Abstract
The diagnosis of feline haemoplasmosis has improved over the years, with several techniques enabling a clear and specific diagnosis, and where polymerase chain reaction (PCR) is considered as the ‘gold standard’. The aim of this study was to survey the prevalence of feline haemoplasmas in 320 cats from the north-central region of Portugal by the use of real-time PCR, as well as to evaluate any associations between infection, clinical presentation and risk factors. The overall prevalence of infection by feline haemoplasmas was 43.43% (139/320), where 41.56% (133/320) corresponded to Candidatus Mycoplasma haemominutum (CMhm), 12.81% (41/320) to Mycoplasma haemofelis (Mhf), 4.38% (14/320) to Candidatus Mycoplasma haematoparvum and 1.25% (4/320) to Candidatus Mycoplasma turicensis. Almost 13% (47/320) of the samples were co-infected, with the most common co-infection being CMhm and Mhf (23.74%). Infection was found statistically significant with feline immunodeficiency/feline leukaemia virus status (P = 0.034), but no significant association was found for breed, sex, fertility status (neutered/spayed/entire), age, clinical status, living conditions (in/outdoor), anaemia status, or the presence/absence of ticks or fleas. Cats from north-central Portugal are infected with all the known feline haemoplasma species, with CMhm being the most common one. Prevalence of all feline haemoplasmas was higher than that reported previously in cats from other European countries, but similar to that described in Portugal for dogs. These data provide a better perspective regarding Mycoplasma species infection in Europe, and new information that helps us better understand feline haemoplasmosis.
Introduction
Haemotropic haemoplasmas are Gram-negative epicellular parasites of erythrocytes that have been re-classified to the genus Mycoplasma owing to results from sequencing of the 16S rRNA genes.1,2 Three different haemoplasma species have been identified in cats:3 –5 (i) Mycoplasma haemofelis (Mhf), known previously as Haemobartonella felis large variant; (ii) Candidatus Mycoplasma haemominutum (CMhm), known previously as Haemobartonella felis; and (iii) Candidatus Mycoplasma turicensis (CMt), discovered in Switzerland in 2005. In 2007, a fourth haemoplasma called Candidatus Mycoplasma haematoparvum-like was identified in cats. 6 In dogs, two different haemotropic mycoplasmas have been identified: Mycoplasma haemocanis (formerly Haemobartonella canis) and Candidatus Mycoplasma haematoparvum (CMhp). 7 Overall, phylogenetic studies have shown that although Mhf and M haemocanis are distinct species, they are closely related, as well as to CMhm and CMhp. 8
Feline haemoplasmosis can be diagnosed by cytology examination of stained blood smears, but cytological diagnosis has poor sensitivity and specificity owing to intrinsic limitations, such as stain precipitates, Howell–Jolly bodies and artifacts caused by slow blood smear drying. Moreover, blood smears can’t differentiate between haemoplasma species, and, because of the cyclic nature of the parasitaemias, the absence of Mhf organisms in blood films does not rule out the diagnosis of haemotrophic mycoplasmosis. 2 Currently, polymerase chain reaction (PCR) techniques have been developed to diagnose Mycoplasma species infections, offering better reliability as they are more sensitive than cytology examination. 9 The use of real-time PCR (qPCR) has been extended as it is more sensitive than conventional PCR and it allows quantification of the amount of haemoplasma DNA present in a cat’s blood, helping to determine the significance of the haemoplasma infection and to monitor the response to treatment. qPCR does not need post-PCR treatment therefore reducing the risk of sample contamination at the diagnostic laboratory. qPCR testing has been beneficial in testing clinically healthy cats being screened as blood donors or for entry into specific pathogen-free colonies. 10
In Europe, prevalence studies carried out in Italy, 11 UK,12,13 Germany,14 –16 Greece, 17 Switzerland18,19 and Spain 20 based on PCR results report an overall prevalence of Mycoplasma species ranging from 7.2% to 30%. CMhm is the most common type of Mycoplasma species ranging from 9.6% to 66.6%, followed by Mhf and CMt with prevalences of infection of 1.3–33.3% and 0.3–2.1%, respectively. CMt infection is associated with co-infection with either, and occasionally both, of the two other feline haemoplasma species, particularly with CMhm, 8 inducing moderate-to-severe anaemia. 19 The nature of the cat’s sampled for these studies included samples from healthy cats to cats suspected of having haemoplasmosis. They also belonged to different geographical locations — a variable that is believed to play an important role in the rates of infection as warmer climates have higher rates of prevalence.18,20 To our knowledge, no previous studies on feline haemoplasmas have been carried out in Portugal. Therefore, the aims of this study were to survey the prevalence of feline mycoplasmas in 320 Portuguese cats by the use of qPCR, as well as to evaluate any associations between infection, clinical presentation and risk factors.
