Abstract
Overview of Musculoskeletal Tumours
Musculoskeletal tumours are much less common in the cat than the dog. They can be broadly divided into those affecting bone and cartilage, and those affecting muscles and soft tissues around bone and joints. There is some overlap, however, since soft tissue tumours may invade and metastasise to bone and cartilage.
Tumours affecting the soft tissues include those derived from muscle and connective tissues. Although both benign and malignant tumours may occur, most are malignant (ie, sarcomas). Soft tissue sarcomas account for approximately 7–9% of all malignant skin and subcutaneous tumours in cats.1,2 Of the primary tumours affecting bone, most are malignant (67–90%) and osteosarcoma (OSA) is the most common, accounting for 70–80%. 3 Other diagnoses include juxtacortical osteosarcoma, chondrosarcoma, ossifying fibroma, chondroma, osteochondroma and osteochondromatosis (multiple cartilaginous exostosis). In addition, there are single case reports of reticulum cell tumour, osteoid osteoma, osteoma, osteocarcinoma and giant cell tumour. Despite these reports, bone tumours remain an uncommon diagnosis in the cat (3.1–4.9 per 100,000 cases).4,5
Musculoskeletal neoplasia is an uncommon but important differential diagnosis for cats presenting with lameness, pain or swellings associated with bones and/or soft tissues.
Other bone lesions may mimic primary tumours such as aneurysmal bone cysts and fibrous dysplasia. Tumours may also invade bone from adjacent soft tissue and these include, most commonly, squamous cell carcinoma, but also adenocarcinoma, fibrosarcoma, lymphosarcoma, rhabdomyosarcoma, haemangiosarcoma, melanoma, meningioma and reticulum cell tumour. Myositis ossificans is not a tumour but has been documented to cause bony proliferation and soft tissue swelling. 6
Soft Tissue Sarcomas
Soft tissue sarcomas are derived from embryonic mesenchymal tissues. Although they include a variety of histological types, they are usually considered as a group as they share similar clinical characteristics and behaviour. Sarcomas may occur at any anatomical site including internal viscera, but this review will focus on those occurring in muscle, connective tissues and also subcutaneous sites (as these may infiltrate deeply into underlying muscle).
In cats, fibrosarcomas are the most commonly reported mesenchymal tumour (Figure 1). Haemangiosarcomas are less common (see box below), and other tumour types including peripheral nerve sheath tumours (PNSTs; see box), rhabdomyosarcomas, malignant fibrous histiocytomas, histiocytic sarcomas, leiomyosarcomas, liposarcomas, synovial sarcomas and myxosarcomas are much less frequently reported than in dogs. Feline restrictive orbital myofibroblastic sarcoma and feline sarcoids are very uncommon but important differentials (see box).

Fibrosarcoma on the dorsum of a 14-year-old male neutered domestic shorthair (DSH) cat. The nodule was alopecic, firm and relatively mobile on palpation
Some important differentials

Regardless of their histological type, all sarcomas are invasive tumours that extend deep into surrounding tissues. The presence of a ‘pseudocapsule’ formed by pressure on and compression of surrounding tissues often leads to inappropriate ‘shelling out’ of sarcomas at surgery. Local tumour recurrence is, therefore, common as tendrils of tumour cells extend far beyond the pseudocapsule (Figure 2). Metastasis is variable and differs between histological types; for example, it is reported for fibrosarcomas and haemangiosarcomas but not for PNSTs. 9 Adequate local control of small tumours (<2 cm) can, therefore, achieve quite long median survival times (608 days), with certain histological types (fibrosarcoma, PNST) leading to a more favourable outcome. 1
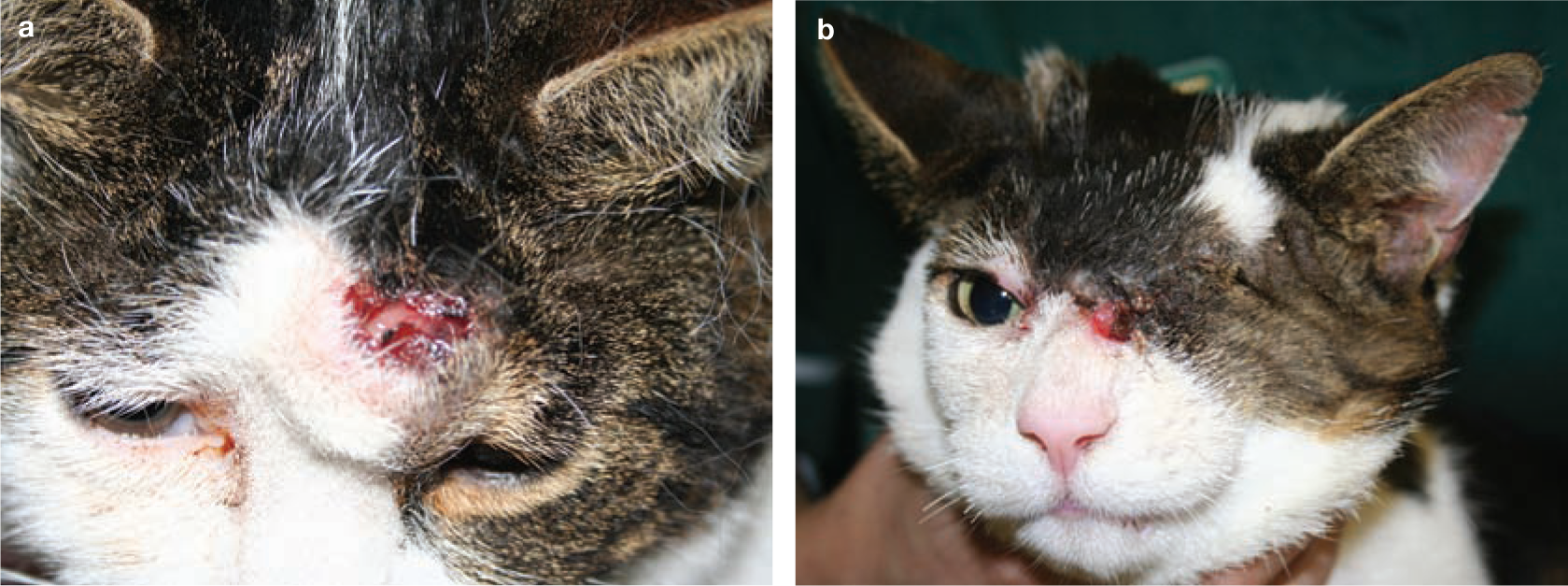
Fibrosarcoma on the head of an 8-year-old male neutered DSH cat. (a) Gross appearance of the tumour above the left eye before surgery. (b) Local recurrence at the medial canthus 4 months after surgery, despite CT imaging prior to surgery, and wide surgical excision including enucleation
