Abstract
Ciclosporin (Atopica oral solution for cats 100 mg/ml; Novartis Animal Health) was recently approved for use in cats with feline hypersensitivity dermatitis. The immunosuppressant effect of ciclosporin on the ability of cats to mount an immune response following vaccination was determined. Thirty-two healthy, immunocompetent adult cats (16 cats/group) were treated with either ciclosporin for 56 days at a dose of 24 mg/kg once daily or sham dosed. Prior to treatment, cats had an adequate antibody response to primary vaccination against feline calicivirus (FCV), feline herpesvirus-1 (FHV-1), feline panleukopenia virus (FPV), feline leukemia virus (FeLV) and rabies. Booster vaccination or novel vaccination with feline immunodeficiency virus (FIV) was administered 28 days after initiation of treatment with ciclosporin. There were no differences between the ciclosporin-treated and control cats for FCV and FPV antibody titers following booster vaccination. There were delays/reductions in antibody response to FHV-1, FeLV and rabies in treated cats; however, adequate protection was achieved in response to all booster vaccinations. Following primary vaccination with FIV, control cats showed a response, but treated cats showed no antibody production. Adverse events commonly associated with ciclosporin treatment, including diarrhea/loose stool, vomiting, salivation and regurgitation, were reported. In adult cats treated with 24 mg/kg/day of ciclosporin (more than three times the therapeutic dose), vaccine titer levels were adequate for protection following booster vaccination. In contrast, treated cats failed to mount a humoral response to a novel (FIV) vaccination, suggesting that memory B-cell immune responses remain intact during repeated high-dose ciclosporin administration in cats, but that primary immune responses are impaired.
Introduction
Feline hypersensitivity dermatitis (FHD), usually manifesting between 6 months and 3 years of age, can be a severely debilitating disease of cats.1,2 Ciclosporin (Atopica oral solution for cats 100 mg/ml; Novartis Animal Health), recently approved for use in cats, is an effective treatment for FHD.3–6 Ciclosporin is a cyclic polypeptide that exerts anti-inflammatory and antipruritic effects in the treatment of allergic dermatitis by binding to the intracellular protein cyclophilin-1, thereby inhibiting calcineurin and selectively preventing the dephosphorylation/activation of nuclear factor of activated T cells. 7 It has the capacity to inhibit the antigen-presenting function of the skin immune system, and to block the recruitment and activation of eosinophils, the production of cytokines by keratinocytes, the function of Langerhans cells, the degranulation of mast cells, and, subsequently, the release of histamine and pro-inflammatory cytokines.
Ciclosporin acts specifically and reversibly on T lymphocytes by preferentially impairing the production of interleukin (IL)-2 and other T-cell-derived cytokines. Inhibition of IL-2 is considered to be the primary mechanism of immunosuppression following ciclosporin therapy. 8 In seronegative cats, immunosuppression can negatively affect the immune response to infectious disease such as toxoplasmosis. 9 IL-2 is required for T cell differentiation into effector and memory cells following antigen-mediated activation, and B cell responses are dependent upon activated T cells. 10 The recrudescence of latent feline herpesvirus-1 (FHV-1) has been observed in cats receiving ciclosporin. 11 The vaccination and booster of cats against common infectious viral diseases is an integral part of clinical practice. However, the impact of concomitant ciclosporin treatment on the response to feline vaccination remains unclear.
The objective of this study was to determine the humoral response (antibody titer) in immunocompetent, healthy cats following primary and booster vaccination with commonly administered commercially available vaccines (feline calicivirus [FCV], FHV-1, feline panleukopenia virus [FPV], feline leukemia virus [FeLV], rabies virus and feline immunodeficiency virus [FIV]) during the concomitant administration of ciclosporin at more than three times the recommended dose.
