Abstract
Overview:
Over 22 Bartonella species have been described in mammals, and Bartonella henselae is most common worldwide. Cats are the main reservoir for this bacterium. B henselae is the causative agent of cat scratch disease in man, a self-limiting regional lymphadenopathy, but also of other potentially fatal disorders in immunocompromised people.
Infection:
B henselae is naturally transmitted among cats by the flea Ctenocephalides felis felis, or by flea faeces. A cat scratch is the common mode of transmission of the organism to other animals, including humans. Blood transfusion also represents a risk.
Disease signs:
Most cats naturally infected by B henselae do not show clinical signs but cardiac (endocarditis, myocarditis) or ocular (uveitis) signs may be found in sporadic cases. B vinsonii subspecies berkhoffii infection has reportedly caused lameness in a cat affected by recurrent osteomyelitis and polyarthritis.
Diagnosis:
Isolation of the bacterium is the gold standard, but because of the high prevalence of infection in healthy cats in endemic areas, a positive culture (or polymerase chain reaction) is not confirmatory. Other compatible diagnoses must be ruled out and response to therapy gives a definitive diagnosis. Serology (IFAT or ELISA) is more useful for exclusion of the infection because of the low positive predictive value (39–46%) compared with the good negative predictive value (87–97%). Laboratory testing is required for blood donors.
Disease management:
Treatment is recommended in the rare cases where Bartonella actually causes disease.
Bacterial properties
Bartonella (previously named Rochalimaea) species are small, vector-transmitted Gram-negative intracellular bacteria that are well adapted to one or more mammalian reservoir hosts.
To date, over 22 Bartonella species have been described, but their role as pathogens of humans and domestic animals is the subject of ongoing investigations (Table 1). 1 The most common species in both cats and humans is B henselae, the agent of cat scratch disease as well as of other potentially fatal disorders in immunocompromised people.
Species and subspecies of Bartonella that are confirmed or potential human pathogens 1
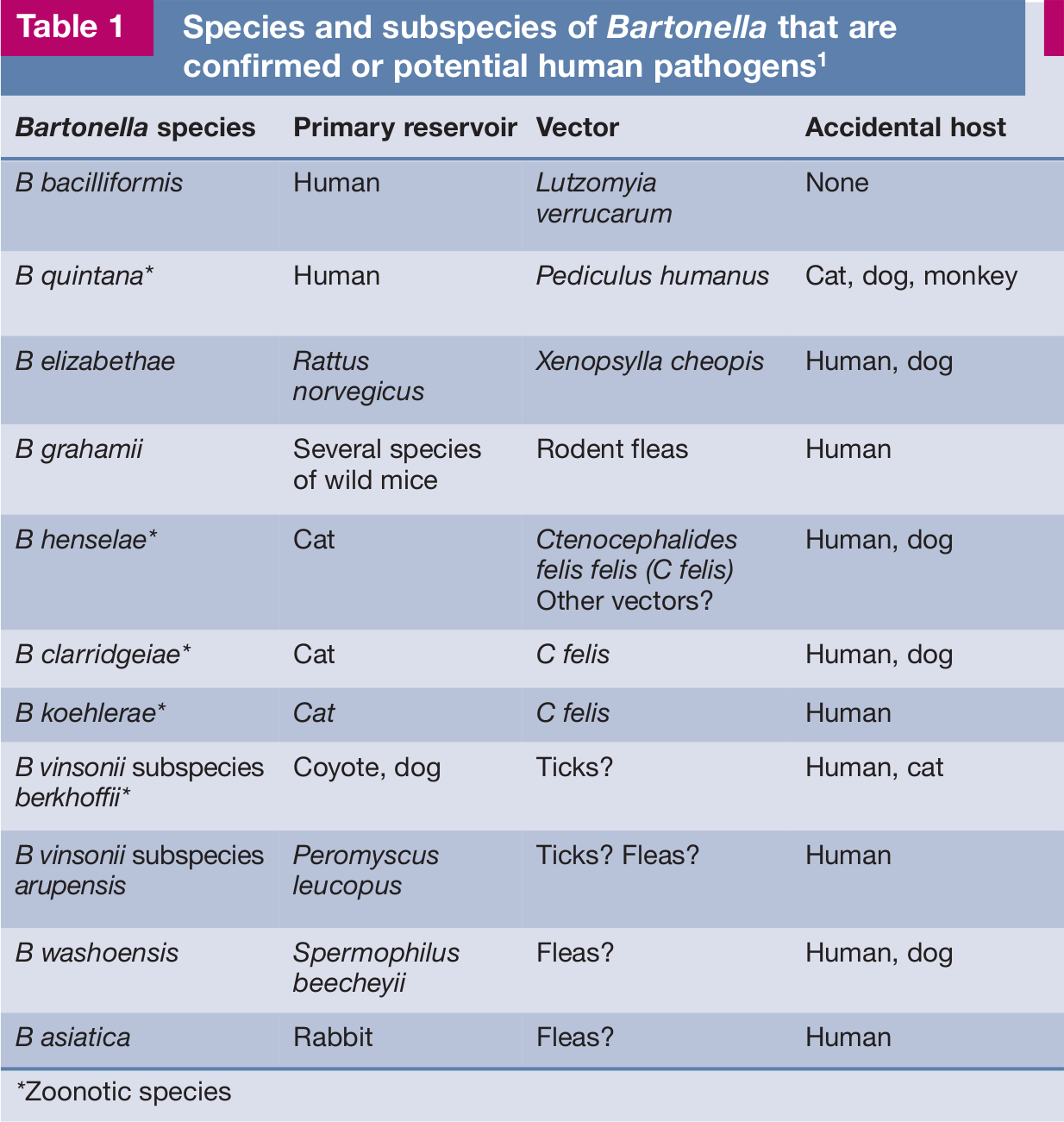
*Zoonotic species
B henselae is naturally transmitted among cats by the flea Ctenocephalides felis felis, or by flea faeces. In the infected cat, Bartonella inhabits red blood cells, which are ingested by the flea, and the bacterium survives in the flea’s gut. Contaminated flea faeces deposited on the skin of the cat end up under the cat’s claws due to self-scratching. A cat scratch is the common mode of transmission of the organism to other animals, including humans. 2

