Abstract
The aim of this study was to describe a series of confirmed and suspected cases of feline panleukopenia virus (FPV) and in-contact cats in an adoption-guarantee shelter in an FPV-endemic area by reviewing shelter records over a 10-month period (January–October 2010). Cats were divided into three groups: in-contact group – asymptomatic cats that were housed with a FPV fecal antigen (Ag)-positive cat/kitten as part of a litter group (n = 66); FPV-survivors group (FPV-infected survivors) – tested FPV fecal Ag-positive and showed clinical signs of FPV, but survived (n = 27); FPV-non-survivors group (FPV-infected non-survivors) – showed clinical signs of FPV and either tested FPV fecal Ag-positive or were housed with an Ag-positive family member, but did not survive (n = 52). Ages ranged from 3 weeks to 3 years, but most were <6 months old (in-contact group: 79%; FPV-survivors group: 70%; FPV-non-survivors group: 85%). A seasonal peak occurred over summer, but cases occurred year-round. Anorexia, dehydration, fever and diarrhea predominated in the FPV-survivors group, and death was preceded by clinical signs of circulatory shock in the FPV-non-survivors group. Housing litters of kittens with their mother was not associated with improved outcome, perhaps because in this population clinical FPV infection was relatively common in queens arriving at the shelter with susceptible litters.
Introduction
Because of the widespread use of highly effective vaccines against feline panleukopenia virus (FPV), which also promote ‘herd immunity’ through viral shedding, 1 veterinarians commonly hold the opinion that the disease has become less prevalent over the last 20 years. 2 FPV is much less prevalent than 20 years ago and it remains rare in private practice. By contrast, the situation seems quite different in animal shelters, which are home to a continual influx of cats of unknown vaccination status, especially during summer and fall when large numbers of kittens with waning maternal immunity are admitted.3,4 As the incubation period is 2–14 days, exposed cats that are clinically healthy but incubating the infection might not show clinical signs until days after they have arrived at a shelter or an adoptive home. 3 Historical mortality rates have varied from 50% to 90%, and large-scale outbreaks have been continuously reported, resulting in the deaths of thousands of cats.3–13 Because of the highly infectious nature of the disease, some animal shelters elect not to treat cats that have clinical signs consistent with FPV, owing to the threat posed to the shelter population. However, the risk of viral transmission in shelters can be mitigated by good population management strategies, including prompt identification of infected cats, adequate cleaning and disinfection protocols, and appropriate quarantine measures.2,3
FPV is environmentally stable, highly contagious and spread by direct contact with the feces, urine and blood of infected cats. Without thorough disinfection with an appropriate disinfectant suitable for non-enveloped viruses, environmental contamination can remain infectious for many months. 14 FPV initially replicates over 18–24 h in the oropharyngeal lymphoid tissues, before viremia ensues over the next 2–7 days, disseminating the virus throughout the tissues. 2 FPV has a tropism for rapidly dividing cells, such as intestinal crypt epithelium, lymphoid tissue and bone marrow stem cells, resulting in diarrhea and immunosuppression. 3 Severe clinical illness is much more likely in young, unvaccinated kittens, and morbidity and mortality peaks at 3–5 months of age, when maternally-derived immunity is waning. 2 In the peracute form, cats can die within 12 h owing to profound septic shock, dehydration and hypothermia, and clinical signs can be minimal or absent. 1 The most common form is the acute form, which often presents with a 3–4 day history of pyrexia, depression and anorexia progressing to vomiting, and sometimes to diarrhea. 2 In most cats, gastrointestinal signs coincide with severe leukopenia, with early neutropenia from neutrophil losses into the gastrointestinal tract, followed by leukopenia from bone marrow suppression. 2 The disease has an acute self-limiting course and cats that survive infection for longer than 5 days usually recover, often over several weeks. 2
The diagnosis of FPV is usually made using a combination of appropriate history, clinical signs, leukopenia on a blood smear or complete blood count, and a positive fecal parvovirus antigen (Ag) test.2,3 Large amounts of FPV can be shed in the feces during the incubation period (24–48 h post-infection) until several weeks after recovery, but viral shedding is usually over in 5–7 days.2,4
Fecal Ag testing can be used in shelters not only as a diagnostic tool but as an aid to population management so that infected animals can be identified and isolated.4,15 The point-of-care tests currently used are designed to detect canine parvovirus Ag in the feces of infected dogs, but most have excellent specificity for the detection of FPV in feline feces.4,15 While false-positive results can occur 1–14 days after the administration of modified live vaccines (MLV), their frequency is dependent on the brand of test used. 3
The purpose of this study was to describe a series of confirmed and suspect cases of FPV and in-contact cats in a large adoption-guarantee shelter in an FPV-endemic area by reviewing the records of diagnosed cases and in-contact cats over a 10-month period.
