Abstract
A 10-year-old domestic shorthair cat showed anorexia, lethargy and ptyalism with hyperammonaemia. Portosystemic shunts were not identified by computed tomography angiography. Biopsy results revealed mild interstinal nephritis and no lesion in the liver. Analysis of urine revealed the presence of a high methylmalonic acid (MMA) concentration. Serum cobalamin (vitamin B12) and serum feline trypsin-like immunoreactivity levels were also markedly low. The cat was diagnosed as having exocrine pancreatic insufficiency (EPI). After 5 weeks of parenteral cobalamin supplementation, serum cobalamin concentration had increased and urinary MMA concentration had decreased. This case suggests that hyperammonaemia may be caused by accumulation of MMA due to cobalamin malabsorption secondary to feline EPI.
Case Report
Hyperammonaemia is one of the major neurotoxins in the complex pathogenesis of hepatic encephalopathy and is associated exclusively with compromised liver function, such as portosystemic shunts or hepatic insufficiency. Hyperammonaemia has been reported rarely in dogs with urea cycle enzyme deficiency,1,2 metabolic disorder of organic acids or hereditary selective cobalamin (vitamin B12) malabsorption.3,4 In cats, there have been only a few case reports of urea cycle enzyme deficiency 5 and cobalamin malabsorption. 6 This report indicates that accumulation of organic acids due to cobalamin malabsorption caused hyperammonaemia secondary to exocrine pancreatic insufficiency (EPI) in a cat.
A 10-year-old neutered male domestic shorthair cat was presented to the Mitsuke Animal Hospital with a 1-month history of anorexia and weight loss. On physical examination, the cat was underweight (3.5 kg, body condition score of 1/5) and was estimated to be 5% dehydrated. Serum biochemistry results revealed an increase in blood urea nitrogen (BUN) [14.3 mmol/l; reference interval (RI) 6.2–11.7 mmol/l] and in calcium (3.0 mmol/l; RI 2.2–2.9 mmol/l). Other blood chemistries, a complete blood count and serum total thyroxine concentration were within the RI. Feline immunodeficiency virus antibody and feline leukaemia virus antigen tests were negative. Ultrasonographic examination of the kidney revealed small renal stones.
First, the cat was treated for dehydration with subcutaneous crystalloid fluids (lactated Ringer solution, twice a week), but it still showed lethargy and ptyalism 39 days after the first visit in the hospital. At that time, biochemical results showed increases in serum alanine aminotransferase (ALT) (236 U/l; RI 22–84 U/l) and in blood ammonia (213 µmol/l; RI 13–45 µmol/l) concentrations. Results of the blood count were within the RI. The cat was treated with intravenous (IV) crystalloid fluids (Ringer solution) and ampicillin (20 mg/kg, IV, q12 h), lactulose (1 ml, PO, q12h) and protein-restricted dry food (Renal Support Diet; Royal Canin) for treatment of hyperammonaemia. The dry food was composed of 24.3% protein, 18.0% lipid, 4.9% fibre, 0.6% calcium and 0.3% phosphorus. After 7 days the condition of the cat had improved and feeding blood ammonia concentration was within the RI. Although lactulose (1 ml, PO, q12h) and protein-restricted food were continued at home, the cat, again, showed lethargy and pytalism on day 74. Biochemical results showed increases in serum ALT (486 U/l) and in blood ammonia (249 µmol/l) concentrations, but the numerical data of the blood count were within the RI. The cat was treated with IV crystalloid fluids (Ringer solution) and ampicillin (20 mg/kg, IV, q12h). After 3 days the condition of the cat had improved and so a feeding tolerance test was performed as follows: after 12 h of the last food and medication, wet food (25 g, Recovery Period Support; Royal Canin) composed of 49.0% protein, 25.5% lipid, 7.8% fibre, 1.3% calcium and 1.1% phosphorus was provided to the cat. Blood samples were obtained before (fasted) and 2 h after feeding, and blood ammonia concentrations were determined using a dry chemistry system (Fuji dry-chem 4000; Fujifilm). Fasted blood ammonia (39 µmol/l) increased markedly after feeding (89 µmol/l). Serum bile acid (2.7 µmol/l) also increased after feeding (13.8 µmol/l), but its concentration was within the RI (<20 µmol/l). Differential diagnoses considered at this point were portosystemic shunts, hepatic insufficiency, urea cycle enzyme deficiency and metabolic disorder of organic acids.
