Abstract

Alimentary lymphoma – and its particular diagnostic challenges
As discussed in part 1, alimentary lymphoma (AL) in cats can be considered clinically as low-grade (LGAL), intermediate- or high-grade (I/HGAL) or, the less commonly identified, large granular lymphocyte lymphoma (LGLL). These diseases differ in clinical presentation, techniques required for diagnosis, response to treatment and prognosis. Here, in part 2, the benefits and limitations of different biopsy techniques are discussed. Special emphasis is given to LGAL, an increasingly recognised clinical problem, and the challenges faced when differentiating this entity from inflammatory disease. The clinician should be aware of the criteria required to diagnose LGAL, both to facilitate communication with pathologists and to optimise the environment for diagnosis of this common disease. Treatment options and prognosis for the different subtypes of AL are also reviewed.
Ultrasound-guided, fine-needle aspiration cytology
Aspiration cytology of diffusely thickened intestinal walls, as are typical of LGAL, can be technically difficult and is usually non-diagnostic. Similarly, cytology of enlarged mesenteric lymph nodes is not helpful in establishing a diagnosis of LGAL as it is not possible to distinguish well-differentiated neoplastic lymphocytes characteristic of low-grade disease from benign lymphoid hyperplasia (Figure 1). 1 Tissue biopsy is required for histological demonstration of disruption of normal lymph node architecture by the neoplastic infiltrate.

Modified Wright-Giemsa stained smear of an ultrasound-guided fine-needle aspirate from the mesenteric lymph node of a cat with low-grade alimentary lymphoma (LGAL). While there is a preponderance of small lymphocytes, it is not possible to distinguish between LGAL and benign lymphoid hyperplasia on cytology. Courtesy of Dr Patricia Martin, Veterinary Pathology Diagnostic Services, University of Sydney
This contrasts with the diagnosis of I/HGAL and LGLL, which can often be made on the basis of aspiration cytology of focal intestinal wall masses, enlarged mesenteric lymph nodes or extraintestinal mass lesions (Figure 2).2–6 This is because of the characteristic morphology of the neoplastic infiltrate (large lymphoblastic cells or large granular lymphocytes [LGLs]), which facilitates differentiation from the background population of lymphocytes. Also, modified Wright-Giemsa stains (eg, Diff Quik; Dade Shearing) used for cytological specimens are more sensitive for detecting LGLs than haematoxylin and eosin (HE) stains used for histological specimens. On cytology, LGLs are identified as large mononuclear cells with moderate amounts of deeply basophilic cytoplasm containing multiple blue or purple granules (Figure 3).5,7 On HE sections, LGLL may be erroneously reported as I/HGAL. Reliable identification of cytoplasmic granules in fixed tissues requires evaluation of Giemsa-stained, plastic-embedded tissue or immunohistochemistry to detect the cytotoxic granule protein, granzyme B.8,9