The aetiology of many feline sarcomas is not known, although a subset (particularly in the USA) are associated with vaccine injections and are, therefore, of huge economic importance to the veterinary profession. These are discussed separately in more detail below as there is a vast literature relating to these tumours.
In a small number of other sarcomas, the aetiology is viral. Up to 2% of fibrosarcomas are caused by feline leukaemia virus (FeLV) DNA provirus combining with a cellular proto-oncogene to form feline sarcoma virus (FeSV), which transforms fibroblasts to produce tumours. 2 FeSV is non-transmissible between cats, relying on FeLV helper virus for replication. FeSV fibrosarcomas occur most often in young cats, are multicentric and grow rapidly, metastasising in up to a third of cases. This is in contrast to non-viral fibrosarcomas, which tend to be solitary and are found in older cats. A virus may also be responsible for another type of low grade skin sarcoma, the feline sarcoid, although it is uncertain whether this uncommon skin condition is truly neoplastic. 13 Feline sarcoma-associated papillomavirus (FeSarPV), a novel type of delta-PV, is proposed as the causative agent, being transferred cross-species from a reservoir host, most likely cattle.13,14
Feline injection site sarcoma
Feline injection site sarcomas (FISSs) were originally called vaccine-associated sarcomas (VASs) after an increased incidence of these tumours was observed 20 years ago in the USA following new recommendations for feline rabies vaccination in Pennsylvania. 18 Subsequently several studies supported the relationship between vaccination (mainly rabies and FeLV) and tumour development.19–21 These tumours have now been reported world-wide22–24 and may occur following injection of other foreign materials as well as vaccines, hence the change of name to FISS.
Pathogenesis and pathology
The injected material induces a chronic inflammatory response that progresses to neoplasia of various histological types (see box on page 46), all of which are characterised by peripheral lymphocytic aggregates, central necrotic foci, and the presence of myofibroblasts and pleomorphic spindle cells, which infiltrate deeply into surrounding tissues.25,26 Compared with non-injection site sarcomas, there are more features of aggressive biological behaviour such as nuclear and cellular pleomorphism, a high mitotic rate and tumour necrosis (Figure 3). Growth factors and cytokines released by the inflammatory cells, in conjunction with changes in oncogenes and tumour suppressor genes, contribute to the neoplastic transformation. PDGF, EGF, TGFb, and their receptors, TGFa, FGFb and c-jun, are expressed in FISSs, as demonstrated immunohistochemically.2,23,27 Mutations in the p53 gene have been identified 28 and immunohistochemical expression of p53 is also abnormal in FISSs.29,30

Histological appearance of typical feline injection site sarcoma (FISS). (a) Low power magnification (x4) showing dense streams and bundles of spindleshaped to fusiform cells with eosinophilic fibrillar cytoplasm and indistinct cell borders among an eosinophilic extracellular matrix. Aggregates of inflammatory cells are present at the periphery of the tumour (arrows). (b) Higher power magnification (x20) showing anisocytosis, anisokaryosis and many mitotic figures. Frequent multinucleated giant cells are present throughout
Presentation
The incidence of FISS is relatively low, with estimates in the USA ranging from 1–3 sarcomas per 10,000 vaccinated cats.27,31 Few data are available for other countries, although a preliminary survey suggests they are also rare in the UK. 24 FISSs are often found in younger cats (6–7 years), although a bimodal distribution is reported, with a second peak at 10–11 years. The history is important to identify their location as being a site of previous injection/vaccination, although the time scale following injection can vary from months to years. 23 FISSs grow rapidly and may be larger at the time of diagnosis than other feline sarcomas (Figure 4), with a central cystic or necrotic cavity (Figure 5).