To determine whether portosystemic shunts existed or not, computed tomography (CT) angiography was performed. No shunt vessels were detected by CT angiography observation. Both prothrombin time (11.3 s; RI 9.3–11.3 s) and activated partial thromboplastin time (31.2 s; RI 20.0–42.0 s) were within the RI. Biopsy samples were obtained from the liver and kidney by exploratory laparotomy. Histopathological examination of the tissues revealed mild interstinal nephritis and no lesion in the liver. Gas chromatography-mass spectrometry (GC-MS) analysis was performed, using a urine sample, to elucidate the possible metabolic disturbance. GC-MS analysis of organic acids in urine indicated the presence of high levels of methylmalonic acid (MMA) (MMA/creatinine: 2.02; RI <0.003) and methylcitric acid (methylcitric acid/creatinine: 0.29; RI <0.001). At that time, a mild proteinuria (30 mg/dl) was found with a urine reagent strip (Uricheck; Godo Shusei). Urine pH was 6.0 and specific gravity was 1.030. At this time, blood count data showed a decrease in red blood cells (5.5 millions/µl, RI 6.0–10.2 millions/µl) and in haematocrit (27.6%; RI 29–48%). However, mean cell volume (50.2 fl; RI 41.5–52.5 fl) and mean cell haemoglobin concentration (33.0 g/dl; RI 30.0–33.5 g/dl) were normal. Blood ammonia concentration (35 µmol/l) was also within the RI. After obtaining the result of the GC-MS analysis, serum cobalamin concentration determined by chemiluminescent enzyme immunoassay (Monolis) and was found to be markedly low (76 ng/l; RI 200–840 ng/l). Moreover, serum feline trypsin-like immunoreactivity (fTLI) concentration determined by radioimmunoassay (Idexx Laboratories) was also low (2.2 µg/l; RI 12.0–82.0 µg/l). On the basis of these findings, the cat was diagnosed as having EPI.
While the fTLI test result was pending, treatment for hypocoblaminaemia was started. Hydroxocobalamin (250 µg, Fresmin; Ajinomoto) was injected subcutaneously once a week for 5 weeks, every other week for 10 weeks, and once a month for 4 months. According to the result of the fTLI test, additional treatment with dried powdered extracts of porcine pancreas (one teaspoon for a meal, Pancreatin; Nipro Pharma) was started. Serum cobalamin concentration increased dramatically after 5 weeks of parenteral cobalamin supplementation and urinary MMA concentration decreased toward the RI (Figure 1a). However, serum cobalamin concentration (204 ng/l) was at the lower end of the RI after discontinuation of parenteral cobalamin supplementation for 3 months (Figure 1a); parenteral administration of hydroxocobalamin at a dose of 250 µg once a month was continued after that. Haematocrit increased and hyperammonaemia was not observed after parenteral cobalamin supplementation (Figure 1b). Body weight increased after parenteral cobalamin and pancreatic enzyme supplementations (Figure 1c). At follow-up 9 months after monthly administration of hydroxocobalamin (250 µg, SC) was started, serum cobalamin (600 ng/l) and blood ammonia (15 µmol/l) concentrations, and haematocrit (34.8%) were within the RI.