Materials and methods
Sample collection
Three hundred and twenty ethylenediamine tetra-acetic acid-anticoagulated blood samples were collected from both stray cats (rescued for animal shelters) and client-owned cats with indoor/outdoor access in Portugal. These samples were collected by veterinarians at the time of clinical presentation and frozen at −20°C until DNA extraction. Data obtained at the time of presentation included age, breed, gender, living condition, geographical region and clinical status (healthy vs unhealthy was determined based on the reason for presentation and physical examination). Data collected in a retrospective manner if available were feline leukaemia virus (FeLV)/feline immunodeficiency virus (FIV) status (determined by SNAP FIV/FeLV Combo Test) and the presence of ticks and/or fleas. Haematology was performed when requested by the attending veterinarian. Packed red cell volume (PCV) value was considered normal when values ranged from 25% to 45%, and anaemia was defined as PCV <25% according to the reference interval of our laboratory.
DNA isolation and qPCR amplification
DNA was obtained from 0.5 ml of peripheral whole blood as described previously. 21 Amplification of Mycoplasma species and the different species in positive samples (Mhf, CMhm, CMt and CMhp) were carried out in a final volume of 20 µl using FastStart Universal SYBR Green Master (Roche), 0.3 µM of each primer (with the exception of reverse primer of CMt at 0.5 µM) and 4 µl of diluted DNA. The thermal cycling profile was 50°C for 2 mins and 95°C for 10 mins followed by 40 cycles at 95°C for 15 s and 60°C for 1 min. The PCR primers were designed with Primer Express 2.0 (Applied Biosystems) based on conserved regions of 16S ribosomal RNA gene for Mycoplasma species. The specificity of each PCR primer pair included (i) in silico analysis; (ii) sequencing the positive controls; and (iii) performing a cross-amplification study in which amplification with specific primers was performed over all the positives of other pathogens available in our laboratory (pathogens that could be in co-infection in cats, mostly vector borne diseases). Expected results were amplification of the specific Mycoplasma-positive sample and no amplification of any of the other pathogens tested. Validation of the PCR included the analysis of clinical samples suspected of being infected by the pathogen and the sequencing of any spurious or unspecific amplification that could occur.