The gross appearance of FISS is typically a large subcutaneous soft tissue mass in the scapular region at a site commonly used for routine vaccination. (a) Fibrosarcoma. (b) Malignant fibrous histiocytoma (being grasped by the author)

Contrast-enhanced thoracic CT image of a 12-year-old male neutered DSH cat demonstrating the cystic appearance of a FISS over the thoracic wall. A soft tissue mass is clearly visible (arrows) laterocaudal to the right scapula, with a non-enhancing, hypoattenuating centre and moderate enhancement at the periphery. It does not appear to invade the deeper muscles, and no obvious periosteal reaction of the underlying ribs is seen
Fibrosarcoma
Myofibroblastic sarcoma
Myxosarcoma
Malignant fibrous histiocytoma
Undifferentiated sarcoma
Osteosarcoma
Chondrosarcoma
Rhabdomyosarcoma
Diagnosis
As the inflammatory component may make cytological interpretation difficult and unreliable, a biopsy is needed for definitive diagnosis. Incisional biopsy is preferred to Tru-cut biopsies to prevent misdiagnosis based on small tissue fragments. The site of the biopsy should be chosen carefully to ensure it can be removed in any excisional surgery likely to follow. The Vaccine-Associated Feline Sarcoma Task Force (VAFSTF) recommendations for biopsy of masses at injection sites are known as the ‘3-2-1 rule’ (see box below). All histologically confirmed FISSs should be reported to the manufacturer and any drug reaction monitoring body appropriate to the country (eg, the Center for Veterinary Medicine, Food and Drug Administration in the USA, or the Veterinary Medicines Directorate in the UK).
Staging
Staging of confirmed tumours should include haematology, biochemistry, feline immunodeficiency virus (FIV) and FeLV status, urinalysis, three-view chest radiographs and abdominal ultrasound, as the most common sites of metastasis are the lungs, followed by lymph nodes and abdominal organs. Local invasion is more of a concern than distant metastasis (which occurs in 0–28% of FISSs) and, consequently, advanced imaging (CT/MRI) of the tumour site is also recommended to determine the infiltrative extent prior to excision (Figure 6).
Vaccine-Associated Feline Sarcoma Task Force

Treatment
Consultation with an oncologist is recommended prior to commencing treatment since FISSs are notoriously difficult to treat effectively. Success rates are higher in the hands of experienced surgeons at referral practices.
The mainstay of treatment remains wide surgical excision of the primary tumour, aiming for 3–5 cm margins of macroscopically healthy tissue and at least one fascial plane beneath the tumour.27,32 To achieve complete excision, amputation of limbs or resection of vertebral spinous processes, ribs or scapulae may be necessary. Tumours excised completely are associated with a longer disease-free interval (DFI) than those with dirty margins,32,33 although the asymmetrical and infiltrative nature of the tumours makes assessment of margins extremely challenging for pathologists. Excised specimens should be marked with India ink or suture tags to provide an anatomical reference to facilitate subsequent treatment. A 3D histology technique to standardise margin evaluation and predict tumour recurrence has recently been recommended. 34
Multimodality therapy is acknowledged to be better than surgery alone and several studies recommend the use of postoperative adjuvant radiotherapy, even after surgical margins appear clear, or as neoadjuvant therapy (Table 1). Despite combining these two treatment modalities, rates of local recurrence remain relatively high (28–45%).35,37–40
Summary of treatments used for feline sarcoma

Median not reached, so range given. ST = survival time, DFI = disease-free interval, Sx = surgery, XRT = radiotherapy, CTX = chemotherapy, Dox = doxorubicin, Doxil = liposome-encapsulated doxorubicin, Cyclo = cyclophosphamide, d = days, mo = months

Imaging of FISS to determine the local extent. (a) Right lateral thoracic radiograph of a 9-year-old female neutered DSH cat (same cat as Figure 3), demonstrating the limitations of radiography for identifying the extent of FISS. A soft tissue mass is visible dorsal to the scapulae (arrows), but its extent cannot be accurately defined. (b) Sagittal and (c) transverse MR images (T1 weighting + gadolinium contrast) showing the same tumour with its extent and cystic nature clearly identifiable (arrows). The mass is isointense to muscle with a hypointense cyst-like region to the left of midline, which was markedly hyperintense on T2
Chemotherapy with doxorubicin has been used in several studies, either alone or in combination with radiotherapy, and other agents such as liposome-encapsulated doxorubicin, ifosfamide, cyclophosphamide and carboplatin have also been tried. The authors favour single-agent doxorubicin, substituting mitoxantrone in cats with reduced renal function. Although it is difficult to compare all the treatment studies for FISS, the current recommendation seems to be a combination of surgical excision, radiotherapy and chemotherapy.23,31 For uninsured cats, however, the cost of this multimodality treatment may be prohibitive and, in such cases, emphasis should be placed on extensive first attempt surgery to delay or prevent local recurrence.
In addition to conventional therapy, some new treatments for FISS are being investigated (see right).
Following treatment, cats should be checked monthly for the first 3 months, then every 3 months for 1 year, looking for local recurrence or metastasis.