Materials and methods
Shelter records for all cats taken into a large adoption-guarantee shelter in Chicago, IL, USA (PAWS Chicago Rescue and Recovery Center), from 1 January 2010 to 31 October 2010 were reviewed. The shelter sourced its intake mainly from the local municipal shelter, but also accepted some animals transferred from other shelters within the same region. Adult cats and kittens that were at least 4–6 weeks old were vaccinated with MLV against FPV, feline calicivirus and feline herpesvirus while they were still at the source shelters. If clinically healthy on an intake veterinary examination at the study shelter, they were re-vaccinated within 24 h of intake using MLV against the same three viruses (Fel-O-Guard Plus 3; Fort Dodge). On intake, kittens were separated from other shelter cats and kittens according to day of intake or by age. Kittens aged 6 weeks or younger were housed with their littermates and mother if possible. All healthy animals were sent to foster homes, depending on availability, within 2–7 days of intake. Foster housing continued until the kittens had received a minimum of two MLV for kittens and one MLV for adult cats, administered by the veterinarians working at the study shelter. Kittens and adult cats were then returned to the study shelter for spaying/neutering, after which they were transferred to an adoption center at another location. The vaccination protocol used by the study shelter did not take into account previous vaccinations that might have been administered at other previous shelters. Kittens were not eligible for adoption until they were at least 8 weeks old. Additionally, a dewormer [Strongid T pyrantel pamoate (Pfizer) and compounded praziquantel] were administered on intake and repeated 2 weeks later. A single dose of flea and heartworm preventative (Revolution selamectin; Pfizer) was applied topically on intake, and all cats and kittens were microchipped on intake. Any unwell animals, or animals that required injectable medications or daily recheck examinations by a shelter veterinarian, were housed at the shelter. Records of monthly shelter intake categorized cats into two age groups: cats (>6 months old) or kittens (≤6 months old). Information was also available regarding whether cats were admitted to source shelters as strays or were owner-relinquished.
Cats/kittens diagnosed with FPV, and all in-contact cats/kittens, were kept strictly isolated from the rest of the shelter population in a dedicated isolation room. During an outbreak, extra precautions were taken to prevent disease transmission, including the following: (i) only assigned staff entered the isolation room and those staff had no contact with other cats in the shelter; (ii) disposable protective gear (gloves, gown, booties) were worn by staff while in the isolation room; (iii) examinations and treatments were performed in the isolation room using a set of equipment (stethoscope, thermometer, cleaner bottles, etc) that stayed only in that room; (iv) in-contact cats/kittens and those that were in an advanced stage of recovery from illness were sent to foster homes, either the homes they came from or a small number identified as experienced with in-contact/recovering cats/kittens that had adequate in-home isolation protocols; (v) after the outbreak, only stainless steel bowls and litterpans from the isolation room were sterilized and kept — everything else, including bedding, was disposed of. The cleaning protocol used in the isolation room consisted of (i) twice-daily cleaning of occupied cages using potassium peroxymonosulfate (Trifectant; Dupont) to spot clean, followed by a water rinse; (ii) using potassium peroxymonosulfate and 1:32 bleach to clean surfaces after examinations were performed; and (iii) a ‘deep clean’ with potassium peroxymonosulfate and 3% bleach used three times once each cage was vacated, and the same procedure was used to clean the whole room once all cats were removed (either died, sent to foster homes or recovered) in an ‘all-in all-out’ protocol.
Intake veterinary examinations were conducted by a staff of approximately three shelter veterinarians and occurred within 24 h of each animal’s arrival at the shelter. The following information was recorded in individual medical records: age, breed, gender (including spay/neuter status) and bodyweight (kg) at intake; physical examination findings by a veterinarian at intake; clinical and medication history during the stay at adoption-guarantee shelter.
