Abstract
A 6-month-old female neutered domestic shorthair cat was examined for pain and swelling of the distal limbs. Swelling and malalignment of both carpi, dorsiflexion of the distal metatarsals and a mild, multicentric lymphadenopathy were detected on examination. Screening blood tests revealed a biclonal gammopathy, hypercalcemia and atypical leukocytes. Survey radiographs revealed lysis and proliferative changes within the physes. Multiple pathological physeal fractures were present, including the distal radii, metacarpal and metatarsal bones. No treatment was attempted and the patient was euthanased. Histopathology revealed infiltration by monomorphic round cells within the liver, spleen, kidney and bone marrow. Immunohistochemistry revealed strong reactivity to CD20. Clonality assays revealed clonal results for the B-cell loci, consistent with B-cell lymphoma. To the authors’ knowledge this is the first report of polyostotic lymphoma with multiple pathological fractures in a cat. The predominantly distal distribution of osseous lesions is rarely reported in dogs and humans.
Case Report
A 6-month-old, 2.6 kg, neutered female domestic shorthair cat was presented for assessment of bilateral carpal swelling and hyperglobulinemia. Eighteen days previously the cat had presented to the referring veterinarian for investigation of a non-weight-bearing left forelimb lameness. Left carpal swelling was identified and symptomatic treatment with 0.3 mg/kg parenteral meloxicam (Metacam; Boehringer Ingleheim) was provided. The cat subsequently developed a right forelimb lameness and examination 12 days after initial presentation revealed bilateral carpal swelling. Symptomatic treatment with oral meloxicam 0.1 mg/kg q24h; doxycycline (Vibravet; Pfizer) 5 mg/kg PO q24h and calcium carbonate (Calci-Tab 600; AFT Pharmaceuticals) 90 mg/kg PO q12h was administered, but there was no improvement. Forty-eight hours later the cat had deteriorated significantly. It was non-ambulatory but was able to stand with assistance; a mild mandibular lymphadenopathy was identified. Hematology and serum biochemistry panels were performed (Table 1). A differential white cell count was not provided; however, cytological examination of peripheral blood showed increased numbers of atypical, medium-sized lymphocytes. In-house tests were negative for feline immunodeficiency virus (FIV) antibodies and feline leukemia virus (FeLV) antigen (AGEN FeLV/FIV Simplify). Treatment with intravenous (IV) fluids (0.45% sodium chloride and 2.5% glucose), prednisolone (Solone; Sigma Pharmaceuticals) 1.8 mg/kg PO q12h and a continuous rate infusion of butorphanol (Dolorex; Merck Animal Health) (0.04 mg/kg/h IV) were administered over 48 h and the cat’s demeanor improved; however, it remained non-ambulatory and was referred for assessment.
Pertinent hematology and biochemistry results 14 and 18 days after initial presentation

The noted ‘Others’ consisted of a population of large round cells
NR = not reported, ALT = alanine aminotransferase, AST = aspartate transaminase, ALP = alkanine phosphatase
Physical examination revealed a small stature and pallor. The cat was unable to stand without assistance. Swelling and malalignment of both carpi, pain on palpation of the distal joints in all limbs and dorsiflexion of distal metatarsal bones in both limbs were noted. A mild, multicentric, non-painful lymphadenopathy was also detected. The patient’s vital signs were within normal limits.
Craniocaudal and lateral radiographs of all limbs (Figures 1, 2 and 3) were obtained under IV sedation. The majority of the physes were abnormal, with patchy lysis and proliferative changes identified. Multiple pathological physeal fractures were present, including both distal radii and the majority of the distal metacarpal and metatarsal bones. There was gross malalignment of both carpi. A lateral spinal radiograph was unremarkable. Overall, the bone density appeared normal. Repeat hematology and serum biochemistry panels were performed (Table 1) and showed a rapidly progressive anemia and leukopenia. Forty-two percent of the leukocyte population consisted of large round cells with scant-to-moderate amounts of deep blue cytoplasm, occasionally with a prominent region of perinuclear pallor. The nuclei were large and round with ropey or stippled chromatin and 1–2 prominent nucleoli. Marked hyperproteinemia was identified, secondary to hyperglobulinemia. There were no clinical signs consistent with hyperviscosity syndrome. Serum protein electrophoresis was performed and a biclonal gammopathy was identified within the beta 2 globulin 64.1 g/l (reference interval 6–10 g/l) and gamma 1 globulin 33.5 g/l (3–25 g/l) regions (Figure 4). Repeat FIV antibody enzyme-linked immunosorbent assay (ELISA) and FeLV antigen ELISA (Idexx Snap FIV/ FeLV Combo test) were performed at a reference laboratory and were negative. A serum coronavirus indirect fluorescent antibody titre was negative (<100).