Materials and methods
Study design
This study was a masked, randomized parallel design study with the individual cat as the experimental unit. The study was conducted in accordance with the US Food and Drug Administration (FDA) Good Laboratory Practice Regulations, 21 CFR Part 58 (except for measurement of the vaccine titers), the target animal Safety Guidelines for New Animal Drugs (Center for Veterinary Medicine [CVM] Guideline 33) and FDA CVM Guideline 104. All procedures were reviewed and approved by the local institutional animal care and user committee, and were in compliance with Novartis Animal Health Animal Welfare Guidelines and the US Department of Agriculture Animal Welfare Act (1996).12–15
Animals
Thirty-two domestic shorthair cats (16 males and 16 females), approximately 7 months old and weighing between 2.4 and 5.0 kg on the day before treatment, were included in a 56 day (8 week) study. These laboratory, purpose-bred cats were in good health as determined by a comprehensive physical examination (including neurological examination) and the review of results of fecal examination for parasites and clinical pathology (hematology, serum chemistry and urinalysis). Cats were acclimated for approximately 4 weeks prior to the start of the study. They were housed individually, each with perches, litter pans and enrichment toys in two biosecure and environmentally controlled rooms (room temperature range 17–26°C; humidity 18–76%; 12 h light/dark cycle). Lab Diet (Laboratory Feline Diet #5003; PMI Nutrition International) and water was available ad libitum except during designated periods.
Investigational treatment administration
A final formulation of ciclosporin microemulsion solution (100 mg/ml, Atopica oral solution for cats; Novartis Animal Health) was used. To account for variability in animal sensitivity to ciclosporin and to establish a worst case scenario, a dose of 24 mg/kg/day was chosen for investigation. Cats were blocked and randomized according to sex and body weight into either an untreated control group (group 1, no treatment) or a ciclosporin-treated group (group 2, 24 mg/kg/day, equivalent to more than three times the maximum exposure), with eight males and eight females per group. Treatment was administered by unmasked individuals who were not involved in any other assessments. For 56 consecutive days (8 weeks) starting on day 0, cats were dosed once daily with ciclosporin or were sham dosed approximately 30 mins after introduction to food following an 8 h fast. Ciclosporin was administered into the back of the oral cavity and followed by 1 ml of tap water to ensure adequate ingestion. Doses were calculated based on the most recent body weight. The control group was sham dosed with an empty syringe followed by a 1 ml rinse of tap water in the same manner as the treated group.
Vaccination
The cats had completed a primary vaccination course (FHV-1, FCV and FPV at 8, 12 and 16 weeks of age – Felocell3, Zoetis; FeLV at 9 and 12 weeks of age – Leukocell2, Zoetis; and rabies at 16 weeks of age, ImRab1; Merial Animal Health), according to the manufacturer’s instructions, approximately 16 weeks before starting treatment with ciclosporin. Pretreatment serum samples confirmed an adequate antibody response to all pretreatment vaccine components, and that cats were immunologically naive for FIV. On day 28, all cats received a primary FIV vaccination (Fel-O-Vax FIV; Boehringer Ingelheim) and booster vaccinations for FHV-1, FCV, FPV, FeLV and rabies.
In vivo observations
Cats were observed twice a day throughout the study for morbidity, mortality, injury, and the availability of food and water. Baseline values for body weight and feed consumption were established during acclimation. During treatment administration, body weight was recorded and feed consumption was calculated on a weekly basis. A staff veterinarian was available, if necessary, to further investigate any abnormalities.
In addition to the general observations (described above), detailed clinical observations were made twice daily, approximately 1 and 7 h post-dosing, by a trained technician masked to treatment. At a minimum, the following was evaluated at each observation: eyes, mucous membranes, respiratory system, circulatory system, autonomic and central nervous systems, somatomotor activity and behavior pattern, and gastrointestinal system (including vomiting and gross fecal assessments [color, quantity, consistency and presence of blood]). Particular attention was paid to the presence of clinical signs indicative of ciclosporin toxicity in cats (lethargy/depression, ataxia, tremors, dehydration, lymph node enlargement, gingival proliferation, hair loss, skin growths, red/swollen pinnae and foot pad hyperkeratosis).