The paper medical and intake records of all cats and kittens that entered the adoption-guarantee shelter during the study period were initially reviewed, and only a subset of records for the following groups of animals were specifically analyzed for this study.
In-contact group (healthy cats that had close contact with FPV-infected cats): these cats/kittens were housed with a FPV fecal Ag-positive cat/kitten as part of a litter group (Snap Parvo test; Idexx), but they did not show any clinical signs of FPV (pyrexia, vomiting, diarrhea and/or clinical signs of circulatory shock).
FPV-survivors group (FPV-infected survivors): all of these cats/kittens had a positive test result for FPV fecal Ag and showed clinical signs of FPV (pyrexia, vomiting, diarrhea and/or clinical signs of circulatory shock), but all survived.
FPV-non-survivors group (FPV-infected non-survivors): these cats/kittens showed clinical signs of FPV (pyrexia, vomiting, diarrhea and/or clinical signs of circulatory shock) and did not survive. All of these cats were either (i) tested for FPV fecal Ag and had a positive result or (ii) were not tested for FPV fecal Ag, but were housed with a family member that had tested positive for FPV fecal Ag.
All cats or kittens with positive fecal FPV Ag tests, or those in contact, were quarantined from the rest of the population in an isolation area for at least 2 weeks after the positive test. All cats with clinical signs received supportive therapy. Any cats requiring 24-h veterinary attention care were transferred to a local 24-h veterinary hospital until recovery, where they were treated in an isolation ward.
Results
Signalment and source details of the in-contact, FPV-survivors and FPV-non-survivors groups are presented in Table 1. Figure 1 shows the distribution of cats in the FPV-survivors and FPV-non-survivors groups by month (1 January 2010–31 October 2010) as % of total feline intake, and also the % of total intake that was kittens (aged ≤6 months old). Figure 2 shows the total number of cats/kittens in the FPV-survivors group and FPV-non-survivors groups, and total feline intake at the study shelter for the same period. In the in-contact group (n = 66), 44 cats underwent fecal Ag testing and all were negative. The remaining 22 cats were not tested. As mentioned previously, in the FPV-survivors group (n = 27), all cats tested positive for fecal Ag. In the FPV-non-survivors group (n = 52), 47 cats tested positive and five cats tested negative on the fecal Ag test. All five of the cats that tested negative were housed with family members that tested positive and showed clinical signs of FPV. One of the five cats was a queen and the rest were kittens.
Signalment and source for cats in the in-contact, FPV-survivors and FPV-non-survivors groups

OR = owner-relinquished; S = stray; DSH = domestic shorthair; DLH = domestic longhair
‘Source’ refers to whether the cats/kittens were OR or S at the time of arrival at the original shelter they were obtained from by the adoption-guarantee shelter used in the study

Percentage of total feline intake comprised of cats and kittens in the FPV-survivors and FPV-non-survivors groups (yellow bars), and percentage of total feline intake comprised of kittens (≤ 6 months; blue bars) at the study shelter by month (1 January 2010–31 October 2010)
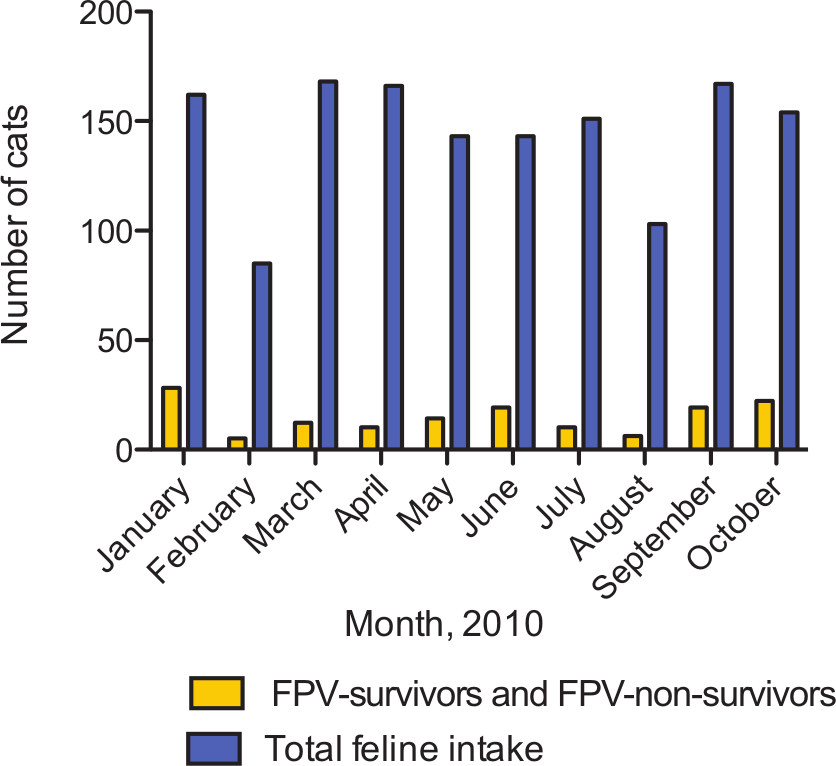
Total number of cats/kittens in the FPV-survivors and FPV-non-survivors groups, and total feline intake at the study shelter by month (1 January 2010–31 October 2010)