Craniocaudal radiograph of the right carpus shows lysis and fracture of the right distal radius (white arrow), lysis and proliferation of the distal ulna (open white arrowhead) and lysis of all distal metacarpals (solid white arrowhead)
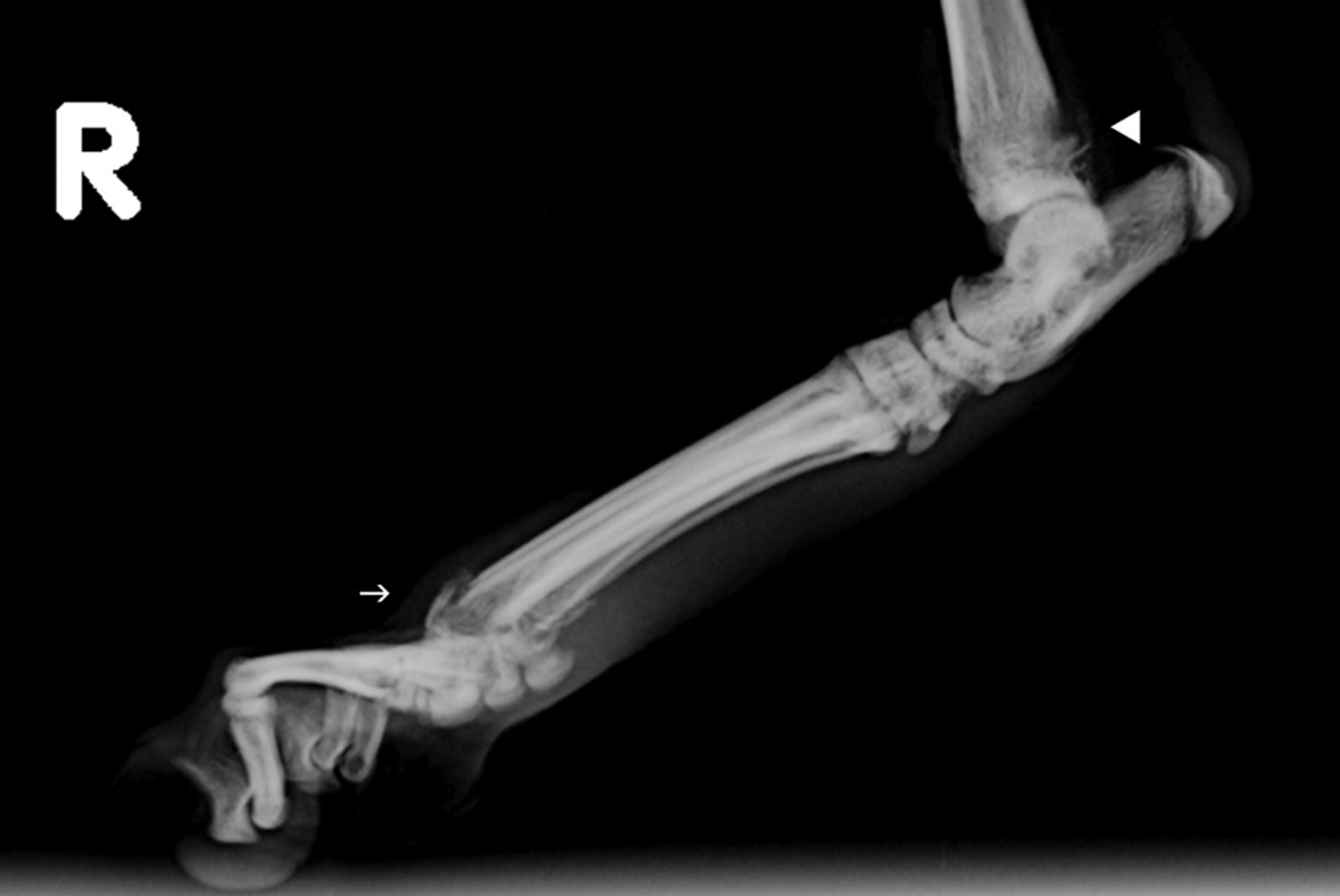
Lateral radiograph of the right distal hind limb reveals lysis and proliferation of the distal tibia (solid white arrowhead) and lysis and pathological fracture of the distal metatarsals (white arrow)
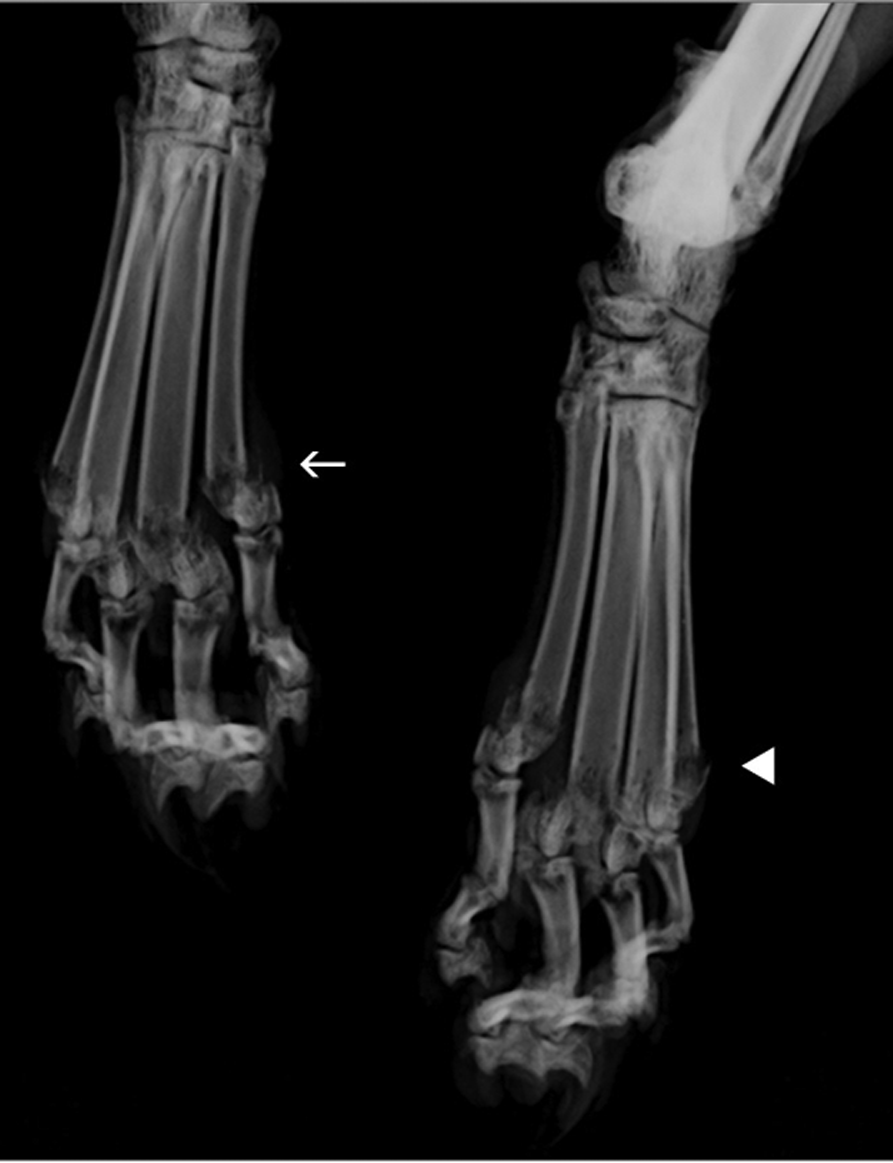
Craniocaudal radiograph of the left and right distal hind limbs. The radiograph reveals lysis and fracture of the left (solid white arrowhead) and right (white arrow) distal metatarsals 2, 3, 4 and 5

Serum protein electrophoresis trace revealing a biclonal gammopathy at the beta 2 and gamma 1 globulin regions
At the owners request the patient was euthanased and tissues samples were collected post mortem. Grossly, the liver appeared normal, the spleen was enlarged and the kidneys were pale with numerous circular hemorrhagic lesions ranging from 1–4 mm within the cortex and medulla. Samples of liver, spleen, kidney, distal radius and ulna were collected for histopathology. The section of liver revealed a significant accumulation of small mononuclear cells with plasmacytic features predominantly in perivascular locations and portal tracts (Figure 5A). Similar cells were evident within sinusoids. These cells had irregular outlines, eosinophilic cytoplasm and small convoluted nuclei with dense chromatin. The mitotic rate was low. Occasional blood vessels displayed mural disruption. The spleen was hypercellular and a similar population of plasmacytoid cells was widely distributed within the red pulp accompanied by extramedullary hematopoiesis and mild lymphoid atrophy in the periarteriolar sheaths. Within the kidney, interstitial accumulations of pleomorphic cells with plasmacytoid features (as