(a) Ileocaecocolic high-grade alimentary lymphoma (HGAL) and (b) a cut surface from the same specimen. Diagnosis of HGAL can often be based on cytology of ultrasound-guided fine-needle aspirates from a focal intestinal mass or an enlarged mesenteric lymph node. (c) Diff Quik cytology of a concurrent abdominal effusion, showing neoplastic round cells and a mitotic figure. Image (c) courtesy of Dr Patricia Martin, Veterinary Pathology Diagnostic Services, University of Sydney
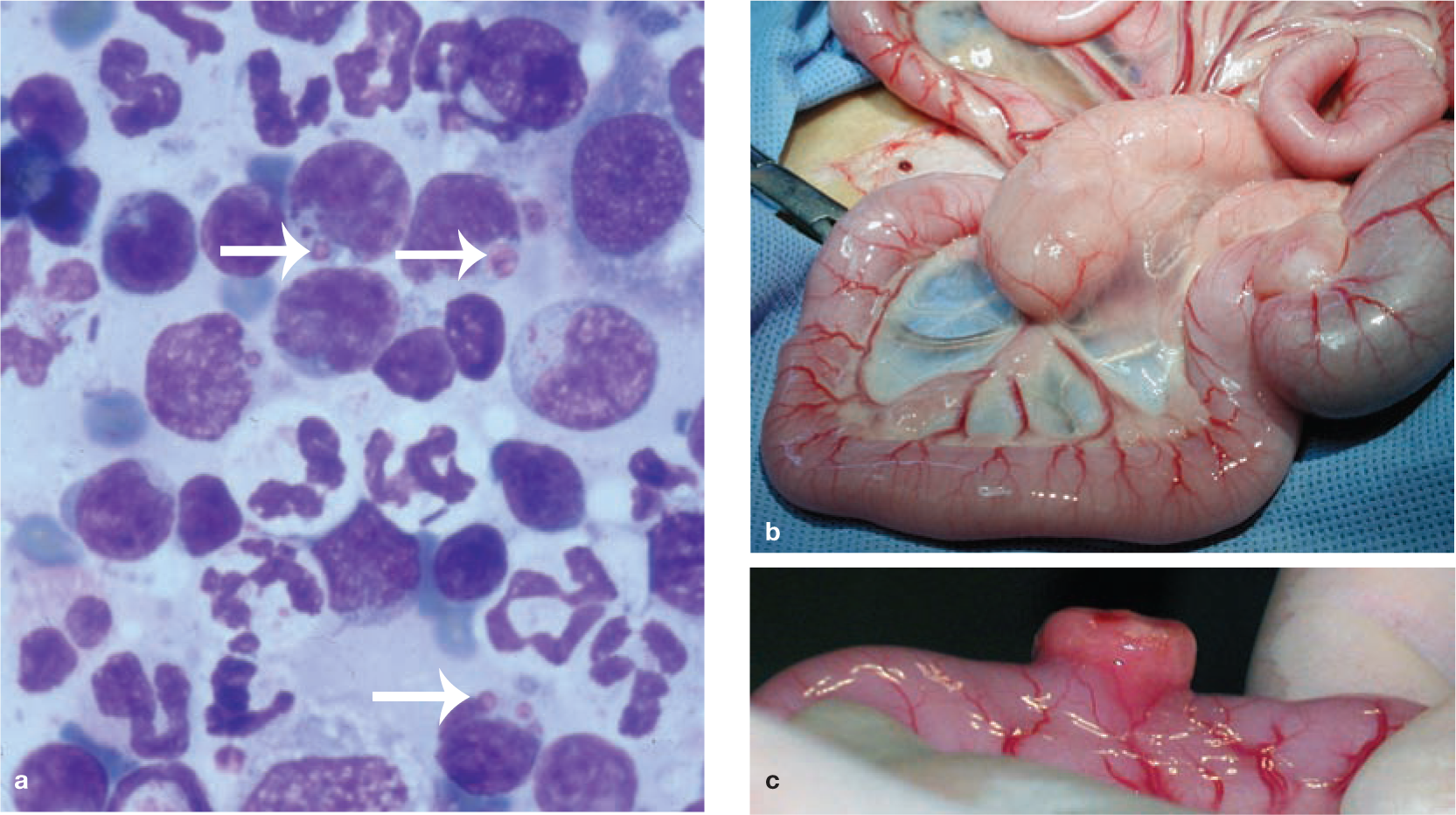
(a) Diff Quik stained smear of a fine-needle aspirate biopsy from an enlarged mesenteric lymph node (b) and intestinal mass (c) in a cat with large granular lymphocyte lymphoma (LGLL). In (a) neoplastic round cells have a basophilic cytoplasm and contain large purple intracytoplasmic granules (arrows). Image (a) courtesy of Dr Patricia Martin, Veterinary Pathology Diagnostic Services, University of Sydney
Diagnostic considerations for LGAL
Intestinal biopsy procurement
Histological evaluation of intestinal biopsies is required for diagnosis of LGAL and for other forms of AL where cytological evaluation is not definitive (Figures 4 and 5). LGAL is typically a diffuse or multifocal disease affecting more than one region of the gastrointestinal tract. There is jejunal and ileal involvement in over 90% of cases, duodenal involvement in over 70% and gastric involvement in 7–40% of cases.1,10,11 The distribution of gastrointestinal involvement in lymphoplasmacytic enteritis (LPE) is similar to LGAL except that gastric involvement is more common in LPE. 11

Histology and immunohistochemistry of intestinal wall biopsies from a cat with B cell HGAL. Heavy infiltration of the lamina propria and submucosa can be seen, destroying the normal architecture of the intestine. Villous and crypt architecture is also severely altered. (a) HE stain; (b) CD3 (T cell marker) stain; (c) CD79a (B cell marker) stain. Note the heavy staining of the neoplastic B cell infiltrate. Courtesy of Associate Professor Mark Krockenberger (image a) and Dr Katherine Briscoe (images b and c), University of Sydney

T cell LGAL. (a,b) HE-stained section of full-thickness intestinal biopsy. There is severe villous blunting and distension. The lamina propria and submucosa are diffusely infiltrated with monomorphous sheets of small lymphocytes. Intraepithelial lymphocytes (IELs) can be difficult to identify in HE-stained sections, but are easily identified in CD3-stained sections (c). This pattern of epitheliotropism with aggregates of neoplastic IELs is common in LGAL and facilitates differentiation from LPE. Courtesy of Associate Professor Mark Krockenberger (image a) and Dr Katherine Briscoe (images b and c), University of Sydney
Given the propensity of LGAL to invade the distal small intestine, if only the stomach and duodenum are biopsied using gastroduodenoscopy, a diagnosis of LGAL could be missed. The current recommendation for endoscopic investigation of enteropathies is to sample both the duodenum and ileum, necessitating both gastroduodenoscopy and ileocolonoscopy. 12 Endoscopic biopsy (EB) of the jejunum is not possible using conventional push endoscopy in cats. Adaptation of double-balloon enteroscopic techniques used in humans, as reported in dogs, would facilitate this procedure. 13 At laparotomy, the standard intestinal regions sampled are duodenum descendens, mid-jejunum and ileum. 14 Laparoscopic intestinal biopsies are usually single site, from the jejunum (Table 1). 15 The utility of this biopsy technique for the diagnosis of LGAL in cats has not been evaluated. Intestinal biopsies obtained at exploratory laparotomy or laparoscopically are full thickness, while optimal EBs include intestinal mucosa and submucosa.