There were weak positive fecal Ag test results recorded for two cats; both were kittens and both were included in the FPV-survivors group. One kitten (6 weeks old) had a weak positive test result on the same day that a littermate had a positive fecal Ag result. Both kittens (FPV-survivors group) had received MLV (Fel-O-Guard Plus 3; Fort Dodge) 10 days previously, and at the time the test was performed were anorexic, lethargic, dehydrated and had lost weight over the previous week. The other kitten (7 weeks old) with a weak positive fecal Ag test result had diarrhea and lethargy (FPV-survivors group), and two littermates tested fecal Ag negative on the same day. All three kittens from this litter were vaccinated with MLV earlier on the day of the fecal Ag tests. One of the two littermates did not develop clinical signs of FPV (in-contact group), but the other littermate developed circulatory shock and died within 24 h of the negative Ag test (FPV-non-survivors group).
Ancillary diagnostic testing was not performed, except in the case of two cats from the FPV-survivors group. In one of the two cats, a blood smear was subjectively assessed by a veterinarian as leukopenic, and in the other cat a complete blood count showed leukopenia (total white cell count: 0.6 × 103/μl; reference interval: 3.5–16.0 × 103/μl).
Of kittens ≤6 weeks (n = 59), 30 were housed with their littermates and their mothers; 27 were housed with their littermates, but were separated from their mothers; and two kittens were admitted to the shelter as singletons. There was a total of 32 litters of kittens included in the study (aged 3–6 weeks at intake; 2–6 kittens/litter; n = 108 kittens), of which eight litters (n = 36 kittens) were housed with their mothers. Of the 36 kittens housed with their littermates and their mothers, 11 (31%) were in the in-contact group, 12 (33%) were in the FPV-survivors group and 13 (36%) were in the FPV-non-survivors group. Of the 72 kittens in litters not housed with their mothers, 32 (44%) were in the in-contact group, 18 (25%) were in the FPV-survivors group and 22 (31%) were in the FPV-non-survivors group. Of 8 queens housed with their litters (estimated age 1–3 years), two (20%) were in the in-contact group, three were in the FPV-survivors group (30%) and three (30%) were in the FPV-non-survivors group.
Table 2 presents fecal Ag test and clinical data for the FPV-survivors and FPV-non-survivors groups. While the data were not suitable for statistical comparison, the median (range) length of time from shelter intake to administration of MLV to a positive fecal Ag test or onset of clinical signs were similar for the FPV-survivors and FPV-non-survivors groups. Cats in the FPV-survivors group took an average of 5 days from a positive fecal Ag test or the onset of clinical signs of FPV until they were clinically healthy on veterinary examination (range 1–9 days). Death occurred an average of 1 day after a positive fecal Ag test or the onset of clinical signs of FPV in the FPV-non-survivors group (range 0–7 days). Table 3 presents the clinical signs recorded for the same groups. Anorexia, dehydration and fever were noted in the records for more than half of the FPV-survivors group. Signs of circulatory shock (pallor, prolonged capillary refill time, collapse) were documented for all of the cats in the FPV-non-survivors group, while fever was documented for just over half of the group.