A licensed veterinarian completed a physical examination, including a neurological assessment, on each cat twice during acclimation, and on days 0, 6, 14, 27, 34, 42 and 55. These examinations were conducted at least 2 h after dosing on that day and included, at a minimum, the evaluation of appetite/general health, body temperature, behavior, gastrointestinal, ocular, musculoskeletal, integumentary, reproductive, lymphatic, nervous, urinary, cardiovascular and respiratory systems, including heart and respiratory rates.
At any time during the study it was possible to withdraw any cat for humane reasons.
Clinical pathology
Blood samples for hematology, coagulation (activated partial thromboplastin time [APTT], fibrinogen and prothrombin time), and clinical chemistry were collected from all cats (fasted overnight) during acclimation and prior to feeding and dose administration on days 7, 28 (prior to vaccination), 35 and 56. Urine was also collected on the same days by replacing litter with Teflon beads for 16–24 h, or by cystocentesis (day 56, 25 cats). All samples were handled and processed according to standard diagnostic laboratory procedures.
Antibody titers
Blood samples for antibody titer determination were collected from all cats (fasted overnight) during acclimation and prior to feeding and dose administration on days 7, 28 (prior to vaccination), 35 and 56. Serum antibody titers were determined using a serum neutralizing assay for FHV-1 and FCV, hemagglutination inhibition for FPV and rapid fluorescent focus inhibition test for rabies (Kansas State Veterinary Diagnostics Laboratory). FeLV neutralizing antibody titers were determined using a FeLV-infected cell culture system using twofold dilutions (Colorado State University [CSU]). Antibody titers against FIV were determined using a commercially available, well-based quantitative enzyme-linked immunosorbent assay (CSU).
A positive biological response to booster vaccination was defined as at least a two-dilution increase in titer when comparing pre- and post-vaccination titers in the same cat for FHV-1, FCV, FPV and FeLV. An adequate rabies vaccination response was defined as a titer of ⩾ 50 (0.5 IU/ml). A positive biological response to primary FIV vaccination was defined as the titer changing from negative (before) to positive (after) on the day 28 FIV vaccine in the same cat.
Statistical analysis
SAS (SAS Institute) was used for all statistical analyses. For endpoints measured once (body weight change) an analysis of variance was conducted. For endpoints measured multiple times during the study and having a pre-treatment measurement (food consumption, clinical chemistry, hematology and urinalysis parameters, and logarithmically transformed vaccine titers), a repeated measures analysis of covariance was conducted. A 2×2 Fisher’s exact test was performed on the ‘negative/positive’ titer data.
Vaccine titers were compared before and after treatment in both the treated and control groups. The analysis was performed separately for each time period comparison. For each model, the treatment effect was tested at a 0.10 level of significance. If the model included the treatment by time interaction it was tested at a 0.10 level of significance; all other effects and interactions included in any model were tested at a 0.05 level of significance.
Results
Observations
All cats completed the study. In addition, there were no vaccine-related adverse events. Daily dosing with >3× ciclosporin for 8 weeks was generally well tolerated. The most frequently observed clinical sign in both groups was related to the gastrointestinal system. ‘Soft stool’ was observed in five control cats, generally as a single occurrence; this is in contrast to 15 cats in the ciclosporin-treated group that were reported as having soft stool on multiple days, sometimes containing blood and mucus, and frequently coincident with diarrhea. Hypersalivation (one control cat; one treated cat) and vomiting/regurgitation (five treated cats) were also observed. Other group differences included a reduced incidence of estrus in the treated females (one treated female VS five control females) and an increase in enlarged lymph nodes in the control group (three control males VS one treated male) and a greater occurrence in the palpation of small peripheral lymph nodes in the treated group (61 observations in the treated group VS 31 observations in the control group). One ciclosporin-treated cat was noted to have a slow or absent ‘startle reflex’ (days 27 and 34), despite a normal pupillary light response. This cat also had slight hindlimb ataxia (day 55), small peripheral lymph nodes (days 6, 14, 34, 42 and 55), a thin body condition (days 42 and 55), and gas- and fluid-filled loops of small bowel (day 42). Other observed abnormalities (eg, erupting teeth, alopecia, feline acne, umbilical hernia) observed across both groups were considered incidental.
