IM) and an identical surgical technique (ligatures for females, knot for males) were used and the same analyses were repeated (similar models with group, period and the interaction between group and period as fixed effects) using the following data: sedation (NRS) and pain scores (DIVAS, 4AVet, MNT), the occurrence of adverse effects, need for supplementary anaesthesia and rescue analgesia.
Ordinal response variables (NRS, 4AVet) were summarised by the median (range), continuous variables (DIVAS, MNT, HR, RR, SpO2) by the mean (SD) and dichotomous variables (adverse effects, supplemental anaesthetics and rescue analgesia) by the frequencies for each group. The analysis was performed in SAS version 9.3 (SAS Institute).
Results
Four hundred and forty-eight kittens were included. Of those, 380 belonged to the PPG group (female: n = 197; male: n = 183) and 68 to the TAG group (female: n = 34; male: n = 34).
Female PPG
One hundred and ninety-seven kittens were recruited: DB-IM group: n = 93; DB-OTM group: n = 84; and MBK-IM group: n = 20. Mean (SD) body weight was 1.01 (0.20) kg and showed no statistically significant difference among the groups (P = 0.2590). Buccal pH was measured in 135/197 kittens and was between 8 and 9 in 131 kittens and <8 in four kittens.
At baseline, not all pain scores were equal to 0 but scores did not differ significantly between treatment groups (Table 2). Twenty-three kittens were excluded for pain assessment because they were too frightened to assess pain accurately.
Comparison of pain assessment data over time

Data are presented as mean (SD) for DIVAS and MNT (newton) scores, and as median (range) for 4AVet scores following prepubertal gonadectomy in female (F) and male (M) kittens randomly assigned to one of the three treatment groups: (1) intramuscular (IM) administration of 60 µg/kg dexmedetomidine plus 20 µg/kg buprenorphine followed by an IM injection of the anaesthetic agent (20 mg/kg ketamine; DB-IM); (2) oral transmucosal (OTM) administration of 80 µg/kg dexmedetomidine plus 20 µg/kg buprenorphine followed by an IM injection of 20 mg/kg ketamine combined with dexmedetomidine (20 µg/kg; DB-OTM); (3) IM injection of a 40 µg/kg medetomidine plus 20 µg/kg buprenorphine plus 20 mg/kg ketamine combination (MBK-IM).
Significant differences between OTM and IM (P <0.05); †significant differences between OTM and MBK (P <0.05)
The anaesthetic protocol used was significantly associated with vomiting (P <0.0001) and with salivation (P = 0.0361) within 15 mins of premedication, with significant differences for vomiting between DB-OTM and DB-IM (P <0.0001) and between DB-OTM and MBK-IM (P <0.0001). Vomiting was most often observed following OTM administration of the premedication: 54/84 DB-OTM, 14/90 DB-IM and 0/20 MKB-IM.
Salivation was observed after 5/84 DB-OTM and 1/90 DB-IM administration of the premedication and in none of the kittens in the MBK-IM group.
The sedation scores 15 mins after premedication were significantly higher in the DB-IM group (4 [1–4]) than in the DB-OTM group (4 [1–4]; P = 0.0015). More kittens were sedated (at least a score of 3) after IM administration (91/93 DB-IM) than after OTM administration (77/84 DB-OTM) of premedication. The mean time to reach a sedation score of 3 was 1.47 (0.48) mins after MBK-IM administration. All kittens reached a surgical plane of anaesthesia.
In every group, at least one kitten required an additional dose of a systemic or local anaesthetic drug: 5/93 DB-IM, 12/84 DB-OTM, 1/20 MBK-IM (P = 0.0872). In all cases, physiological parameters (HR, RR, SpO2 and T) decreased from baseline values during anaesthesia. No significant differences among groups (Table 3) were detected for HR (P = 0.1278), RR (P = 0.8336) and SpO2 (P = 0.7070).
