Abstract
Rationale:
This Report was developed by the Feline Vaccination Advisory Panel of the American Association of Feline Practitioners (AAFP) to provide practical recommendations to help clinicians select appropriate vaccination schedules for their feline patients based on risk assessment. The recommendations rely on published data as much as possible, as well as consensus of a multidisciplinary panel of experts in immunology, infectious disease, internal medicine and clinical practice.


Introduction
The AAFP produced the first organization-driven vaccination guidelines in 1998. These were updated in 2000 and again in 2006. 1 Each version has offered a comprehensive review of the literature and has provided recommendations for vaccine protocols based on known science along with some extrapolation between studies and between species when feline studies were not available. This Report has used the same criteria.
The practicing veterinarian is in the best position to determine how to put these Guidelines into practice for an individual patient. The veterinarian should undertake a clinical risk/benefit assessment for each animal and discuss recommended vaccination schedules with the owner so that they can make an informed choice. The assessment should include discussion on the likelihood of exposure, the health and lifestyle of the animal, and the risks related to vaccination.
The Advisory Panel recognizes that situations differ in different countries, and that every country will have slightly different issues and priorities; thus these Guidelines will not necessarily be applicable to every country and the practitioner must interpret accordingly.
The three international panels that have produced feline vaccination guidelines (AAFP, World Small Animal Veterinary Association and European Advisory Board on Cat Diseases) recommend that an annual health examination be performed irrespective of whether vaccines are administered. While the optimal frequency of health examinations for cats is unknown, it is generally accepted that healthy adult cats should be examined at least once a year. In the past, annual veterinary visits were structured around vaccinations as the primary focus. With the increasing body of knowledge about duration of immunity (DOI) from vaccinations, their potential adverse effects, and the increased awareness of pet owners about these issues, it is clear that vaccination no longer justifies the need for annual visits.

Practitioners are encouraged to help cat owners understand the value of regular health care and that it ideally should be proactive rather than reactive. A useful approach is for health care to be tailored to the various feline life stages, which improves early recognition of potential health-related issues and can facilitate treatment. 2
A Pet Owner Guide, discussing the risks and benefits of vaccination, is included as Appendix 2 (pages 807 and 808).
Vaccination principles
Vaccination plays an important role in the control of infectious diseases, both for an individual as well as for the cat population (ie, herd health). Some vaccine antigens are also used to lessen the potential for zoonotic spread of disease (eg, rabies). The benefits of routine and widespread vaccination are clear: the incidence of serious disease caused by highly pathogenic organisms, such as feline parvovirus (panleukopenia), can be reduced in populations in which widespread vaccination is practised. However, the level of protection conferred by a particular vaccine in an individual patient varies. The quality of vaccine-induced immunity in any patient is influenced by a complex interaction of factors unique to the individual patient, the patient’s environment, and the nature of the vaccine and pathogen. Precisely predicting either the outcome of vaccination or subsequent exposure to a pathogen is difficult (or impossible) and, therefore, vaccination should never be offered as a guarantee of protection.
The risk of infection and subsequent development of disease varies with a number of factors including the age and health of the cat, magnitude of exposure to the infectious agent, the pathogenicity of individual agents, the geographic prevalence of infection and the vaccination history of the cat. Some of the factors that negatively affect an individual animal’s ability to respond to vaccination include interference from maternally derived antibodies (MDA), congenital or acquired immuno- deficiency, concurrent disease or infection, inadequate nutrition, immunosuppressive medications, chronic stress and an aging immune response. Additionally, some vaccinal agents (eg, FPV) will induce a much stronger protective immune response than others (eg, feline herpesvirus [FHV-1]). As vaccine-afforded protection against both infection and disease is thus variable and not absolute, exposure to infected animals and infectious agents should be minimized, even after vaccination.

Kittens are generally more susceptible to infections than adult cats are and typically develop more severe disease (Figure 1). Thus, they represent a principal primary target population for vaccination. As part of a routine health care program, the vaccination needs of all cats, including adults, should be assessed at least once a year, in conjunction with a comprehensive physical examination and consultation, modifying vaccination recommendations as necessary on the basis of altered risk/benefit ratio.

Kittens are more susceptible to infection than adult cats are, and are a principal primary target population for vaccination. Courtesy of Dr Deb Givin

Vaccination is a medical procedure, and the decision to vaccinate, even with core vaccines (see box above), should be based on a risk/benefit assessment for each cat and for each vaccine antigen. Vaccination may indeed be beneficial, but it is not innocuous, and the benefit of vaccinating an animal (eg, the induction of clinically meaningful immunity) must be balanced against the risk of adverse events, likelihood of exposure and severity of disease. Where practical, every effort should be made to ensure that cats are healthy prior to vaccination; however, concurrent illness should not necessarily preclude vaccination.
The overall objectives of vaccination are shown on the right.
General information on types of feline vaccines
Vaccines, including different products licensed to protect against the same pathogen, are not necessarily alike. Different vaccine technologies may directly influence efficacy, safety, DOI and route of administration of individual products. Awareness of fundamental differences is necessary.