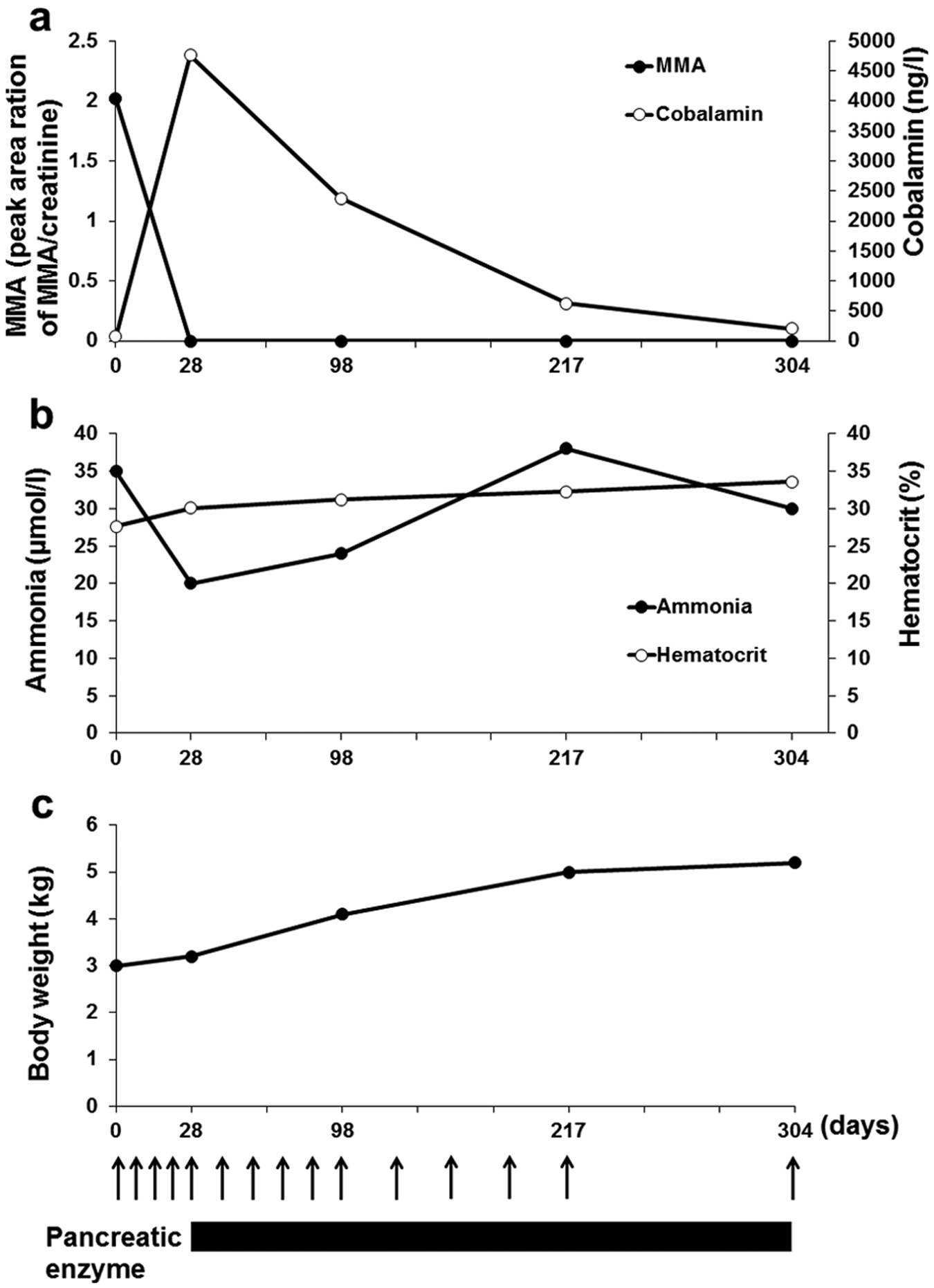
Changes after parenteral injection of hydroxocobalamin (250 µg, SC, arrow) and pancreatic enzyme application in the meal (one teaspoon for a meal, black horizontal bar). x-axis indicates days after starting clinical treatment. (a) y-axis indicates concentrations of urinary methylmalonic acid (MMA) [reference interval (RI) <0.003] and serum cobalamin (RI 200–840 ng/l). (b) y-axis indicates blood ammonia (RI 13–45 µmol/l) concentration and haematocrit (RI 29–48%). (c) y-axis indicates body weight
A diagnosis of EPI was made on the basis of low serum fTLI concentration, which is a specific marker for EPI in cats. 7 Although the marked clinical sign of feline EPI is weight loss followed by diarrhoea, polyphagia and vomiting, 8 the cat in the present case had no clinical signs of digestive disorders. Thompson et al also reported that weight loss was a clinical sign in only 19% of cats with EPI, 9 but that the case of weight loss increased depending on the age of cats. In senior aged cats, the weight loss was observed in 70% of cases of EPI. 9 Therefore, EPI should be considered as a differential diagnosis in senior cats with weight loss.