Prognosis
Recurrence at the local site, metastatic spread, large tumour size, high histological grade, excision by less experienced surgeons and axial site of tumour have all been associated with a worse prognosis. 47 Early diagnosis and referral for radical first attempt excision offer the best chance of long term control.
Prevention
VAFSTF recommends that vaccination should be carried out in the distal limbs so that surgical amputation is possible should a FISS develop. To monitor which vaccines cause the most tumours, the current standard is for FeLV vaccination to be given as distally as possible in the left hind limb and rabies to be given as distally as possible in the right hind, with other vaccinations to be given in the right shoulder, avoiding the midline or interscapular space. A recent study, however, monitoring the location of FISSs before and after introduction of the recommendations, showed that although the number of FISSs detected in the cranial part of the body has decreased, not all practitioners are following the guidelines. 48 A high proportion of tumours still developed in the interscapular region and lateral abdominal sites, which may be attributable to aberrant placement of injections intended for pelvic limbs. 48 VAFSTF also recommends that individual cats should not be vaccinated more frequently than necessary and only against infectious agents to which they have a realistic risk of exposure.
The current recommendation for FISS seems to be a combination of surgical excision, radiotherapy and chemotherapy. The authors favour single-agent doxorubicin, substituting mitoxantrone in cats with reduced renal function.
Bone Tumours
As OSA is the most common feline bone tumour, it is discussed in detail below, along with bone neoplasia resulting from metastatic spread to distant sites. Other, less common bone tumours are summarised in Table 2.
Summary of tumours affecting bones in the cat

Osteosarcoma
OSA is defined as a tumour consisting of malignant mesenchymal spindle cells capable of producing an extracellular matrix of bone or osteoid.
Pathogenesis and pathology
Most commonly, OSA is of medullary origin. Although parosteal OSAs are also described, these tumours arise from periosteal connective tissue and are, therefore, not truly of bone origin. In general, feline OSAs are slow growing compared with those in dogs, meaning that median survival times in cats (treated with amputation) are more than double those of dogs (treated with amputation and adjunct chemotherapy): approximately 24–44 months versus 8–18 months, respectively (Table 3).2,51 If metastasis occurs it is usually late in the disease process. There is a lack of long term follow-up in many reports, however, and it should also be noted that some cases can still develop widespread metastatic disease. 2
Comparative aspects of feline and canine OSA

On histopathology, feline OSAs are characterised by a population of hyperchromatic, pleomorphic tumour cells with an angular or spindle shape. Multinucleate giant cells (osteoclasts) and mitotic figures occur, but frequencies of these findings vary.
Human and canine OSAs have been sub-classified into the following histomorphological subtypes: osteoblastic, fibroblastic, chondroblastic, telangiectatic and mixed. Within these species there is no significant association between histological subtype and survival, but the histopathological grade appears to have prognostic value.
In the cat, chondroblastic, fibroblastic, telangiectatic and giant cell types of OSA have been described.6,50 More recently, a histopathological grading system has been proposed in which tumours are graded based on presence or absence of tumour cells in blood vessels, number of mitoses, absolute number of neoplastic cells, and degree of matrix production, necrosis and pleomorphism. 52 However, this histopathological grading system does not seem to be a significant prognostic indicator for survival time or DFI.
The only reported genetic change that may affect the pathogenesis of feline OSA is a mutation in the coding region of the p53 gene,53,54 but this has yet to be described as having any prognostic value.
Although mainly a spontaneous tumour, OSA has also been described secondary to forelimb fracture55,56 and radiotherapy. 2
Presentation
Over 300 cases of feline OSA are reported in the literature and, among these, no sex or breed predilection has been consistently detected. The majority of affected cats are domestic short- or longhair cats, but Siamese, Persian and Maine Coon breeds are also described.
OSA occurs most commonly in older cats (mean age approximately 10 years) and there does not appear to be the bimodal age distribution that is seen in canine counterparts. Despite this, the age range varies widely (from 3.5 months to over 17 years); therefore, OSA cannot be excluded in young or very old cats.
OSAs can be classified into the following subgroups according to anatomical location: (1) appendicular skeleton, (2) axial skeleton and (3) extraskeletal. Within these groups a variety of sites have been described (see box on page 50). Skeletal OSA makes up about 60% of all cases, being distributed approximately equally between appendicular and axial sites. In cats, OSA is more common in the diaphyseal than the metaphyseal region of the bone. Of the appendicular OSAs, the hind limbs are more likely to be affected compared with the fore limbs.
Clinical presentation can vary and is very dependent on the anatomical location of the tumour. Appendicular OSA is often associated with bony swelling or limb pain/chronic lameness, whereas axial OSAs commonly cause dental problems, skull deformities, firm swellings and nasal discharge (Figure 7).