The following terminology is used throughout these Guidelines to describe types of vaccines: inactivated (killed), modified-live (attenuated) and recombinant. The attributes of each vaccine type are summarized in Table 1.
Examples of different types of feline vaccines and their attributes
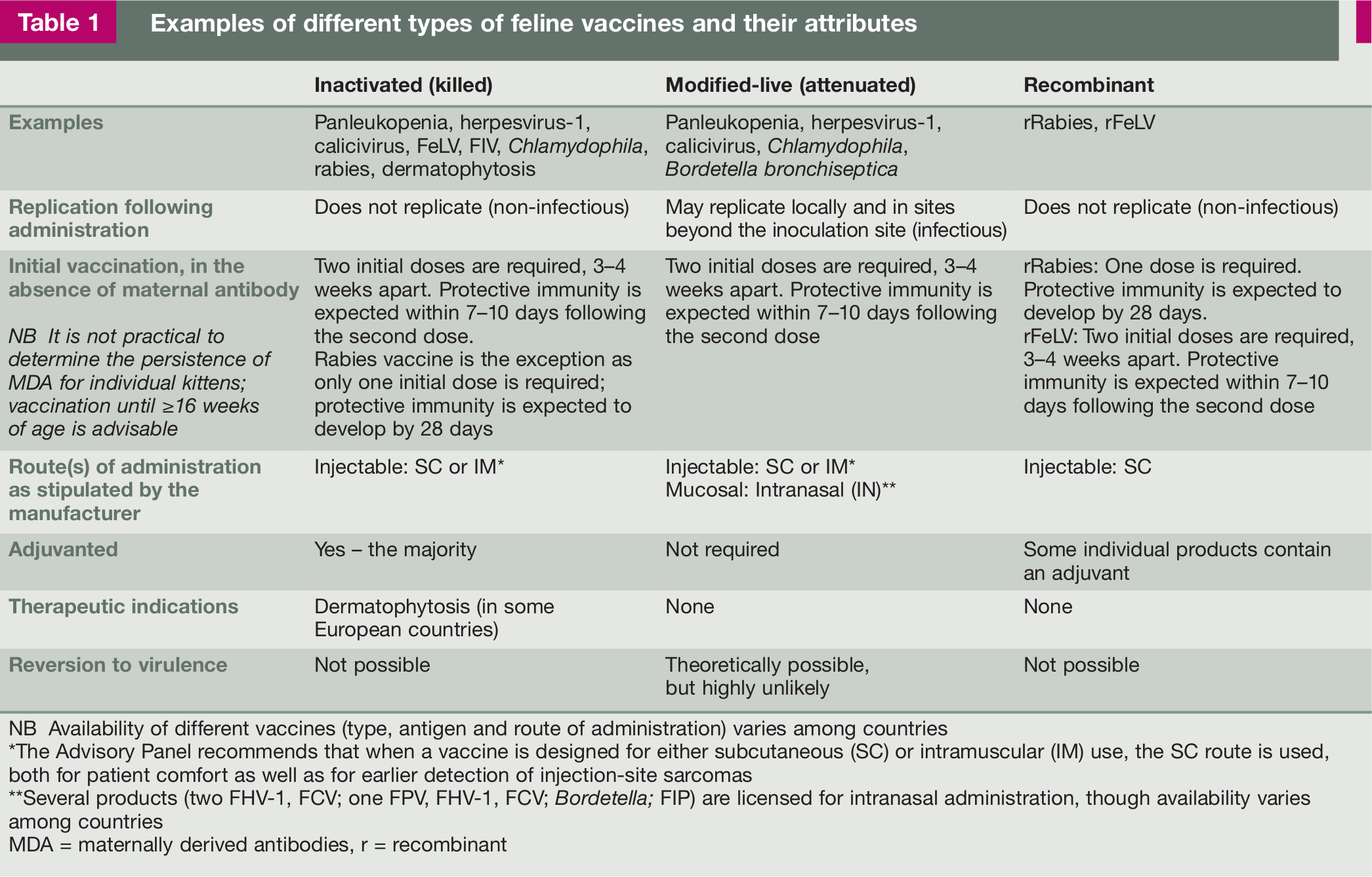
NB Availability of different vaccines (type, antigen and route of administration) varies among countries
The Advisory Panel recommends that when a vaccine is designed for either subcutaneous (SC) or intramuscular (IM) use, the SC route is used, both for patient comfort as well as for earlier detection of injection-site sarcomas
Several products (two FHV-1, FCV; one FPV, FHV-1, FCV; Bordetella; FIP) are licensed for intranasal administration, though availability varies among countries
MDA = maternally derived antibodies, r = recombinant
Characteristics of vaccine types have been reviewed as recently as 2011. 3 All veterinary vaccines, prior to licensing, are subjected to testing for efficacy, safety, potency and purity. Testing methods may vary among different manufacturers and licensing authorities. While all licensed vaccines need to meet minimum efficacy standards, the level of protection induced can vary depending on many factors, including the method used to manufacture the product. For further information on licensing, readers should refer to the 2006 Guidelines (see box on page 786) and to individual licensing authorities (United States Department of Agriculture [USDA]; Canadian Food Inspection Agency [CFIA]; Veterinary Medicines Directorate [VMD], Department for Environment, Food, and Rural Affairs [DEFRA], UK; European Medicines Agency [EMA], EU).
The principal differences between inactivated, modified-live and recombinant vaccines are discussed below.
Risk/benefit assessment
In assessing the risk for an individual cat, information about the cat, the environment and infectious agents to which the cat will be realistically exposed needs to be considered. Specifically, questions need to be asked that address the cat’s lifestyle as well as the lifestyle of any other cats in the same household. Queries should also be posed regarding other sources of exposure, such as excursions outside the home, boarding and travel.
Patient
Age is an important element in assessing an individual’s risk profile. Most infectious diseases are more prevalent in kittens, and kittens less than 6 months old are generally more susceptible to infection and disease than adult cats are. Kittens, therefore, represent a principal primary target population for vaccination.
MDA provide important protection for the kitten, but may also interfere with, or neutralize, vaccines. As the level of MDA varies among individuals, the age at which a kitten may be able to respond to vaccination will also vary, and in some cases may be 16 weeks or older. While information is available on the variability of MDA as pertains to FHV-1, FCV and FPV, limited data is available for other antigens; thus the role of MDA in interference with vaccination against rabies, FeLV or other pathogens is unknown. Stopping a vaccination course too early (when MDA are still interfering) is thought to be the single most common cause of vaccination failure in kittens.
Patient’s environment