Internal controls were added to all the samples analysed and all the PCRs performed. Negative controls include negative extraction control for the DNA extraction and a negative template control for the PCR. Positive qPCR controls were obtained from clinical samples that had been amplified previously and sequenced to confirm the pathogen. The sequences of the primers used and assay temperatures (dissociation curve analysis) are shown in Table 1.
Primer sequences for Mycoplasma real-time PCR assays and species-specific melting temperatures
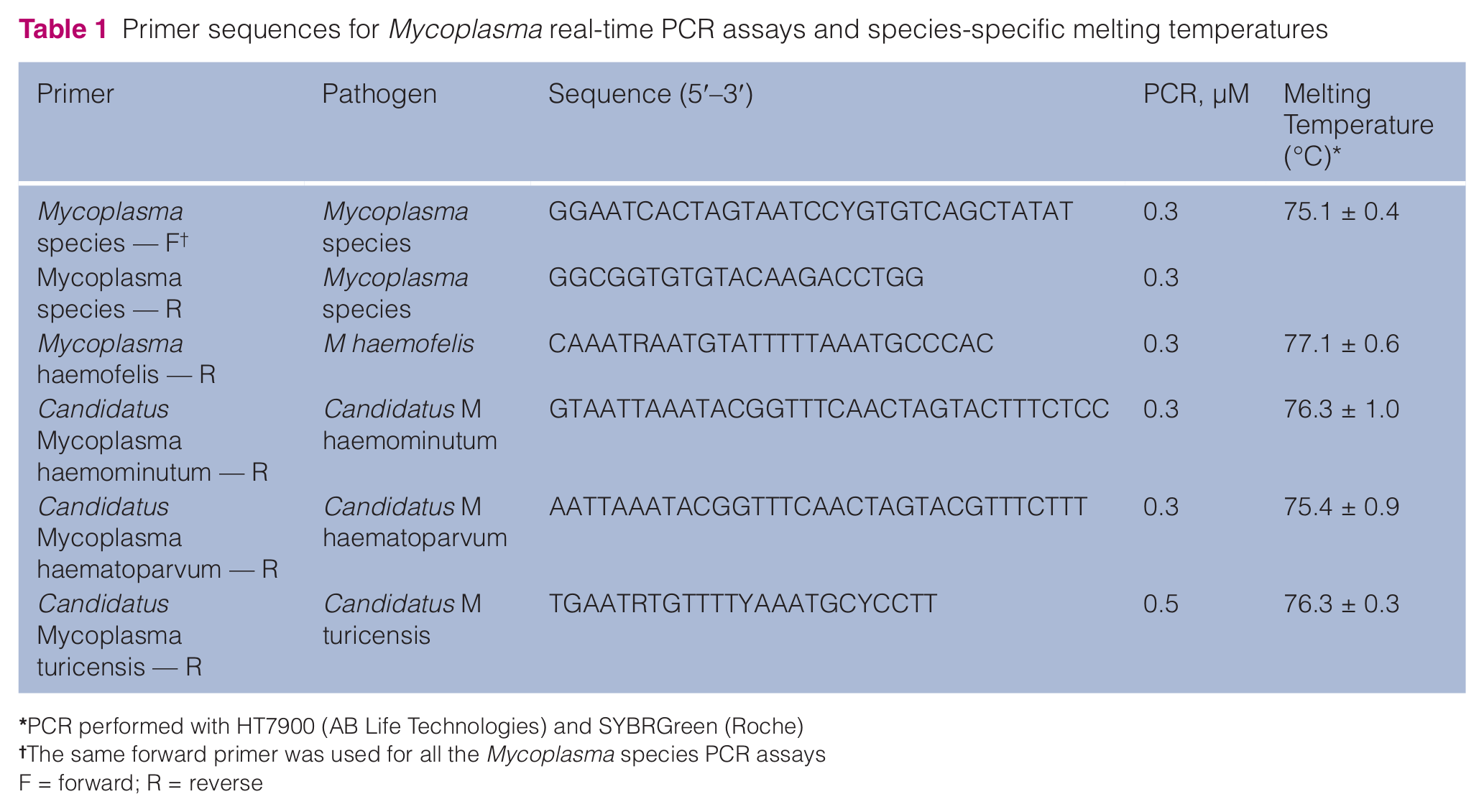
PCR performed with HT7900 (AB Life Technologies) and SYBRGreen (Roche)
The same forward primer was used for all the Mycoplasma species PCR assays
F = forward; R = reverse
The eukaryotic 18S RNA Pre-Developed TaqMan Assay Reagents (Applied Byosystems) was used as internal reference for cat genomic DNA amplification to ensure proper qPCR amplification of each sample and that negative results corresponded to true negative samples rather than to a problem with DNA loading, sample degradation or PCR inhibition. The use of an endogenous control (18S RNA) that amplified between cycle threshold 19–21 in all samples corroborate that similar amounts of genomic DNA were used in all PCRs.
Statistical analysis
Difference between groups was tested for significance by χ2 analysis (cell frequencies >5) or Fisher’s exact test (cell frequencies ≤5). A P-value <0.05 was considered statistically significant. For the observed prevalence, the 95% confidence interval (CI) was calculated. Potential risk factors were evaluated by calculating the odds ratio with a 95% CI, and P-value <0.05 was considered statistically significant. All available data were analysed using MedCalc Software. 22
Results
A total of 320 cats (175 males and 136 females), ranging in age from 0.5 to 20 years (median 4.23 years) from the north and north-central regions of Portugal were included in the study. Most samples came from the district of Aveiro (36.87%; 118/320). The other samples came from Oporto 14.37% (46/320), Coimbra 13.12% (42/320) and 35.62% from Braga, Bragança, Leiria, Viana do Castelo, Vila Real and Viseu (114/320).
