Abstract
A 4-year-old female neutered British Shorthair cat was presented as an emergency owing to progressive apathy, anorexia, adipsia, weight loss and weakness. Clinical findings showed severe weakness, collapse, weak pulse, bradycardia, hypovolaemia and hypothermia. Blood examinations revealed marked metabolic acidosis, hyponatraemia, hyperkalaemia, hyperphosphataemia, hypercalcaemia, hypochloraemia and azotaemia. The diagnosis of feline hypoadrenocorticism was based on low cortisol and aldosterone plasma levels before and after synthetic adrenocorticotropic hormone administration. Initial treatment consisted of intravenous fluid therapy. After stabilisation a combination of fludrocortisone and prednisolone was given orally. One year after diagnosis the cat is free of clinical signs and in good condition.
Case Report
A 4-year-old female neutered British Shorthair cat with a body weight of 3.4 kg and a body condition score of 4/9 was referred to the Small Animals Clinic, University of Giessen, as an emergency owing to lethargy, weakness, anorexia, adipsia and weight loss. On the day of consultation the cat’s health status declined drastically compared with the previous days. Four weeks previously the cat developed polyuria and polydipsia, and was treated by the referring veterinarian with cefovecin sodium (Convenia; Pfizer, dose unknown) without any clinical improvement. The cat had not received exogenous oral or topical glucocorticoids. The cat became progressively weak, lethargic and stopped eating and drinking; the owners had been giving food and water via a syringe for the previous 7 days. Furthermore, intermittent vomiting was noticed by the owners. They also described an uncoordinated gait/ataxia that had begun a few weeks previously and had worsened progressively. The cat lived exclusively indoors. It was dewormed every 3 months, vaccinated annually and was fed a commercial cat food.
On physical examination, the cat was initially attentive and able to stand with support, but unable to walk. With continued handling, the cat became progressively weaker until she was unable to move and went into sternal recumbency. Mucous membranes were mildly pale and dry, capillary refill time was prolonged (>3 s) and there was abnormal skin tenting; 10% dehydration was estimated. The femoral pulse was not palpable bilaterally. The cat was bradycardic with a heart rate of 120 beats per minute, but with a regular heart rhythm. She showed a mild tachypnoea with a respiration rate of 44 breaths per minute. Body temperature was 32.7°C. Neurological examination was unremarkable.
Blood gas analysis (Table 1) revealed a marked metabolic acidosis with partial respiratory compensation and marked hyponatraemia and hyperkalaemia. The sodium:potassium ratio was 15. Haematology showed a mild lymphocytosis {8.5 × 109/l, [reference interval (RI) = 1.5–7.0 × 109/l]} and lack of a stress leukogram [neutrophils 4.5 × 109/l (RI 2.5–12.5 × 109/l); monocytes 0.11 × 109/l (RI 0.04–0.85 × 109/l)], which was unexpected in a chronically ill and stressed cat. Clinical biochemistry revealed, in addition to the electrolyte abnormalities, a severe azotaemia [urea 22.3 mmol/l (RI 7.1–10.7 mmol/l); creatinine 599 µmol/l (RI 0–168 µmol/l)] and hyperphosphataemia [3.7 mmol/l (RI 0.8–1.9 mmol/l)]. Thoracic radiography (Figure 1) revealed a small cardiac silhouette with a vertebral heart score of 6.2 (RI 6.7–8.1), pulmonary hypoperfusion and a narrow caudal vena cava. A blood pressure reading could not be detected in any limb using the Doppler method. Unfortunately, cystocentesis and urinalysis were not possible because, ultrasonographically, the urinary bladder was empty. An adrenocorticotropic (ACTH) stimulation test was performed by injecting 125 µg synthetic ACTH (Synacthen; SIGMA-TAU) intravenously. Serum cortisol and serum aldosterone levels were measured at baseline and 60 mins after stimulation by a commercial laboratory (Biocontrol, Ingelheim, Germany) by chemiluminescence immunoassay method for quantitative measurement of cortisol and with a radioimmunoassay for quantitative determination of aldosterone.
