Abstract
A pericardial cyst developed in a 2-year-old male neutered Maine Coon cat following surgery for an incidentally diagnosed congenital peritoneopericardial diaphragmatic hernia. The cyst caused no clinical signs in the cat, although clinical findings included positional right-sided cardiac tamponade and compression of thoracic structures, associated with a cardiac arrhythmia and axis deviation on electrocardiography. Extensive assessment of the cyst included radiography, echocardiography, computed tomography, exploratory thoracotomy, electrocardiography, histopathology and fluid analysis. Surgical removal of the cyst was curative, and the arrhythmia and axis deviation resolved. This report details case management from initial diagnosis to long-term follow-up, adding to the limited body of literature available on feline pericardial cysts. This is also the first report to associate cardiac arrhythmia with a pericardial cyst.
Case Report
A 2-year-old male neutered Maine Coon cat was presented for echocardiography for hypertrophic cardiomyopathy screening. One episode of coughing was reported in the 2 weeks prior to echocardiography. The cat was in poor body condition, weighing 6.7 kg and with a body condition score (BCS) of 3/9, despite an excellent appetite, regular anthelmintic treatment (emodepside/praziquantel [Profender; Bayer]) and negative status for feline immunodeficiency virus/feline leukaemia virus.
On physical examination, there was a shifting of audible cardiac sounds from the ‘typical’ sternal location to a lateral thoracic location on auscultation. Abdominal palpation was unremarkable, with faeces palpable in the colon, and a normal renal size and shape. Echocardiography revealed dorsal displacement of the heart and the presence of peristaltic intestinal loops within the pericardial space. The right atrium was compressed and there was mild cardiac tamponade, but no evidence of pleural or abdominal effusion, jugular venous distension or hepatic venous congestion. Conscious thoracic radiography (Figure 1a) revealed enlargement of the cardiac silhouette, dorsal displacement of the trachea, microhepatica and large bowel loops filled with faeces tracking to the level of the cardiac silhouette, consistent with a diaphragmatic defect. A peritoneopericardial diaphragmatic hernia (PPDH) was suspected. The lung fields were reduced in size but had a normal pattern.

Right lateral thoracic radiographs. (a) At the time of original diagnosis of peritoneopericardial diaphragmatic hernia, demonstrating an enlarged cardiac silhouette consistent with cardiomegaly. The cardiac silhouette merges with the diaphragmatic outline and loops of large bowel can be seen within the pericardium (white arrow). (b) Immediate postoperative radiograph demonstrating pneumopericardium and a clearly defined diaphragm. (c) Radiograph demonstrating dorsal displacement of the cardiac silhouette by a large soft tissue opacity structure (white arrow) adjacent to the sternum. (d) Follow-up radiograph revealing no abnormalities. The diaphragmatic outline is well defined, the cardiac silhouette is unremarkable and the lung fields are normal
Surgical repair of the defect was performed 3 days later under general anaesthesia. Premedication with methadone (0.2 mg/kg) followed by induction with alfaxalone and maintenance with sevoflurane was utilised. A ventral midline coeliotomy was performed and a large defect was found in the centre of the diaphragm, with right and central hepatic lobes, along with the associated biliary tract, herniated into the thoracic cavity. The contents of the hernia were reduced, adhesions from the liver to the diaphragm and pericardium were released by blunt dissection, and the defect was repaired using 4-0 polyproplene (Prolene; Ethicon) in a continuous pattern. Bilateral thoracostomy drains (14 gauge × 20 cm; Mila International) were placed and removed 24 h post-operatively as non-productive. Recovery was uneventful, and post-operative thoracic radiography confirmed resolution of the defect (Figure 1b), and the patient was discharged the following day with a 5 day course of meloxicam (0.05 mg/kg once daily [Metacam; Boehringer Inghelheim]).