Feed consumption and body weight
Treated cats consumed significantly less (P ⩽0.004) food than control cats prior to and for the first week of treatment, and they consumed more (P ⩽0.0625) food than control cats for the final 4 weeks of treatment. An apparent decrease in food consumption was noted in both the treated and control groups, for both sexes, in week 4 following vaccination on day 28 (Figure 1). There were no other differences in food consumption between groups.
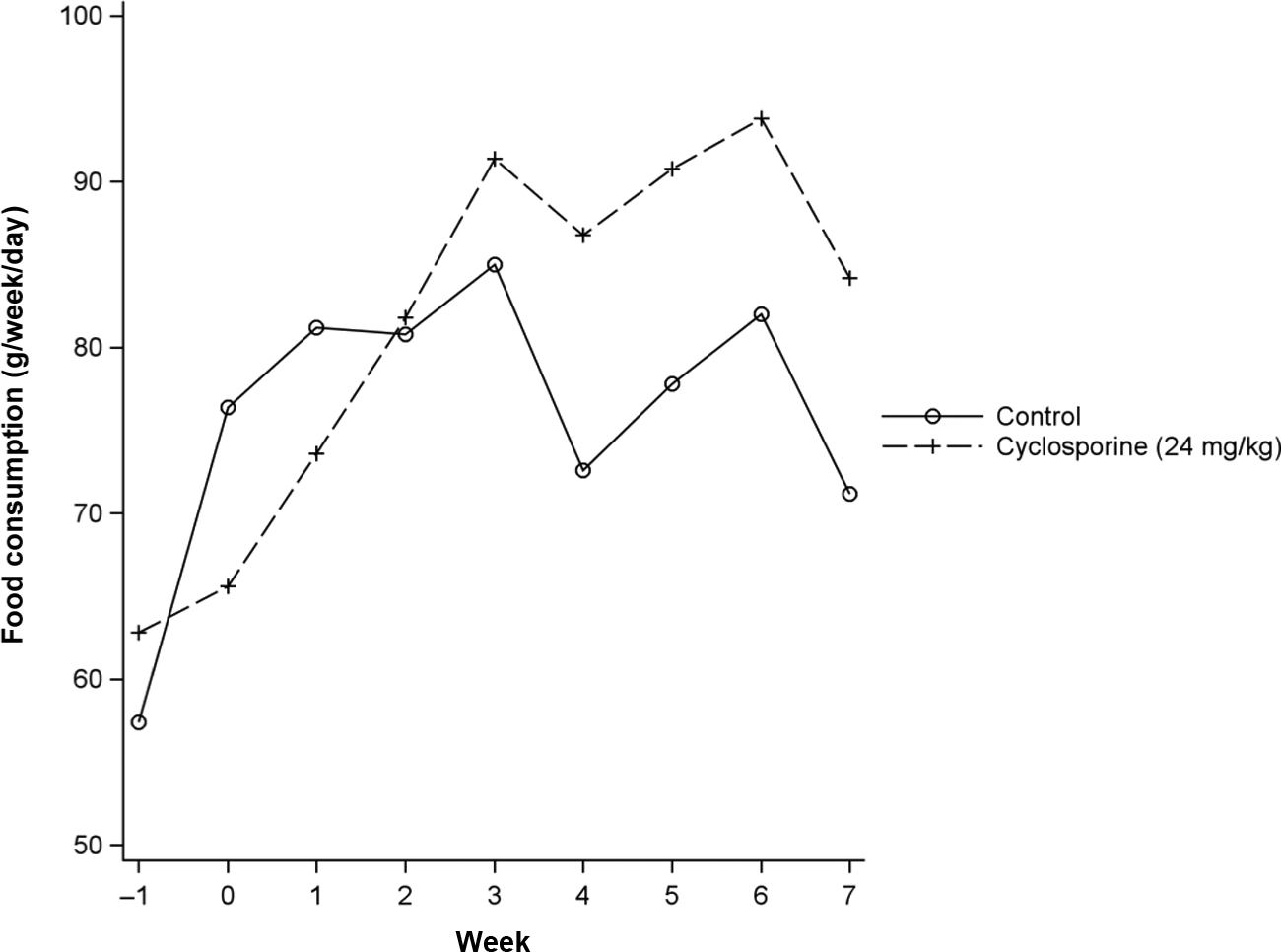
Treated cats consumed significantly less (P ⩽0.004) food than control cats prior to and for the first week of treatment, and they consumed more (P ⩽0.0625) food than control cats for the final 4 weeks of treatment. An apparent decrease in food consumption was noted in both the treated and control groups, for both sexes, in week 4 following vaccination on day 28
There was a significant treatment-by-sex interaction for body weight change (P <0.0001). There was also a significant decrease in weight for treated males compared with control males (P <0.0001), while there was no significant difference in weight change within the female study population (P = 0.2223). Following 8 weeks of treatment, treated males weighed 14.0% less than controls, while treated females weighed 5.7% more than controls (Table 1).