Blood gas analyses (bold entries indicate values outside the reference interval)
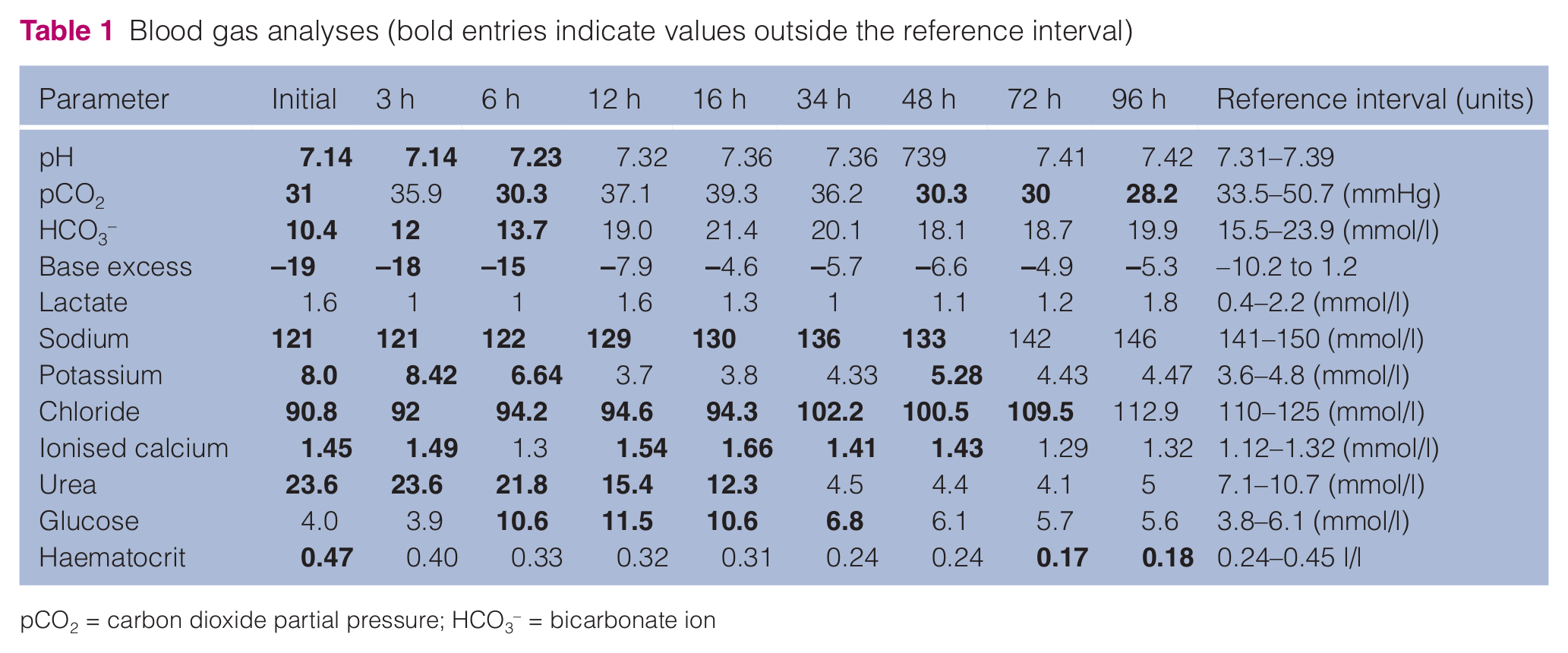
pCO2 = carbon dioxide partial pressure; HCO3− = bicarbonate ion

Thoracic radiograph after initial stabilisation
Initial treatment consisted of intravenous (IV) fluid therapy and of rewarming the cat. A bolus injection of 20 ml/kg 0.9% saline and 6 ml/kg hydroxyethyl starch, given over 1 h, was instigated. Sodium bicarbonate at a dose of 1 mmol/kg to correct the metabolic acidosis and 1 ml 10% calcium–gluconate as a cardioprotective agent were administered IV. After rehydration, 0.9% saline spiked with 40% glucose was continued at 6 ml/kg/h. Body temperature increased slowly to 37.8°C over a period of 12 h. The cat was monitored closely (blood gas analyses every 3 h; continuous electrocardiography monitoring), and, over the next 12 h, several blood gas parameters normalised, but a marked hyponatraemia persisted (Table 1). Clinically, the cat became more alert, was able to move into sternal recumbency and drink with assistance. The cat’s heart rate increased to 180 beats per minute and systolic blood pressure was measured repeatedly to be >100 mmHg. The next day treatment for hypoadrenocorticism was started with 0.025 mg fludrocortisone/cat twice daily PO (Astonin H 0.1 mg; Merck) and 0.3 mg/kg prednisolone once daily PO (Prednisolone 5 mg; CP-Pharma). Fluid therapy was continued as before with electrolyte replacement according to blood gas analyses findings. ACTH stimulation test results confirmed the diagnosis of hypoadrenocorticism, with both cortisol and aldosterone levels below the detection limit of the assay at baseline and after stimulation (Table 2). Ultrasonographic investigation identified the left adrenal gland with a diameter of 0.3 cm, while the right adrenal gland could not be visualised. Over the next few days the cat stabilised and all clinical signs resolved. In the course of the cat’s hospitalisation haematology showed a moderate, non-regenerative anaemia [haematocrit 0.17 l/l (RI 0.24–0.45 l/l); haemoglobin 3.6 mmol/l (RI 4.9–9.3 mmol/l]; reticulocytes 24.3 × 109/l]. The cat was discharged with the same dosages of fludrocortisone and prednisolone as given during hospitalisation.
Adrenocorticotropic hormone stimulation test

RI = reference interval
Re-examination after 6 days revealed normal behaviour and no abnormality was detected during physical examination. Blood testing showed only mild electrolyte abnormalities. Anaemia was now regenerative [haematocrit 0.21 l/l (RI 0.24–0.45 l/l), haemoglobin 4.0 mmol/l (RI 4.9–9.3 mmol/l), reticulocytes 123.2 × 109/l]. Fludrocortisone was continued at the initial administered dose, while prednisolone was discontinued. When, 2 weeks later, polyuria and polydipsia resumed, prednisolone was reinstituted and clinical signs resolved. The dosage of prednisolone was decreased slowly (to 0.2 mg/kg once daily) over the following months, during which time the cat gained weight (current body weight 4.7 kg vs 3.4 kg at initial presentation). The cat is doing well 1 year after initial presentation.