Mean (SD) heart rate, respiratory rate and SpO2 (%) during surgery and rectal temperature at the end of surgery in female and male kittens randomly assigned to one of the three treatments groups

End of surgery
The anaesthetic protocol used had a significant effect on the incidence of at least one postoperative adverse effect (P = 0.0065). Postoperative adverse effects were most often observed in the DB-OTM group (23/84) compared with the DB-IM group (12/93; P = 0.0162) or the MBK-IM group (0/20; P = 0.0113). Salivation (20/84 DB-OTM, 2/93 DB-IM), excitation (8/84 DB-OTM, 5/93 DB-IM) and vomiting (3/84 DB-OTM, 6/93 DB-IM) were observed. Eight kittens (7/84 DB-OTM, 1/93 DB-IM) showed signs of salivation and excitation; one kitten in the OTM group salivated, vomited and showed excitation.
Sedation scores 3 h postoperatively were significantly higher in the DB-OTM group (1 [0–4]) compared with the DB-IM group (1 [0–4]; P = 0.0010) or MBK-IM group (1 [0–4]; P = 0.0432). More kittens in the DB-OTM group (34/84) than in DB-IM group (19/92) or MBK-IM group (5/20) were still sedated (at least a score of 2) at 3 h postoperatively, and these kittens received atipamezole. All kittens recovered uneventfully. Time to sternal recumbency did not differ between anaesthetic protocols, but time to eating in recovery was significantly different between anaesthetic protocols (P = 0.0133; Table 4). Time to eating in the DB-IM group (193 [40] mins) was significantly shorter than time to eating in the DB-OTM group (214 [38] mins) (P = 0.0102). At 6 h postoperatively, almost all kittens (191/194) were completely awake (score of 0); three kittens (2/92 DB-IM, 1/83 DB-OTM) were still slightly sedated (score of 1) without significant differences among groups (P = 0.5875).
Mean (SD) recovery time (sternal recumbency and eating) in mins after prepubertal gonadectomy for female and male kittens randomly assigned to one of the three treatment groups
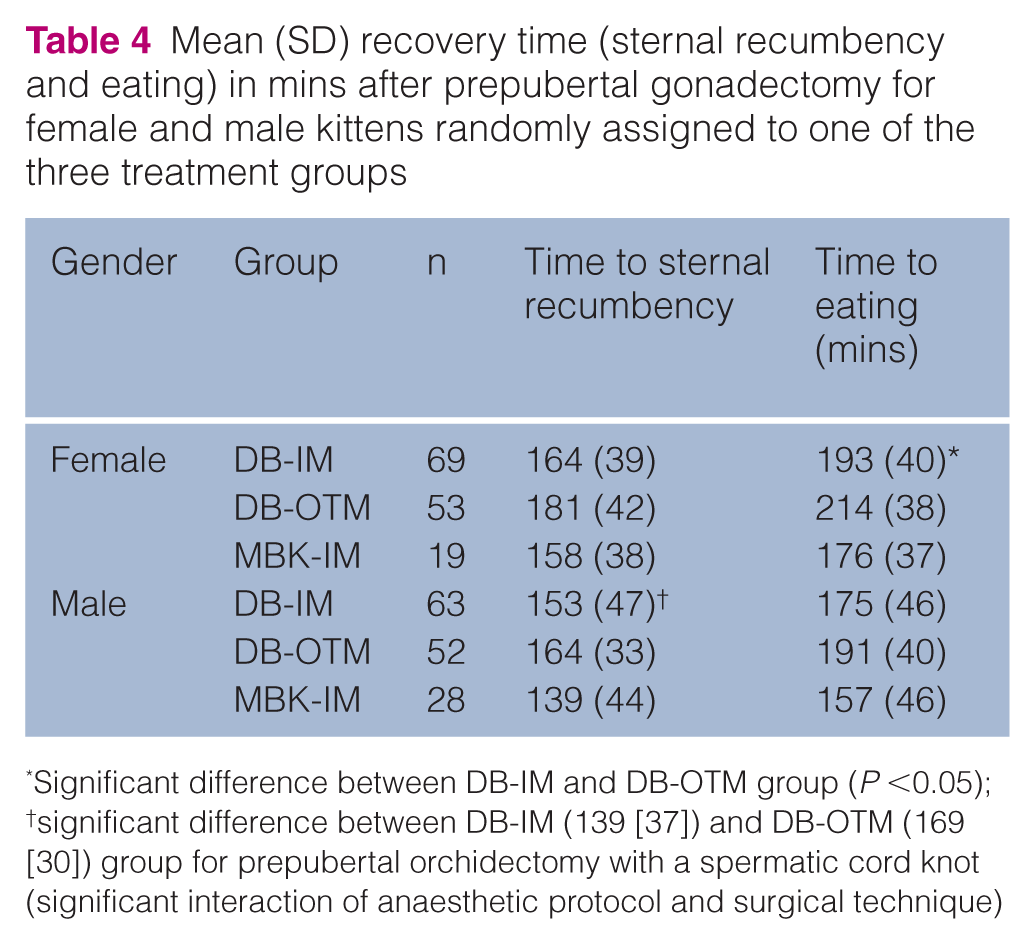
Significant difference between DB-IM and DB-OTM group (P <0.05); †significant difference between DB-IM (139 [37]) and DB-OTM (169 [30]) group for prepubertal orchidectomy with a spermatic cord knot (significant interaction of anaesthetic protocol and surgical technique)