B henselae has been experimentally transmitted among cats by transferring fleas fed on naturally infected cats to specific pathogen-free (SPF) cats, and by intradermal inoculation of excrement collected from fleas fed on B henselae-infected cats. 2 This has demonstrated that both the vector and the cat – through scratches – may transmit the organism. Infection is amplified in the flea hindgut, and B henselae can persist in the environment in flea faeces for at least 9 days. 3 Blood transfusion also represents a risk: cats have been experimentally infected with B henselae and B clarridgeiae by intravenous or intramuscular inoculation with infected cat blood. 4
B henselae transmission did not occur when infected cats lived together with uninfected cats in a flea-free environment. Transmission consequently does not occur through bites, scratches (in the absence of fleas), grooming, or sharing of litter boxes and food dishes. Furthermore, transmission could not be demonstrated between bacteraemic female cats and uninfected males during mating, or to the kittens of infected females either during gestation or in the neonatal period, again in flea-free environments. 5
Ticks may also act as vectors for transmission among cats, human beings, dogs and other mammalian hosts: trans-stadial transmission of B henselae was demonstrated in Ixodes ricinus. 6
Epidemiological evidence and experimental studies have demonstrated the important role of fleas in the transmission of B henselae and B clarridgeiae among cats. Three other species, B koehlerae, B bovis and B quintana, have been isolated from cat blood, but the modes of transmission and the reservoir potential of these species in felids have not been established. In addition, B vinsonii subspecies berkhoffii DNA was detected in the blood of a cat. 7
Epidemiology
Bartonella species have a worldwide distribution, with highest prevalences in areas where conditions are most favourable for arthropod vectors, mainly fleas. In Europe, many studies have been carried out and the antibody prevalence in cats has ranged from 8–53% (Table 2).
Antibody prevalence of Bartonella infection in the feline population in European countries

Pathogenesis
Chronic bacteraemia mainly occurs in young cats, under the age of 2 years. 20 Young experimentally infected cats maintained relapsing B henselae or B clarridgeiae bacteraemia for as long as 454 days. 21 Immune system avoidance via intracellular location, frequent genetic rearrangements and alteration of outer membrane proteins are considered important factors for the maintenance of persistent bacteraemia. The location within erythrocytes and vascular endothelial cells is believed to protect Bartonella also from antimicrobial agents.
