Abstract
Complications associated with the Stamey percutaneous loop cystostomy catheter (Cook Medical), including exposure of the most proximal side-hole and leakage of urine from the bladder, were encountered following percutaneous placement in three cats. In all cats, surgical exploration for removal of the catheter was performed.
Feline idiopathic cystitis (FIC) may be obstructive or non-obstructive in its presentation, but urethral obstruction is far more common in male cats, with no difference reported between intact and castrated males. 1 Persistently increased urethral closure pressures may result in recurrent urethral obstructions, even after the urethral plug is removed. 2 For cats with obstructive FIC that are azotemic or where there is concern for detrusor atony, an indwelling urethral catheter is recommended to maintain urine flow, allow appropriate diuresis and to keep the bladder small. Indwelling catheters require the cat be hospitalized and may contribute to urethral inflammation.3,4
A novel treatment protocol was proposed recently in male cats with obstructive FIC, whereby indwelling urethral catheters were not placed and cats were treated with analgesics and decompressive cystocentesis when needed. A successful outcome (appropriate urination within 72 h) was observed in 67% of the 12 cats. 5 Percutaneous cystostomy drainage using a tube inserted through the abdominal wall provides an alternative means of drainage that does not require catheterization of the urethra or repeated cystocentesis. Cystostomy tube placement may also be required in those cats where placement of a urethral catheter proves impossible owing to urethral obstruction, stricture or trauma. These tubes may be preferable to repeated decompressive cystocentesis and its associated bladder trauma. All previous reports of cystostomy tube placements in cats and dogs referred to surgical placement, including cystopexy in order to reduce the risk of leakage.6–11 However, percutaneous cystostomy is performed routinely in humans 12 and has been reported in goats,13,14 and hence has potential as an option for treating cats with obstructive FIC and urethral obstruction resulting from other conditions.
The Stamey percutaneous loop suprapubic catheter (Cook Medical) is commercially-available, easy to place and the locking loop mechanism (activated by tightening a string running from the catheter tip to the hub; Figure 1) is designed to ensure the catheter end and drainage holes are retained within the bladder. 15 We observed varying results in clinical patients following placement of cystostomy tubes and therefore commenced a clinical trial to evaluate the efficacy of percutaneous cystostomy catheters for management of cats with obstructive FIC in order to ascertain if cats with cystostomy tubes to have fewer recurrent obstructive episodes compared to those with traditional urethral catheter placements. This communication documents our observations in a clinical patient identified by retrospective review of our medical record database, the first cat enrolled in the aforementioned clinical trial, and a third research cat in which we evaluated the intravesicular pressure required to produce leakage following placement of the Stamey catheter.
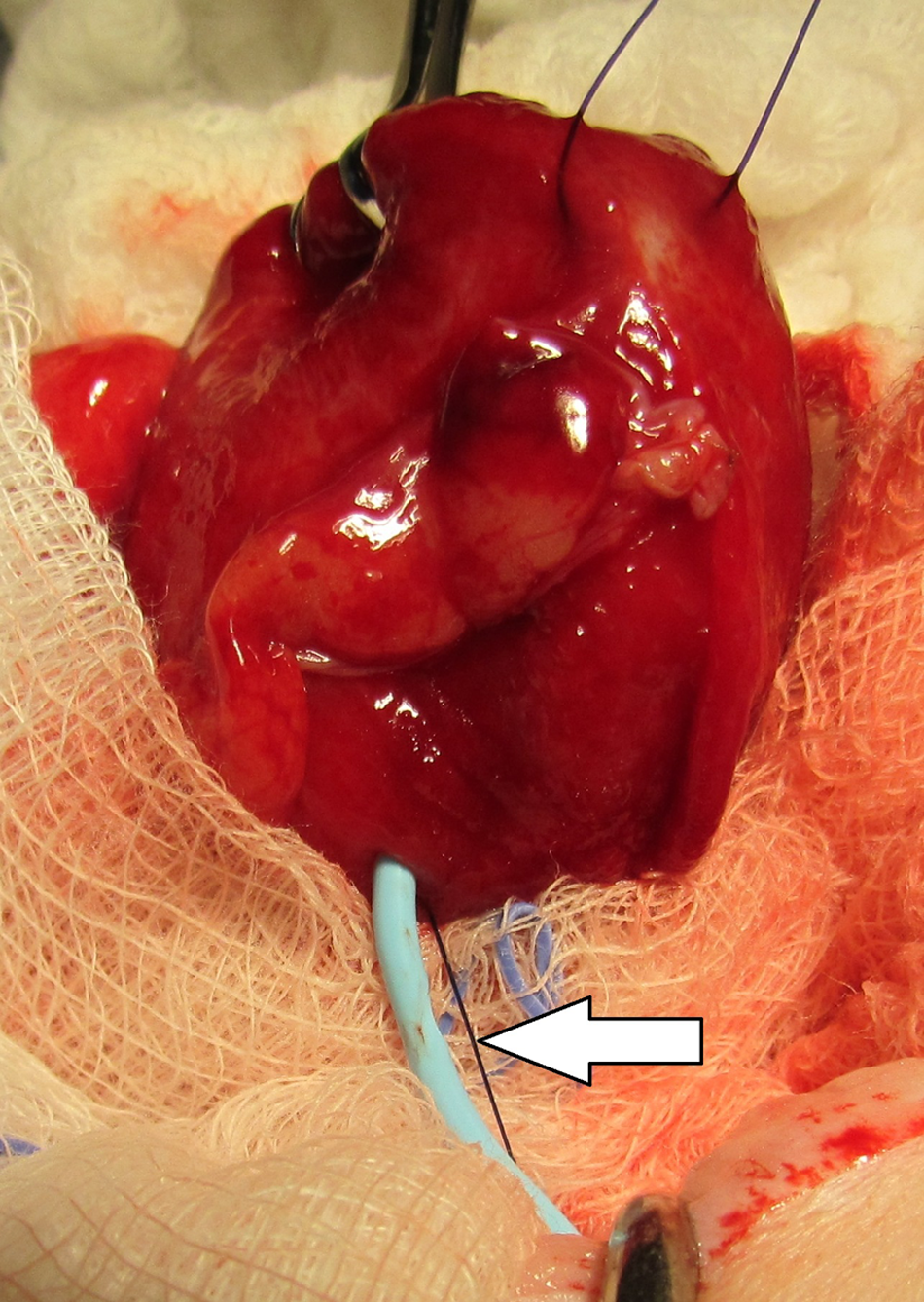