Table 2 shows the DIVAS, 4AVet, MNT and MNT wound scores at the various time points. No significant differences were detected between the three groups at 3 h postoperatively (DIVAS P = 0.6790, 4AVet P = 0.1568, MNT P = 0.3953, MNT wound P = 0.5869) and at 6 h postoperatively (DIVAS P = 0.3606, 4AVet P = 0.9910, MNT P = 0.3405, MNT wound P = 0.5907). Rescue analgesia was administered in one kitten of the DB-IM group 3 h postoperatively and in five kittens (3/78 DB-IM, 2/79 DB-OTM) 6 h postoperatively without significant differences among groups (P = 0.7995).
Female PPG and TAG
The female PPG kittens from the DB-IM group with ligatures as surgical technique (n = 23) were compared with 34 female TAG cats. Mean (SD) body weights were 1.00 (0.21) kg and 2.61 (0.34) kg, respectively.
Three PPG cats vomited after premedication (P = 0.2552), whereas one TAG cat salivated after premedication (P = 1.000). In both groups, all cats were sedated (at least a score of 3) at 15 mins after premedication (P = 0.7284), and all cats reached a surgical plane of anaesthesia after ketamine injection. Only 1/23 PPG and 2/34 TAG cats needed an additional systemic anaesthetic drug to allow ovarian isolation (P = 0.5478).
Significant differences in postoperative adverse effects were not detected between PPG (3/23) and TAG (1/34) cats (P = 0.6315). At 3 h postoperatively, the sedation score was significantly higher in PPG (1 [0–4]) compared with TAG (0 [0–2]) cats (P = 0.0220). More PPG kittens (6/23) than TAG cats (2/31) were still sedated (at least a score of 2) and thus received atipamezole. Also, DIVAS pain scores at 3 h postoperatively were significantly higher in PPG (17 [11]) compared with TAG (6 [9]) cats (P = 0.0011; Table 5). Only one cat (TAG) received rescue analgesia at 6 h postoperatively without a significant difference between PPG and TAG (P = 0.4615).
Comparison of pain assessment data over time, presented as mean (SD) for DIVAS and MNT (newton) scores, and presented as median (range) for 4AVet scores in female cats randomly assigned to the prepubertal gonadectomy (PPG) group and the gonadectomy at traditional age group (TAG), both anaesthetised with the DB-IM protocol

Significant difference between PPG and TAG cats (P <0.05); †no statistical analysis between PPG and TAG cats due to use of different probe tip for PPG and TAG cats
Male PPG
One hundred and eighty-three kittens were recruited: DB-IM: n = 78; DB-OTM: n = 76; and MBK-IM: n = 29. Mean (SD) body weight was 1.04 (0.21) kg and showed no statistically significant difference among the groups (P = 0.1000). Buccal pH was measured in 100/183 kittens and was between 8 and 9 in 98 kittens, and <8 in two kittens.