As the host-adapted reservoir of B henselae, cats display minimal pathogenic effects after experimental infection. Gross necropsy findings were unremarkable in experimentally infected cats but some histopathological changes emerged: follicular hyperplasia of lymph nodes and spleen, lymphocyte and plasma cell infiltrates in liver, heart, kidney and eye, and pyogranulomatous inflammation in liver, spleen, kidney, heart and lymph nodes.5,21
Immunity
The antibody response to B henselae has been investigated for the identification of vaccine candidates. The kinetics in response to B henselae antigens in chronically infected experimental cats is highly variable in degree and duration.2,21,22 Reinfection by a different strain of B henselae is possible, as supported by the isolation of unrelated bacterial clones from the same cat at different times. 23 Antibodies are, therefore, considered not protective, and Bartonella species antibody positive cats may be infected. 17 A cell-mediated response was not evident in investigated experimentally infected cats. 21
Clinical signs
Cats naturally infected with Bartonella species usually do not show clinical signs. Both experimental and natural infection studies have tried to establish an association between clinical signs and infection, but a link has not been unequivocally proven.
Experimental Infection
Exposure to infected fleas does not result in clinical signs [
Natural Infection
The role of Bartonella as a cause of clinical signs is even more unclear after natural infection despite a plethora of studies. Studies based on antibody detection are of limited value because antibody only proves exposure, and not necessarily an active infection. Moreover, there is cross-reactivity between different Bartonella species and strains that may or may not cause clinical signs. Because of the high percentage of infected healthy cats in endemic areas an association between clinical signs and B henselae infection is not easy to demonstrate.
It has been suggested that Bartonella infection could play a role in chronic gingivostomatitis, but the prevalence of antibodies or organisms was not higher in diseased cats than in control populations [
Cats positive for both feline immunodeficiency virus (FIV) and Bartonella antibodies had in one study an increased risk of lymphadenopathy [
An association between Bartonella antibodies and urinary tract disease or haematuria was found in two studies [
Pearce et al did not find any difference in antibody prevalence between healthy cats and cats with seizures or other neurological conditions.
32
A non-controlled retrospective study reported Bartonella DNA and antibodies in cerebrospinal fluid from cats with CNS disease [
No difference in Bartonella antibody prevalence was found between healthy cats and those affected by uveitis, but in some cases evidence of Bartonella species exposure was reported in cats with uveitis responsive to drugs considered effective against Bartonella [
No difference in Bartonella antibody prevalence was found in cats affected by anaemia, but in cats positive for Bartonella antibodies a significant association with hyperglobulinaemia was seen [
A study based on serology and culture did not find an association between Bartonella infection and chronic rhinosinusitis. 40 Also no link was reported between Bartonella infection and pancreatitis, based on the finding that cats with normal feline pancreatic lipase immunoreactivity values and cats with elevated values did not show any difference in Bartonella antibody prevalence. 41