Fecal antigen (Ag) test and clinical data for cats in the FPV-survivors and FPV-non-survivors groups

Adult cats and kittens that were at least 4–6 weeks old were vaccinated with modified live vaccines (MLV) against FPV, feline calicivirus and feline herpesvirus while at the source shelters. They were re-vaccinated within 24 h of intake at the shelter where the study was performed using MLV against the same three viruses (Fel-O-Guard Plus 3; Fort Dodge). Three kittens in the FPV-survivors group were unvaccinated because they developed clinical signs on the day of intake before vaccine was administered. Nine kittens in the FPV-non-survivors group were unvaccinated because they were 3 weeks old on admission to the study shelter
Clinical signs recorded for the FPV-survivors and FPV-non-survivors groups

Records incomplete for 11 cats
Rectal temperature >38.3°C in kittens/cats ≤6 weeks old; >39.2°C in kittens/cats >6 weeks old 24
Pallor, prolonged capillary refill time, collapse
Supportive therapies used included fluid therapy (lactated Ringer’s solution administered either SC or IV); antimicrobial drugs — metronidazole (15 mg/kg q24h PO, SC or IV), doxycycline (10 mg/kg q24h PO) or enrofloxacin (5 mg/kg q24h PO, IM, IV); H2 receptor antagonists — famotidine (0. 5–1mg/kg q12–24h SC); anti-emetic drugs—metoclopramide (0.2–0.4 mg/kg q8h SC or IV) or dolasetron (0.6 mg/kg q24h IV); and appetite stimulants — diazepam (0.05–0.15 mg/kg q24h IV) or mirtazapine (3.75 mg q72h PO).
Discussion
This is the first article to describe FPV using real-life cases from a shelter in an endemic area. The shelter studied was an adoption guarantee shelter and while the animals selected for intake were healthy at the time of selection, many developed clinical signs of FPV very soon after. The data documented in this study are shaped by the ability of the shelter to select healthy animals (and avoid taking in overtly unhealthy animals) and the constant influx of new animals. Therefore, the data described here might not reflect the situation in other shelter types, such as municipal shelters where animals are not purposely selected before intake, or sanctuaries, where the population is more stable and often contains more mature, fully-vaccinated cats.
The most profound determinant of survival in cats exposed to FPV is the protective antibody titer carried by each cat at the time of exposure.16,17 Treatment regimens such as those described here are supportive and are only likely to be successful if serum FPV levels at least approach protective levels. In this population, there did not seem to be any clear protection afforded by keeping young kittens (≤6 weeks old) with their mothers, perhaps because anti-FPV immunoglobulins are unlikely to be present in the milk of unprotected queens, and susceptible queens can act as a source of infection to their litters. Six of the eight nursing queens documented in this study developed clinical signs of FPV and three of those eight queens died of FPV.
Most of the animals that were included in this study were kittens, and the proportion of kittens admitted to the shelter peaked over summer, undermining the ‘herd immunity’ of the population with the entry of large numbers of susceptible animals, reflecting the findings of a much earlier study. 18 There were also substantial numbers of adult cats included, including 30% cats in the FPV-survivors group and 15% cats in the FPV-non-survivors group. One study that investigated protective antibody titers against viral diseases in feral cats in Florida reported that one third of the cats did not have protective titers to FPV, and our study confirms that protection from FPV cannot be assumed for adult cats in shelters. 19 Another more recent study of cats entering a Florida shelter reported that approximately 60% of cats were seronegative for antibodies against FPV. 20 There were no clear trends regarding stray and owner-relinquished status in our study population, perhaps because the population consisted mostly of kittens, which could have limited the advantage that possible vaccination by relinquishing owners might have provided because of interference by maternally-derived antibodies (MDA). The interference with vaccine-induced seroconversion by MDA was demonstrated in a very recent study that detected significant titers of MDA in kittens at 8 and 12 weeks of age, and in some 20-week-old kittens, preventing over a third of kittens from seroconverting after triple vaccination. 21 Interestingly, a recent French epidemiologic study reported that FPV infection was more common in owned cats than in free-roaming cats, perhaps because they congregated around human settlements, thus facilitating viral transmission. 22 The practice of revaccinating cats within 24 h of intake to the study shelter could have positive or negative consequences, depending the recent vaccination status of incoming cats. In cats that had been recently vaccinated using an effective vaccination regimen, it is possible that interference caused by the resultant antibodies could occur when they were re-vaccinated at the study shelter, thereby reducing protection levels. However, if cats arrived unvaccinated, or if they had been vaccinated but the vaccine was ineffective owing to improper storage, handling or MDA, revaccination would have been a necessary part of intake protocol.