Comparison of full-thickness with partial-thickness intestinal biopsies for diagnosis of LGAL
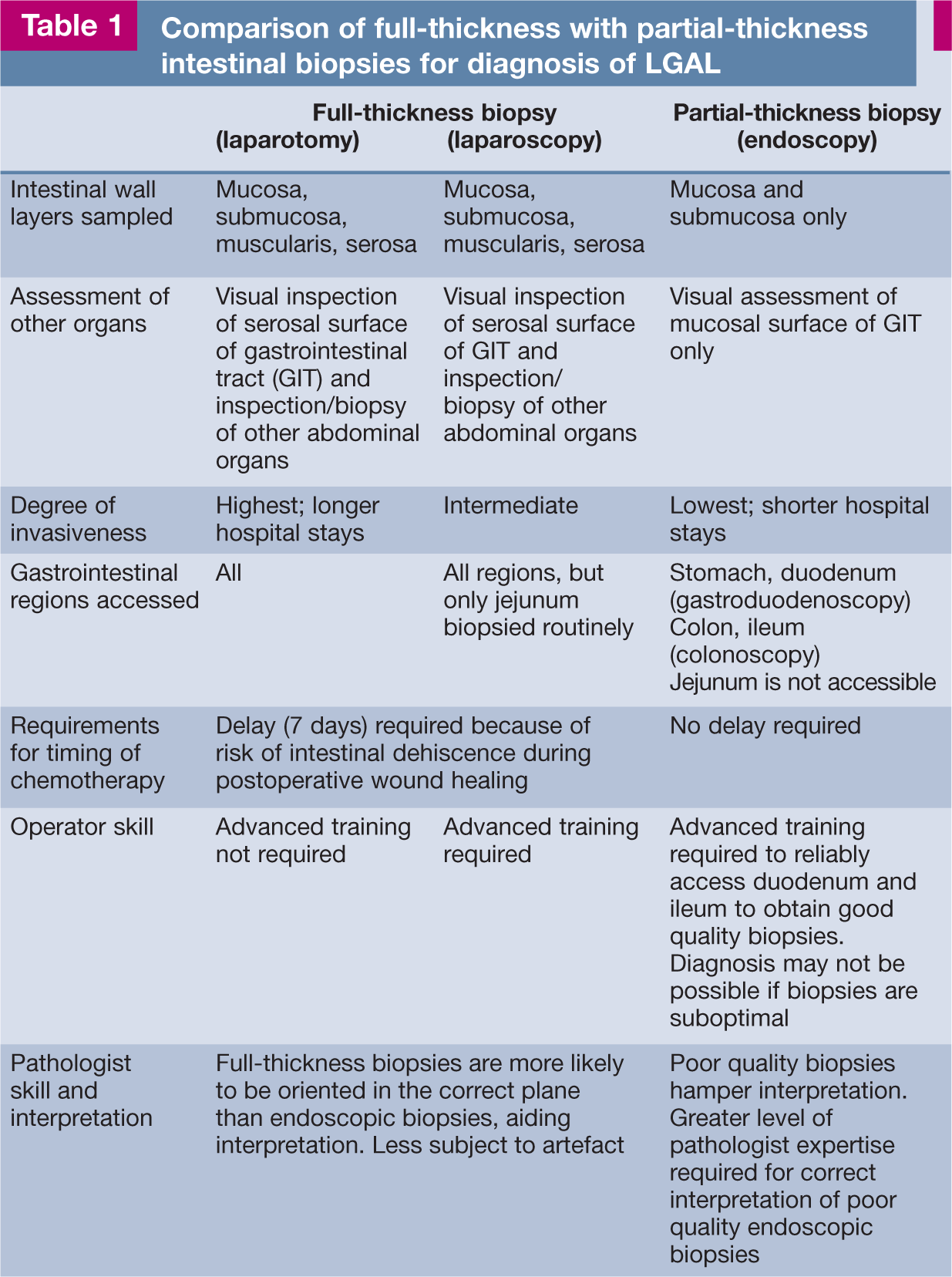
While EBs are minimally invasive, diagnosis of LGAL by EB requires significant expertise on the part of both endoscopist and pathologist, as well as appropriate laboratory tissue processing (Table 1). Full-thickness biopsy should be considered when these factors are not optimal. In one small series, histological evaluation of full-thickness biopsies of the gastrointestinal tract was found to be more sensitive than EBs procured by gastroduodenoscopy for the diagnosis of LGAL. 10 However, in that study, technical difficulties may have hampered the quality of EB specimens, as some cats underwent only ‘partial’ duodenal assessment or blind duodenal biopsy. The quality of EB samples has a profound effect on their sensitivity for identifying certain lesions. 16 Substantially fewer biopsy samples are needed to establish a diagnosis as the quality of the tissue increases. It has been shown that if six ‘marginal’ or ‘adequate’ quality duodenal or gastric EBs are taken, as defined by the presence of at least one villus and subvillus lamina propria, correct histological diagnosis is very likely to be achieved. 16 Optimal histological processing, including biopsy orientation, positioning and staining, is also essential for correct interpretation. 17
Further studies are required to compare the results of ileal EB and full-thickness biopsy specimens in the diagnosis of LGAL in cats.
Histological features
In contrast to higher grades of AL, the histological diagnosis of LGAL is not straightforward as neoplastic infiltrates of small lymphocytes are often morphologically indistinguishable from those present in the gastrointestinal tract of healthy cats or, in particular, cats with LPE. In a histopathological review of LPE and LGAL, there was disparity in diagnosis between two pathologists for 8/12 cases of moderate to marked LPE, requiring assessment by a third pathologist to reach a consensus diagnosis. 11 In another study, the use of adjunctive immunophenotyping and analysis of clonality of lymphoid infiltrates by polymerase chain reaction (PCR) resulted in 10/19 cases diagnosed as inflammatory on histological evaluation of HE sections being reclassified as T cell lymphoma, while three cases of T cell lymphoma were reclassified as inflammatory. 18