At baseline, mean pain scores were not equal to 0 (Table 2). Only 4AVet baseline values in kittens assigned to the DB-OTM group were significantly higher than the values in kittens assigned to the DB-IM group (P = 0.0296) or MBK-IM group (P = 0.0008). Seventeen kittens were not assessed for pain due to their frightened reaction to the investigator.
The anaesthetic protocol used was significantly associated with vomiting after administration of the premedication (P <0.0001) with significant differences between groups. Of the 76 kittens in the DB-OTM group, 55 kittens vomited in contrast to 15 of the 77 kittens in the DB-IM group (P <0.0001) and no kittens in the MBK-IM group (P = 0.0010). Salivation was observed occasionally, irrespective of the anaesthetic protocol used (P = 0.3258): 2/76 DB-OTM, 0/78 DB-IM and 1/29 MBK-IM. Sedation scores (15 mins after premedication) were significantly higher after IM administration (4 [2–4]) than after OTM administration (4 [0–4]; P = 0.0461). More kittens from the DB-IM group (77/78) than from the DB-OTM group (69/76) were sedated (at least a score of 3) at 15 mins after premedication. Mean time to reach a sedation score of 3 was 1.42 (0.63) mins after MBK-IM administration. All kittens reached a surgical plane of anaesthesia.
A surgical plane of anaesthesia was reached in all kittens without the need for an additional local or systemic drug. In all cases, physiological parameters (HR, RR, SpO2 and T) decreased from baseline values during anaesthesia. No significant differences among groups (Table 3) were detected for HR (P = 0.2348), RR (P = 0.0605) or SpO2 (P = 0.9033).
The anaesthetic protocol used had a significant effect on the incidence of at least one postoperative adverse effect (P = 0.0100). Postoperative adverse effects were more often observed in the DB-OTM group (22/73) compared with the DB-IM group (9/76; P = 0.0219): salivation (13/73 DB-OTM, 2/76 DB-IM), excitation (10/73 DB-OTM, 6/76 DB-IM) and vomiting (4/73 DB-OTM, 4/76 DB-IM). In the MBK-IM group, excitation was observed in one kitten. Six kittens (4/73 DB-OTM, 2/76 DB-IM) were observed with signs of salivation and excitation. Sedation score at 3 h postoperatively was significantly higher in the DB-OTM group (1 [0–4]) compared with the DB-IM group (1 [0–4]; P = 0.0002) and the MBK-IM group (0 [0–4]; P = 0.0010). More kittens in the DB-OTM group (26/75) were still sedated (at least a score of 2) and received atipamezole compared with kittens in the DB-IM (13/77) and the MBK-IM group (1/29). At 6 h postoperatively, all but three kittens were completely awake (score of 0), whereas those three kittens (2/75 DB-OTM, 1/75 DB-IM) were slightly sedated (score of 1) without significant differences among treatment groups (P = 0.5037). All kittens recovered uneventfully. There was a significant interaction between the anaesthetic protocol and surgical technique for time to sternal recumbency in recovery (P = 0.0240). For prepubertal orchidectomy with a spermatic cord knot, time to sternal recumbency did significantly differ between the anaesthetic protocols (P = 0.0029). Time to sternal recumbency in the DB-IM group (139 [37] mins) was significantly shorter than in the DB-OTM group (169 [30] mins; P = 0.0024). There was no significant difference between the anaesthetic protocols in time to eating (P = 0.1349; Table 4) and in pain scores at 3 h postoperatively (DIVAS P = 0.7101, 4AVet P = 0.5108, MNT P = 0.8635) and at 6 h postoperatively (DIVAS P = 0.4933, 4AVet P = 0.3409, MNT P = 0.8873; Table 2). Rescue analgesia was only deemed necessary at 6 h postoperatively in four kittens (2/78 DB-IM, 2/76 DB-OTM).
Male PPG and TAG
The male PPG kittens from the DB-IM group with knot as the surgical technique (n = 39) were compared with 34 male TAG cats. Mean (SD) body weight was 1.04 (0.21) kg (PPG) and 3.32 (0.44) kg (TAG).
