Abstract
GM1 gangliosidosis is a fatal, progressive neurodegenerative lysosomal storage disease caused by mutations of the β-galactosidase (GLB1) gene. In feline GM1 gangliosidosis, a pathogenic mutation (c.1448G>C) of the feline GLB1 gene was identified in Siamese and Korat cats previously diagnosed with the disease in the USA and Italy, respectively. The present study demonstrated the same mutation in a Siamese cat that had been diagnosed with GM1 gangliosidosis in Japan in the 1960s. The mutation was confirmed using DNA extracted from stored paraffin-embedded brain tissue by a direct sequencing method and a polymerase chain reaction–restriction fragment length polymorphism assay. This pathogenic mutation seems to have been distributed around the world.
Short Communication
GM1 gangliosidosis is a fatal, progressive neurodegenerative lysosomal storage disease caused by mutations of the β-galactosidase (GLB1) gene. 1 Feline GM1 gangliosidosis was first reported in Siamese cats in 1971 in Japan 2 and the USA 3 as different occurrences at the respective locations. Subsequently, the disease was reported in mixed-breed cats in the UK4–6 and Korat cats in Italy. 7 In general, affected cats manifest neurological signs of progressive motor dysfunctions starting from 4 to 6 months of age and die prematurely at approximately 1 year of age. A pathogenic mutation has been identified as a single nucleotide substitution from guanine to cytosine in exon 14 at nucleotide position 1448 (c.1448G>C) in the coding region of the feline GLB1 gene, resulting in the substitution of arginine with proline at amino acid position 483 (p.R483P) in Siamese cats in the USA. 8 The disease in Korat cats has been demonstrated to be caused by the same mutation.9,10
Among Siamese cats with GM1 gangliosidosis diagnosed in Japan, several affected cats from one family were reported in 1969 as having cerebral lipidosis similar to human Tay-Sachs disease, a variant of GM2 gangliosidosis. 11 This feline disease was subsequently revealed, in 1970, to be GM1 gangliosidosis. 12 As mentioned, a definitive biochemical diagnosis established by a deficiency of β-galactosidase activity with the accumulation of GM1 ganglioside in brain tissue was described in 1971, 2 but the pathogenic mutation(s) in this family had yet to be determined.
In the present study, genomic DNA extracted from stored brain tissue of a Siamese cat with GM1 gangliosidosis, born in Japan in the 1960s, was analysed to determine whether it has the same pathogenic mutation as that (c.1448G>C) identified previously in Siamese and Korat cats with GM1 gangliosidosis in the Western world. This article also discusses the significance of the mutation.
Genomic DNA was extracted from paraffin-embedded cerebral tissue of an 8-month-old Siamese cat with GM1 gangliosidosis. The affected cat was one of three animals diagnosed with the disease histopathologically and biochemically that belonged to a family of Siamese cats with six clinically-affected members born between 1966 and 1967 after the backcross mating of a pair of cats.2,11,12 Stored genomic DNA from whole blood of a mix-breed, clinically healthy adult cat was used as a control. DNA was used for polymerase chain reactions (PCRs) in direct DNA sequencing and the PCR–restriction fragment length polymorphism (RFLP) assay.
Direct sequencing of a 106-base pair (bp) DNA fragment, including position 1448 in exon 14 of the feline GLB1 gene, was performed using forward (5’-AGC GAA GTT ACG TGA TCA CTC T-3’, c.1370–1391) and reverse (5’-CTA TGG CAG ATA CAT CAA TGA-3’, c.1445–1475) primers designed based on the exonic sequence of the feline GLB1 gene (GenBank accession number AF029974). The PCR fragment was purified by agarose gel electrophoresis and sequenced in the forward and reverse direction.