Histological criteria distinguishing LGAL from LPE include, in the former, the relative absence of mixed lymphoid and granulocytic cells and their replacement with monomorphous sheets of neoplastic lymphoid cells involving the lamina propria. In early disease, neoplastic cells form lamina propria ‘patches’, which are discrete regions of lymphocytic infiltration within some villi but not others.8,19–23 As disease progresses, lamina propria ‘bands’ of lymphocytes spanning the crypt–villous junction are seen, followed by ‘villous laminal propria obliteration’ by dense, monomorphic, lymphocyte infiltrates. In the most severe lesions ‘villous and crypt laminal propria obliteration’ occurs due to complete lymphocytic infiltration and formation of a band of lymphocytes beneath the crypt epithelium but above the muscularis mucosae. 8
Epitheliotropism, characterised by increased numbers of IELs, is a feature of both LGAL and LPE.11,12,18,24–26 However, particular patterns of epitheliotropism, including the formation of ‘nests’ (≥5 clustered IELs) or ‘plaques’ (≥5 adjacent epithelial cells overrun by IELs) in the villous or crypt epithelium, are highly specific for LGAL.8,11,18,20,23,27,28 Other histological features of LGAL that help to differentiate this disease from LPE include extension of the lymphocytic infiltrate into layers deep to the mucosa,11,18 more severe disruption to villous and crypt architecture (Figure 5), 11 intravascular lymphocytic infiltrates 18 and the presence of neoplastic cellular infiltrates in mesenteric lymph nodes.19,22,29

Immunophenotype
Immunophenotyping assists in the differentiation of LGAL from LPE. A monomorphic lymphocytic population supports a diagnosis of lymphoma, while a mixed lymphocytic population supports a diagnosis of inflammation. Of 32 cats diagnosed with lymphoma based on HE-stained sections, immunohistochemical stains revealed that, in five cats, the ‘neoplastic’ infiltrate was composed of a mixed population of small B and T lymphocytes and plasma cells. On this basis, all five cases were reclassified as having inflammatory enteropathies. 26 Importantly, LGAL cannot be diagnosed on the basis of T cell phenotype alone, since expansion of T cell populations in intestinal mucosal-associated lymphoid tissue can occur in inflammatory intestinal disease in cats.11,18,30 An additional feature of CD3-stained intestinal sections is that IELs can be more clearly visualised than in HE-stained sections.
Clonality testing
Determination of clonality of T cell populations in lymphocytic intestinal infiltrates is a useful diagnostic tool when the distinction between LGAL and LPE remains ambiguous after histological evaluation and immunophenotyping.8,18,30,31 The clonality of infiltrates of T lymphocytes in intestinal sections can be determined by assessment of T cell receptor gamma (TCRG) V–J junctional diversity.18,30–32 Similarly, B cell clonality can be determined by PCR of immunoglobulin heavy chain (IgH) variable regions.32–34
During development in the thymus, T cells rearrange their antigen receptor genes TCRA, TCRB, TCRG and TCRD to form two lineages, αβ and δγ T cells. Most αβ T cells rearrange TCRG before rearrangement of TCRA and TCRB. The TCRD gene is deleted from the genome during rearrangement of the TCRA gene. Thus, TCRG gene rearrangements occur in both αβ and δγ T lymphocytes. During this process the V-domains are somatically rearranged in a process called V–J recombination, where the V-region is randomly and imprecisely joined to the J-region. Random nucleotides are added at the joining sites, further enhancing diversity and length polymorphism. This length polymorphism can be visualised by conventional PCR of the resultant hypervariable region of the V-domain, known as the complementarity determining region 3 (CDR3), using primers directed against relatively conserved framework regions. Amplified products are analysed using heteroduplex gel electrophoresis and clonality is determined by the number and size of the bands in duplicate samples run side by side.