Pedigree cats represented only 14.6% of the cohort, which included 26 Persian, 11 Siamese, three Norwegian Forest, two Sphynx, one Birman and one Chartreaux. The remaining 85.4% were domestic shorthair cats. Females represented 43.7% (136/311) of the population, whereas males represented 56.2% (175/311); 40.4% (55/136) of the females and 46.8% of the males were PCR-positive. Over half the population of cats had outdoor access [61.1% (189/320)] and 60.5% (184/320) were classified as sick when presented to the veterinary clinic, 44.5% (82/184) of whom were PCR-positive for feline haemoplasmas. Regarding antibiotic treatment, information was available for only 21 cats (6.25%). Antibiotics used in these 21 cats were amoxicillin, esperamicin, enrofloxacin, doxycycline and cefalexin, but only eight cats were receiving enrofloxacin or doxycycline as treatment for mycoplasmosis.
Table 2 summarises all the data for breed, sex, fertility status (neutered/spayed/entire), indoor/outdoor status, age, clinical status, PCV, presence/absence of ticks or fleas and FIV/FeLV status.
Sample characteristics. Data from animals that had no available information were not analysed
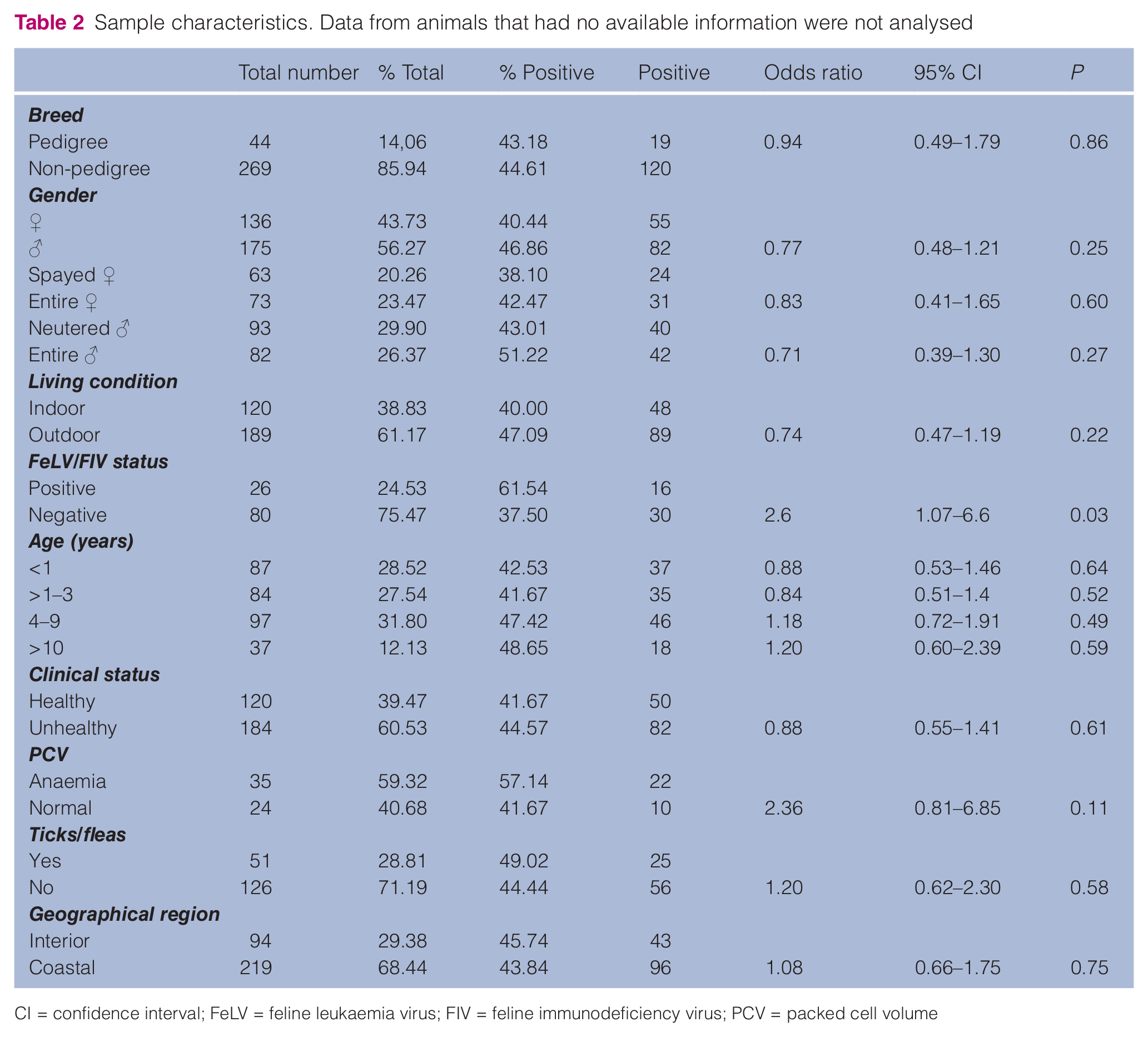
CI = confidence interval; FeLV = feline leukaemia virus; FIV = feline immunodeficiency virus; PCV = packed cell volume
The overall sample prevalence for Mycoplasma species infection was 43.44% (139/320): 41.56% (133/320) were infected with CMhm; 12.81% (41/320) with Mhf; 4.38% (14/320) with CMhp and 1.25% (4/320) with CMt. Forty-two samples were co-infected with two types of Mycoplasma species, with the most common co-infection being CMhm + Mhf; four samples were co-infected with three types of Mycoplasma species and one sample presented positive results for all the Mycoplasma species tested. Overall co-infection sample prevalence was 14.68% (47/320). The results are summarised in Table 3.
