Abstract
Healthy cats of three cat breeds — Sphynx (n = 11), British Shorthair (n = 15) and Ragdoll (n = 15) — were included in this study. All cats underwent an ultrasonographic examination to assess renal length, cortical thickness, medullary thickness and corticomedullary ratio. Of all ultrasonographic measurements, renal length showed the highest variation. For all ultrasonographic dimensions, individual and kidney side (left vs right) variation were much more pronounced than interbreed variation. Sphynx cats tended to have larger kidneys (4.09 ± 0.33 cm) than British Shorthair (3.77 ± 0.43 cm) and Ragdoll cats (3.87 ± 0.41 cm). British Shorthair cats, however, tended to have a thinner cortex (0.67 ± 0.13 cm) and medulla (0.76 ± 0.18 cm) than Sphynx (0.76 ± 0.14 cm and 0.90 ± 0.25 cm, respectively) and Ragdoll cats (0.75 ± 0.13 cm and 0.91 ± 0.22 cm, respectively). However, statistical tests did not reveal significant differences between these cat breeds. The corticomedullary ratio was similar for the three cat breeds (Sphynx: 0.93 ± 0.43; British Shorthair: 0.91 ± 0.26; Ragdoll: 0.88 ± 0.31). The left kidney (3.83 ± 0.42 cm) was significantly smaller than the right kidney (3.99 ± 0.40 cm) and showed a thicker medulla (left: 0.93 ± 0.21 cm, right: 0.79 ± 0.22 cm), and thus a lower corticomedullary ratio (left: 0.80 ± 0.23, right: 1.01 ± 0.32). For the cortical thickness, no significant difference was observed between the left (0.71 ± 0.14 cm) and right kidney (0.74 ± 0.14 cm).
Introduction
Kidney disease is a common health problem in cats. Ultrasonography is a useful diagnostic imaging modality in the investigation of kidney disease as it can provide important anatomical information concerning the renal size, shape and internal architecture.1 –4
Kidney disease can cause changes in renal size, shape and contour. Alterations in renal size may precede other ultrasonographical changes and can be helpful to differentiate acute kidney injury (AKI) from chronic kidney disease (CKD). In general, reduced renal size suggests the presence of CKD, whereas increased renal size is compatible with AKI.5 –9 Renal length can be measured on ultrasound with a high degree of reproducibility in both humans and dogs,10 –12 and is found to be a good reflection of the true length, although there seems to be a tendency for renal length to be underestimated.10,13 Nevertheless, there is one study in cats where no significant difference could be found between sonographic and actual (necropsy) renal measurements. 14 In dogs and cats, general values for organ measurement (ie, renal length) have showed their limits. These limiting factors include imprecise definition of borders, difficulty in achieving standard planes of section, the need to extrapolate measurements for objects which extend outside the sector and scanning artifacts.10,13 Previous studies in dogs and cats revealed that renal length is influenced by different variables such as gender, age and bodyweight.5,6,14 –16 In dogs a positive relationship exists between renal length and bodyweight.5,17 Although there is less variation in body size between different cat breeds, it has been suggested that Persian cats and Chinchillas tend to have smaller renal lengths. 18
In veterinary medicine, little attention is given to cortical and medullary thickness. Only a few studies describe the cortical and medullary thickness of normal feline kidneys.13,14 In human medicine, cortical thickness is used to differentiate between CKD and AKI. Cortical thinning is often present in the case of CKD and can precede changes in kidney size.9,19,20 Moreover, cortical thickness may be related more closely to estimated glomerular filtration ratio than renal length in patients with chronic kidney disease. 20 It is not known if this is also true for veterinary patients. Previous studies performed in humans indicated that cortical thickness is also influenced by many variables such as age, gender, race, bodyweight/body mass and height.12,19,21 Therefore, it is necessary to establish normal values in healthy cats before cortical thickness and corticomedullary ratio can be used for the ultrasonographic detection of kidney disease.
The purpose of this study was to determine renal length, cortical thickness, medullary thickness and corticomedullary (CM) ratio in healthy cats, and to evaluate if a significant difference exists between cat breeds concerning these dimensions.