Clonal lymphocyte populations produce one or two sharp bands that are consistent in duplicate samples; oligoclonal populations produce three bands; while polyclonal populations produce a broad band, smear or ladder of bands. Pseudoclonal populations contain one or two bands that are of different sizes or are non-reproducible in duplicate analyses. Neoplastic populations of T lymphocytes are clonal or oligoclonal, while inflammatory populations are polyclonal. 30
In three studies, determination of lymphocyte clonality by PCR was 78–90% sensitive in the detection of intestinal T cell lymphoma, where clonal or oligoclonal T cell populations were considered to be neoplastic.8,30,31 In one of these studies, 22/28 cats were found to have clonal rearrangements of the TCRG gene, while three had oligoclonal rearrangements. In comparison, polyclonal rearrangements were detected in 3/3 cats with normal intestinal histology and in 9/9 cats with LPE. 30
Performing concurrent T and B cell clonality analysis can increase the sensitivity of detection of T cell lymphomas due to cross-lineage IgH gene rearrangements.18,32 As clonality is not always specific for malignancy, this technique cannot be used as a stand-alone diagnostic test for LGAL. A diagnostic approach that combines the assessment of histological features, immunophenotype and clonality analysis is optimal to distinguish LGAL from LPE, especially where EBs are submitted. 18 Lymphocyte lineage should be based on immunophenotypic assessment rather than clonality determination by PCR if the results of these two techniques are divergent. 30


Chemotherapy protocols and response to therapy
LGAL
Most cats with LGAL show an excellent response to treatment with oral, slow-alkylating agents and prednisolone.1,27,43,44 Combinations of oral prednisolone and chlorambucil used for induction, and median ST or remissions achieved, are listed in Table 2.1,18,27,43,44 Complete remission (CR), defined as complete resolution of clinical signs for ≥30 days, occurred in 70% of cases of LGAL in two studies.1,27 For cats achieving CR, median durations of remission of 26 and 29 months are reported.43,44 In these studies, since the assignation of anything less than a CR was considered arbitrary due to insufficient data, any cats with a partial response (PR) were included in the ‘no response’ category. In contrast, another report found a lower CR rate of 56%. 43 A key difference in this study was that a third (partial) response category was included – defined as >50% but less than 100% response. However, 95% of the 41 cats achieved either CR or PR with median remission durations of 29 and 14 months, respectively. This would be considered a good outcome, even for cats with PR.