The PCR–RFLP assay was performed to confirm the presence of c.1448G>C in genomic DNA of the affected cat. The PCR was carried out targeting an 82-bp sequence around the mutation site using forward (5’-GAG CCA CTC TGG ACC TTC TG-3’, c.1412–1431) and reverse (5’-TCA ATG ATT TCA AGG TAG GAC CAG C-3’, c.1469–1482+11) primers designed based on the exonic and intronic sequences of the feline GLB1 gene (AF029974 and ACBE01328632 respectively). To detect c.1448G>C, the amplification product was digested by a restriction endonuclease, HaeIII (New England Biolabs). Both the PCR and digested products were subjected to electrophoresis in 4% agarose (Agarose 21; Nippon Gene). The mutation was judged based on 2-, 47- and 35-bp, fragments as a result of the digestion site (GG|C
The direct sequencing data for the affected Siamese cat and control mixed-breed cat were compared with those in the GenBank database, ie the wild-type (AF006749) and mutant (AF029974) sequences. The affected Siamese cat was found to be homozygous for the c.1448G>C mutation, whereas the control cat was homozygous for the wild-type sequence at position 1448 (Figure 1). The PCR–RFLP assay also demonstrated that the affected cat was homozygous for the mutant sequence (GGC
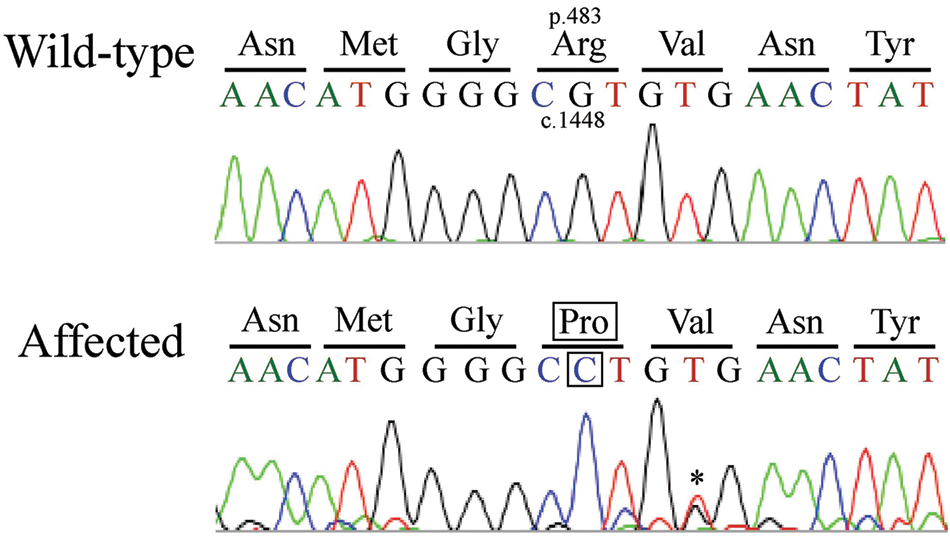
Partial genomic sequence electropherograms of exon 14 in the feline GLB1 gene from the wild-type and affected cats. The guanine at the coding nucleotide position 1448 is substituted with a cytosine in the affected cat (c.1448G>C). The nucleotide substitution causes the substitution of an arginine at amino acid position 483 with a proline (p.R483P). *This signal was estimated as a thymine, although noise from guanine-like signal interfered with the reading

Polymerase chain reaction–restriction fragment length polymorphism (PCR–RFLP) assay using agarose gel electrophoresis in the wild-type and affected cats. Amplified DNA without digestion (N) and DNA digested with the restriction endonuclease HaeIII (D) were analysed simultaneously. Lane M shows molecular size markers. bp = base pairs
The results of direct DNA sequencing and a PCR–RFLP assay demonstrated the pathogenic mutation in a Siamese cat diagnosed with GM1 gangliosidosis in Japan in the 1960s to be c.1448G>C, the same as that previously identified in Siamese and Korat cats with the disease in the USA and Italy, respectively.3,7,8 Only a specimen from one of several affected Siamese cats was available, but it is strongly suspected that the GM1 gangliosidosis in these animals was caused by the common c.1448G>C mutation because all the affected cats were born to the same parents. 11
Cats had been thought to be domesticated in ancient Egypt, where they were considered sacred, some time before 1600 BC, 13 but recent genetic data points to multiple domestication events in separate locations in the Near East 10,000 years ago. 14 Subsequent gradual human migration then spread domestic cats across the globe. Modern cat pure-breeds range from the earliest fancy breeds, including Egyptian Mau, Persian, Siamese, and Korat, established around the seventeenth century to the most recent breeds, such as American Curl and Selkirk Rex, established during the late twentieth century.13,14 The Siamese and Korat breeds originate from Thailand. 13
Based on the history of cat domestication and breed establishment, the occurrence of the same pathogenic mutation at distant locations in the Siamese and Korat breeds suggests that the mutation originates from the Southeast Asian Siamese region and has been distributed in the population of these breeds around the world. Actually, the carrier frequency is high (16.7%, 38/227) in Korat cats from a number of countries. 11 The results of the present study suggest that the mutation may be present in Siamese cats in the world. Furthermore, as one of the large groups of breeds is derived completely, or in part, from Southeast Asian ancestors, 13 GM1 gangliosidosis caused by the same mutation may also occur in those breeds (Oriental Shorthair, Balinese, Havana Brown, Birman, Burmese, Singapura and others). Thus, as c.1448G>C has possibly spread to related pure-breeds around the world, a PCR-based diagnostic test is important for rapid differential diagnosis in cats suspected of having the disease and/or for genotyping to control and prevent the mutation in pure-breeds related to Siamese and Korat cats. The PCR–RFLP assay developed in the present study and a real-time PCR assay reported previously 10 can assist with this.
We might make further progress by tracking where/how many of these cases have been seen in different countries and the chronology of their occurrence.
Footnotes
Acknowledgements
The authors are grateful to Dr Hiroshi Mochizuki, Professor Emeritus at Osaka Prefecture University, for information regarding feline GM1 gangliosidosis identified in Japan in the 1960s.
Funding
This study was supported financially by a grant (number 21658109, OY) from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
Conflict of interest
The authors declare that there is no conflict of interest.