Population density and opportunity for exposure to other cats (eg, whether the cat is free-roaming or has access to the outdoors) are among the most critical issues affecting risk of exposure to an infectious agent. Cats and kittens living in multiple-cat households and environments (eg, boarding, breeding, foster or shelter facilities) are likely to have a substantially higher risk of infection than are cats living indoors in one- or two-cat households. Furthermore, the introduction of new cats into a household poses a potential risk – not only to the cat entering the household, but also to the whole group because of possible exposure to new infectious agents. The immunosuppressive effects of stress inherent in the change of social demographics may also result in recrudescence and an increased susceptibility to infection and disease. Conversely, cats that are naturally exposed to infectious agents after vaccination may have an opportunity for ‘natural boosting of immunity’ that may not be afforded to cats kept alone.
Indoor cats generally have a low risk of exposure to infectious agents, particularly where the agent in question is only transmitted by direct contact among cats. However, they may also be exposed to infection from other cats in the household (ie, subclinically infected or carrier cats), or by indirect transmission of pathogens brought in from outside on owners’ clothing, shoes, etc. In theory, strictly indoor cats may be more susceptible to developing panleukopenia because they do not receive boosting through the possibility of natural exposure. It is important to ask owners about other exposure that indoor cats may have, such as supervised visits out of doors (eg, on harness/leash, in the garden, etc), visiting other cats in an apartment building, balconies or roof gardens, visiting cats that belong to other family members, and staying in boarding facilities. Fostering shelter cats alters the risk for the resident cats, both through potential direct exposure to infectious agents as well as through stress-induced immunosuppression.
Trap–neuter–return (TNR) and other special situations are discussed on pages 791–794.
Geographic distribution of infectious agents may result in substantially different risks of exposure for cats living in different areas (eg, rabies). Questions regarding future travel should be included in determining the risk of exposure to specific infectious agents. Periodic housing in boarding facilities, shelters or breeding facilities or other multiple-cat households also places cats at increased risk of exposure to a variety of infectious agents, although the risk will vary substantially between different situations.
Infectious agent
Independent agent-associated variables, such as virulence, strain variation and mutation, challenge dose and stability in the environment, influence the outcome of infection. These are difficult to assess objectively.
Recommendations for vaccination of household pet cats
Developing universal guidelines for vaccination of household pet cats is complicated by the lack of a clear definition of what is, and what is not, a ‘pet cat’. What follows are reasonable recommendations, based on scientific evidence and expert advice, applicable to most cats presented to private practitioners. Differences in cat population density, introduction of new cats, and exposure risk are dynamic variables that the veterinarian must take into consideration when recommending any vaccine for any cat. It is advised that veterinarians reassess risk factors for exposure to infectious disease at each visit (at least once a year), as changes in factors such as the health of the animal or its lifestyle may dictate changes to vaccination needs.
Table 2 summarizes vaccination recommendations for household pet cats.
Recommendations for vaccination of household pet cats