Remission and survival data, and induction chemotherapy protocols for cats with LGAL
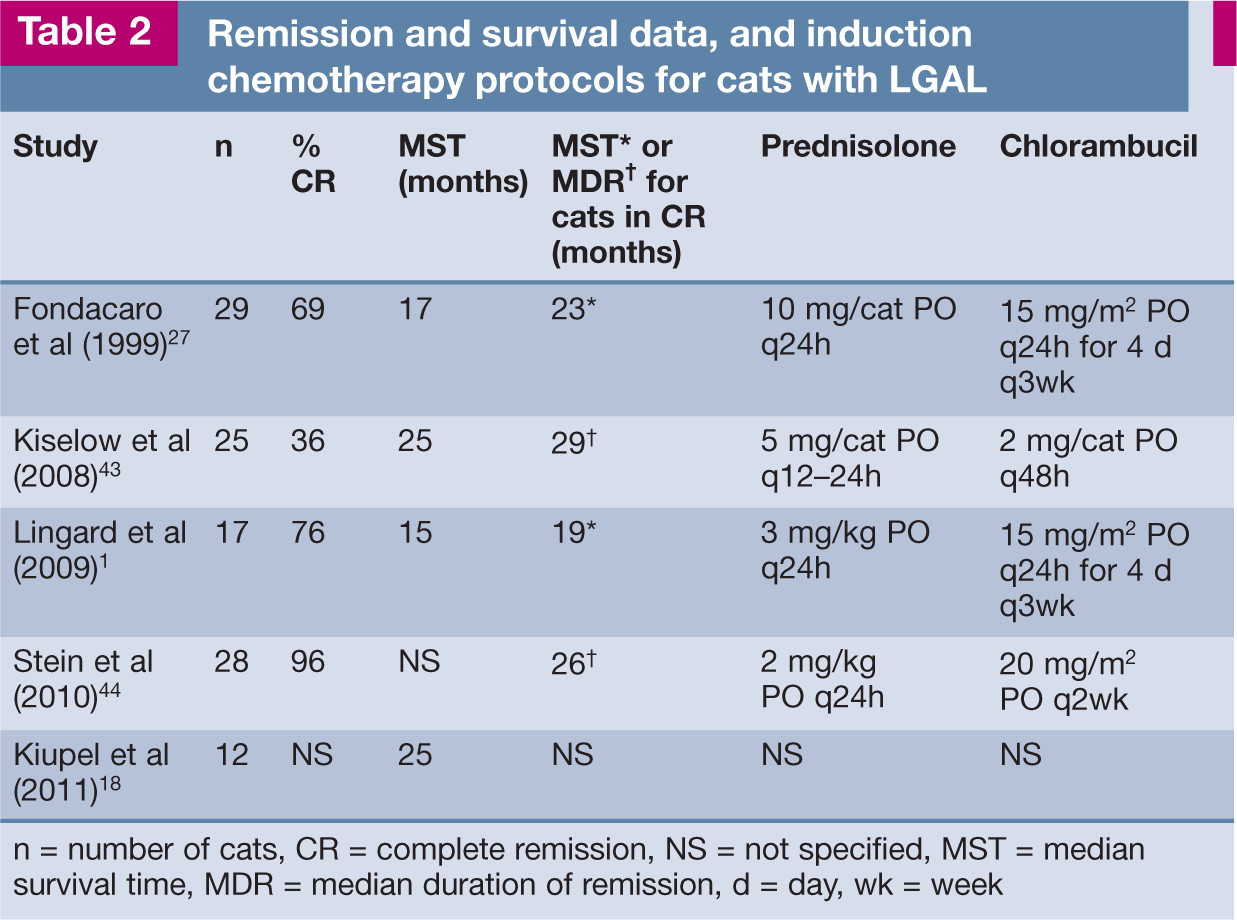
n = number of cats, CR = complete remission, NS = not specified, MST = median survival time, MDR = median duration of remission, d = day, wk = week
Reported doses of prednisolone to maintain remission are 5 mg q24h PO or 1–3 mg/kg q48h PO.1,27,43 Cyclophosphamide (10 mg/kg PO every 3–4 weeks, 225 mg/m 2 PO every 3 weeks or 200–250 mg/m 2 PO given on days 1 and 3, every 2 weeks) in combination with prednisolone at induction or maintenance doses has been used as a rescue protocol for cats that come out of clinical remission.27,44,48 The median ST of 12 cats in which remission was re-induced using cyclophosphamide was 29 months, while for four other cats the median duration of second remission was 9 months.27,44
I/HGAL
There is little precise information regarding remission rates and treatment responses for I/HGAL since, in many studies, all anatomical forms (mediastinal, multicentric, alimentary and extranodal) are considered as a single group for analysis. A median ST of 7–10 months is expected with chemotherapy protocols that include doxorubicin.3,37,38,40,42,45 In a series of 28 cats with AL treated with a COP protocol, of which 25 cases were HGAL, the median ST was 50 days. 4 For cats achieving CR (32%), the median remission time was 7 months. In another report of 21 cats with AL, where cytological and ultrasonographic findings were consistent with HGAL in most cases, treatment with a CHOP-based protocol (COP plus doxorubicin [hydroxydaunorubicin]) gave a median ST of 9 months and 38% of cats achieved CR. 3 Surgical resection of an intestinal mass prior to chemotherapy has not been demonstrated to improve survival compared with chemotherapy alone.4,49
Traditionally, CHOP-based chemotherapy protocols have involved 1 or more years of chemotherapy.42,45,50 In one study, COP and doxorubicin for a total of 6 months gave median remission times of 9.5 months compared with 3 months for cats treated with COP alone. 38 It was postulated that shorter CHOP-based protocols (6 months) are adequate for the treatment of feline lymphoma. 