The cat was routinely re-assessed 4 months later, at which time no further concerns were reported and the cat was reported to have recovered without complications; body weight had increased to 8.1 kg, with a BCS of 5/9. An arrhythmia with a regularly irregular rhythm was detected on cardiac auscultation, with good strength matching pulses and a heart rate of 200 beats per minute, which had not been present postoperatively following PPDH repair. The remainder of the physical examination was otherwise unremarkable. Electrocardiographic (ECG) assessment (Figure 2a, performed in right lateral recumbency) revealed a bigeminal rhythm, with normal sinus beats followed by a triggered supraventricular premature complex (SVPC) with a constant coupling interval. There was a left axis deviation, consistent with left anterior fascicular pattern (QRS complex of normal duration, with small Q wave and tall R wave in leads I and aVL, and deep S wave in leads II, III and aVF).

Standard six-lead electrocardiographic analysis at 50 mm/s and 20 mm/mV revealing (a) bigeminal rhythm, with normal sinus beats, followed by triggered supraventricular premature complexes, with a constant coupling interval and a left axis deviation (–70°) consistent with left anterior fascicular pattern (QRS complex of normal duration, with small Q wave and tall R wave in leads I and aVL, and deep S wave in leads II, III and aVF). (b) Normal sinus rhythm following surgical correction of the pericardial cyst
Echocardiography revealed a large anechoic structure associated with the pericardium (Figure 3a,b). The structure appeared to be lateralised to the right, with positional tamponade of the right atrium in right lateral recumbency. On thoracic radiography (Figure 1c), the cardiac silhouette was elevated from the sternum by a soft tissue opacity with dimensions of 8 cm × 3 cm. The right atrium and right ventricle were obscured by the opacity. The trachea was displaced dorsally and the ventral diaphragm was poorly visible.

Echocardiography. (a) Rotated right parasternal short-axis view demonstrating an anechoic cystic structure adjacent to the right ventricle (RV). (b) Right parasternal long-axis view, demonstrating an anechoic cystic structure adjacent to the RV, causing tamponade of the right ventricular outflow tract. (c) Right parasternal short-axis view at the level of the heart base 6 months following surgical removal of the cyst, demonstrating normal structures and no further cystic structures. (d) Right parasternal long axis four-chamber view demonstrating normal chamber dimensions and no cystic structures 6 months following surgical removal. Ao = aorta; LA = left atrium; LV = left ventricle; RA = right atrium; RVOT = right ventricular outflow tract; PV = pulmonary valve; PA = pulmonary artery
Owing to the lack of overt clinical signs, the cat was monitored over the following months. Repeat echocardiography 3 months later confirmed the continued presence of the cystic structure, with an increase in size and continued positional tamponade. Thoracic computed tomography angiography (Figure 4) revealed a large fluid-filled, non-contrast-enhancing, cyst-like lesion, of approximately 9 cm × 4 cm in size in the right ventrolateral aspect of the thorax, extending from the fifth rib to the diaphragm. At the level of the sixth/seventh rib, a thin capsule-like structure and a small amount of fat could be seen separating the cyst-like structure from the cardiac silhouette. The rim of the lesion was thin and contrast enhancing, and was in close proximity to the caudal vena cava and the diaphragm. The ventral aspect of the right middle and entire caudal aspect of the left cranial lung lobes were soft tissue enhancing, suggestive of secondary atelectasis from compression. The sternal lymph nodes were enlarged.

Computed tomography angiography images demonstrating a large non-contrast-enhancing structure (white arrow) located on the right (R) side of the cardiac silhouette on transverse (a) and dorsal (b) views. Note the marked left mediastinal compression
While no clinical signs were apparent at this stage, surgical removal was discussed owing to concerns about the compression of intrathoracic structures. Furthermore, the cyst had appeared to increase in size over this period of time, and elective removal was advised while the patient was asymptomatic to allow for definitive diagnosis and to prevent decompensation. A right sixth intercostal exploratory thoracotomy was performed 24 h later. Premedication with 0.2 mg/kg methadone was followed by general anaesthesia using alfaxalone for induction and sevoflurane for maintenance with an intercostal block using lidocaine performed prior to the first incision. The patient was maintained on intravenous fluid therapy at 6 ml/kg/h during the procedure, and reduced to maintenance fluid rates on recovery. An infusion of fentanyl was initiated following the induction of anaesthesia for analgesia, and positive pressure ventilation was given while the thoracic cavity was open. Thoracotomy revealed an enlarged, fluid-filled, cystic structure adherent to the pericardium by a stalk (Figure 5a). The structure was removed en bloc and subphrenic pericardiectomy was performed. The thoracic cavity was lavaged and routinely closed with 2-0 polydioxanone (PDS; Ethicon) circumcostal sutures, 3-0 PDS (Ethicon) for muscle layers and 4-0 poliglecaprone (Monocryl; Ethicon) for subcutaneous and intradermal sutures. A 14-gauge, single lumen, silicone chest drain (Mila International) was then placed and negative pressure reintroduced. The patient was managed post-operatively with a 5 day course of meloxicam (0.05 mg/kg once daily; Metacam), recovered uneventfully and was discharged 4 days later.