A few case reports concern B henselae-associated endocarditis or myocarditis. Fatal aortic and mitral valve B henselae-associated endocarditis was reported in two cats in the USA.42,43 Moreover, B henselae anterior mitral valve leaflet vegetative endocarditis was successfully treated in a cat presenting with a grade III/IV systolic heart murmur and signs of aortic embolisation (lethargy and weakness in the hind limbs, weak femoral pulses, pelvic pain, increased serum creatine kinase activity). 44 This case report confirms that Bartonella species may be a cause of blood culture-negative endocarditis, as suspected. 45 Bartonella-associated myocarditis was suspected in a cat presenting with supraventricular tachycardia responsive to antibiotic therapy. 46 Post mortem evidence of pyogranulomatous myocarditis and diaphragmatic myositis associated with B henselae infection was also obtained in two cats (Figure 1). 47
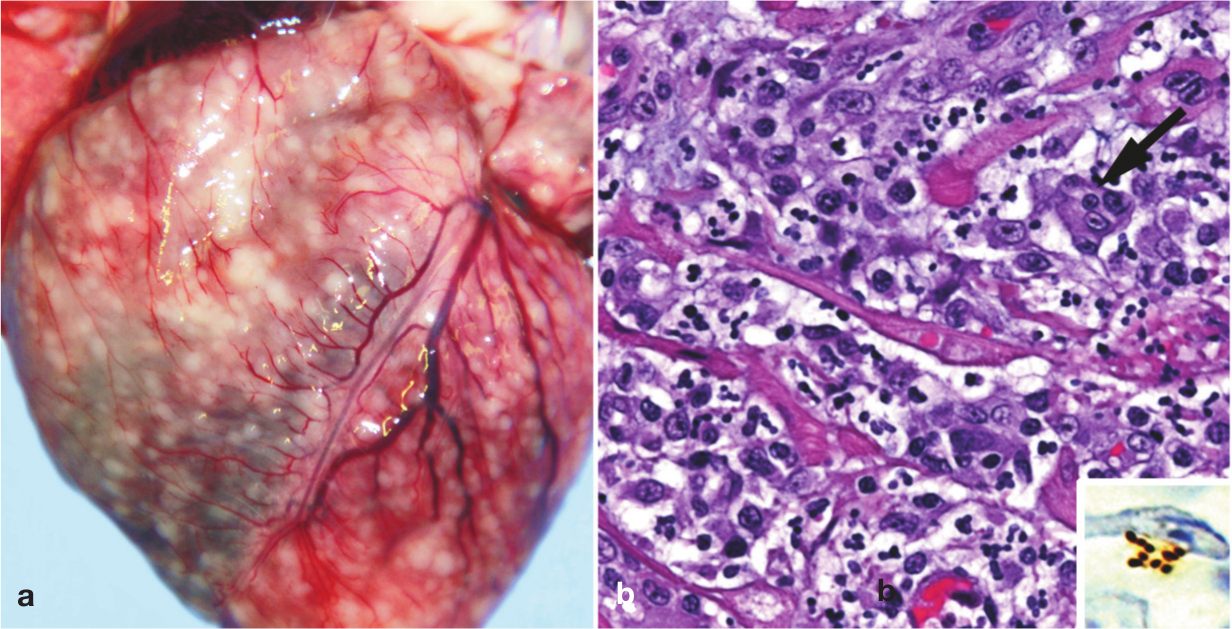
Gross and histological findings in two cats from a North Carolina shelter that died after a litter of flea-infested kittens was introduced to the shelter. (a) Coalescing granulomas distributed throughout the myocardium of an approximately 9-week-old female kitten. (b) Pyogranulomatous myocarditis in an 8-month-old castrated male cat, which had been co-housed with the flea-infested kittens. Macrophages, with a rare multinucleated giant cell (arrow), are particularly numerous towards the upper left of the image; haematoxylin and eosin stain. Inset: Cluster of short bacilli in an inflammatory focus are immunoreactive (brown) for B henselae-specific monoclonal antibody; immunohistochemistry with diaminobenzidine chromogen and haematoxylin counterstain. Reproduced, with permission, from Varanat et al (2012) 47


Immunohistochemical identification of B henselae in a case of cat scratch disease. With Warthin-Starry silver stain, these coccobacillary pathogens can be seen singly, in small clumps or in chains in necrotic foci. Courtesy of Dharam Ramnani, Webpathology.com
Lameness and pain during limb palpation were observed in a cat affected by recurrent osteomyelitis and polyarthritis associated with B vinsonii subspecies berkhoffii infection and bacteraemia. 7
In conclusion, most cats naturally infected by B henselae do not show clinical signs. The identification of Bartonella infection in cats with disease should prompt a critical assessment of the role of the infection in the causation of the clinical signs and the exclusion of other compatible diagnoses.
Diagnosis
Bartonella laboratory testing is required for feline blood donors, for pet cats belonging to immunosuppressed persons, or when a human Bartonella-related disease is diagnosed in a cat’s home.
Isolation of the bacterium is the gold standard, but because of the high prevalence of infection in healthy cats in endemic areas, a positive culture is not confirmatory, and other compatible diagnoses must be ruled out.
The disease is, therefore, diagnosed on the basis of exclusion, and by assessing the response to therapy. The ex juvantibus inference about disease causation from the observed response to a treatment may apply to uveitis, endocarditis, myocarditis, osteoarthritis and multifocal central nervous system (CNS) disease, which are considered compatible with feline bartonellosis.
PCR may be used on samples of blood, aqueous humour, cerebrospinal fluid or tissues, and several gene targets have been studied. To reduce false-negative test results, repeated blood cultures are required or PCR performed on more than one kind of biological sample (blood, lymph node, oral swab).