Mean body weight (BW) change

All means are arithmetic group means
Clinical pathology
The clinical pathology data were generally unremarkable and similar among both groups for the pre-study samples collected, indicating that all cats were clinically healthy. The overall group least square mean values remained within the laboratory normal reference ranges for all variables assessed.
Absolute lymphocyte counts were transiently increased in treated cats on day 7 relative to controls, but by day 28, lymphocyte counts were significantly lower in treated cats compared with control cats, as were eosinophils (Table 2). Treated cats also demonstrated elevated mean values for red blood cells, hemoglobin, hematocrit, neutrophil counts, fibrinogen counts and APTT values. Treatment-associated alterations in serum biochemistry were generally of small magnitude and considered to be of little biological importance. However, treated cats had significantly higher glucose, cholesterol, creatinine and blood urea nitrogen, and significantly lower total protein than control cats. While urinalysis results were also similar between groups at the different sampling time points, overall the pH and total volume were significantly different. Further, transient glucosuria was observed in five cats (two control cats and three treated cats), of which two treated cats had concurrent hyperglycemia.
Lymphocyte and eosinophil counts in both treatment groups at all treatment time points

LS = least square
All means are LS mean
Lymphocyte concurrent reference interval: 1.0–9.3 ×103/ml
Eosinophil concurrent reference interval: 0.0–5.0 ×103/ml
Vaccine titers
Response to pretreatment vaccination
All cats responded to the primary pretreatment vaccination courses of FCV, FHV-1 (with the exception of one control cat), FPV, FeLV and rabies. A declining antibody titer was seen before booster vaccination for FCV and FHV-1, with a varying pattern for the other antigens (Figure 2). Although on day 7 treated cats had significantly lower titers for FHV-1 (P = 0.0266) and significantly higher titers for FPV (P = 0.0163) when compared with control cats, there were no significant differences in antibody titers for any antigens (P >0.10) immediately prior to booster vaccination on day 28 (Table 3).

All cats responded to the primary pretreatment vaccination courses of feline calcivirus (FCV), feline herpesvirus (FHV)-1 (with the exception of one control cat), feline panleukopenia virus (FPV), feline leukemia virus (FeLV) and rabies. Following booster vaccination, increases in antibody titers over day 28 levels were observed for (a) FCV, (b) FHV-1, (d) FeLV and (e) rabies in both treatment groups. (c) There was no apparent increase in antibody titer in either group for FPV. All cats were seronegative for feline immunodeficiency virus (FIV) before vaccination on day 28. Following vaccination against FIV, the control cats demonstrated a significant increase in FIV titer between days 35 and 56 when compared with treated cats. (f) No increase was observed from days 28 to 35 in treated cats
Antibody titers following booster vaccination

LS = least square; FCV = feline calicivirus; FeLV = feline leukemia virus; FHV-1 = feline herpesvirus-1; FPV = feline panleukopenia virus
All means are LS mean
P value for treatment main effect; the treatment-by-time interaction was non-significant (P = 0.2627); therefore, no individual timepoint analysis was performed
Response to booster vaccination during treatment
Following booster vaccination, increases in antibody titers over day 28 levels were observed for FCV, FHV-1, FeLV and rabies in both treatment groups (Figure 2a, b, d, e). There was no apparent increase in antibody titer in either group for FPV (Figure 2c). There were no significant differences in post-booster antibody titers between treated and control cats for FCV or FPV at any time point assessed (Table 3). However, control cats demonstrated a more marked increase in titer following booster vaccination to FHV-1, FeLV and rabies than the treated cats. By day 56 the pooled mean titer value for FeLV in treated cats was lower (based on least squares means) than the control cats. However, by day 56, for both FHV-1 and rabies, the pooled titer mean values for the treated group were not significantly different than the control cats.
