Abstract
A 4-month-old female kitten presented with chronic lower urinary tract signs and
Case Report
A 16-week-old female domestic shorthair kitten, weighing 2.28 kg was referred to Southpaws Specialty Surgery for Animals for evaluation of recurrent cystitis and a suspected bladder mass. The kitten had had clinical signs of hematuria and stranguria since being acquired at 5 weeks of age and was presented to the referring veterinarian for suspected constipation and pollakiuria. Physical examination identified a suspected bladder mass and in-house urinalysis showed pyuria and hematuria. The kitten was prescribed amoxicillin/clavulanic acid at 16 mg/kg q12h (Noroclav; Norbrook) and meloxicam (Metacam; Boehringer Ingelheim) for 10 days without improvement. A urine culture via cystocentesis revealed >100,000 colony forming units (cfu) of
Four weeks later the kitten presented to Southpaws Specialty Surgery for persistent cystitis despite appropriate antibiotics. Upon clinical examination, the kitten was bright and alert. She was small for her age, but appropriately proportioned. Vital signs were within normal ranges. Her bladder was firm and semi-distended upon palpation. No other abnormalities were present. Abdominal ultrasound revealed pronounced, diffuse thickening of the bladder wall (up to 0.52 cm), most prominent within the trigone region (Figure 1). No discrete bladder masses or other abnormalities were identified on abdominal ultrasound. Urinalysis via cystocentesis revealed a urine specific gravity of 1.046 and a pH of 6, with 2+ protein and an active sediment of >1000 ×106/l red blood cells and >1000 ×106/l white blood cells, but no ketones, glucose, bilirubin or bacteria were seen microscopically. A urine culture was not performed at this time, and the amoxicillin/clavulanic acid was continued. Feline leukemia and feline immunodeficiency virus enzyme-linked immunosorbent assay were negative. As the kitten was too small for cystoscopy to acquire mucosal biopsies and examine the lower urinary tract, an exploratory celiotomy and ovariohysterectomy was performed. The goal of surgery was to acquire full thickness biopsies to further characterize the bladder wall thickening, with the intention to direct therapy to the underlying cause and allow more accurate prognostication. At surgery, the bladder mucosa was grossly thickened with soft plaque-like proliferations covering the entire mucosal surface. Full-thickness bladder wall samples were acquired for histopathology and aerobic bacterial culture with sensitivity. The ureteral insertions into the trigone region were normal, as was the remainder of the abdomen. The kitten was discharged the following day on amoxicillin/clavulanic acid and buprenorphine (Temgesic; Rickitt-Benkiser) sublingually for analgesia.

Ultrasound image of the bladder at the kitten’s initial visit to Southpaws Specialty Surgery for Animals. The bladder wall was diffusely thickened and irregular, measuring up to 0.52 cm in sections and most prominently in the trigone region (not shown). The bladder contained a moderate amount of urine (8 MHz, curvilinear probe)
Culture of the bladder wall grew

A low-power photomicrograph of the submucosa markedly infiltrated and expanded by diffuse sheet of large histiocytic cells, consistent with von Hansemann cells (hematoxylin and eosin stain)

A high-power photomicrograph of the submucosa revealing the von Hansemann cells containing abundant cytoplasm containing numerous fine periodic acid-Schiff (PAS)-positive granules. The von Hansemann cells have a low mitotic rate, are relatively uniform and have central nuclei with small or insignificant nucleoli (PAS stain)
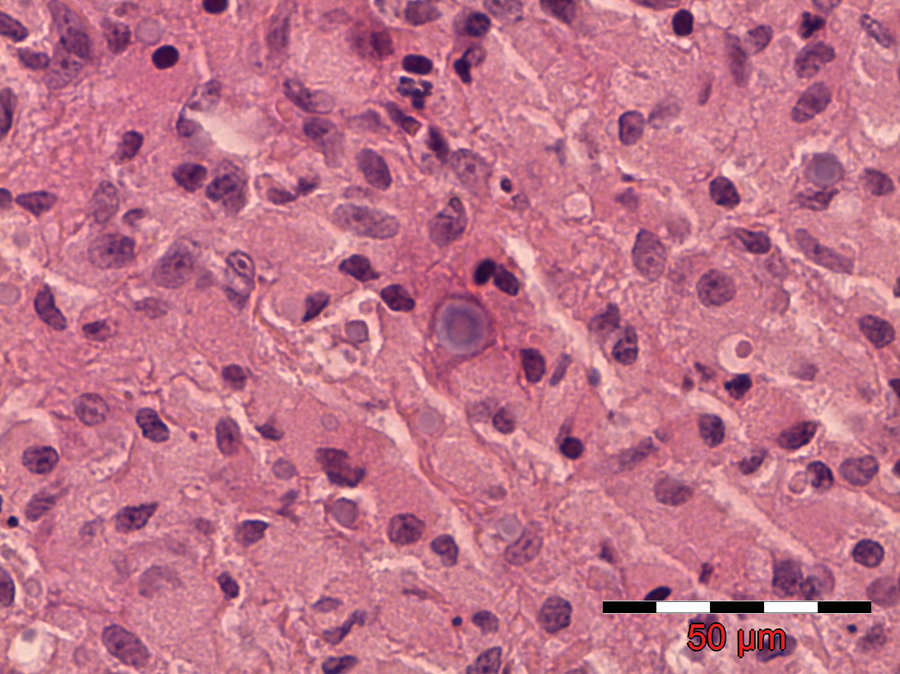
A high power hematoxylin and eosin-stained photomicrograph of Michaelis–Gutman bodies, which are basophilic, mineralized, targetoid concretions (5–10 µm in diameter) situated between the von Hansemann cells or within the cytoplasm

