Abstract
The aim of this prospective study was to characterise patient characteristics and the histories of cats with acquired nasopharyngeal stenosis (ANS), and to describe the use of a removable silicone stent for treatment. ANS was diagnosed in 15 cats with clinical signs present for a median of 4 months. Clinical signs included stertor and inspiratory difficulty, nasal discharge, sneezing, dysphagia, regurgitation, vomiting and anorexia. Radiographs revealed a dorsal deviation or deformation of the caudal part of the soft palate in 10 of the cats, a soft tissue density across the cranial nasopharynx in four and no abnormality in one. The stenosis was initially dilated with a Kelly forceps in 10 of the cats and by balloon dilatation in five. A segment of a 24 Fr silicone thoracic catheter was used for the stent in five cats; in the other 10 cats a segment of a 28 Fr catheter was used. The stent was removed after 3 weeks in 12 cats and after 4 weeks in the other three. Endoscopy revealed an adequate nasopharyngeal diameter in all of the cats. At both 3 and 10 months after surgery the response was considered to be satisfactory, with complete resolution of clinical signs in 14 cats and improvement in the remaining cat. The treatment of ANS by stenosis dilatation followed by temporary stenting with a silicone stent is a rapid, safe, economical and effective procedure.
Introduction
The nasopharynx is the upper portion of the pharynx between the choanae of the nasal cavity and the intrapharyngeal ostium. 1 Benign acquired nasopharyngeal stenosis (ANS) is an obstruction caused by scar tissue formation between the nasopharynx and the oropharynx. It is relatively uncommon in cats: in five retrospective studies describing nasal and nasopharyngeal disorders in cats, ANS was diagnosed only 5/77, 2 1/24, 3 0/75, 4 0/53 5 and 0/27 6 times. As with other nasopharyngeal disorders, ANS causes signs of upper airway obstruction, which can range from stertorous breathing, gagging, and dyspnoea to open-mouth breathing, vomiting, dysphagia and weight loss. Various methods of treatment have been reported, including surgical excision of the stenotic membrane, 7 a mucosal advancement flap 8 and balloon dilatation.9–11 The need for repeated interventions and the occurrence of complications have been reported for all of these techniques – the greatest problem being recurrence of the stenosis. The purpose of this study was (1) to characterise the signalment, history, clinical signs and radiographic features of cats with ANS; (2) to describe the use of a removable silicone stent for treatment; and (3) to evaluate medium- and long-term results after removal of the stent.
Materials and methods
Animals
Clinical history
Between January 2010 and October 2012, benign ANS was diagnosed in 15 cats: six males (two intact) and nine neutered females, with a mean age of 4.0 years (range 0.5–13.0 years). Thirteen were domestic shorthairs, one was a Siamese and one was a Maine Coon.
Clinical signs had been present for a median of 4 months (range 1–24 months) and included stertor and inspiratory difficulty in all 15, nasal discharge and sneezing in seven, dysphagia in six, regurgitation and vomiting in six, and anorexia in four. All of the cats had been treated with antibiotics and glucocorticoids, with only mild, transient improvement in seven of them. One cat was treated surgically by excision of the stenosing tissue, as described by Mitten, 7 with transient improvement, but recurrence of symptoms within 30 days. Four cats were treated by forceps dilatation of the stenosis and two by balloon dilation, following which they were free of clinical signs for a median of 17.5 days (range 10.0–40.0 days) before stertorous breathing recurred.
Head and thoracic radiographs were obtained in all 15 cats; abdominal radiographs were obtained in seven. Radiographs revealed a dorsal deviation or deformation of the caudal part of the soft palate in 10 of the cats (Figure 1a), a soft tissue density across the cranial nasopharynx in four and no abnormality in one. There was oesophageal dilation alone in four cats, oesophageal and gastric dilation in three, and generalised gastrointestinal meteorism in three.

Right lateral radiograph of the skull in cat 15. (a) The stenotic area corresponds to the dorsal deviation of the soft palate (white arrow). (b) The stent (white arrow) is positioned across the stenotic area with at least 3–5 mm of soft palate extending caudal to it to allow for nasopharyngeal closure during swallowing. The white arrowhead indicates the piece of tubing (cut from a butterfly infusion needle tube) used as a cushion to prevent damage to the soft palate by the suture
Patient characteristics and history are listed in Table 1.