There were no significant differences in the number of positive biological responders for any antigen, except for FeLV. For this antigen, significantly more cats in the control group were considered positive biological responders over the course of the treatment period (P = 0.0121) and following booster vaccination (P = 0.0121).
Response to novel vaccination during treatment
All cats were seronegative for FIV before vaccination on day 28. Following vaccination, the control cats demonstrated a significant increase in FIV titer (from a pooled mean of 1.0 to 8.5) between days 35 and 56 when compared with treated cats (P <0.0001). No increase was observed from days 28 to 35 (Figure 2f). Treated cats remained at a titer of 1 throughout the study.
There were three negative responders in the control group, while all treated cats were negative responders on day 56. This difference in biological response (change from ‘negative’ prior to vaccination to ‘positive’ following vaccination) titer data was significant (P <0.0001).
Discussion
Although ciclosporin has been used in cats for some time, ciclosporin oral solution (Atopica for Cats) has only recently been approved for the control of FHD in cats of at least 6 months of age and 1.4 kg in body weight.6,16 As allergic dermatitis is a chronic condition, treatment at a dose of 7 mg/kg/day should be given for at least 4–6 weeks, after which the dose regimen can be tapered to maintain the desired therapeutic effect. Given that ciclosporin is an immune modulatory medication, the long-term effect of ciclosporin on vaccine titers is unclear. 17 The results of this study show that even at more than three times the approved ciclosporin dose, cats are able to mount a memory humoral immune response to booster vaccination. However, a primary humoral immune response was not observed within 4 weeks of exposure to a novel antigen.
Few adverse reactions to ciclosporin have been reported in cats, but gastrointestinal signs (diarrhea, vomiting and anorexia) are the most frequently observed reactions.4–6,18–20 Here, following daily administration of more than three times the approved dose over an 8 week period, there was an increased incidence of diarrhea/soft stools, vomiting and salivation in treated cats. There was also a decrease in mean body weight in treated male cats (14% less than controls) and an increase in mean body weight in treated female cats (5.7% greater than controls) over the course of the study. Pooled food consumption was decreased during weeks 0 and 1, with a steady increase through weeks 4–7 relative to controls. In contrast, in a 6 month target animal study, male and female cats dosed with ciclosporin at 24 mg/kg/day gained weight over the course of the study. 19 However, Steffan et al 20 showed that cats had a tendency for slight and temporary weight loss on initial daily dosing at 7 mg/kg for up to 28 days. The weight loss was attributed to the combination of ciclosporin-related gastrointestinal disturbance and the effects on food intake; the majority of cats with weight loss and treated with ciclosporin tended to regain weight, resulting in no overall weight loss by the end of the study (up to 84 days). 20
Treatment with 24 mg/kg/day of ciclosporin resulted in changes in clinical pathology parameters. Some of the changes were consistent with secondary effects of mild diarrhea, such as dehydration (elevated erythrocyte count, hemoglobin and hematocrit) or may have been a result of mild skin inflammation (increased neutrophil and fibrinogen counts), as several cats had skin lesions described as feline acne, which may account for these increased values. The transient change in blood glucose with accompanying glucosuria observed in two treated cats may be stress-related; however, a drug related effect cannot be ruled out. 21 In addition, APTT was prolonged. This has been previously reported in cats treated with at least twice the approved dose of ciclosporin. 19 Prolonged APTT may indicate a coagulation factor deficiency, although it may also increase in liver disease, as the liver is the source of most coagulation factors. 22 It is not clear why APTT was increased in this study; however, all liver parameters measured as part of the clinical pathology assessment were within normal ranges.