Intraoperative view in case 1. The Stamey locking loop catheter has withdrawn partially from the bladder, exposing the most proximal side-hole and the black suture used to activate the locking loop mechanism (white arrow)
Case descriptions
Case 1
An 8-year-old male neutered Siamese cat (5.5 kg) was presented for straining to urinate. He passed a small amount of blood-stained urine during physical examination, but it was not possible to express his bladder. Abdominal radiographs revealed multiple radiodense cystic calculi. Anesthesia was induced with midazolam (1.8 mg IV) and ketamine (50 mg IV), and supplemental oxygen was administered by face mask. A 3.5-French polypropylene catheter (Covidien) was placed through the urethra into the bladder with difficulty, after multiple attempts. Once the bladder was emptied (approximately 60 ml of markedly bloody urine), attempts to pass a 3.5-French red rubber catheter (Bard Medical) were unsuccessful. The 3.5-French polypropylene catheter could not be reinserted into the bladder, so an 8-French Stamey percutaneous loop suprapubic catheter (Cook) was placed aseptically through the right abdominal wall and secured to the skin. The catheter was connected to a closed collection system. Mild peritoneal effusion was detected sonographically following the procedure, but the cat recovered uneventfully. The following morning, the cat was anesthetized once again and a urethrogram revealed rupture of the penile urethra, multiple small filling defects within the urethra, bladder mucosal irregularity and filling defects within the urinary bladder. A celiotomy was performed for the purpose of removing the cystic calculi by cystostomy and evaluating the lower urinary tract. When the abdomen was explored, a small amount of blood-stained fluid was present and it was immediately obvious the Stamey catheter had partially dislodged from the bladder such that the proximal side-hole had migrated into the peritoneal cavity. The cystostomy catheter was withdrawn and the small exit hole in the bladder wall closed with a cruciate suture of 4-0 polydioxanone. A perineal urethrostomy was also performed. This cat recovered uneventfully following surgery.
Case 2 (first cat enrolled in clinical trial)
A 4-year-old male castrated domestic longhair cat (7 kg) was presented for hematuria. On physical examination, a large (8 cm diameter), firm, painful and non-expressible urinary bladder was palpated. Sandy material was noted around the penis. An abdominal radiograph revealed no obvious urethral or cystic calculi, and obstructive FIC was suspected as the cause of the urethral obstruction. The cat was sedated with 0.4 mg oxymorphone and 1.4 mg midazolam intravenously (IV), and supplemental oxygen was administered via a face mask. Propofol was titrated to effect to maintain sedation. Urethral obstruction was relieved by flushing sterile saline through a 3.5-French open-ended polypropylene catheter (Covidien), which was introduced into the urethra and then removed. In order to avoid the necessity for maintenance of an indwelling urethral catheter, a 6-French Stamey percutaneous loop suprapubic catheter (Cook Medical) was placed under fluoroscopic guidance, using an 0.032-inch through-the-catheter guide wire, and its position within the bladder confirmed by injection of radio-opaque contrast. The catheter was secured to the skin of the abdomen using a finger trap suture of 4-0 nylon and connected to a closed collection system for continuous drainage of the bladder.