Analysis of organic acids in urine revealed the presence of methylmalonic aciduria. Methylmalonic aciduria have been reported in cats with cobalamin malabsorption.6,10 Cobalamin is an essential cofactor for the activity of methylmalonyl-CoA mutase and methionine synthase. 11 Methylmalonyl-CoA mutase converts methylmalonyl-CoA to succinyl-CoA, which may subsequently enter the tricarboxylic acid cycle. Therefore, deficiency of cobalamin is associated with reduced activity of these enzyme systems and the resulting alteration in urinary concentrations of the products of these enzyme systems.
Urinary MMA concentration decreased dramatically after parenteral administration of cobalamin. This result is consistent with that reported in cats with small intestinal disease and hypocobalaminaemia. 12 The decrease in MMA indicates that the extremely low serum cobalamin concentration reflected a cobalamin deficiency state. Cobalamin absorption requires the formation of cobalamin-intrinsic factor complexes. Intrinsic factor is a cobalamin-binding protein that is only produced by the pancreas in cats and is necessary for ileal absorption of cobalamin. 13 Thus, cats are prone to cobalamin malabsorption secondary to EPI due to insufficient production and secretion of intrinsic factor.7,9
Hyperammonaemia was not identified after decrease in urinary MMA concentration, suggesting that hyperammonaemia was caused by accumulation of MMA. However, the relationship between the blood concentration of ammonia and the urinary concentration of MMA is currently unknown. As the mechanism for hyperammonaemia is methylmalonic acidaemia, it has been proposed that the accumulation of excessive amounts of MMA results in indirect inhibition of the urea cycle through the decreased synthesis of N-acetylglutamate — an essential activator of the carbamyl phosphate synthetase 1 reaction.14,15
The increases in serum ALT were associated with hyperammonaemia. This may reflect functional damage of hepatocytes secondary to accumulation of MMA although histopathological examination did not detect observable damage in the liver. Although elevations of hepatic enzymes have been reported,7,9 the results of serum biochemical test are generally unremarkable in EPI.
Normocytic normochromic anaemia was identified in the cat in the present case and hematocrit increased after parenteral cobalamin supplementation. It is suggested that anaemia might be influenced by serum cobalamin concentration. Similar to the present report, normocytic normochromic anaemia has been identified in 46% cats with EPI and inflammatory lesions of EPI accompanying diseases were speculated as a cause for anaemia. 9 The effects of cobalamin supplementation on the periodic blood count, urinary MMA and serum cobalamin concentrations have never been reported in cats with EPI. Further studies are necessary to identify the origin of anaemia and the mechanisms of the curing action of cobalamin.
Hydroxocobalamin supplementation was an effective medical therapy for hypocobalaminenia in a cat with EPI. Cobalamin deficiency may lead to villous atrophy and intestinal inflammation, with resultant failure to respond to a pancreatic enzyme alone. 8 In addition, abnormalities in amino acid metabolism has been detected in cats with cobalamin deficiency. 16 Therefore, cobalamin levels should be checked routinely in cats with EPI and lifelong cobalamin supplementation started.AQ1: Please check the edits made to the sentence beginning ‘Therefore, cobalamin levels…’, in particular the insertion of ‘started’ at the end of the sentence.
In conclusion, this report indicates that cobalamin malabsorption secondary to feline EPI caused hyperammonaemia through the accumulation of MMA. The prognosis for feline EPI with methylmalonic aciduria is not known; therefore, long-term follow-up is needed to evaluate the treatment response and survival time.
Footnotes
Acknowledgements
We thank Dr Takio Kitazawa and Dr Eiji Uchida (Rakuno Gakuen University, Hokkaido, Japan) for critical reading of the manuscript.
Funding
The authors received no specific grant from any funding agency in the public, commercial or not-for-profit sectors for the preparation of this case report.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