Additional considerations when vaccinating household pet cats
Because vaccination requirements and risk of exposure to infectious agents vary among household pet cats, individual vaccination protocols will vary. The following recommendations address some alternative situations and offer insights on vaccination of pet cats using non-core vaccines.
Pet cats that spend most (or all) of their lives outdoors are at greater risk of exposure to most infectious diseases compared with predominantly indoor pet cats (Figure 2). Offsetting this is the natural boosting of immunity they may receive if they are exposed to infectious agents. Among outdoor adult cats, exposure risk for rabies, FeLV and FIV is generally higher than for indoor cats. In addition to the conventional vaccines recommended in Table 2, FIV vaccination could be considered for outdoor cats. (See accompanying Disease Information Fact Sheet on FIV – details on page 799.)

Pet cats that spend any time outdoors are at greater risk of exposure to many infectious diseases compared with indoor-only pet cats. Images courtesy of Dr Terry Curtis (a), Dr Margie Scherk (b) and Karen James (c)
Recommendations for vaccination of shelter-housed cats
Generally, shelter-housed cats (Figure 3) can be considered to be at especially high risk of exposure to infectious disease. Endemic disease, high rates of turnover, stress and sustained exposure are contributing factors. Vaccination in shelters should be limited to those diseases that are likely to be transmitted within the shelter itself. For diseases of concern in shelters (notably FPV and upper respiratory infections), vaccines may be indicated at an earlier age, and be administered at shorter intervals compared with schedules for pet cats. Rapid onset of protection is critical; therefore, administration of FPV, FHV-1, FCV vaccines should be considered for all cats at the time of (or ideally, before) intake.

Generally, cats in shelters, whether individually (a) or group (b) housed, are at especially high risk of exposure to infectious disease. Images courtesy of UC Davis Koret Shelter Medicine Program Team Members
Table 3 summarizes vaccination recommendations for shelter-housed cats.
Recommendations for vaccination of shelter-housed cats
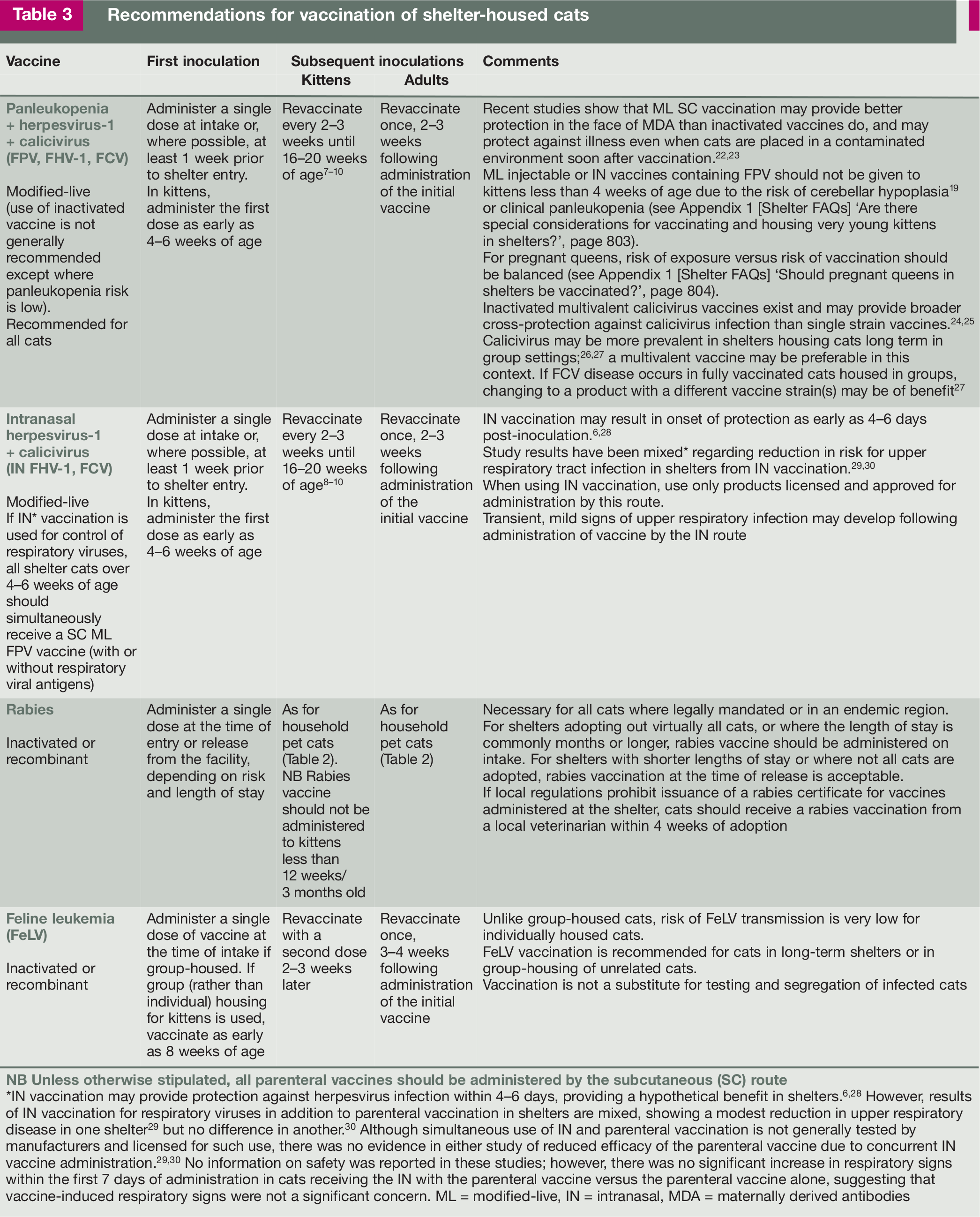
IN vaccination may provide protection against herpesvirus infection within 4–6 days, providing a hypothetical benefit in shelters.6,28 However, results of IN vaccination for respiratory viruses in addition to parenteral vaccination in shelters are mixed, showing a modest reduction in upper respiratory disease in one shelter 29 but no difference in another. 30 Although simultaneous use of IN and parenteral vaccination is not generally tested by manufacturers and licensed for such use, there was no evidence in either study of reduced efficacy of the parenteral vaccine due to concurrent IN vaccine administration.29,30 No information on safety was reported in these studies; however, there was no significant increase in respiratory signs within the first 7 days of administration in cats receiving the IN with the parenteral vaccine versus the parenteral vaccine alone, suggesting that vaccine-induced respiratory signs were not a significant concern. ML = modified-live, IN = intranasal, MDA = maternally derived antibodies
Additional considerations when vaccinating shelter-housed cats
Recommendations for vaccination of cats in trap–neuter–return programs
Most community cats (Figure 4, ie, free-roaming unowned feral and stray cats) lack protective antibody titers against FPV, FHV-1 and rabies.9,35 In one study, the vast majority of feral cats vaccinated once at the time of TNR surgery developed protective antibody titers against FPV and FCV by the time they were re-trapped for testing 2–3 months later, regardless of whether inactivated or modified-live vaccines were used. 35 In contrast, only inactivated vaccines resulted in a high rate of protective antibodies against FHV-1. 35