described above) were present (Figure 6A). Multiple sections of the bone were examined; however, cell morphology was not well preserved as a consequence of the demineralization process. These revealed increased cellularity with areas of disruption and necrosis in the physeal plate. Within the bone marrow there was a diffuse infiltrate of plasmacytoid cells similar to those described in the kidney, liver and spleen (Figure 7). The erythroid and myeloid tissues were markedly decreased, presumably secondary to myelophthisis. The histomorphological hallmark of all of the specimens was the marked infiltration by a monomorphic round cell population centered on the portal areas in the liver, the red pulp of the spleen, the renal interstitium and diffusely throughout the bone marrow. Immunohistochemical staining of the liver, kidney and spleen for coronavirus antigens was performed using antibodies against FIPV3-70 mAb (Custom Monoclonals). It was negative on two occasions whilst the positive control was positive.
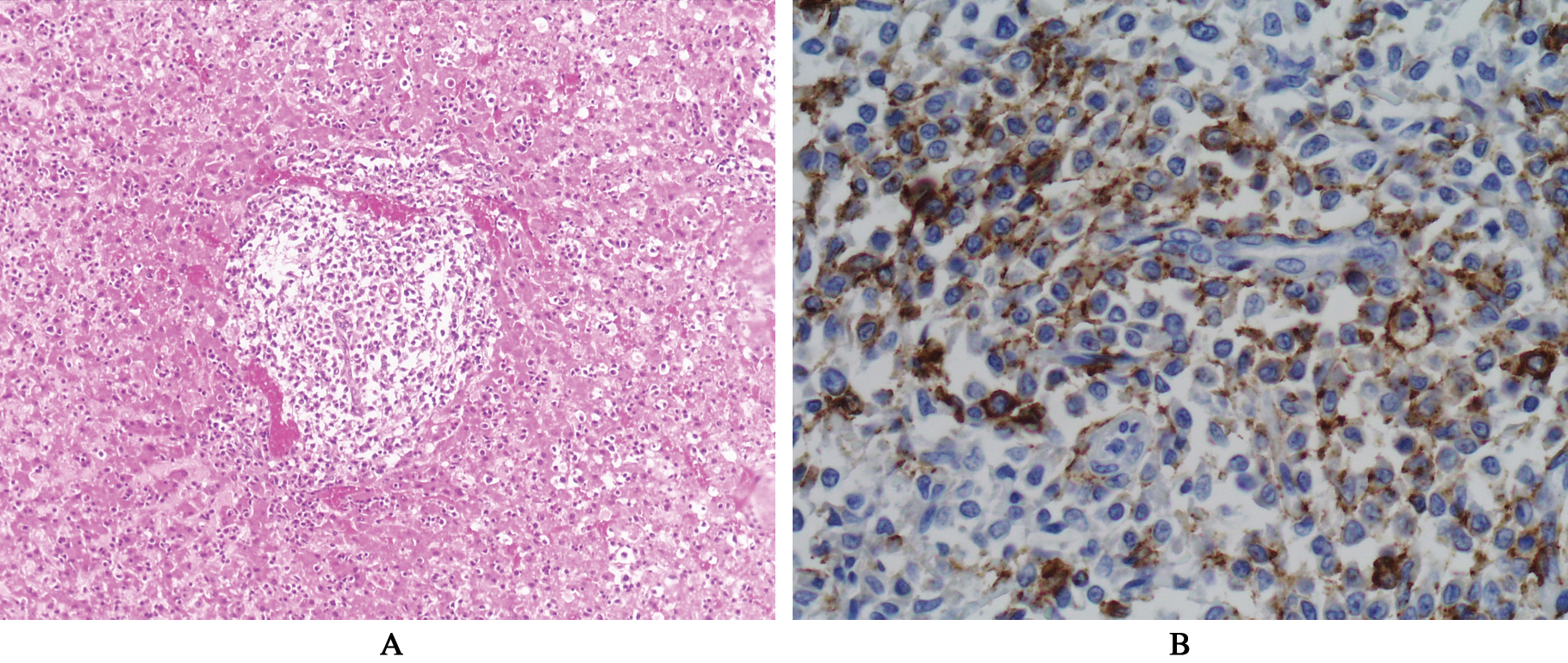
(A) Histopathology of the liver revealing perivascular infiltrates of neoplastic lymphocytes. Hematoxylin and eosin stain (10×). (B) Histopathology of the liver showing portal cell infiltrates of neoplastic lymphocytes stained with CD20 (40×)

(A) Histopathology of the kidney showing interstitial infiltrates of neoplastic lymphocytes. Hematoxylin and eosin stain (20×). (B) Histopathology of the kidney showing interstitial infiltrates of neoplastic lymphocytes stained with CD20 (40×)

Histopathology of the bone marrow showing infiltrates of neoplastic lymphocytes. Hematoxylin and eosin stain (40×).