Materials and methods
Three different cat breeds were involved in this study: 15 Ragdoll (three intact males, two neutered males and 10 intact females) with a mean age of 30 months (range 12–75 months) and a mean weight of 4.08 kg (3.0–5.1 kg); 15 British Shorthair (six intact males, two neutered males and seven intact females) with a mean age of 34 months (range 11–78 months) and a mean weight of 3.98 kg (range 2.85–5.65 kg); and 11 Sphynx (five neutered males, five intact females and one neutered female) with a mean age of 27 months (range 9–47 months) and a mean weight of 3.79 kg (range 2.7–4.6). Only healthy cats were included. The cats were considered healthy on the basis of physical examination, routine urinalysis (urine specific gravity, urinary protein-to-creatinine ratio, urinary sediment and urinary dipstick) and blood examination (serum creatinine concentrations and serum urea nitrogen concentrations) and absence of ultrasonographic abnormalities at the level of the kidneys. The presence of hyperechoic cortices and/or medullary rim signs was considered as a normal finding if no other abnormalities were observed.
All cats underwent an ultrasonographic examination without sedation. The cats were placed in dorsal recumbency and were restrained manually by two assistants. The hair was not clipped because most cats were show cats. Alcohol and acoustic coupling gel were used to provide an appropriate acoustic contact between the transducer and the skin. High frequency (7.5–12 MHz) linear transducers (iU22; Philips) were used for scanning. For each kidney, the radiologist was asked to obtain sagittal, dorsal and transverse scans. The length (in cm) was determined on the sagittal sonogram (Figure 1), which is characterised by the appearance of two bright parallel bars formed by cross-sectioned pelvic diverticula. The cortical and medullary thickness (in cm) were defined as the distances from the renal margin to the corticomedullary junction, the interface between the hyperechoic renal cortex and hypoechoic renal medulla, and from the corticomedullary junction to the renal pelvis, respectively. The edge of the renal pelvis was defined as being the interface between the hypoechoic medulla and the hyperechoic renal pelvis fat. The measurements were taken from a dorsal sonogram in the middle of the kidney and were orientated perpendicular to the long axis (Figure 2). When a medullary rim sign was present, it was included in the measurements of the medulla.

Sagittal sonogram of a normal feline kidney. The green line from 1 to 2 shows the measurement of the renal length

Dorsal sonogram of a normal feline kidney. The green lines indicate the measurements of the cortical (1–2) and the medullary thickness (3–4)
Afterwards, the CM ratio was calculated as being the ratio of the cortical and medullary thickness. To assess the variability linked with the cat breed (interbreed variation), the individual cat within the same breed (individual variation) and the two (left vs right) kidneys within the cat (kidney side variation) a random effects model with cat nested in breed and kidney nested in cat was fitted and variance components for each of the three sources of variation were obtained by restricted maximum likelihood. Further analysis was based on a linear mixed model with animal as random effect, and breed and kidney side as categorical fixed effects. F-tests, at a global significance level of 5%, were performed to evaluate if renal length, cortical thickness, medullary thickness and CM ratio were significantly influenced by the cat breed and/or side (left vs right) of the kidney.
Results
The ultrasonographic findings of the three different cat breeds are summarised in Table 1.