Community cats in TNR programs (a,b) should receive FPV, FHV-1, FCV and rabies vaccines at the time of surgery. Images courtesy of International Cat Care
In the same study, nearly all cats developed high antibody titers against rabies after a single dose of inactivated rabies vaccine. 35 Vaccine licensing studies have demonstrated 3–4 year DOI following a single vaccine administered to laboratory kittens. This suggests that, while the first rabies vaccine may only be recognized by regulatory agencies as valid for a single year, it is likely that vaccinated cats are protected for much longer.
It is the recommendation of the Advisory Panel that cats in TNR programs receive FPV, FHV-1, FCV and rabies vaccines at the time of surgery.
Recommendations for vaccination of cats housed in breeding catteries
Breeding catteries are variable in size, population and the nature of available facilities. The cat population may number less than 10 individuals or more than 50. Cats of various ages and life stages are typically present and many catteries continue to house retired breeding individuals that have been neutered. Some also contain household pets that may or may not have access to outdoors. The facilities may be sophisticated enough to allow for segregation of subpopulations or all individuals may be housed together. Generally, the medical and vaccination history of the residents is well known, but some diseases, such as upper respiratory tract disease, may be endemic.
Vaccination programs should be limited to those diseases that are relevant to the cattery and should be determined by analysis of risk factors. When assessing the level of disease risk in catteries, factors to consider include:
Rate of population turnover.
Population size and density.
Number of litters/year (Figure 5).
Presence of endemic disease.

The number of litters produced per year in a breeding cattery is one of the key factors in determining the level of disease risk, and devising an appropriate vaccination program. Courtesy of Betsy Gaither
Transmission of infectious diseases is facilitated by group living, young kittens mixing with older kittens and adults, contact during mating, introduction of new cats, and movement of cats into and out of the cattery (eg, queens going to other catteries for breeding, return of previously sold cats, travel for cat shows or other exhibitions). Catteries assessed as low risk would be considered similar to pet homes (Table 2), whereas catteries assessed as high risk would be considered similar to shelters (Table 3), pet stores, etc. In high-risk environments, vaccines may be used at an earlier age than in pet cats, particularly for control of endemic upper respiratory tract disease.
In general, vaccination may be started at an earlier age than in the pet cat population and revaccination intervals may be shortened. Breeders should be encouraged to work with a veterinarian to develop a comprehensive wellness program that includes appropriate vaccinations for their specific situation. Vaccination records should be kept for each individual in the cattery that include all relevant information (eg, antigen, brand, date, vaccination site, adverse events, etc). Management and husbandry have an important impact on the health of individual cats in catteries. Relevant references and resources should be consulted.36,37
Table 4 summarizes vaccination recommendations for cats in breeding catteries.
Recommendations for vaccination of cats in breeding catteries

