Abstract
Chronic myelogenous leukaemia was diagnosed in a 7-year-old male neutered domestic shorthair cat. Leukocytosis (74,900/µl) – mature neutrophilia, eosinophilia and basophilia – was observed. Bone marrow aspiration revealed hypercellularity with proliferation of cells of myeloid lineage. An underlying condition leading to leukocytosis was not identified. The severe leukocytosis did not respond to antibiotic therapy. Based on these findings, chronic myelogenous leukaemia was diagnosed. Because of the absence of clinical signs, the cat was monitored without treatment until 7 months after diagnosis, when it developed pruritic skin lesions. Pruritus was controlled with oral prednisolone. Forty-two months after diagnosis, the cat developed nasal lymphoma, which was treated with radiation therapy, resulting in complete remission. The cat was still in good physical condition 63 months after diagnosis, despite the persistence of marked neutrophilia, eosinophilia and basophilia.
Case Report
Chronic myelogenous leukaemia (CML) is characterised by clonal expansion of granulocytes in peripheral blood and bone marrow. CML is thought to arise from a multipotent haematopoietic stem cell and is categorised as a chronic myeloproliferative disease (CMPD) according to the World Health Organization classification. 1 Although there have been several reports of other CMPDs, such as polycythaemia vera and essential thrombocythaemia in cats, to date, there is no published report on CML in cats.2–4 In this report, we describe the case of a cat with marked granulocytosis persisting for longer than 5 years, without any specific underlying disease.
A 7-year-old male neutered domestic shorthair cat was presented to a primary care hospital because of acute vomiting and decreased appetite. Complete blood count (CBC) revealed marked leukocytosis (112,600/μl), but other values for CBC and blood chemistry were within reference intervals. The cat was treated with supportive therapy and the clinical signs resolved. The cat was examined 3 weeks later, and it still showed marked leukocytosis (107,100/μl) but was asymptomatic. The cat was referred to the Veterinary Medical Center of the University of Tokyo for further investigation. At the time of physical examination, the cat was alert, responsive and moderately obese (body weight: 5.75 kg; body condition score: 4/5).
CBC measured using a multiple automated haematology analyser (pocH-100iV diff; Sysmex) revealed marked leukocytosis (74,900/μl) (Table 1). Other haematological parameters were within the normal reference intervals (Table 1). Cytological analysis of the peripheral blood smear showed the leukocytosis to consist of neutrophilia (62,000/μl), monocytosis (2000/μl), eosinophilia (7700/μl) and basophilia (200/μl). The majority of neutrophils were mature and well-segmented (Figure 1a). Occasional vacuolated monocytes were seen. Eosinophils were mature, well-granulated and slightly larger than normal (Figure 1b). Basophils were larger than neutrophils and contained bluish cytoplasmic granules that varied in size and number (Figure 1c). A small number of immature myeloid cells corresponding to neutrophilic myelocytes, promyelocytes and myeloblasts were observed (Figure 1d). Occasional large platelets were seen.

Cytological findings in peripheral blood collected from a cat with chronic myelogenous leukaemia. Peripheral blood smear at initial presentation at × 40 objective (a) and × 100 objective (b–d). (a) Peripheral leukocytes were mainly mature neutrophils. (b) Eosinophils were abundant with granules. (c) Basophils (arrows) contained bluish cytoplasmic granules of various size and numbers. (d) Immature myelocytes were occasionally seen. (a–d) Wright–Giemsa stain. Bars = 25 μm (a), 10 μm (b–d)
Blood biochemical profile, analysed using an automated biochemical analyser (DriChem 7000 V; FUJIFILM) showed values within the reference intervals (Table 1). Tests for feline leukaemia virus antigen and feline immunodeficiency virus antibody were both negative (Snap FeLV/FIV Combo Test; Idexx). Diagnostic imaging tests, including thoracic and abdominal radiographs and abdominal ultrasound, were generally unremarkable except for moderate diffuse splenomegaly.
For bone marrow examination, general anaesthesia was introduced by an intravaneous injection of propofol (6 mg/kg; Propofol, Maruishi) and maintained by isoflurane inhalation after premedication with an intravaneous injection of butorphanol (0.1 mg/kg; Vetrophale, Meijiseika). Cytological analysis of bone marrow aspirates from the femur and the humerus revealed hypercellularity with proliferation of cells of myeloid lineage, resulting in an increased myeloid to erythroid ratio (6.17) (Figure 2a,b; Table 2). The proportions of eosinophilic and basophilic precursors appeared increased (Figure 2c). The number of megakaryocytes was adequate. Neutrophilic granulocytes were shown to be positive for myeloperoxidase (Figure 2d). Cellular atypia suggesting dysplastic change was not apparent in any of the cell lineages.