Patient characteristics and clinical history

DSH = domestic shorthair; DLH = domestic longhair; SF = spayed female; NM = neutered male; M = male; Ab = antibiotics; St = steroids; FD = forceps dilatation; BD = balloon dilatation
Surgical approach
After endoscopic examination, stenosis dilatation was performed by forceps or balloon, followed by the placement of a removable silicone stent. All of the procedures were performed under general anaesthesia, by the same person (DDL). The study protocol was in compliance with institutional guidelines for research on animals and with the informed consent of the owners.
The preanesthetic evaluation included signalment, history including treatments, clinical signs and their duration, haematologic findings, and head and thoracic radiographs.
Surgical procedure
The cat was premedicated with intramuscular methadone (0.2 mg/kg) and ketamine (1 mg/kg), and general anaesthesia was induced with intravenous propofol (1 mg/kg) and maintained after intubation with isoflurane in oxygen.
A plain lateral radiograph of the skull was first made to evaluate the nasopharynx, and the shape and orientation of the soft palate. Nasopharyngeal endoscopy was performed with the cat in ventral recumbency, using a flexible fibrescope (5.2 mm diameter × 85 cm length; K. Storz 60001VL). The endoscope was passed through the mouth into the caudal oropharynx then reflexed dorsally over the soft palate to allow visualisation of the nasopharynx and choanae. Nasopharyngeal stenosis was recognised as a narrow opening surrounded by a smooth membrane between the end of the soft palate and the choanae. The diameter of the opening was estimated by comparing it with the tip of the endoscope forceps (1.5 mm closed, 3.5 mm open) passed through it under endoscopic guidance.
The stenosis was dilated by one of two techniques, depending on its location. If the stenosis was in the caudal nasopharynx, the cat was placed in dorsal recumbency and the soft palate was retracted to expose it (Figure 2a). A closed curved Kelly forceps was inserted through the orifice (Figure 2b) and then carefully opened to dilate it to a diameter of 8–10 mm (Figure 2c). Bleeding was moderate and of short duration, and blood and mucus were carefully aspirated before placement of the stent.

With the cat in dorsal recumbency, the soft palate is (a) retracted to allow direct visualisation of the stenosis, and (b) a closed curved Kelly forceps is inserted through the opening and then (c) carefully opened to dilate it to a diameter of 8–10 mm. An endoscopic biopsy forceps with alligator jaws is (d) inserted through the nostril into the nasopharynx and then (e) retracted until its tip can be grasped with forceps and brought into the oral cavity. (f) The silicone stent is then firmly grasped with the alligator jaw forceps and brought into the nasopharynx
If the stenosis was too far cranial to be adequately visualised and treated in this manner, dilation was achieved by inserting a balloon dilatation catheter in an orthograde fashion, as described by Glaus et al. 11 The procedure was monitored via the endoscope retroflexed over the soft palate. A lubricated valvuloplasty balloon dilatation catheter with an inflated diameter of 12 mm (Tyshak II PDC520; Numed Canada) was passed through the nostril and along the ventral nasal meatus and the choanae to the nasopharynx. Once the balloon was positioned in the stenosis, it was inflated and deflated two or three times, and the effect was evaluated endoscopically.
After dilation of the stenosis, a removable silicone stent was inserted and secured in the nasopharynx to prevent recurrence of the stenosis. The stent was created by cutting a 1.5–2.0-cm segment from a silicone thoracic catheter incorporating a radiopaque line (Argyle Silicone Thoracic Catheter; outer diameter 24 Fr for cats younger than 1 year and 28 Fr for older cats). The length of the stent was initially determined by measuring, on a lateral radiograph, the distance between the caudal border of the vomer bone and the caudal edge of the soft palate, then subtracting about 3 mm. Further shorting could be made after stent placement. The stent was then positioned as follows. With the cat in dorsal recumbency and the soft palate retracted, an endoscopic biopsy forceps with alligator jaws (Olympus FB211D.A) was inserted through the nostril to the nasopharynx (Figure 2d) and then retracted until the tip could be grasped with the forceps and brought into the oral cavity (Figure 2e). The silicone stent was then grasped (Figure 2f) and positioned in the nasopharynx by drawing back the forceps and simultaneously gently pushing the stent until it stopped against the vomer bone and the choanae. Its correct placement was ascertained by radiography and nasopharyngoscopy (Figure 1b).