ML = modified-live, IN = intranasal, MDA = maternally derived antibodies
Additional considerations when vaccinating cats in breeding catteries
Vaccine adverse events
Although the administration of biological products can never be entirely free of risk, in general currently available feline vaccines have an excellent safety record. It is important to report any known or suspected negative events associated with vaccination, recognizing that a temporal relationship between an event and vaccine administration does not necessarily imply causality. In the United States, veterinarians are requested to contact the manufacturer (Veterinary Technical Services) of the vaccine(s) considered to be involved; veterinarians may also report known or suspected adverse events directly to the US Department of Agriculture. In other countries procedures may vary, but, in general, veterinarians should contact the manufacturer and notify the appropriate regulatory agency to report a vaccine adverse event (eg, the Canadian Centre for Veterinary Biologics [Canada]; the Veterinary Medicines Directorate [UK]; the European Medicines Agency [EU]. (See Appendix 1 [Adverse Event FAQs] on page 805 for specific reporting forms and instructions.)
The most commonly reported vaccine reactions are lethargy, anorexia and fever for a few days after vaccination, or local inflammation at the site of injection.4,42,43 Rarely anaphylaxis is seen. Because vaccines are biologically active products, occasional adverse reactions associated with vaccination are inevitable. It should be recognized, however, that establishing causality is often difficult, especially if the suspected reaction is delayed (days or weeks). 43

Prevalence and type of adverse reactions
Although post-vaccinal adverse events in cats are considered rare, the true prevalence is likely to be underestimated due to under-reporting by both veterinarians and owners. 44 In the most substantial survey to date, adverse reactions were reported for all cats presented to Banfield Pet Hospitals in the United States between 2002 and 2005. 4 During this period, more than 1.25 million doses of various vaccines were administered to nearly 0.5 million cats. Adverse reactions within 30 days of vaccination were reported at a rate of 51.6/10,000 cats vaccinated (0.52%), with 92% of these reactions occurring within the first 3 days. Clinical signs described for 1699 of 2560 cats with vaccination-associated adverse events included lethargy (± pyrexia) in 54%, local pain or swelling at the vaccine site (25%), vomiting (10%), facial or periorbital edema (6%) and generalized pruritus (2%). Death was reported in four cats, and in at least two of these it was attributed to anaphylaxis.
Although the vaccines used were predominantly from one manufacturer, no vaccine type was found to be significantly more likely to cause local reactions. Administration of multivalent FPV, FHV-1, FCV and Chlamydophila vaccines was significantly more likely to be associated with lethargy (± pyrexia) than administration of vaccines without the Chlamydophila component. The risk of an adverse reaction was greatest in cats around 1 year of age and/or increased as the number of vaccines administered concurrently increased. 4 In another extensive study specifically investigating local post-vaccine reactions, a prevalence of 0.23% was reported. 45 Previous large studies have suggested adverse vaccine reaction rates of around 1–3%,46–48 but some variation in prevalence can be expected with the use of different products, administration of multiple vaccines at the same appointment, and surveillance methods.

Hypersensitivity reactions
Anaphylaxis and allergic reactions
Anaphylaxis is perhaps the best characterized immune-mediated hypersensitivity (type I) reaction to vaccination, but it is rare (approximately 1–5/10,000 vaccines).4,46 In cats it may manifest as vomiting, diarrhea, respiratory distress, facial or generalized pruritus, facial swelling and collapse.1,43,49
A careful risk assessment is needed when considering the revaccination of cats with a history of anaphylaxis. In cats that have experienced an allergic reaction with true anaphylaxis, revaccination should usually be avoided. Vaccine excipients (inactive ingredients) are thought to cause most type I hypersensitivity reactions. 4 Hence, where revaccination is considered necessary, using a different vaccine formulation and premedicating with an antihistamine and glucocorticoids 20–30 mins prior to vaccine administration is recommended, followed by close observation of the patient for several hours.1,4
Depending on geographic location, the requirement to vaccinate cats for rabies may take precedence over medical considerations. Veterinarians are urged to contact the appropriate authorities to determine what the local status is when concerns arise and whether the individual may be excused from vaccination. (See also 2006 Guidelines, Appendix 1: Certificate of Exemption from Rabies Vaccination – details on page 786.)
Other reactions
While other forms of hypersensitivity reactions (types II, III and IV) almost certainly occur in cats after vaccination, these are rarely documented. Some forms of local reaction probably reflect type IV reactions. Polyarthritis is occasionally seen after FCV vaccination. Rarely it may represent a form of type III reaction, but it is mainly due to co-infection with field virus or vaccine virus itself.50,51 (See Appendix 1 [General FAQs] ‘What is the cause of lameness occasionally seen after FCV vaccination?’, page 802.)
Update on feline injection-site sarcomas (FISS)
Vaccine-associated sarcoma was first recognized as an issue in cats in the early 1990s. While initial studies suggested a risk of sarcoma development in around 2/10,000 doses of vaccine administered, 52 which increased to 13–36/10,000 doses in other studies,53–55 current estimates based on larger epidemiologic studies (published between 2002 and 20074,45,56) suggest that the risk of sarcoma development following vaccination is actually very low (probably well below 1/10,000 doses of vaccine).4,45
Although initial reports linked development of sarcomas at vaccination sites with the use of inactivated rabies 57 or FeLV vaccines, 52 and aluminum-based adjuvants, more recent studies found no relationship between vaccine type, brand or use of inactivated versus modified-live vaccines and the risk of subsequent sarcoma formation.56,58,59 The impact of using the canarypox-vectored rabies vaccine is still unclear. One retrospective study of histopathology samples showed no reduction in the prevalence of FISS after the introduction of this vaccine; however, the types of vaccine used were not reported. 58 In a recently published case control study it was suggested that there may be a lower risk of inducing sarcomas with this vaccine than with other rabies vaccines. 59 Many of these studies have also clearly shown that injections other than vaccines also have the ability to induce sarcoma formation.
No studies have been published that define objective methods for reducing the risk of FISS in individual cats presented for routine vaccination. Based on our current understanding of this problem, it is likely that vaccines are not uniquely implicated in the development of injection site sarcomas in cats.56,60 FISS risk following vaccination likely results from a complex interaction of multiple extrinsic (eg, frequency and number of vaccines administered over time, composition of the injected product, etc) and intrinsic factors (eg, genetic predisposition, tissue response following injection, etc). The presumed relationship between types of vaccine, inflammation at the site of vaccination 61 and subsequent FISS development appears complex at best and, if involved, is likely only one among many factors that contribute to FISS development.