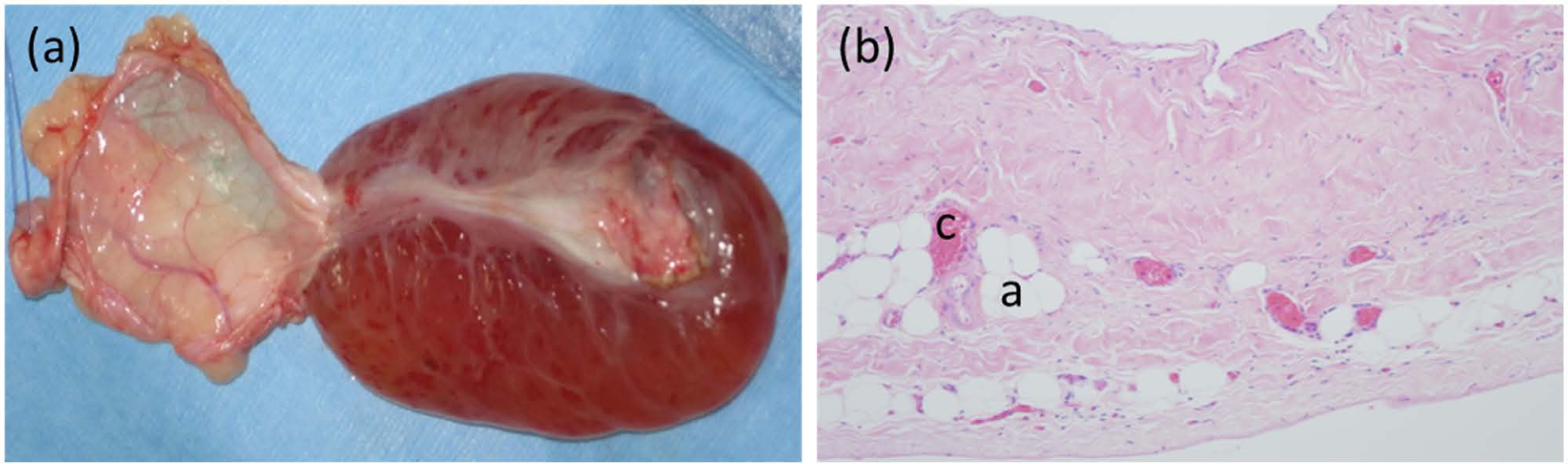
(a) Cystic structure, removed en bloc with its pericardial attachments (to the left of the image). (b) Histopathology reveals a wall of connective tissue lined on both the inner and outer surfaces by flattened mesothelial cells. The fibrous connective tissue wall was segmentally infiltrated with lobules of adipose tissue (a) and congested capillaries (c) (magnification x 10)
Fluid collected aseptically from within the cystic structure had low cellularity; centrifuged preparations revealed a mixed population of cells consisting of 60% large vacuolated macrophages that exhibited erythrophagocytosis or contained intracytoplasmic haemosiderin, 30% small lymphocytes and 10% intact neutrophils in a moderate background of red blood cells. Aerobic culture of the fluid was negative for bacteria.
Histopathology of the cyst (Figure 5b) revealed a wall of fibrous connective tissue lined on both inner and outer surfaces by flattened mesothelial cells. The wall of the cyst contained lobules of adipose tissue, congested blood vessels and dilated lymphatic vessels. The pathological findings were consistent with a cyst.
On reassessment 6 months later, there were no abnormalities on physical examination, echocardiography (Figure 3c,d) or thoracic radiography (Figure 1d). ECG revealed a normal sinus rhythm with resolution of the axis deviation and arrhythmia seen initially (Figure 2b).