38 An example of a currently recommended 25-week CHOP-based protocol is given in Table 3. 51 In COP-treated cats, doxorubicin was shown not to be an effective rescue protocol for I/HGAL. 52 Other rescue protocols (eg, CCNU, MOPP) are generally associated with a 40–50% response rate and a short median duration of second remission of 1.5–2 months. 51
CHOP-based chemotherapy protocol for cats with lymphoma 52

A complete blood count (CBC) should be performed before each chemotherapy treatment. If the neutrophil count is <1500 cells/µl, delay chemotherapy for 5–7 days, repeat a CBC and, if the neutrophil count has risen above 1500 cells/µl, administer the drug
Prednisolone is continued at 1 mg/kg q48h PO from this point forward
LGLL
LGLL has the poorest prognosis of all forms of AL and, from limited reports, appears to be minimally responsive to standard chemotherapy protocols. Of 24 cats receiving combination chemotherapy (COP [n = 20], CHOP [n = 4]), the overall response rate was 30% (one CR, six partial remission) with a median ST of only 45 days. 6
Radiation protocols and response to therapy
Targeted abdominal irradiation has recently been evaluated for the treatment of feline AL.53,54 In a pilot study, eight cats with I/HGAL (n = 5) or multicentric lymphoma confined to the abdomen (n = 3), which were assessed to be in remission following treatment with a 6-week CHOP-based chemotherapy protocol, received whole-abdomen radiation therapy in 10 daily fractions of 1.5 Gy. 53 Two cats relapsed following treatment, one of which was subsequently diagnosed with LGLL, and another was euthanased for unrelated reasons. The other five cats were all in remission at the time of publication, with durations ranging from >266 to >1332 days. In a retrospective study, 10/11 cats with AL (six LGAL, four I/HGAL, one LGLL) responded to whole-abdomen radiation (8 Gy over 2 days) as a rescue therapy. Median ST overall was 355 days and median ST post-radiation therapy was 214 days. Radiation therapy was well tolerated in both studies.
Further investigations of the use of abdominal radiation for the treatment of feline AL are warranted.