Hypoadrenocorticism or Addison’s disease is a rare disease in cats, with approximately 40 cases reported in literature.1 –12 In contrast to Addison’s disease in dogs, there is no evidence for sex, age or breed predisposition in cats.3,10 Age at presentation ranges between 1.5 and 14.0 years (median 4.0 years). The majority of patients are domestic shorthair and longhair cats.
The diagnosis is usually confirmed by measuring cortisol at baseline and following exogenous ACTH stimulation. While aldosterone in response to ACTH administration has been measured in healthy cats,13,14 to our knowledge this is the first hypoadrenocorticoid cat in which aldosterone has also been measured. Not surprisingly, both values were below the detection limit of the assay, confirming that hypoaldosteronism was responsible for the marked electrolyte abnormities seen in this cat. Other causes of electrolyte abnormalities, such as effusion, other endocrine diseases or urinary problems could therefore be excluded.15,16
In contrast to dogs, where the majority of cases are thought to be caused by primary immune-mediated adrenal destruction, the cause of adrenal insufficiency in cats is largely unknown.3,4,6,8,10–12 There are few case reports that found some causes, such as bilateral destruction of the adrenal glands by an infiltrating lymphoma in two cats 2 and two cases as a result of trauma.5,6 Unfortunately, only one adrenal gland could be visualised with ultrasonography in the present case, which was considered small, so the cause remains unclear. 13
This cat’s history and physical examination findings were typical for cats suffering from hypoadrenocorticisim.1 –12 In decreasing order, anorexia (100%), lethargy (93%), weight loss (86%), vomiting (36%), waxing–waning course of illness (29%), previous response to symptomatic therapy (21%), polyuria/polydipsia (21%), dehydration (92%), weakness (85%), hypothermia (77%), slow capillary refill time (38%), weak pulse (38%), collapse/inability to rise (23%), a painful abdomen (23%) and bradycardia (15%) have been reported 10 and were almost all seen in the present case. This cat also had typical clinicopathological abnormalities, including hyperkalaemia (90%), hyponatraemia (100%), a markedly decreased sodium:potassium ratio, hypochloraemia (90%), azotaemia (100%), hyperphosphataemia (100%), hypercalcaemia (10%), lymphocytosis (20%), lack of a stress leukogram and anaemia (30%).3,4 It was thought that anaemia was masked initially by haemoconcentration based on high-grade dehydration.