The stent was positioned to lie across the site of the stenosis, but with at least 3–5 mm of soft palate lying free caudal to it to permit nasopharyngeal closure during swallowing.
To prevent migration of the stent, a single suture of 3-0 non-absorbable filament (Lene Vetsuture 2CN) was placed in the middle of the soft palate, perpendicular to its long axis, through the soft palate, the stent and the soft palate again. Before the knot was tied, a 1 cm segment of tubing from a butterfly infusion needle was placed beneath it to lessen the pressure on the soft palate mucosa (Figure 3).
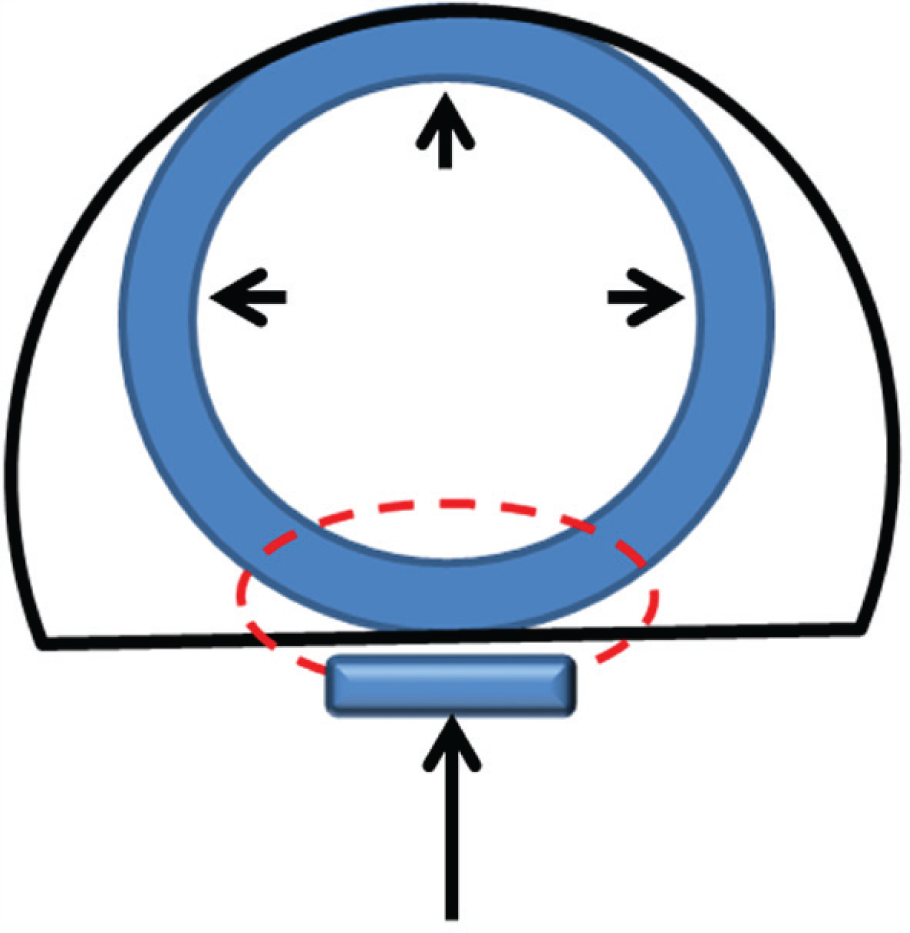
Schematic drawing of a cross-section of the nasopharynx at the level of the suture used to prevent stent migration. Red line = suture, black line = nasopharynx, long arrow = cushion tube, arrowheads = stent in place
Following surgery, prednisolone was administered orally at a dose of 0.5 mg/kg twice daily for 3 days, then once daily for 2 weeks; amoxicillin–clavulanic acid was administered orally at a dose of 20 mg/kg twice daily for 2 weeks. The stent was removed under anaesthesia by cutting the suture and simply extracting it after soft palate retraction, after 3–4 weeks; the patency and size of the airway were checked regularly by endoscopy after stent removal.