While vaccines are not uniquely implicated in sarcoma formation, there are certain actions that can be taken to reduce the risk of FISS, as summarized in Table 5. Courtesy of Albert Lloret
Table 5 provides a brief review of considerations and management options for the reduction of FISS risk, taken from current publications. None of these suggestions are known to prevent or cure FISS.
Summary of considerations and management options for FISS risk reduction

FISS = feline injection-site sarcoma, SC = subcutaneous, IM = intramuscular
When considering vaccine type, the Advisory Panel recommends that the following be taken into consideration. Recent studies demonstrate that all vaccines carry some risk of inducing FISS, as do at least some other injectable products. Although current information as outlined above does not clearly show differences in risk of FISS development between modified-live and inactivated vaccines, some Advisory Panel members consider that, on balance, risk might be mitigated by the use of modified-live vaccines. There are also other factors that may influence the choice of live versus inactivated vaccines (see Table 6 and Appendix 1 [General FAQs], page 803). Overall, however, the Advisory Panel concluded that, at the current time, there is insufficient information to make definitive recommendations to use particular vaccine types to reduce the risk of FISS.
Indications for the use of inactivated vaccines in cats

ML = modified-live, DOI = duration of immunity
Post-vaccination monitoring
The Advisory Panel recommends that clinicians and their staff instruct clients to monitor the vaccine site for swelling or lumps in order to detect potential sarcomas while they may still be removed successfully. Biopsy of any mass present is warranted if it (a) remains present 3 months after vaccination; (b) is larger than 2 cm in diameter; or (c) is increasing in size 1 month after vaccination (the ‘3-2-1 rule’ – see Table 5). It is recommended that multiple needle biopsies or an incisional wedge biopsy are obtained to reduce the risk of harvesting non-representative biopsy material and to minimize the risk of tracking tumor cells outside of the future surgical field.


Administration of FPV, FHV-1, FCV vaccine subcutaneously below the right elbow. Courtesy of Dr Susan Little

Administration of FeLV vaccine subcutaneously below the left stifle. Courtesy Dr Susan Little
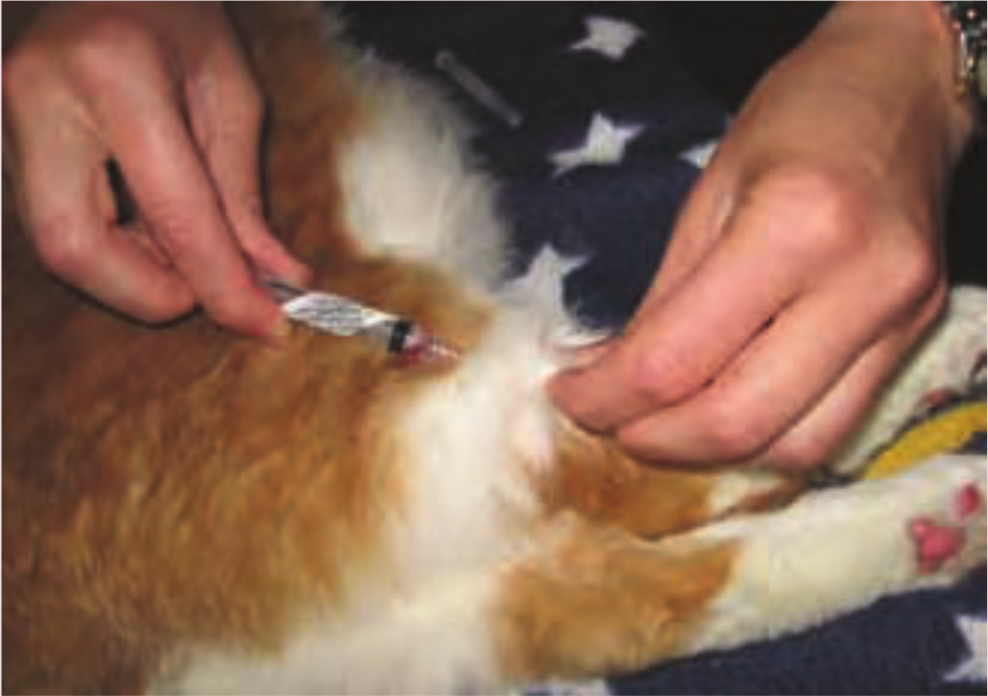
Administration of rabies vaccine subcutaneously below the right stifle. Courtesy Dr Susan Little