Haemoplasma detection by real-time PCR

The highest sample prevalence of infected cats was found in Viseu (73.33%, P = 0.02, odds ratio: 3.65, 95% CI = 1.13–11.73) (Table 4). FIV/FeLV status was statistically significant (P = 0.03, odds ratio: 2.6, 95% CI = 1.07–6.62) when comparing clinically sick cats (15/82) and healthy cats (1/50) with positive Mycoplasma species qPCR (Table 2). No significant P-value was found for breed, sex, fertility status (neutered/spay/entire), age, clinical status, presence/absence of ticks or fleas, living conditions or anaemia status in the overall population.
Prevalence of Mycoplasma infection by geographical region

CI = confidence interval; ND = no data
The majority of the samples that amplified Mhf were co-infected with CMhm alone (33/41), although multiple co-infections with additional species were also detected (3/41 CMhm + Mhf + CMhp and 1/41 Mhf + CMhm + CMhp + CMt). There was a statistical significance when comparing breeds (pedigree vs non-pedigree) with an odds ratio of 2.25 (95% CI 1.01–5.01) and a P value of 0.04 for cats infected with Mhf.
Only 14 samples were positive for CMhp. All the samples that amplified for this type of haemoplasma were co-infected (14/320): nine with CMhm, one with CMt+CMhm and three with CMhm+Mhf. Only four samples were positive for CMt. Two of these samples were co-infected, and 3/4 samples belonged to sick cats with outdoor access.
Discussion
This study documents the occurrence of Mhf, CMhm, CMhp and CMt in naturally-infected cats from the north-central area of Portugal. To our knowledge, no previous studies have reported on the prevalence of feline haemoplasmas in Portugal. Previous studies have only been conducted on canine samples, reporting a prevalence for haemoplasma infection of 40% 23 which is similar to that found in our study for the cat population. The prevalence rates observed in Portugal are higher than those of other European countries, such as Spain20,24 Greece, 17 Italy, 11 the UK,12,13,19 Switzerland 18 or Germany14–16 which range from 7.2% to 30%. Differences in prevalence among countries could be explained by geographical variations, such as climate, vector distribution and the cat population per se. Moreover, direct comparisons of prevalence results are of limited value because of sample characteristics, inclusion criteria, diagnostic techniques, different statistical methods or a combination of them all, resulting in discrepancies between studies. Most of the cats in our study were cats with outdoor access, which means that they have a higher chance of Mycoplasma species infection as a result of their living style (more exposure to vectors like ticks and fleas which could transmit Mycoplasma species, higher chance of FIV/FeLV transmission, etc).
In our Portuguese samples CMhm was the most prevalent haemoplasma found, as has been described previously.25,26 In fact, Reynolds and Lappin 27 suggest that CMhm may be a primary pathogen in some naturally-infected cats. Some studies indicate that CMhm infection is less pathogenic when compared with other feline mycoplasmas,4,18 and some reports, such as one from Spain, 20 as well as our study, have failed to demonstrate an association between anaemia and Mycoplasma species infection. A limitation to our study was that a complete blood count was not performed for all 320 cats.