The cat recovered well until 24 h after placement of the cystostomy tube. At that time the catheter was closed to allow the bladder to fill in order to see if the cat could void in a normal fashion. However, when this was done, serosanguinous fluid began leaking from the cystostomy tube entrance site; an abdominal ultrasound confirmed free intra-abdominal fluid and a pocket of subcutaneous fluid surrounding the cystostomy tube.
uroperitoneum and partial migration of the Stamey catheter out of the bladder allowing urine leakage through the proximal side-hole (Figure 1). The catheter was removed and the entry hole repaired with a single suture of 4-0 polydioxanone. The cat’s recovery was uneventful and was managed with analgesics, diet and environmental therapy.
Case 3
Owing to the problems associated with the first case in our clinical trial, a research trial was commenced with the aim of evaluating the safety of Stamey catheter placement and withdrawal, with emphasis on the bladder pressure that may be needed to produce leakage of urine while the tube was in place and after it was removed. The subject was a 12-month-old female domestic shorthair cat (2.9 kg). The cat was premedicated with 0.15 mg oxymorphone and 0.06 mg atropine subcutaneously. Anesthesia was then induced with 15 mg/kg propofol in combination with 0.75 mg/kg midazolam IV and endotracheal intubation performed. Anesthesia was maintained with a non-rebreathing circuit connected to the endotracheal tube and isoflurane 1–5% in 100% oxygen.
A 6-French Stamey percutaneous loop suprapubic catheter (Cook Medical) was placed according to the manufacturer’s instructions and its position within the bladder was confirmed by injection of a radiographic contrast agent. The locking loop mechanism was then engaged and injection of radiographic contrast repeated. At this stage, contrast material could be seen escaping from the bladder into the peritoneal cavity. The locking loop mechanism was disengaged and an attempt made to withdraw the catheter from the abdomen prior to making another attempt at placement. Substantial resistance to removal of the catheter was encountered; therefore, a small surgical incision was made into the abdomen. The catheter was then withdrawn and it became apparent that the locking loop mechanism had snared the omentum (Figure 2). The catheter was released from the omentum and placed directly into the bladder for pressure testing.

Photograph of case 3 following incision into the abdominal wall to allow withdrawal of the Stamey catheter. The locking loop mechanism has snagged omentum (white arrow), preventing easy removal
A 6-French double lumen urinary catheter (Cook Urologic) was placed into the urethra so the tip of the catheter was situated just cranial to the urinary trigone. This catheter was then connected to pressure transducers to obtain intravesical pressures (Urivision Janus V Urodynamic System). Sterile water was infused at 4 ml/min and the tube site was evaluated for leakage. Sterile water was observed leaking from the insertion site around the catheter before the cystic pressure had reached 30 mmHg. In addition, gentle traction on the catheter allowed dislodgement of the proximal side-hole out of the abdomen with subsequent brisk leakage (Figure 3). The bladder was then emptied, the catheter removed and saline infusion repeated. Leakage from the catheter insertion site was observed at an intravesicular pressure between 15 and 17 mmHg.

Photograph of Stamey percutaneous loop cystostomy catheter in situ during urodynamic evaluation in case 3. Gentle traction on the catheter causes dislodgement, with exposure of the most proximal side-hole and locking loop suture, allowing drainage of urine from the bladder (white arrow) into the peritoneal cavity or tissues of the abdominal wall
Discussion
Our experience in these three cats leads us to conclude that the locking loop-style catheter has a high risk of partial dislodgement from the bladder in cats when used in the method described in this report. This risk is compounded by the fact that the catheter remains partially contained within the bladder while the proximal side-hole is exposed, thereby creating a conduit which allows urine leakage from the bladder into the peritoneal cavity or subcutaneous tissues. The leakage arising from this catheter complication is unlikely to resolve spontaneously, which might otherwise occur if the cystostomy tube migrated completely out of the bladder and the hole could ‘self-seal’. The risk might be ameliorated by securing the cystostomy tube in a position that does not allow it to pull out of the bladder when traction is applied or the position of the bladder changes in relation to the body wall, but this is difficult when placing it percutaneously and defeats the purpose of using a non-surgical approach. Our observations in case 3 are also cause for concern as there seems to be potential for the catheter snagging intra-abdominal structures, such as omentum, leading to the necessity of a surgical approach to remove the catheter, and for leakage at relatively low pressures both while the catheter is in situ and also following its removal. Normal maximum detrusor pressures in cats range from 68 to 73 mmHg, depending on the level of sedation.16,17 Urine leakage was observed at substantially lower pressures in case 3 of this study, raising concerns as to the safety of either leaving the Stamey catheter in place when allowing the bladder to fill to observe normal micturition or following acute withdrawal.
Conclusions
In light of these observations, we encourage clinicians to use extreme caution when considering the percutaneous locking loop-style cystostomy catheter in cats. The successful use of cystostomy tubes in cats with urethral obstruction would be contingent on further clinical evaluation of a variety of commercially-available catheters. Investigation of modifications to either catheter design or insertion methods that reduce the risk of dislodgement and leakage is warranted.
Footnotes
Funding
Treatment of case 2 and the research study described in case 3 was supported financially by a grant from the Companion Animal Health Foundation, University of California-Davis.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