Ultrasonographic dimensions of the Ragdoll, British Shorthair and Sphynx

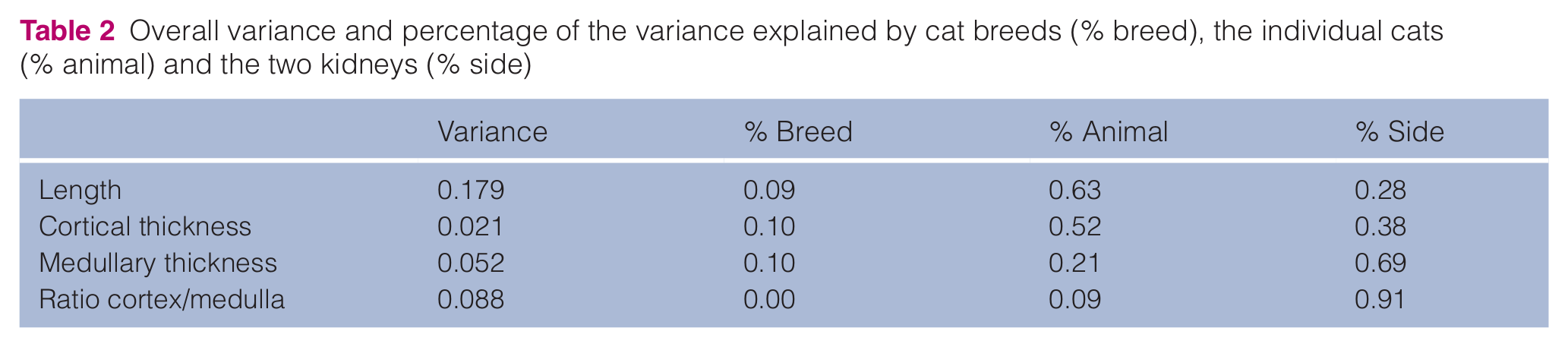
The highest variation was observed for the renal length (0.179), followed by the CM ratio (0.088), and the medullary (0.052) and cortical thickness (0.021). The CM ratio showed the lowest interbreed variation percentage (0.000). For all ultrasonographic measurements the individual and kidney side variation was much more pronounced than the interbreed variation (Table 2), which means that when two cats of a different breed are considered, the variation between those cats is caused mainly by individual variation and less by interbreed variation.
Overall variance and percentage of the variance explained by cat breeds (% breed), the individual cats (% animal) and the two kidneys (% side)
None of the ultrasonographic measurements — renal length, cortical thickness, medullary thickness and CM ratio — revealed significant differences (P >0.01) between the three cat breeds. Nevertheless, Sphynx cats (4.09 ± 0.33 cm) tended to have larger kidneys than British Shorthair (3.77 ± 0.43 cm) and Ragdoll cats (3.87 ± 0.41 cm). British Shorthair, however, tended to have a smaller cortical (0.67 ± 0.13 cm) and medullary (0.76 ± 0.18 cm) thickness than Sphynx (0.76 ± 0.14 cm respectively 0.90 ± 0.25 cm) and Ragdoll cats (0.75 ± 0.13 cm respectively 0.91 ± 0.22 cm). The CM ratio was similar for the three cat breeds (Sphynx: 0.93 ± 0.34, British Shorthair: 0.91 ± 0.26, Ragdoll: 0.88 ± 0.31).
Contrary to the cat breed, the side (left vs right) of the kidney had a significant influence on the renal length (P <0.001), the medullary thickness (P <0.001) and the CM ratio (P <0.001), but not on the cortical thickness (P = 0.19). The left kidney (3.83 ± 0.42 cm), in general, was smaller than the right kidney (3.99 ± 0.40 cm) and showed a thicker medulla (left: 0.93 ± 0.21 cm, right: 0.79 ± 0.22 cm) and thus a lower CM ratio (left: 0.80 ± 0.23, right: 1.01 ± 0.32) (Table 3). The cortical thickness was similar for the left (0.71 ± 0.14 cm) and right (0.74 ± 0.14 cm) kidney (Table 3).
Ultrasonographic dimensions (cm) of the left and right kidney

Discussion
Previous ultrasonographic studies reported that the normal size of the feline kidney varies between 3.0 and 4.3 cm, and can even be up to 5.3 cm.4,13,14,17,22,23 This corresponds to our results where the mean renal length was 4.09 ± 0.33 cm in Sphynx, 3.77 ± 0.43 cm in British Shorthair and 3.87 ± 0.41 cm in Ragdolls.
No significant differences could be found between the different cat breeds. Although we tried to have three comparable groups concerning the age, bodyweight and gender, differences were still present between the three cat breeds. It is mentioned in the literature, that feline kidneys become smaller with advancing age, while neutered cats have somewhat smaller kidneys than intact cats, and male cats have larger kidneys than females.6,15,16 Unfortunately, owing to the low number of cats, statistical comparison was not possible and the influence of age, gender and bodyweight on our results cannot be excluded.