Discussion
PPDH is one of the most common congenital diaphragmatic abnormalities in cats and dogs and is proposed to occur as a result of a failure of closure of the septum transversum, or failure of fusion of the septum transversum to the pleuroperitoneal folds during normal fetal development.1,2 The prevalence of PPDH ranges from 0.06% to 1.45%, and is higher in domestic longhair, Himalayan and Maine Coon cats (as in this case) than in domestic shorthair cats.3–5
Feline pericardial cysts are reported rarely in the literature, with more data available on canine cases, and very limited imaging, histopathology or case follow-up.1,5,6 In one large retrospective study on feline patients with PPDH, one cat had a pericardial cyst associated with entrapped hepatic tissue 1 year after surgery, which was diagnosed after the cat presented with respiratory distress after acute trauma. 5 A primary pericardial cyst has also been reported in a DSH cat with right-sided cardiac tamponade, associated with a continuous pedicle from the liver passing through a small PPDH. 1 In another case report, cysts found within the pericardium were found to originate from the right lateral lobe of the liver, which was incarcerated within the pericardium. 6 In addition, a pericardial cyst has been described as an incidental finding in a cat with hyperthyroidism. 7 Histopathology of the cyst in the present case was consistent with pericardial structures, and no hepatic elements were present. Differential diagnoses for the cause of the cyst in this case included a suture reaction from the previous surgery, although this was unlikely due to the distant location of the cyst in relation to previous suture sites in the diaphragm. A congenital cyst was considered; however, no evidence of the cyst was seen at original surgery. Neoplasia, hydatid cysts and entrapped glandular tissue were also considered, but pathology was not consistent with these as a cause. Ultimately, it was suspected that the cyst may have formed from adipose and haematopoietic tissue lining the pericardial sac as a consequence of activation following surgical repair of the PPDH defect and impaired lymphatic drainage leading to fluid formation.
Pericardial cysts are relatively rare in the human population, with an overall incidence of 1:1,000,000 in all age groups. 8 While rare, these cysts account for approximately 7% of all non-malignant mediastinal masses. 9 A significant majority of human pericardial cysts are incidental radiographic findings; 10 however, they have been associated with chest pain and dyspnoea as a consequence of cardiac tamponade. 11 Diagnosis is based on computed tomography, and the treatment of choice is definitive surgery, even in asymptomatic cases, to prevent complications and aid in diagnosis, as was performed in this case. 10
Left anterior fasicular patterns can be seen on feline ECGs for many reasons, including left ventricular hypertrophy, hyperkalaemia, ventricular pre-excitation and altered heart position within the thorax. 12 In this case, the most likely cause was altered intrathoracic heart position, secondary to compression from the cyst, based on resolution of the left axis deviation following removal of the cyst, and no evidence of alternative causes. While an association between left anterior fascicular block and cardiac hypertrophy has been reported in the literature, 13 no evidence of hypertrophy was seen on follow-up echocardiography in this case; therefore, it was not considered a differential. SVPCs are caused by ectopic foci within the atria due to either re-entry, abnormal automaticity or triggered activity.12,14 In this case, owing to the bigeminal rhythm and constant coupling interval between sinus beat and SVPC, triggered activity was considered most likely. Resolution of the SVPCs following removal of the cyst suggested a corresponding cause and effect from compression of the heart, leading to increased automaticity of this atrial focus, which resolved with excision of the cyst. While an interesting clinical finding, this electrical abnormality was not considered haemodynamically significant.
Conclusions
To the best of our knowledge, this is the first case report to provide thorough clinical details of a feline pericardial cyst from initial diagnosis with multiple imaging modalities, along with histopathological and cytological assessment. Long-term follow-up was also available in this case. Furthermore, we believe that this is the first example of a pericardial cyst associated with electrical arrhythmias as opposed to signs associated with right-sided cardiac tamponade, as previously described. 1 Of note in this case is the resolution of arrhythmia and electrical axis deviation following surgical removal of the cyst.
Footnotes
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
Funding
The authors received no specific funding or agency grant in the public, commercial or not-for-profit sectors for the preparation of this case report.