Tissue was sent to the Leukocyte Antigen Biology Laboratory at University of California, Davis. Immunohistochemical staining for CD18, CD3, CD20, CD79a and MUM-1 was performed on liver, spleen, kidney and bone. CD18 is a broad leukocyte marker, whilst CD3 is specific for T lymphocytes. CD20 and CD79a are the two most commonly used pan-B cell-specific markers. MUM1 is expressed in B lymphocytes and plasma cells. The most notable immunohistochemical finding was strong membranous and cytoplasmic reactivity to CD20 within the red pulp of the spleen, liver, kidney and bone marrow (Figures 5B and 6B). Approximately 5% of portal lymphocytes displayed strong reactivity to CD3. CD18 revealed weak cytoplasmic staining. The CD79a and MUM-1 staining were difficult to interpret because of spurious staining patterns. CD79a revealed strong nuclear staining; however, the correct staining should be on the cell surface. This is a known issue with this antibody and it occurs inconsistently. MUM-1 showed strong cytoplasmic staining; however, the correct staining should be nuclear, as it is a transcription factor. Immunohistochemistry within the bone marrow was somewhat compromised by decalcification. However, neoplastic cells with a similar staining pattern for CD20 as described in the liver and spleen were found in the bone marrow. Clonality assays of the liver revealed clonal results for the immunoglobulin heavy chain locus (IgH framework 2 and IgH framework 3), which indicates rearrangement of a B-cell antigen receptor gene. Clonality testing of the bone marrow failed, presumably because of DNA damage during the decalcification process. Taken together, these results are consistent with a B cell lymphoma with plasma cell differentiation involving the spleen, liver, kidney and bone.
Lymphoma is one of the most common neoplasms in cats and in a study of benign and malignant tumours occurring in cats up to 1 year of age, it was the most frequent diagnosis.1,2 A bimodal age distribution has been reported, with an initial peak of incidence at approximately 1–3 years. 