Vomiting after administration of the premedication was seen in PPG (6/39) as well as TAG (2/34) cats (P = 0.3223). Salivation was observed in two TAG cats (P = 0.2265). Adequate sedation was reached 15 mins after premedication in both groups (P = 0.3331), and a surgical plane of anaesthesia was reached after ketamine injection. Only one cat (TAG) needed an additional local anaesthetic drug during surgery (P = 1.0000).
Significant differences in postoperative adverse effects were not detected between PPG (5/38) and TAG (2/34) cats (P = 0.6845). At 3 h postoperatively, the NRS sedation score was significantly higher in PPG (1 [0–4]) than in the TAG (0 [0–1]) group (P = 0.0017): more PPG kittens (5/39) than TAG cats (0/34) were still sedated (at least a score of 2) and received atipamezole. Furthermore, 4AVet values were significantly lower in PPG (0 [0–3]) compared with TAG (1 [0–4]) cats (P = 0.0267) at 3 h postoperatively (Table 6). DIVAS score 6 h postoperatively was significantly lower in PPG (2 [2]) than in TAG (6 [8]) cats (P = 0.0181; Table 6). There was no need for rescue analgesia.
Comparison of pain assessment data over time, presented as mean (SD) for DIVAS and MNT (Newton) scores, and median (range) for 4AVet scores in male cats randomly assigned to the prepubertal gonadectomy (PPG) group and the gonadectomy at traditional age group (TAG), both anaesthetised with the DB-IM protocol

Significant difference between PPG and TAG cats (P <0.05); †no statistical analysis between PPG and TAG cats due to use of different probe tip for PPG and TAG cats
Discussion
There were no anaesthesia-related mortalities in this study. This finding is in agreement with previous reports using similar drug combinations for neutering kittens.1,4,10,14 Furthermore, there were no clinically relevant differences in any of the anaesthetic/analgesic parameters between PPG and TAG.
Although the criteria (analgesia, sedation and loss of consciousness) for balanced anaesthesia 5 were fulfilled in all three anaesthetic PPG protocols, the current results are in favour of the IM administered protocols (DB-IM and MBK-IM). Compared with IM premedication (DB-IM), fewer sedative effects and more pre-anaesthetic adverse effects were observed following OTM administration of the premedication (DB-OTM). To facilitate the clinical interpretation of the sedative and analgesic effects following different routes of administration, a pharmacodynamic 30 and pharmacokinetic 31 study were performed to compare OTM and IM administered dexmedetomidine plus buprenorphine in experimental adult cats. The results suggested that dexmedetomidine combined with buprenorphine was not as well absorbed from the oral mucosa as from the muscular injection site. 31 This finding parallels the fact that higher doses of dexmedetomidine were selected for sedation in the DB-OTM group compared with the DB-IM group. Although the alkaline pH of the buccal saliva of the kittens would suggest a high uptake of such drugs,18,19 the simultaneous administration of the pharmaceutical drug formulations of dexmedetomidine hydrochloride and buprenorphine hydrochloride, and the rather acid pH of their combined solution (ie, 4.383), were likely to affect OTM absorption negatively. Furthermore, some kittens seemed to resent OTM administration, possibly due to the taste of the solution.
After MBK-IM administration, adverse effects were rarely observed, likely due to the rapid onset of sedation and quick induction of anaesthesia which reduces activation of the vomiting centre. 12 On the other hand, it should be mentioned that adverse reactions (vocalising, withdrawal, biting) during the injection were more prominent in the MBK-IM group than in the DB-IM group (data not shown), precluding single handler injections for the former period. This observation has been reported previously with similar dissociative drug combinations. 12
Recovery times following PPG were considered long relative to the short duration of surgery. 9 These observations were also reported in cats aged from 2 months to 6 years. 14 Atipamezole was not administered at the end of (or shortly after) surgery, although routine use of a postoperative alpha-2 adrenergic antagonist probably would have shortened recovery time without inducing adverse effects4,13 and without compromising postoperative pain relief. 4 A shorter recovery time reduces the risk of hypoglycaemia as well as of hypothermia, 4 which is why an alpha-2 adrenergic antagonist may be beneficial, particularly in kittens.