Cytological findings of the bone marrow collected from a cat with chronic myelogenous leukaemia. Bone marrow smear at initial presentation at × 20 objective (a) and × 100 objective (b–d). (a) Bone marrow was hypercellular. (b) Increase in number of cells of granulocytic lineage was observed. (c) Increased number of eosinophilic (arrow) and basophilic precursors (arrowhead). (d) Most neutrophilic granulocytes were positive for myeloperoxidase. (a–c) Wright–Giemsa stain. (d) Myeloperoxidase stain. Bars = 25 μm (a), 10 μm (b–d)
Myelograms obtained by bone marrow aspiration

NA = not available
The reference intervals are based on
To exclude the possibility of reactive leukocytosis secondary to bacterial infection, the cat was treated with amoxicillin (17 mg/kg, PO, q12h; Pacetocin, Kyowa) and orbifloxacin (3.5 mg/kg, PO, q24h; Victas, DS Pharma Animal Health) for 2 weeks. However, the leukocyte count remained high (70,200/μl) after antibiotic therapy, which was, therefore, discontinued. As no underlying cause for the leukocytosis could be identified, CML was diagnosed. Although marked leukocytosis persisted, the cat was asymptomatic. Thereafter, monthly evaluations of physical status and CBCs were performed.
During the follow-up period, the total leukocyte counts ranged between 68,000 and 126,700/μl. Eosinophil counts ranged from 4200–22,000/μl, while basophil count remained below 1000/μl until 16 months after diagnosis, after which it gradually increased, varying between 1000 and 12,700/μl until 63 months after diagnosis (Figure 3). Packed cell volume (range 33–49%), haemoglobin concentration (12.0–15.0 g/dl), total red cell count (5.88–8.81 × 106 cells/µl) and platelet count (350–800 × 103 cells/µl) in each examination were generally within reference intervals.
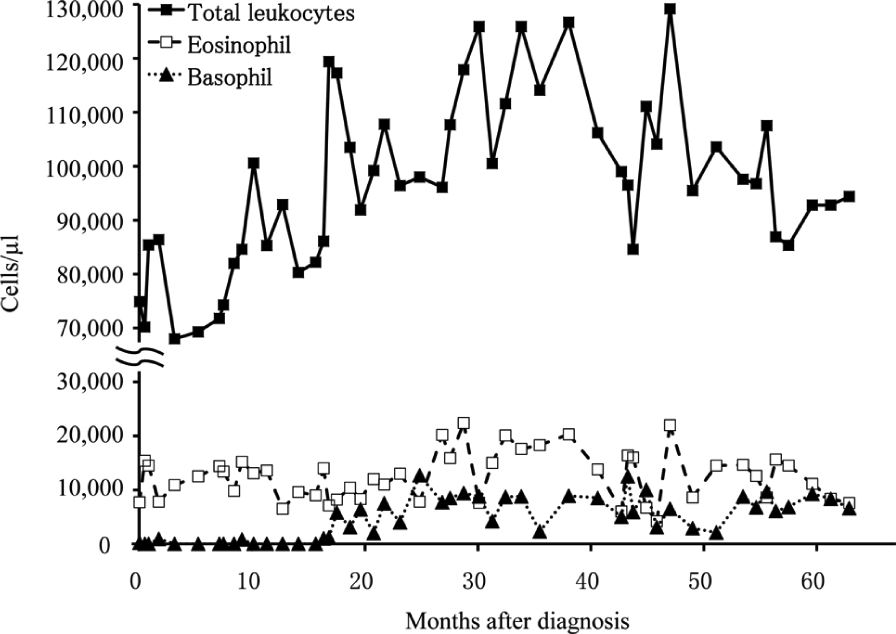
Sequential counts of total leukocyte (black square), eosinophils (white square) and basophils (black triangle) in a cat with chronic myelogenous leukaemia
The cat developed intense pruritus 7 months after diagnosis. Physical examination revealed multifocal areas of epidermal erythema and exfoliation around the face, neck and limbs. Scraping of the skin lesions with erythema and hair loss revealed cornified keratinocytes and leukocytes microscopically. The majority of the leukocytes were neutrophils; however, some eosinophils and basophils were also observed. Most of the leukocytes did not show degeneration, and bacteria or fungi were not apparent. No ectoparasite was found. Thus, the cat was treated with prednisolone (0.8 mg/kg, PO, q24h; Predonine, Shionogi) for 2 weeks on a trial basis. The skin lesions and pruritus resolved, and the dosage was tapered and discontinued 4 months later. Because of the recurrence of skin lesions, prednisolone treatment was re-instated (0.4–0.8 mg/kg/day), which helped control the skin condition until 63 months after diagnosis (time of report).