Normally, both kidneys should be of similar size.13,17,24 However, in previous studies in dogs and cats, the right kidney was often larger than the left kidney.5,6,22 This corresponds to our results where the right kidney (3.99 ± 0.40 cm) was significantly longer than the left one (3.83 ± 0.42 cm). In humans, Kubodera et al did not find a significant size difference in cortical thickness, medullary thickness or the CM ratio. 21 This is in contrast to our results where the left kidney had a thicker medulla and lower CM ratio than the right one. The difference in measurements between the left and right kidneys is probably a result of technical limitations. True sagittal, dorsal and transverse scans are very important, as measurement of renal geometric determinants may be inaccurate if the beam slices the kidney at an oblique angle.1,15 Sometimes the caudal and cranial poles of the kidney are difficult to visualise. 14 Feline kidneys are also extremely mobile. Mild pressure on the abdominal wall by the transducer may be sufficient to displace the kidney or to alter its orientation. This is true mainly for the left one, as the right kidney sits in the renal fossa of the caudate lobe of the liver and is therefore less mobile. The right kidney, however, is traversed on its ventral surface by the descending duodenum and ascending colon.1,4,24 Gas within these bowels reflects sound and prevents transmission to and from the kidney. For practical purposes, the dimensions of the left and right kidneys in cats can be considered to be similar.
The cortical thickness of healthy cats measures 0.2–0.5 cm on the basis of anatomic evaluations. 13 This does not correlate with our findings where the mean cortical thickness measured 0.76 ± 0.14 cm in Sphynx, 0.67 ± 0.13 cm in British Shorthairs and 0.75 ± 0.13 cm in Ragdolls. These values are in between the results of Park et al 14 (0.47 ± 0.08 cm) and Walter et al 13 (0.82 ± 0.14 cm). Why British Shorthair cats tended to have thinner cortices, though the difference was not significant, is unclear. A limitation of our study is that the cats were not clipped, especially for the British Shorthair cats, which had very thick hair coat. We used plenty of water, alcohol and acoustic coupling gel to provide an appropriate acoustic contact between the transducer and the skin, and to avoid a significant loss of acoustic transmission. Nevertheless, we cannot exclude that this would not have had an influence on the accuracy of our measurements. Further anatomical/histological research is needed to evaluate the relevance of having a thicker versus thinner cortex in healthy cats. The results we obtained for the medullary thickness (Sphynx: 0.90 ± 0.25 cm; British Shorthair: 0.76 ± 0.18 cm; Ragdoll: 0.91 ± 0.25 cm) were higher than the results found by Walter et al 13 (0.59 ± 0.06) and Park et al 14 (left kidney: 0.55 ± 0.07; right kidney: 0.50 ± 0.07). These authors determined the cortical and medullary thickness on a sagittal sonogram, whereas we used a dorsal sonogram. As kidney width (sagittal plane) differs from kidney height (dorsal plane), the scan plane used to perform the measurements has an influence on the measurements of the cortical and medullary thickness. In addition, the dimensions of the renal medulla are not always clearly defined owing to the unclear borders of renal sinuses. In our study seven cats (three Ragdolls, two British Shorthairs and two Sphynxs) showed a medullary rim sign. Cats with a medullary rim sign are expected to show a thicker medulla as this sign was included in the measurements of the medulla. In our study, cats with a medullary rim sign had a mean medullary thickness of 1.00 ± 0.26 cm, whereas cats without a medullary rim sign had a mean medullary thickness of 0.82 ± 0.21 cm.
Although cortical thickness, medullary thickness and CM ratio showed a wide range, overall variance was lower than for renal length. Renal length is not only a larger numerical measurement but, as already mentioned, is also influenced by many factors, such as age, gender and bodyweight.6,15 These factors can cause not only individual, but also interbreed, variation as there exists small (ie, Singapura) and larger (ie, Maine Coon) breeds of cats. Further research is necessary to evaluate the influence of these parameters on the CM ratio.
Conclusions
Although mild differences in renal length and CM ratio were present between the cat breeds tested, significant differences could not be established. The only significant differences were in kidney size.
Footnotes
Funding
This research received no grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