Supportive treatment
For stabilisation of cats presenting acutely with AL, intravenous fluid therapy, blood products, antimicrobials and antiemetics may be required. Many cats with AL have a low body condition score and are inappetent or anorexic at presentation, necessitating provision of enteral or parenteral nutrition. Consideration should be given to placement of oesophagostomy or gastrostomy tubes during general anaesthesia for intestinal biopsies. In the postoperative period, appetite stimulants such as mirtazapine or cyproheptadine may be beneficial until glucocorticoid therapy can be initiated. When gastrointestinal ulceration is identified or suspected, treatment with proton-pump inhibitors (eg, omeprazole) or H2-antagonists (eg, ranitidine or famotidine) and mucosal protectants (eg, sucralfate) is indicated.
A further consideration is dietary modification for treatment of concurrent LPE or dietary intolerance. The diet should be gluten-free and contain a novel protein component that is single source or hydrolysed. 55 The carbohydrate component should also be single source and highly digestible (eg, cooked rice). Although evidence of efficacy is currently lacking, use of prebiotics and probiotics may be considered. Folate deficiency should be treated with oral folic acid supplementation. Parenteral cobalamin supplementation is indicated if the serum cobalamin concentration is subnormal (see Case Notes). Because cobalamin deficiency itself can result in inflammatory infiltration of the gastrointestinal mucosa and villous atrophy, the response to chemotherapy may be suboptimal in cats with untreated hypocobalaminaemia. 56 In cats showing relapse of clinical signs, serum cobalamin concentration should also be measured routinely.
Footnotes
Key points
Case notes
![]() ). Intestinal wall layering was normal. Thoracic radiographs were unremarkable.
). Intestinal wall layering was normal. Thoracic radiographs were unremarkable.
Cytology of an ultrasound-guided mesenteric lymph node aspirate revealed 92% small lymphocytes, 5% medium lymphoid cells, 2% neutrophils, 1% plasma cells and occasional large lymphoid cells. At laparotomy, the ileum and distal jejunum were diffusely thickened and biopsies were obtained from the stomach, duodenum, jejunum, ileum, liver, mesenteric lymph nodes and pancreas. An oesophagostomy tube was placed for enteral nutritional support during the postoperative period.
On histology of the jejunum and ileum, abnormalities in villous architecture, including widening of the base, shortening and fusion, were noted. The lamina propria of the villi and crypts was distended by an infiltrate of large numbers of small lymphocytes arranged in sheets. The infiltrate showed positive staining with CD3 and no staining with CD79a. Clusters of IELs were identified in the villous epithelium on CD3-stained sections. In the lymph node, there was an expanded paracortical zone comprising small lymphocytes that stained positively for CD3. The liver and pancreas were normal.
• WHAT IS YOUR ASSESSMENT?
1 What does the cytology of the mesenteric lymph node suggest?
(a) LGAL with metastasis to the lymph node.
(b) Benign lymphoid hyperplasia.
(c) HGAL (given the presence of occasional large lymphoid cells).
(d) Answers (a), (b) or (c) are equally likely.
(e) It could be (a) or (b).
2 In cases where differentiation between LPE and LGAL cannot be made on routine HE-stained intestinal sections, what further diagnostic approach would you recommend?
(a) Cytology of a repeat ultrasound-guided fine-needle aspirate of the enlarged mesenteric lymph node.
(b) Immunohistochemistry of intestinal biopsy sections using markers for CD3 and CD79a.
(c) PCR of intestinal biopsies for detection of T cell and B cell clonality.
(d) No further diagnostic tests are necessary – a therapeutic trial with prednisolone is indicated as LPE and LGAL are treated the same way.
(e) Approach (b) interpreted together with the HE-stained intestinal biopsy sections. If still not diagnostic, proceed to (c).
Sooty remained in remission for 19 months before vomiting and weight loss recurred. The results of clinical investigations were consistent with relapse of LGAL. A rescue chemotherapy protocol was commenced comprising 15 mg prednisolone q24h PO and 50 mg cyclophosphamide once every 3 weeks. Sooty achieved clinical remission for a further 11 months before relapsing, at which point euthanasia was performed.
Funding
The authors received no specific grant from any funding agency in the public, commercial or not-for-profit sectors for the preparation of this review article.
Conflict of interest
The authors declare that there is no conflict of interest.