To investigate the cause of pruritus, we examined plasma histamine concentration using a histamine enzyme-linked immunosorbent assay kit (Immunotech; Medical & Biological Laboratories), which is used for measurement of histamine concentrations in dogs.5,6 Blood from three healthy blood donor cats was used as control. These blood samples were taken for routine blood testing and the excess blood was utilised. Plasma histamine concentration was 18.4 ng/ml and 9.7 ng/ml at 35 and 38 months after diagnosis, respectively, which was higher than levels in the controls (1.37, 1.78 and 2.44 ng/ml).
Forty-two months after diagnosis, the cat developed epistaxis that did not respond to antibiotic therapy. Computed tomography scan revealed a mass in the nasal cavity and destruction of the nasal septum. Histological analysis showed that the mass consisted of necrotic tissue and a sheet of neoplastic lymphocytes with polygonal nuclei and scarce cytoplasm. The mass was diagnosed as nasal lymphoma. Further staging by thoracic and abdominal radiography and abdominal ultrasound identified no evidence of metastasis. The cat received radiation therapy with 4 MV photons, with a total dose of 36 Gy, given in weekly 6 Gy fractions for 6 weeks. The epistaxis resolved a few weeks after the initiation of therapy. The cat was still in good physical condition and showed no evidence of recurrence at the time of publication.
CML has rarely been reported in the field of veterinary science, possibly because the disease is poorly characterised and difficult to diagnose owing to the lack of methods available to show clonal proliferation of proliferating leukocytes. Therefore, CML can only be diagnosed after careful exclusion of other causes of leukocytosis, such as inflammation, immune-mediated diseases or other neoplasias. 7
This cat developed epistaxis 42 months after diagnosis of CML and was then found to have nasal lymphoma. As there is no genetic marker for CML in cats, its diagnosis can be made after ruling out possible underlying diseases causing leukocytosis. Paraneoplastic neutrophilia has been reported in lymphoma. Thus, there might be a possibility that the cat already had occult nasal lymphoma before the diagnosis of CML. However, as nasal lymphoma is generally locally invasive, it was unlikely that such type of lymphoma persisted for more than 3 years without any clinical signs. Moreover, the remarkable leukocytosis persisted after the achievement of complete remission of the nasal lymphoma. For these reasons, we conclude that the cat could be diagnosed with CML, irrespective of the subsequent development of lymphoma.
Human CML is characterised by the presence of reciprocal translocation between chromosomes 9 and 22, which produces the so-called Philadelphia chromosome, resulting in creation of the
Human CML is divided into three phases: chronic, accelerated and blast phases, with most patients being diagnosed in the chronic phase. Consistent with the haematological findings in this case, clinical signs of this condition in the chronic phase in humans are vague and non-specific; the clinical signs include anorexia, easy fatigability and weight loss, and peripheral blood features include mature neutrophilia with eosinophilia and basophilia.8,10 Although anaemia is a common finding in human CML, the cat did not present with anaemia during the observation period of more than 5 years. Without chemotherapy, progression into the accelerated phase and, finally, the blast phase is inevitable in most human cases of CML. The blast phase is defined as a condition with presence of >20% blast cells in bone marrow or peripheral blood, large clusters of blasts in bone marrow or extramedullary disease. 11
Skin lesions with pruritus are a common finding in human CMPDs because of the overproduction of basophils and blood histamine. 12 In this cat, the histamine concentration was much higher than that in the healthy controls, possibly associated with basophilia and leading to intense pruritus.
In this case, severe leukocytosis continued for more than 63 months with no evidence of blast crisis. The fundamental abnormalities characteristic of CML did not change and the cat seemed to remain in the chronic phase. Continued collection and publication of data from similar cases is needed to characterise feline CML.
Footnotes
Funding
This study was supported by the Japan Society for the Promotion of Science, KAKENHI 23381082.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.