Follow-up
A telephone interview with the cat’s owner was conducted at 3 and at 10 months after stent removal. The owner was asked to rate the cat’s respiratory and digestive functions as (1) worsened or not improved; (2) improved, with some limitations such as intermittent noisy breathing; or (3) satisfactory with complete resolution of clinical signs.
Results
Benign ANS was diagnosed in 15 cats. Patient characteristics and history are listed in Table 1. No previous treatment had resulted in lasting improvement.
Endoscopic examination revealed the diameter of the stenotic nasopharynx to be from 1.5 to 4 mm (mean 3 mm). The stenosis was dilated with a Kelly forceps in 10 of the cats and by balloon dilatation in five. The mean duration of dilatation by Kelly forceps was 4.2 mins (range 3–6 mins) and the mean duration of dilation by balloon catheter was 8.6 mins (range 7–12 mins). In all cases the dilation achieved was considered to be adequate.
In five cats (⩽1 year old) a segment of a 24 Fr catheter was used for the stent; in the other 10 cats a segment of a 28 Fr catheter was used. The mean time required for stent placement was 4.4 mins (range 3–6 mins); hence, the duration of the entire procedure was 9–13 mins. Bleeding was moderate and self-limiting in all cats.
In two cats, moderate-to-severe swelling of the soft palate developed after placement of the suture to prevent stent migration, but no treatment was needed and the exaggerated swallowing during eating disappeared after 2–3 days. In three cats there was moderate, self-limiting nasal bleeding following orthograde insertion of the forceps used to grasp the stent and to place it in the nasopharynx.
All cats began to eat soft food regularly 1–2 days after stent placement; stertorous breathing also disappeared in this interval. Two cats had occasional nasal discharge and sneezing associated with food intake, probably because the stent was too long or in too caudal a position, interfering with closure of the intrapharyngeal opening during swallowing. The associated discomfort did not appear to be severe and the stent was left in place for 3 weeks. Nasal discharge disappeared shortly after stent removal in both cats.