The Michaelis–Gutman bodies stain strongly for calcium with Von Kossa stain (high power, Von Kossa stain)

Fluorescent in situ hybridization of the bladder wall. The results reveal many inflammatory cells, but no invasive bacteria are seen
Hematuria, stranguria and pollakiuria persisted postoperatively and urine culture obtained via cystocentesis 12 days postoperatively revealed persistent growth of
Malakoplakia is an unusual chronic granulomatous inflammatory response most commonly affecting the genitourinary system, particularly the bladder. This condition has been well recognized in human patients since it was first identified in 1901 by von Hansemann and later described by Michaelis and Gutmann in 1902.1,2 The term malakoplakia was coined by von Hansemann in 1903 and derived from the Greek words meaning malakos (soft) and plakos (plaque) to describe the soft, yellow–brown mucosal plaques associated with the disease.1,2 There have been numerous cases (>400) in the human literature involving not only the bladder and kidneys, but the colon, prostate, lungs, tongue, skin and lymph nodes, with clinical signs varying according to the organ affected.2–6 Age of onset varies from juveniles to the middle-aged, with bladder lesions appearing to be more common in females.2,4,6 Malakoplakia is confirmed by the presence of pathognonomic microscopic mineralized target or ‘owl-eye’ cytoplasmic basophilic inclusions (calcospherites), known as Michaelis–Gutmann bodies (MG bodies) within large von Hansemann macrophages, which have abundant finely granular eosinophilic PAS-positive cytoplasm.1,7 The composition of MG bodies appears to be calcium, mucopolysaccharide and lipid, which probably represents calcification of undigested bacteria due to ineffective phagocytosis.1,6–9
The veterinary literature contains only pathological descriptions of the disease: three describing naturally-occurring disease, of which two involve incidental post mortem findings in pigs describing malakoplakia of the lymph nodes, and one with systemic involvement.10,11 Most recently, Bayley et al 12 described a kitten with a similar presentation to the one described herein. The kitten presented initially for stranguria and tenesmus which failed to respond to antibiotic therapy; the kitten was euthanased 3 weeks later. Malakoplakia of the bladder was identified at post-mortem examination. The present report provides valuable information on the management of patients with such a condition as malakoplakia.
The exact pathogenesis of malakoplakia remains obscure, but most agree that bacteria play a significant role in the development of the disease, but whether they act as primary or secondary agents is still debatable.
13
The majority of cases involving the urinary tract occur with coliform cystitis with or without concurrent immune suppression, and successful treatment of the infection generally results in resolution of the malakoplakia.4,5 A number of studies have identified
Malakoplakia bears some resemblance to granulomatous colitis in Boxers and leptomeninigitis in Beagles, which are also associated with
Prolonged antimicrobial therapy is the mainstay of treatment for malakoplakia. Antibiotics with the ability to penetrate macrophages, such as trimethoprim-sulfamethoxazole and fluoroquinolones, appear to have the most success, and treatment is less successful with beta-lactams, which do not penetrate the cells.5,9,15,16,20 Our kitten was treated initially for 4–6 weeks without improvement, with the most appropriate antibiotics based upon culture and sensitivity testing. Clinical improvement only occurred once treatment with TMS had started. Instillation of neosporin into the bladder and cholinergic agents, such as bethanechol, that increase the cyclic-GMP levels within macrophages have been used in humans with variable success.3,9,13,14,21 As the disease has been associated with immune suppression, discontinuation of immune suppressant drugs is generally recommended.4,6 Survival depends upon the organs affected and the extent of the lesion as there can be multiple organ involvement and subsequent organ failure (ie, hydronephrosis due to renal obstruction).3–5 Plaques within the bladder appear to be the most amenable to treatment, but other cases require a multimodal therapeutic approach, including systemic antibiotics and, potentially, surgery to remove large masses or re-routing of ureters.3–5,9,14
Conclusions
Malakoplakia is unusual in humans and greater awareness of it is needed in veterinary medicine, especially as its presentation may be similar to neoplasia. Treatment can be successful with prolonged antibiotics that penetrate macrophages.
Footnotes
Acknowledgements
The authors thank Lisa Lang and Dr Karen Brenner for their expert assistance in managing the kitten. The authors thank Dr Anthony Mutsaers for critical review of the manuscript.
Funding
The authors received no specific grant from any funding agency in the public, commercial or not-for-profit sectors for the preparation of this case report.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