The transient increase followed by a decrease in the lymphocyte counts in treated cats, combined with the increased incidence in the number of palpated small peripheral lymph nodes, may reflect immunosuppression following prolonged exposure to ciclosporin. However, no treatment-related changes in lymphocyte counts were reported following 6 months exposure to five times the approved dose of ciclosporin. 19 Wisselink and Willemse 6 actually reported an increase in total white blood cell counts from clinical cases of allergic dermatitis. Clinical samples were taken following 4 weeks of ciclosporin exposure so that their results are more likely due to the pre-existing inflammatory skin condition rather than to ciclosporin. While the differences in lymphocyte count and increased incidence of small peripheral lymph nodes may be incidental, an immunosuppressant effect of ciclosporin cannot be discounted.
The presence of antibodies (FCV, FPV, FeLV, FHV-1 and rabies) prior to vaccination on day 28 confirmed that the included cats were immunocompetent. Cats were able to respond to booster vaccination following 28 days of treatment with more than three times the approved dose of ciclosporin. Although the vaccine titers for FeLV and FHV-1 were significantly lower in the ciclosporin-treated cats for at least one time point post-vaccination compared with controls, the titers remained adequate for protection. Indeed, simply the presence of specific antibodies to FHV-1, FCV or FPV has been suggested to be predictive of resistance to infection. 23 Therefore, while it is reasonable to assume that these cats retained the ability to mount a protective immune response despite exposure to the immunosuppressive effects of ciclosporin, it should be noted that the cats were not challenged with the respective pathogens unequivalently, proving that the titer responses were sufficient. In contrast, ciclosporin-treated cats failed to develop antibody titers to novel FIV vaccination.
Common commercially available vaccines of different preparations (modified live, inactivated/adjuvant and killed) were used in this study. Different vaccine technologies may directly influence efficacy and safety, such that the lack of responsiveness to the FIV antigen (present as a killed viral preparation) in the ciclosporin-treated animals could be due to the vaccine preparation itself. However, the difference in response to booster and novel vaccination in this study was more likely due to the mode of action of ciclosporin. Feline lymphocytes exposed to ciclosporin in vitro produce less IL-2, IL-10, inteferon-gamma, and granulocyte-macrophage colony stimulating factor production. 24 The down-regulation of these pro-inflammatory cytokines, in particular IL-2, inhibits T cell activation and results in the immunosuppressive effects of ciclosporin.8,25 The production of antibodies by B cells in response to novel antigen requires the help of activated T cells, and studies have shown that calcineurin inhibitors affect the humoral immune response by interfering with T-helper signals, but not by targeting B cells directly. 26 Therefore, the suppression of T cell help following exposure to novel FIV antigens in ciclosporin-treated cats likely explains their inability to mount a humoral response to primary vaccination. In contrast, memory B cells are able to produce antibody in response to antigen exposure, independently of T cell help. 27 Therefore, cats that had developed specific immunity before ciclosporin treatment were able to respond to booster vaccination in a T-cell independent manner. The clinical signs of hypersensitivity dermatitis in cats initially appear between 6 months and 3 years of age; therefore, most cats will present in the clinic with signs of allergic dermatitis after they have received their kitten vaccinations and developed a good memory response to vaccination.
Conclusions
In adult cats treated with 24 mg/kg/day of ciclosporin (more than three times the approved dose), vaccine titer levels were adequate for protection following booster vaccination with FCV, FPV, FeLV, FHV-1 and rabies. In contrast, ciclosporin-treated cats failed to mount a primary humoral response to a novel vaccination (FIV). These results suggest that immune response to vaccination during repeated ciclosporin administration depends upon prior exposure to vaccine before starting treatment. Naive cats may not develop protective titers during treatment.
Footnotes
Conflict of interest
All authors are employees of Novartis Animal Health US, Greensboro, NC, USA.
Funding
This work was supported by Novartis Animal Health US, Greensboro, NC, USA.