Serology (IFAT or ELISA) is more useful for exclusion than for confirmation of the infection because of the low positive predictive value (39–46%) compared with the good negative predictive value (87–97%) [

Treatment
Treatment is recommended in the rare cases where Bartonella has actually caused disease (eg, endocarditis).

Current therapeutic strategies in cats (Table 3) are based on in vitro studies and human bartonellosis. Data from controlled efficacy studies in cats are lacking. A cat affected by recurrent osteomyelitis and polyarthritis associated with B vinsonii subspecies berkhoffii genotype II infection and bacteraemia recovered after therapy with azithromycin (10 mg/kg PO q48h for 3 months) and amoxicillin–clavulanate (62.5 mg PO q12 for 2 months) [
Suggested treatment for bartonellosis in cats
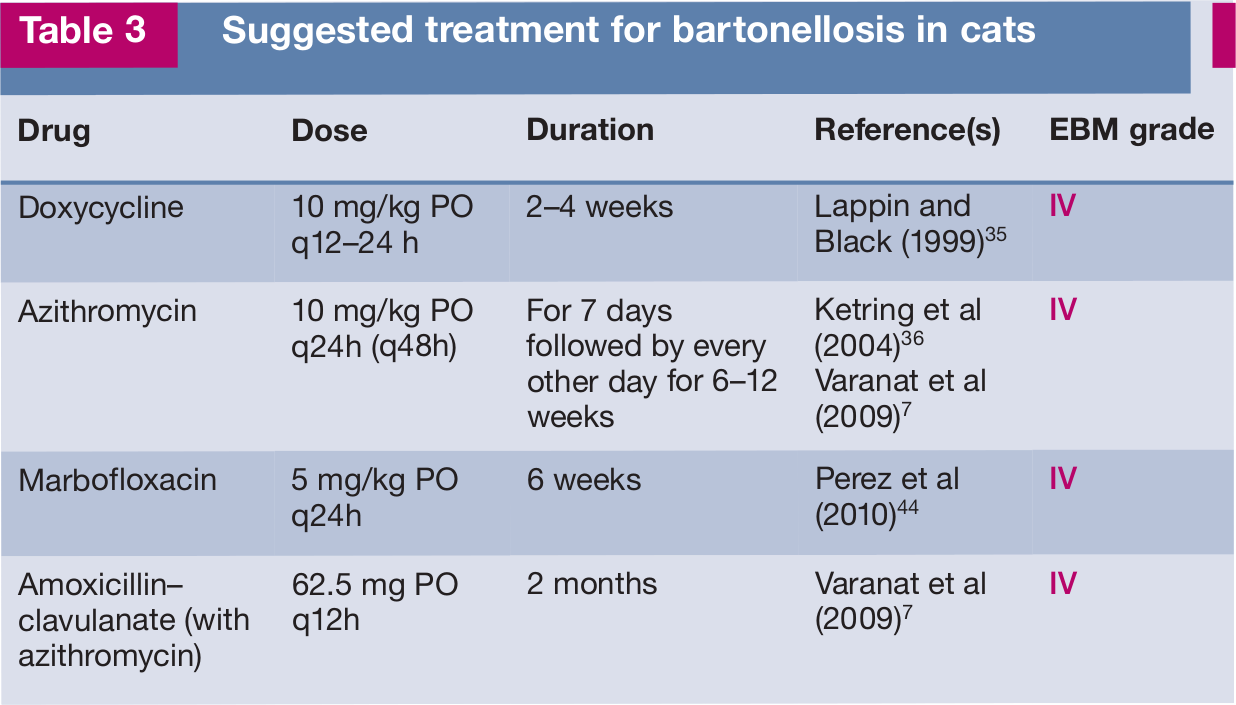
After natural or experimental infection with B henselae or B clarridgeiae, healthy cats have been treated to eliminate bacteraemia and many drugs have been evaluated: doxycycline, amoxicillin, amoxicillin-clavulanate, enrofloxacin, erythromycin and rifampicin [
Prevention
Based on transmission studies to date, strict flea (and tick) control is the only successful preventive measure. There is no vaccine available against Bartonella infection.
Footnotes
Key Points
Funding
The authors received no specific grant from any funding agency in the public, commercial or not-for-profit sectors for the preparation of this article. The ABCD is supported by Merial, but is a scientifically independent body.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