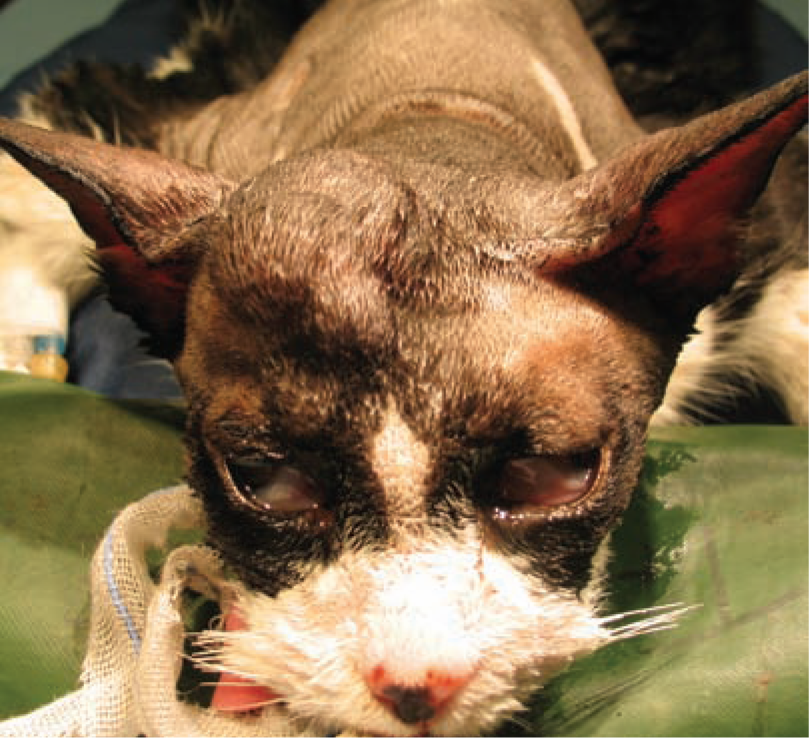
Axial (skull) osteosarcoma visible as an irregular, firm mass over the right side of the cranium
Diagnosis
The radiographic appearance is variable, with changes ranging from purely lytic to proliferative (Figure 8). In one study, 93% of appendicular OSAs were metaphyseal, with a primarily lytic or ‘moth-eaten’ appearance. 3 An ill-defined transitional zone between neoplastic and normal tissue is also described. The frequency of pathological fracture ranges from single case reports to up to two-thirds of all cases. Histopathology is required to make a definitive diagnosis of feline OSA (Figure 9) and methods used to obtain bone for analysis are described in Table 4.
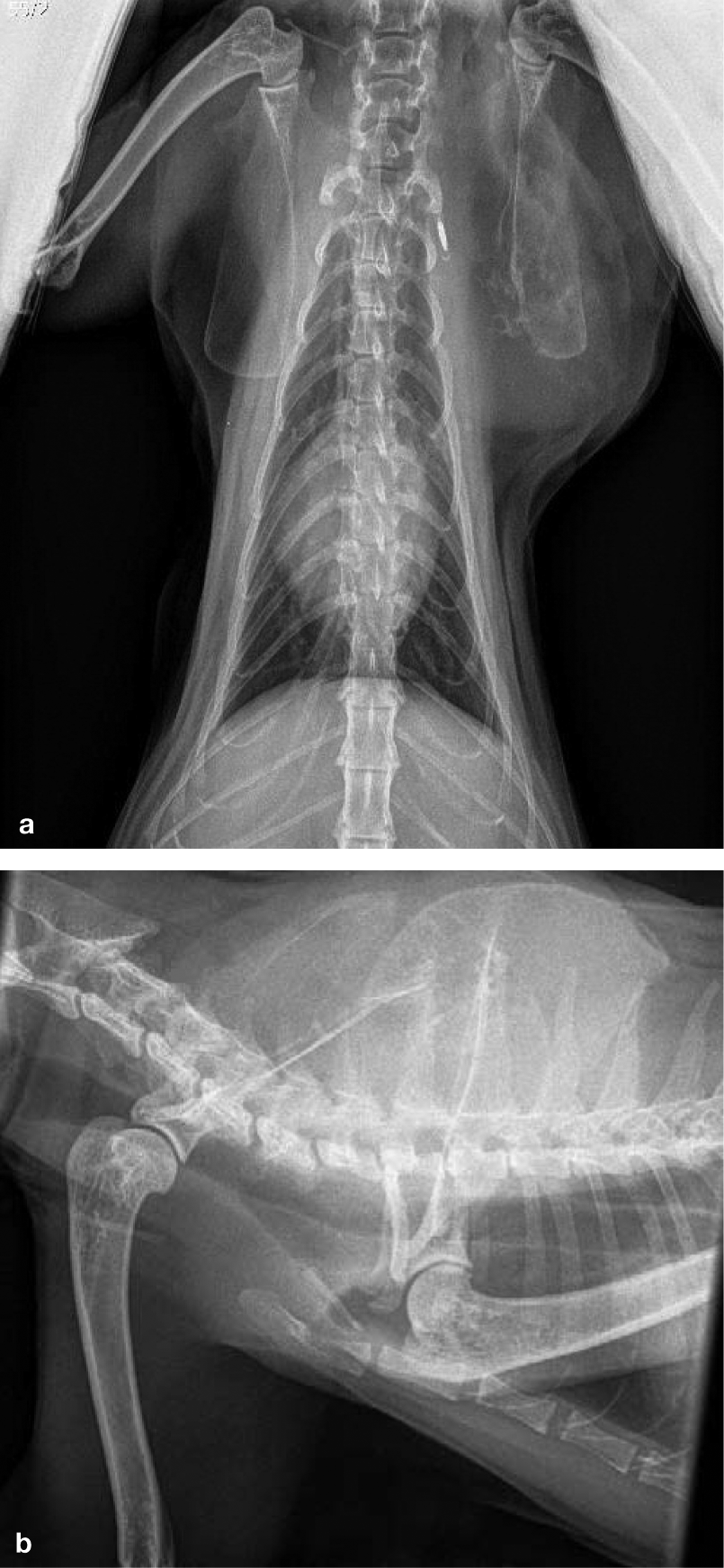
Scapular osteosarcoma. (a) Dorsoventral and (b) right lateral radiographs of a 13-year-old female neutered DSH cat. The left scapula is markedly distorted by a large expansile lytic lesion centred on the dorsal surface. There is extensive surrounding soft tissue swelling causing displacement of the scapula away from the trunk and some irregular, mainly medial, periosteal reaction. There is no evidence of invasion into ribs/vertebrae underlying the soft tissue swelling, but the mass is closely apposed to the microchip
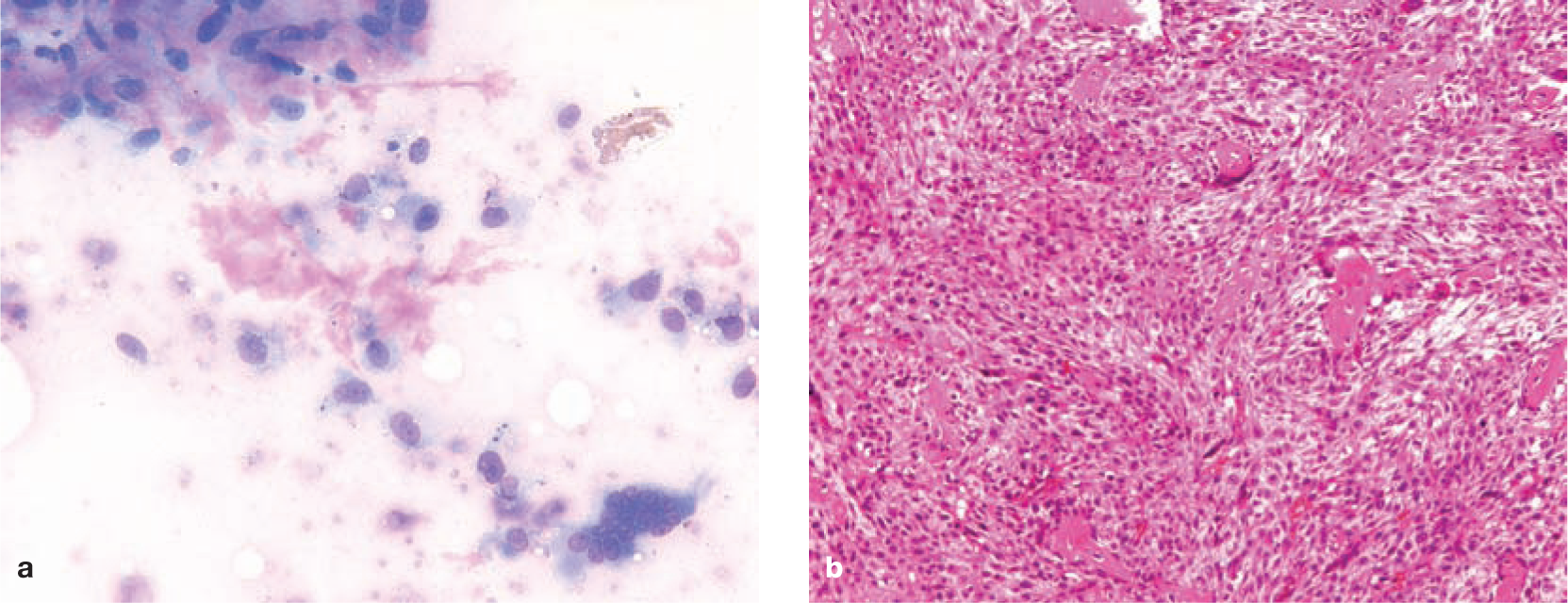
Scapular osteosarcoma. (a) Cytological appearance (x100 magnification) and (b) histological appearance (x20 magnification). On cytology, many large to giant spindle cells are present in loosely cohesive groups. There is marked anisocytosis and anisokaryosis, with open chromatin and prominent nucleoli. Cytoplasm is moderately basophilic with some vacuolation and eosinophilic secretory inclusions. Eosinophilic matrix is also visible. On histology, sheets of pleomorphic, spindle-shaped to polygonal mesenchymal cells are present, destroying the normal bone architecture. There are deposits of osteoid with mineralisation and cartilaginous islands, and multifocal areas of necrosis
Methods used to obtain bone for analysis

Staging
Full haematology, biochemistry, feline virus serology and urinalysis should be undertaken as standard procedures, although results are usually within normal limits. No links to FeLV/FIV have been found, although the number of cats tested in the case series available is low.3,43 Despite the low frequency of abnormalities, it is important to conduct these tests to rule out concurrent disease that may affect treatment options and/or prognosis. Likewise, thoracic radiography and abdominal ultrasound are also advised; however, the risk of distant metastases at the time of diagnosis is much lower than in the dog.