Different methods for pain assessment were combined, since a validated method in cats was not available when this study started in 2010. Besides DIVAS and 4AVet pain scores, MNT was measured as a more objective method to evaluate postoperative pain in cats. 32 Due to the use of a probe tip with a different diameter, MNT pain scores between PPG and TAG cats were not compared. To obtain consistent pain scores, a single person assessed pain.4,25,33 Frightened kittens were excluded from pain assessment based on the inability to palpate the surgical site and the lack of differentiation between fear and stress behaviours and pain behaviours. 34 It should also be noted that preoperative baseline values were not equal to zero. Anxiety and stress (following the transportation, new environment, strange person [NP]) will likely have resulted in an overestimation of these baseline pain scores.
In the postoperative period, on the other hand, the cat’s level of sedation and consciousness would affect pain assessment in the opposite direction. 23 For this reason, sedation was assessed, and, if needed, atipamezole was administered before pain was assessed. For all protocols, pain scores remained low at 3 and 6 h postoperatively, and very few kittens required rescue analgesia. Although some significant differences in DIVAS values between the PPG and TAG group were observed, pain scores were low in both groups, and thus the clinical relevance of the statistical differences is questionable. The low scores in the present study using anaesthetic protocols based on pre-emptive and multimodal analgesia are likely to support the finding that most kittens and young adult cats experienced mild pain following gonadectomy if provided with adequate analgesia.4,14 For practical considerations, analgesia was only evaluated up to 6 h postoperatively. Also, for animal welfare reasons, it was beneficial that (frightened) kittens only spent a minimal period of time at the Faculty. Pain in the later postoperative period was further assessed at the shelter (PPG) or at home (TAG). Caretakers (PPG) and owners (TAG) were requested to fill out the 4AVet pain scale up to 48 h postoperatively, since it was the most applicable method for lay persons to assess pain. Pain behaviour in all kittens and cats was diminishing according to the caretakers (PPG) and owners (TAG; data not published). Despite the variety in evaluators, this finding is not unimportant because caretakers or owners are well placed to evaluate the level of pain or anxiety experienced by their own cats. 35
This clinical study was subject to a number of limitations in terms of data collection. Since a single person (NP) performed all procedures related to anaesthesia and surgery, data collection was not blinded. The data of kittens initially enrolled in the large-scale project on PPG were not included in the present study. Lower doses of dexmedetomidine (DB-OTM and DB-IM) and ketamine were used at that time, but since an inadequate surgical plane of anaesthesia was reached in many cases, doses of dexmedetomidine and ketamine were gradually increased. Other research groups also used higher doses of ketamine 10 or medetomidine-ketamine 1 in kittens. It is quite likely that slightly lower doses of dexmedetomidine and ketamine would have been sufficient towards the end of the research project when surgical times were shorter. 9 Two different non-steroidal anti-inflammatory drugs (NSAIDs) were used in the protocols for PPG. However, it is advisable to use meloxicam as an NSAID for PPG in kittens, since, if necessary, its administration can be continued orally beyond 24 h postoperatively, whereas it is advised that carprofen is only administered once. 36
Conclusions
There were no anaesthesia-related mortalities in the present study, and all anaesthetic protocols for PPG provided a surgical plane of anaesthesia and analgesia up to 6 h postoperatively. Despite its painless and easy administration, DB-OTM cannot be recommended as an ideal route for premedicating kittens. The MBK-IM protocol seems the most attractive in a shelter medicine environment.
Footnotes
Acknowledgements
The authors would like to thank the shelters and cat owners for their participation in this research project. Thanks are also due to Michael Dixon and Polly Taylor for their advice about the mechanical threshold testing.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
Funding
This research (RT 09/12 Sterycat) was funded by the Belgian Federal Public Service (FPS) Health, Food Chain Safety and Environment.