3 Lymphoma typically involves lymphoid tissue, including lymph nodes, spleen and bone marrow. Extra-nodal lymphoma may arise in almost any tissue in the body and includes renal, (para) nasal, central nervous system, ocular, laryngeal and cutaneous forms. 4 Lymphoma may occur as a primary tumor of bone but most frequently occurs in association with multicentric lymphoma or leukemia. 5 Lymphoma tends to infiltrate and replace bone marrow but bone destruction is rarely recognized radiographically or as a cause of clinical signs. 6 In a recent report of feline extranodal lymphoma there were no reports of bone involvement in 149 cases. 4 To our knowledge this is the first report of polyostotic lymphoma with multiple pathological fractures in a cat. The predominantly distal distribution of osseous lesions is rarely described in dogs and humans. 7
The principle differential diagnoses for this patient were multiple myeloma and feline infectious peritonitis. Immunohistochemistry for leukocyte markers and coronavirus antigens were not supportive for either of these diagnoses. The cat in this report had a biclonal gammopathy and hypercalcemia. Monoclonal and biclonal gammopathies are rarely reported paraneoplastic syndromes in feline lymphoma. 8 Hypercalcemia is uncommonly reported in cats with lymphoma. 9 Hypercalcemia was detected on the initial biochemistry panel but had resolved at the time of the second panel. This may reflect treatment with IV fluids, prednisolone or a spurious result. Unfortunately, ionized calcium was not assessed. Azotemia was detected on the initial biochemistry and subsequently resolved 4 days later at the time of the second biochemistry panel. Potential explanations for the azotemia include pre-renal causes such as hyperviscosity secondary to hyperglobulinemia and hypovolemia, and renal causes such as hypercalcemia, acute kidney injury secondary to meloxicam and lymphoma infiltration of the kidneys. Resolution of the azotemia following IV fluid therapy and prednisolone is more suggestive of a pre-renal cause.