Mhf has been classified as the most pathogenic Mycoplasma species in cats. 9 Cats during the acute phase of infection with Mhf develop anaemia as a result of extravascular erythrophagocytosis by macrophages in the liver, spleen, lungs and bone marrow. In chronic phases of infection anaemia may not be present. 2 Conversely, various studies of naturally-infected cats, including ours, have failed to demonstrate an association between anaemia and Mhf infection.12,20,25 Different results might occur owing to different cat populations, the presence of different isolates of Mhf involved co-infections with other Mycoplasma species and the underlying health status of the cats. Most of the cats infected with Mhf in our study were co-infected with other types of Mycoplasma species, making it difficult to explain any association with the risks factors included. In our study, Mhf + CMhm co-infection was the most common co-infection, with a sample prevalence of 23.7% (95% CI 16.3–33.3%), although it was considered rare in previous articles with prevalences ranging from 0.2% to 4.9%.11,12,15,16,18,28,29
Regarding CMhp infection, we have detected, by means of a qPCR assay, molecular evidence of infection in 4.37% (14/320) of cats. All of the qPCR CMhp-positive cats were co-infected with other types of Mycoplasma species, with the most common co-infection being CMhp + CMhm (9/14) followed by triple co-infections with CMhp + CMhm + Mhf (3/14). These results are higher than the prevalence rate reported by Sykes et al, 6 who reported a 0.7% prevalence in anaemic and non-anaemic cats from the USA. Other studies performed in the USA 30 or Italy 11 have failed to find any evidence of CMhp infection in cats. Further prevalence studies should be performed in different countries to understand the pathogenicity of this species of Mycoplasma.
In the case of CMt infection, 2.8% (4/139) of cats were positive for CMt, a sample prevalence rate similar to that reported in UK 19 and greater than those observed in other European studies of 1.1%, 16 0.8%, 13 0.7%, 15 0.5%, 20 0.4% 5 and 0.3%. 11 It has been described that cats infected with CMt have significantly lower blood copy numbers than cats infected with Mhf or CMhm as these cats become qPCR-negative as early as 45 days after infection, while cats infected with Mhf or CMhm remain qPCR-positive even after 85 days after infection. 9 In fact, eight cats in our study were receiving or had received antibiotic (enrofloxacin, doxycycline) treatment before qPCR analysis, and five of these cats remained qPCR-positive for CMhm, indicating that long-term carrier status is common in comparison to infection with other Mycoplasma species, as reviewed by Tasker. 9
There was no significant difference in overall haemoplasma sample prevalence between healthy and unhealthy cats, which could be explained by chronic infection detection rather than an acute infection, and the presence/absence of other diseases which may not have been reported. It has been reported that 10% of healthy cats can be infected with feline haemoplasma at any point in their lives. 9 These data should be kept in mind when interpreting qPCR results, as a positive result does not always correlate with clinical disease. A diagnosis of mycoplasmosis should be based on the patient’s physical findings, biochemistry and haematological tests, as well as qPCR results. 31
Results regarding the geographical region have to be interpreted with caution. Although microclimate variations within the same climate area could influence the epidemiology of haemoplasma infections, 11 it is not safe to assume that one district is more at risk of having a higher prevalence of a specific type of haemoplasma because not all districts had the same sample size (sample size was relatively low in most of the districts). Furthermore, indoor or outdoor habitat may function as a cofactor in different geographic areas.
Contradictory results have been reported on whether concomitant FIV and FeLV infections could predispose to haemoplasma infections, favouring the chronic carrier state or worsen the clinical course.6,11,13,32,33 In our study, being FIV- or FeLV-positive was a risk factor for haemoplasma infection, which is in agreement with previous studies, 11 but results should be interpreted carefully as FIV/FeLV status was only determined in 33.12% of the cats in this study.
Conclusions
Cats from north-central Portugal are infected with all the known feline haemoplasma species, with CMhm being the most common one. The sample prevalence of all feline haemoplasmas was higher than that reported previously in cats from other European countries, but similar to that described in Portugal for dogs. These data provide a better perspective regarding Mycoplasma species infection in Europe and new information that helps us better understand feline haemoplasmosis.
Footnotes
Acknowledgements
Thanks to the veterinarians, cat owners and veterinary clinics that willingly provided the samples for this research. Special thanks to Lisete Vieira who collaborated with the collection of blood samples.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