The clinical signs reported in this population (Table 3) showed some clear trends, with cats in the FPV-survivors group mainly showing anorexia, dehydration, fever and diarrhea, but none showing signs of circulatory shock, while all cats in the FPV-non-survivors group showed signs of circulatory shock before they died. While circulatory shock is almost certainly an indicator of grave prognosis, a prospective study design would be required to confirm this.
The fecal Ag test used in this study has excellent specificity, but Ag shedding can be intermittent, thus limiting the sensitivity of the test as a screening tool.2,15,23 This is the most likely explanation for four Ag-negative cats in this study that died with clinical signs of FPV and were housed with family members that tested Ag-positive and showed clinical signs of FPV infection. There were also two kittens with weak positive Ag test results, which could possibly be attributed to post-vaccination Ag shedding. 4 However, in these cases, the kittens showed clinical signs of FPV, they were housed with littermates with clinical signs of FPV and a littermate of one of the two kittens had positive fecal Ag test results, so it is likely that the weak positive results were not vaccine-related. Additionally, weak positive results are very unlikely with the brand of fecal Ag test used in this study. 4 Combining intake dates and positive fecal Ag test dates also enables the likely site of infection to be located. In this study, the cats in the FPV-survivors group were all shedding Ag in feces 11 days or less after intake, so it is likely that many (but not all) were infected before they were admitted to the study shelter. However, in the FPV-non-survivors group, although the median time of diagnosis by fecal Ag testing was 7 days after intake, some cats were diagnosed up to 43 days after intake, in which case the likely source of infection was inadvertent fomite spread by workers in the study shelter (Table 2), as the incubation period is 2–14 days. 3
This retrospective study had several limitations. The determination of which cats were allocated to each group was based on fecal Ag testing, the presence of clinical signs of FPV and contact with a family member known to Ag-positive. Fecal Ag-positive results could have been due to post-vaccination viral shedding, as most cats in this study received MLV, but, as mentioned earlier, this is unlikely with the brand of test used. 2 Also, it is likely that, while Table 2 is a faithful representation of the medical records for the population studied, as fecal Ag testing was usually prompted by the onset of clinical signs of FPV, viral shedding might have commenced some days before the test was performed. It is possible that some cats that should have been included in the study might not have met the study criteria because Ag test results were not available, or medical records were incomplete. These cats could have been cats that had mild or absent clinical signs, as cats with more severe clinical signs would be more likely to have undergone diagnostic testing for FPV. The small sample size used dictated a descriptive study rather than statistical comparisons between groups. More useful prognostic information might have been extracted from a larger dataset by comparing groups statistically.
Conclusions
FPV is a relatively common, highly infectious and often fatal infection of kittens and adult cats admitted to shelters in endemic areas, and cases can occur year-round. Caseload was dominated by kittens, and in this shelter-based population housing litters with their mothers did not improve kitten survival. While some cats that tested fecal Ag-positive survived the infection, in those that succumbed to the disease, death was preceded by signs of circulatory shock. Education of shelter workers and foster carers regarding the typical signs of FPV and the likely acute nature of their onset could help to improve outcomes. Ideally, this should be combined with physical examinations on all cats and kittens at intake followed by daily rounds to monitor for clinical signs of disease. Early intervention should be performed as soon as clinical signs are detected in the population to confirm the diagnosis with fecal antigen testing and blood smears, quarantine all in-contact cats, and isolate and treat all cats with clinical signs, or make disposition decisions as appropriate.
Footnotes
Acknowledgements
We are grateful for the assistance of Kristen Hall CVT and PAWS Chicago Rescue and Recovery Center, Chicago, IL, USA.
Funding
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