Proximal humerus
Distal femur
Proximal tibia
Digits
Skull
Pelvis
Ribs
Vertebrae
Scapula
Subcutaneous (including interscapular) Mammary gland
Eye
Spleen
Liver
Duodenum
Kidney
Stomach
Treatment
Given the lower incidence of metastases, feline appendicular OSA is usually managed with wide surgical excision (eg, limb amputation).
Adjunctive chemotherapy and radiotherapy are not frequently described and as yet there is no evidence to support adjunct therapy for appendicular OSA following surgical excision. Longer survival times are possible if surgery is combined with these modalities for managing axial OSA, but these results are based on very small numbers of cats. 57 A feline nasal OSA treated by partial surgical excision followed by high dose per fraction radiotherapy achieved over 2 years of complete remission. 58
Supportive care with analgesics is very important. The limited information available about the use of bisphosphonates in cats suggests that they may be beneficial in reducing bone resorption 59 and potentially managing hypercalcaemia; 60 however, they have not yet been trialled in cats with OSA.
Prognosis
Axial OSAs tend to carry a worse prognosis than those affecting appendicular sites, with reports of a median survival time of 5.5 months in six cats 3 and a mean survival time of 6.07 months in 21 cats. 57 However, in cases where complete surgical excision was possible (eg, tail amputation for a vertebral axial OSA), survival was improved (mean survival time 16 months), 57 suggesting larger studies are required to see if statistical significance can be reached.
The comparatively better prognosis for cats with appendicular OSA is reflected by the fact that many cats have a prolonged survival after local therapy. Of 11 cats with postoperative follow-up data available, five were deceased and had a median survival time of 49 months, and six were still alive 13–64 months postsurgery. 3 In another study of 15 cases of appendicular OSA, one of the five cases treated with amputation died 5 months later (due to metastatic disease) but the remaining four were alive up to 26 months postsurgery. 6
Extraskeletal OSA has a variable outcome but, of a group of 55 cats with a mean survival time of 12.67 months, the extraskeletal OSA group had an improved outcome compared with the axial OSA group. 57 This may not be due to an intrinsic difference in tumour behaviour, but rather may simply reflect the fact that a subcutaneous location may facilitate surgical excision. 57
Metastatic Disease (Including Digital Carcinoma)
Several feline tumours are known to metastasise to bone (see box). In the cat, the most common site for metastases are the digits, with pulmonary adenocarcinomas or squamous cell carcinomas being the most common tumours spreading to this site.
Pulmonary neoplasia with metastases to the digits
Pathogenesis and pathology
The exact pathogenesis of metastasis to the digit is unknown. However, digital metastases are extremely rare in the dog and metastases from human bronchogenic carcinoma are usually limited to bones proximal to the knees or elbows, suggesting that cats possess a unique mechanism that makes them more prone to distal (digital) metastases. One explanation for this species difference may be an increased blood supply in cats, which facilitates heat loss through their pads. 61 The finding in one study that the distal phalanx was the site of metastases in all 36 cases with no evidence of dew claw involvement supports this theory. 62
Presentation
This disease is most common in middle- to old-aged cats that present with digital swelling, claw deviation, fixed exsheathment, growth of the claw into the digital pad and lameness. The majority of affected cats are domestic shorthairs although other breeds are described (domestic longhair, Persian, unspecified purebred). No sex predilection has been determined. Interestingly, some cats will present with no signs of respiratory disease despite having intrathoracic masses; hence, thoracic and distal limb radiographs are indicated in middle- to old-aged cats presenting with the above clinical signs. Other differential diagnoses would include traumatic injuries, nail bed infections or primary digital neoplasia.
Diagnosis
Radiographs of the affected digit(s) may show loss of the nail, bone lysis that may cross the joints between P3 and P2, and, in some cases, extensive necrosis of P3 (Figure 10). Extensive periosteal proliferation is also possible. Radiographs of the thorax may reveal a pulmonary mass (primary tumour). Diagnosis is suspected based on the combined findings from thoracic and distal limb radiography; however, any suspicious lesions should be biopsied for histopathological confirmation.
Cats appear relatively prone to distal (digital) metastases – possibly because of an increased blood supply, which facilitates heat loss through their pads.