The stent was removed after 3 weeks in 12 cats and after 4 weeks in the other three. In all the cats endoscopy revealed an adequate nasopharyngeal diameter and moderate mucosal inflammation associated with moderate mucus accumulation. In three cats there was moderate mucosal hyperplasia just caudal to the choanal opening, probably due to chronic irritation by the cranial edge of the stent. None of the cats was reported to have stertorous breathing at 3 and 10 months by telephone interview. At both 3 and 10 months after surgery, the response was considered satisfactory in 14 of the cats, but qualified in the remaining cat by stertorous breathing during intense physical activity. The findings and results are listed in Table 2.
Findings and results
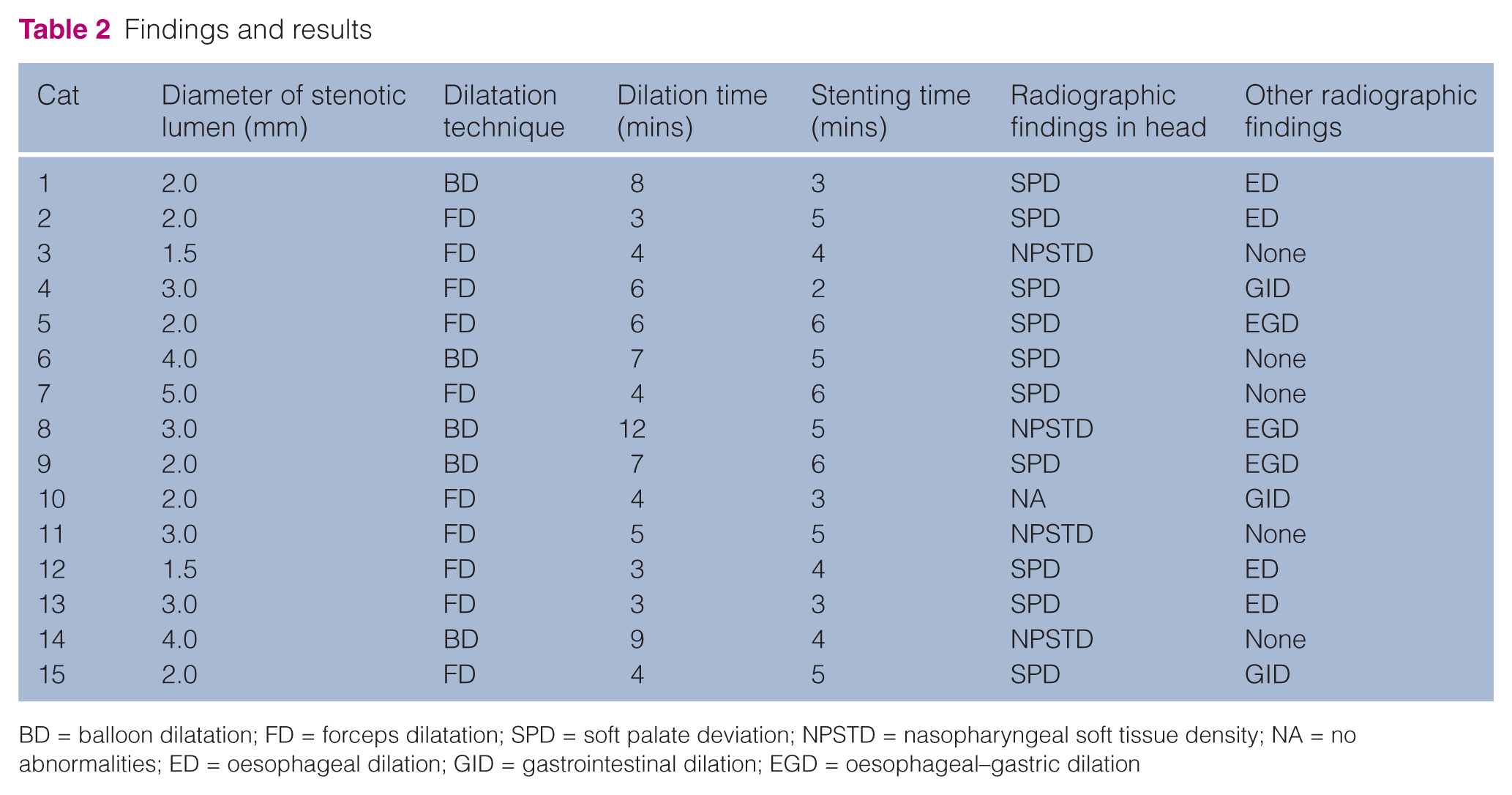
BD = balloon dilatation; FD = forceps dilatation; SPD = soft palate deviation; NPSTD = nasopharyngeal soft tissue density; NA = no abnormalities; ED = oesophageal dilation; GID = gastrointestinal dilation; EGD = oesophageal–gastric dilation
Discussion
ANS is considered to be rare in humans and is most often secondary to tonsillectomy, adenoidectomy, uvulopalatoplasty, radiotherapy or disease such as tuberculosis or syphilis. 12 There were only three cases among 100,000 tonsillectomies and adenoidectomies during 10 years at one human hospital. 13 It has been described in a small number of dogs and cats, with stertorous breathing being more common in cats because they avoid open-mouth breathing.7,11,14 Occlusion of the nasopharynx is usually caused by a membrane due to scar formation following an infectious respiratory disease or vomiting with aspiration of gastric contents into the nasopharynx. 15
ANS is diagnosed by radiographs and endoscopy. Laterolateral radiographs are useful for detecting processes in the nasopharynx8,9,15 and may suggest the underlying disease, but usually do not provide a specific diagnosis. 2 Although the two rami of the mandible overlie the rostral nasopharynx in lateral projection, a nasopharyngeal abnormality was correctly identified in radiographs in 14/15 cases. The stenotic membrane may be seen radiographically as a soft tissue opacity within the nasopharynx dorsal to the soft palate; in this series of cats this was often associated with dorsal deviation or a deformation of the soft palate at the site of attachment. The latter feature was present in 10/15 cats and thus can be considered suggestive of ANS. Gaseous dilation of the thoracic oesophagus was observed in 10 of the cats, but it disappeared spontaneously after nasopharyngeal dilatation and placement of the stent in all cats. The dilation of the oesophagus, and also the stomach and intestine, observed in some cases was likely the result of chronic swallowing of air associated with inspiratory dyspnoea and dysphagia.