Regions indicated in green are recommended. Those in red are key sites that should be avoided. Image ©iStockphoto.com/GlobalP

Legal considerations associated with vaccination
Veterinarians in most countries are permitted to use professional judgment in the selection and use of licensed vaccines. Reference to these Guidelines, therefore, is appropriate when developing vaccination protocols for individual patients even though the guidance may vary from the manufacturer’s label recommendations or data sheet (eg, annual revaccination vs triennial revaccination for core vaccines).
Rabies vaccination of cats
Where rabies vaccination of cats is required, veterinarians may not have discretion to vary from the manufacturer’s recommendations or from requirements set forth by regulatory agencies. Rabies vaccination requirements vary from country to country and can vary significantly within individual countries. In locations where feline rabies vaccination is required by law, veterinarians are obligated to be familiar with and follow legal requirements when administering rabies vaccines. Rabies vaccination recommendations contained in these Guidelines do not constitute vaccination requirements.
Medical record documentation of vaccination
At the time of vaccine administration, the following information should be recorded in the patient’s permanent medical record:
Vaccine(s) recommended for this patient.
Date of vaccine administration.
Identity (name, initials or code) of the person administering the vaccine(s).
Vaccine name, lot or serial number, expiration date, and manufacturer of vaccine(s) actually administered.
Site and route of vaccine administration.
Concurrent medications/therapy.
Recommendations for future vaccinations.
Adverse events should be recorded in a manner that will clearly alert all staff members during future visits. Risks and benefits of vaccination should be discussed with the owner so that they can make an informed choice. Consent should be documented in the medical record to demonstrate that relevant information was provided to the client and that the client authorized the procedure.

Supplemental Material
Click here for Supplementary Material
disease information fact sheet: Feline herpesvirus 1
Supplemental Material
Click here for Supplementary Material
’The immune response to vaccination: a brief review’
Supplemental Material
Click here for Supplementary Material
Pet Owner Guide
Supplemental Material
Click here for Supplementary Material
Pet Owner Guide (high resolution PDF)
Supplemental Material
Click here for Supplementary Material
disease information fact sheet: Feline calicivirus
Supplemental Material
Click here for Supplementary Material
disease information fact sheet: Feline panleukopenia
Supplemental Material
Click here for Supplementary Material
disease information fact sheet: Rabies
Supplemental Material
Click here for Supplementary Material
disease information fact sheet: Feline leukemia virus
Supplemental Material
Click here for Supplementary Material
disease information fact sheet: Feline immunodeficiency virus
Supplemental Material
Click here for Supplementary Material
disease information fact sheet: Feline infectious peritonitis
Supplemental Material
Click here for Supplementary Material
disease information fact sheet: Chlamydophila felis
Supplemental Material
Click here for Supplementary Material
disease information fact sheet: Bordetella bronchiseptica
Footnotes
Appendix
Acknowledgements
The AAFP would like to thank Boehringer Ingelheim for its sponsorship of these Guidelines and for its commitment to helping the veterinary community develop projects that will improve the lives of cats. Sponsorship does not imply endorsement of the sponsor’s products or services by the Association. The content of these Guidelines has been solely created by the Feline Vaccination Advisory Panel members. The sponsor has provided financial support directly to the Association and has had no role in the creation of content.
Conflict of interest
RBF consults with IDEXX Laboratories (Westbrook, ME), Merial Ltd (Duluth, GA) and Elanco (Greenfield, IN). RMG is a co-investigator on a project funded by MSD. KFH runs a shelter medicine residency program at UC Davis sponsored by Boehringer Ingelheim. JKL has worked with various pharmaceutical, vaccine and diagnostic test companies over the years to complete research studies, student and house officer training, to present at veterinary conferences, and to serve on expert advisory panels.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