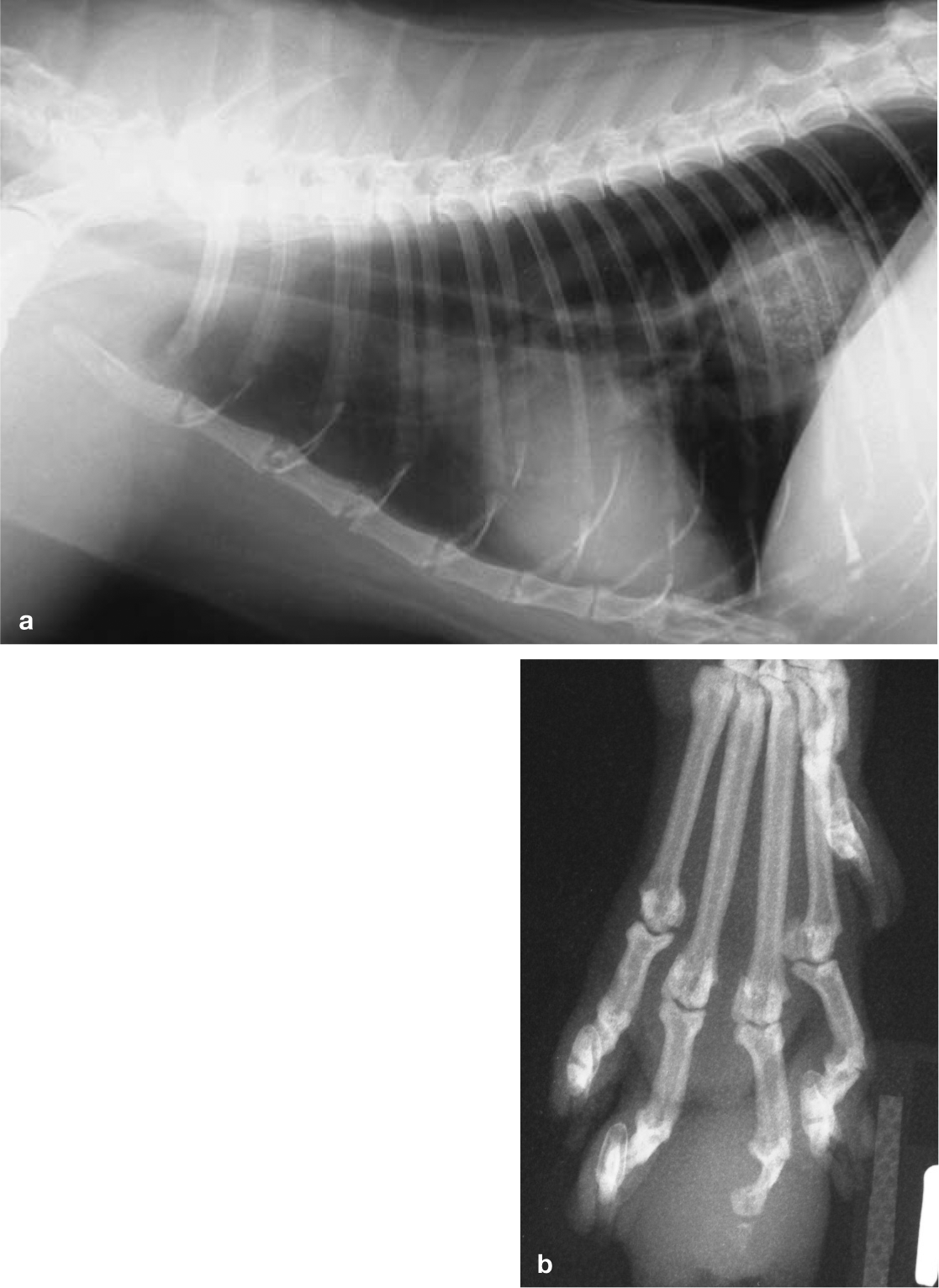
Thoracic and distal limb radiographs from a 21-year-old male neutered DSH cat presenting with lameness. (a) Right lateral view showing a soft tissue mass in the caudodorsal lung field (diagnosed as bronchial carcinoma on aspiration cytology). (b) Dorsopalmar view of the left foreleg showing soft tissue swelling of the third digit, extensive lysis of P3 and loss of nail, indicative of tumour metastasis to this site
Primary pulmonary neoplasia:
–Squamous cell carcinoma
–Pulmonary carcinoma
–Pulmonary adenocarcinoma
Primary mammary carcinoma
Eyelid carcinoma
Pancreatic carcinoma
Staging
Routine haematology, biochemistry, feline virus serology and urinalysis are worthwhile to evaluate the patient for concurrent disease, while thoracic imaging is of obvious importance. Abdominal imaging (usually via ultrasonography) is advisable as concurrent extraskeletal metastases (lymph nodes, lung, spleen, kidney, skeletal muscle and heart) are described.
Treatment
Generally, therapeutic strategies are not reported. Digital amputation could be considered but as multiple digits are usually affected this is rarely palliative.
Prognosis
Overall the prognosis is poor for cats with metastatic digital tumours, with median survival times of 1–2 months being reported.
Key Points
Feline injection-site sarcomas (FISSs) are aggressive tumours that are best treated with a multimodality approach: radical surgery, radiotherapy and chemotherapy.
Vaccination in the distal limbs is recommended by the Vaccine-Associated Feline Sarcoma Task Force to allow a greater chance of curative surgery (amputation) should a FISS develop.
Feline osteosarcoma (OSA) is slower growing and carries a better prognosis than its canine counterpart.
Amputation alone may allow prolonged survival in cases of feline OSA.
Pulmonary neoplasia with digital metastasis is an important differential if an older cat presents with digital swelling, claw deviation, fixed exsheathment, growth of the claw into the digital pad and lameness.
Footnotes
Acknowledgements
The authors would like to thank Donald Sheahan, the Diagnostic Imaging Service (University of Glasgow) and the Veterinary Diagnostic Service (University of Glasgow) for contributing suitable figures for use in this article.
Funding
The authors received no specific grant from any funding agency in the public, commercial or not-for-profit sectors for the preparation of this review article.
Conflict of interest
The authors declare that there is no conflict of interest.