The severe non-regenerative anemia, thrombocytopenia and neutropenia were pertinent hematological findings in this case. These changes were most likely attributable to myelophthisis owing to infiltration of large numbers of neoplastic lymphocytes within the bone marrow. The circulating atypical lymphocytes most likely represented a neoplastic population; however, in the absence of further tests, such as flow cytometry and polymerase chain reaction for antigen receptor rearrangement, a reactive population cannot be excluded.
Should the diagnosis have been obtained ante-mortem, systemic chemotherapy with a multi-agent protocol utilizing vincristine, cyclophosphamide, doxorubicin and prednisolone could have been attempted. External beam radiation therapy centered on lytic bony lesions could have been utilized concurrently for palliation of bone pain. Given the severe and widespread nature of the disease euthanasia would also have been a valid option.
A review of the literature identified two feline cases of lymphoma in which the vertebral bodies were involved.10,11 One patient had lymphoma of the cauda equina involving the body of L7 and the cranial sacrum. 10 Surgical decompression yielded a diagnosis but did not lead to clinical improvement, hence the cat was euthanased. The second patient had a large anaplastic spinal B-cell lymphoma of the cervical spinal region. Treatment was not attempted and euthanasia was performed. On post-mortem examination an extradural mass at the C1—C2 spinal cord segment was identified with neoplastic cells in the adjacent vertebral bodies, surrounding skeletal muscle, axillary lymph node and femoral bone marrow. 11 Neither had polyostotic involvement nor pathological fracture.
There are 13 reported cases of polyostotic lymphoma in dogs, ranging in age from 15 weeks to 10 years.12–17 Nine of these patients were less than 2 years of age. Four cases involved appendicular sites (primarily long bones), three involved axial sites and six involved both appendicular and axial sites. Only one patient had involvement of distal sites as seen in this case. Eight patients had pathological bone fractures.
Osseous involvement of lymphoma is reported uncommonly in the human literature in both adults and children and is typically associated with non-Hodgkin’s lymphoma and acute lymphoblastic leukemia.18–20 Pathological fracture is uncommon even in patients with involvement of the long bones.21,22
In the case presented here the histopathology, immunohistochemistry and clonality were consistent with lymphoma of the B-cell phenotype. The lineage assignment of feline lymphomas has been performed mainly by immunohistochemistry. 23 Similar to this study, previous studies have reported cases determined as B cell lymphoma without expression of CD79a. Therefore, identification of B-cell lymphoma in cats should require another B-cell marker, such as CD20 or BLA-36 in addition to CD79a. 23 Clonality is a fundamental property of neoplasia, hence evaluation of the clonal nature of a population of lymphocytes can facilitate the diagnosis. The assessment of clonality is commonly used as an adjunctive test for the diagnosis of suspected B-cell neoplasia in humans. 24 In cats, reported sensitivity and specificity for clonality testing at the IgH (B cell) locus have been reported at 68–89% and 75%, respectively.25,26
Although an isolated case, based on the findings reported here, feline lymphoma should be considered when polyostotic lesions are detected, particularly in a young patient.
Footnotes
Funding
The authors received no specific grant from any funding agency in the public, commercial or not-for-profit sectors for the preparation of this case report.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