Immediate diagnosis of nasopharyngeal disorders without inducing trauma is the major advantage of endoscopic examination of the nasopharynx and the choanae. 6 The flexible endoscope enables localisation of a stenosis and estimation of the diameter of the opening. 16 It also facilitates positioning of a stent and follow-up examination if necessary. Flushing and aspiration of mucus and blood via the endoscope greatly improves the visualisation of the stenosis.
Techniques that have been used to relieve nasopharyngeal stenosis include transpalatal reconstruction with or without mucosal flap rotation, balloon dilatation and intraluminal stenting.7–11,14,17 The greatest problem is recurrence and long-term success has only been obtained with transpalatal resection.14,16 The main complications are breakdown of the repair due to excessive tension and recurrence of stenosis due to scar tissue.
Treatment of nasopharyngeal stenosis by balloon dilatation brought only transient improvement initially, 17 but later achieved long-term resolution in small groups of cats after one or more interventions combined with glucocorticoid administration.10,11
Intraluminal stenting can be temporary or permanent, and different types of stent have been used. A braided wire endoprosthesis placed after removal of stenotic tissue led to obstructive inflammation. 17 Balloon-expandable metallic stents placed in three dogs and three cats 14 were effective in 5/6 animals, although some additional procedures were needed. The authors concluded that such stents are only appropriate if the stenosis is at the junction of the hard and soft palate because a rigid metallic stent located over the soft palate can interfere with swallowing.
The authors of a more recent report described a full-thickness defect in the palate of two toy breed dogs 4 and 20 months following nasopharyngeal application of a covered metallic stent. 18 The owner of one of these dogs complained of continuous nasal discharge after placement of the stent. Examination revealed that the fractured caudal end of the stent had eroded through the palate and caused ulceration of the tongue.
Erosion of tissue around stents in the respiratory and gastrointestinal tracts in humans is common and may be related to incorporation of the stent in the surrounding tissues and subsequent fracture of metallic wires.19,20
In both human and veterinary medicine metallic self-expanding or balloon-expanding stents are commonly used for treating airway obstruction. However, they may fracture or erode into adjacent structures, causing mucous retention and granulation tissue. This is especially of concern in patients with benign airway disease in whom a stent may have to function for years. In humans with benign airway obstruction the complication rate for metallic stents approaches 45%. 21 Thus, the US Food and Drug Administration advised that in patients with benign airway disorders metallic stents should be considered only after thoroughly exploring all other treatment options, such as surgical procedures or placement of silicone stents. 22
Removable stents cut from rubber catheters or feeding tubes were used to treat choanal atresia in two cats,23,24 but we are not aware of other reports of the use of such stents to treat ANS in cats. Although choanal atresia and nasopharyngeal stenosis are different conditions, both frequently relapse following treatment, and repeated efforts are often required. 12
In humans, nasal stenting plays an important role in the success of endoscopic and transpalatal choanal atresia repair techniques, for many authors affirm that stenting prevents postoperative stenosis.25–28 The material used for the stent varies and the length of time it is left in place can range from several weeks to months.
We have shown that treatment of ANS by stenosis dilatation followed by temporary stenting with a silicone stent is a rapid, safe, economical and effective procedure. It is suitable in both young and adult cats as the first approach, as well as for treatment of recurrent stenosis after one or more dilatation procedures. None of the cats included in this study has required any further treatment or re-examination, and there was no difference in the outcome between balloon dilatation and forceps dilatation. Furthermore, the same results were obtained in patients with ANS being treated for the first time and in those treated previously.
Conclusions
Severe nasopharyngeal stenosis is a rare, but severe, complication of nasopharyngeal inflammation in cats. We have described a simple and effective technique employing stenosis dilatation and use of a removable silicone stent, with very favourable results.
Footnotes
Acknowledgements
We are grateful to Dr Anjop J Venker-van Haagen and Dr BE Belshaw for their invaluable advice and editing.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