Besides the mentioned clinicopathological abnormalities, this cat had a marked metabolic acidosis. The presence of (generally mild) metabolic acidosis is well known in hypoadrenocorticoid dogs, 3 but has, to our knowledge, never been described in cats. About 50% of dogs with hypoadrenocorticism have a mild metabolic acidosis; severe metabolic acidosis (serum bicarbonate between 9 and 12 mmol/l) is an infrequent finding in dogs (<10%). 3 This cat had a severe metabolic acidosis with partial respiratory compensation. The main mechanisms for these findings are thought to be the decreased renal excretion of hydrogen ions due to hypoaldosteronism fortified by hypotension, and poor perfusion of tissues and a decreased glomerular filtration rate. 3 We decided to administer sodium bicarbonate to correct severe metabolic acidosis and to lower serum potassium concentration more quickly. The bicarbonate deficit was estimated by the following formula: 0.3 × body weight (kg) × (24 × patient bicarbonate). One fourth of the calculated dose was given slowly IV. Potential complications resulting from sodium bicarbonate administration include metabolic alkalosis, hypokalaemia, hypocalcaemia, hypercapnia and paradoxical intracellular or central nervous system acidosis, which can cause respiratory arrest. On this account sodium bicarbonate therapy should be reserved exclusively for life-threatening cases.
A retrospective study by Bell et al. described 49 cats with sodium to potassium ratio <27. 16 Interestingly, none of the cats suffered from hypoadrenocorticism. Hypoadrenocorticism was excluded with either a negative ACTH stimulation test, a lack of clinical signs compatible with hypoadrenocorticism or resolution of decreased sodium:potassium ratio after therapy other than glucocorticoids or mineralocorticoids. At least two of these three criteria had to be met. Systems affected were, in decreasing order, gastrointestinal, urinary, cardiorespiratory and endocrine systems; rarely, other organ systems, such as the eye or skin were reported. 16 Only recently a cat with hypoadrenocorticism and marked hypoglycaemia has been reported. 8 We assumed that hypoglycaemia resulted from a combination of lack of cortisol (reduction of glucose mobilisation) and anorexia. The present case had low normal glucose at initial presentation and mild hyperglycaemia during hospitalisation (Table 1), likely due to stress.
Microcardia as a sign of dehydration, hypovolaemia and hypotension has been reported in 87% of cats when thoracic radiographs were available. This was equally seen in the present case. This is not surprising as most cats were markedly hypovolaemic, as manifested by the physical examination.4,6,7
All things considered, the clinical and clinicopathologic findings (lateral recumbency, decompensated hypovolemic shock, hypotension, electrolyte shifts and metabolic acidosis) were indicative for an Addisonian crisis. This seems to be a rare condition in cats, as, to our knowledge, it has never been described before, but is well known in dogs suffering from hypoadrenocorticism. The term describes a life-threatening condition that is characterised by hypotension, dehydration, hyperkalaemia, hyponatraemia and acidosis. 3 All of these findings were recognised in the present case. Initially, blood pressure was not detectable in any leg by Doppler method, so it had to be assumed that this cat was markedly hypotensive. After stabilisation, systolic blood pressure increased to >100 mmHg at every subsequent measurement.
Conclusions
With appropriate treatment, prognosis for long-term survival, once the Addisonian crisis has been managed, seems favourable.3,4,6,7,12 Still, it has to be mentioned that most case reports (including this one) have been reported during the life time of the described cats so that, to our knowledge, there are no long-term survival times published yet.
Footnotes
Funding
The authors received no specific grant from any funding agency in the public, commercial or not-for-profit sectors for the preparation of this case report.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
