Abstract
Testicles of 30 mongrel cats were analyzed histologically and morphometrically, divided into three groups: G1 (1–2 years old), G2 (over 2 and up to 4 years old) and G3 (over 4 and up to 6 years old). After orchiectomy and histopathology, the morphometric parameters studied were: thickness of the tunica albuginea (72 μm) and seminiferous epithelium (77.19 μm), perimeter (53.81; 90.57 μm), (54.80; 101.07 μm); area (174.23; 494.55 μm2), (176.68; 629.70 μm2); maximum diameter (14.94; 28.02 μm), (14.76; 31.66 μm); minimum diameter (13.25; 21.92 μm), (13.30; 24.52 μm); and shape factor (index for regularity of the format) (1.36; 1.36), (1.39; 1.35) of the nucleus and cytoplasm of spermatogonia and Leydig cells, respectively. The results can be used for comparative studies and contribute knowledge concerning the height of the seminiferous epithelium, thickness of the tunica albuginea and size of spermatogonia and Leydig cells.
Introduction
The knowledge of the reproductive function of domestic cats is of great importance because of similarities between the reproductive anatomy of Felis catus and the majority of wild felines. As orchiectomy is one of the most common procedures performed in companion animal surgery, domestic cat gonads are easy to obtain.The increase in studies related to male gonads of this species has scientific relevance, particularly regarding morphology, for which data is scarce, in order to complement the literature and allow for detailed comparison of healthy testicles with those that exhibit alterations and, thus, allow planning of adequate treatment. 1
Therefore, the objectives of this study were to measure the thickness of the tunica albuginea and height of the seminiferous epithelium, and analyze the perimeter, area, maximum and minimum diameter and form factor of the nucleus and cytoplasm of spermatogonia and Leydig cells of adult domestic cats to contribute new information to the scientific literature on histology and animal reproduction.
Materials and methods
Animals
Thirty male, 1- to 6-year-old domestic cats of different breeds and mean body weight of 3 kg were used in this study. The animals were patients of the Obstetrics and Animal Reproduction Sector of the Veterinary Hospital ‘Governador Laudo Natel’ of the School of Agricultural and Veterinary Sciences of São Paulo State University (UNESP), Jaboticabal, SP, Brazil.
All procedures in this study are in accordance with the ethical principles of experimentation, protocol 025893-08 and were approved by the Ethics Committee on Animal Use at UNESP/Jaboticabal.
The animals selected did not have any history of reproductive diseases and were considered healthy after physical and clinical examination. After the selection of animals for the study, they were divided into three groups: group 1 (G1), 1–2 years old; group 2 (G2), over 2 and up to 4 years old; and group 3 (G3), over 4 and up to 6 years old (Table 1).
Experimental groups of domestic cats classified according to age

Orchiectomy
The animals were subjected to 8–12 h of food, and 4–6 h of water fasting. Acepromazine (0.2 mg/kg of body weight, intramuscularly) was used as premedication. Analgesia was obtained with meperidine (0.01 mg/kg of body weight, subcutaneously). Ten minutes later, dissociative anesthesia was performed using an association of tiletamine and zolazepam (10 mg/kg of body weight, intramuscularly).
The surgical technique employed has been described. 2 Bilateral orchiectomy was performed, but only the right testicle was collected for this study. For the postoperative period, topical administration of rifamycin on the surgical wound every 12 h for 5 days and benzathine penicillin (40,000 UI/kg of body weight, intramuscularly) in alternate days for a total of three applications, were prescribed. Finally, ketoprofen (1 mg/kg of body weight, orally, q24h) was prescribed for 3 days, for analgesia. Wound healing was achieved by secondary intention.
Histology procedures
After orchiectomy, the testicles were macroscopically evaluated to eliminate gonads with alterations.The right testicle was identified and washed in running water to remove excess blood. Two transverse sections were performed on the proximal and distal ends of the testicle using a scalpel, without cutting it completely. They were then placed in sterile universal containers, immersed in Bouin’s solution at room temperature.
After 3 h, two other transverse sections were performed at equal distances from the first two and the gonad was re-immersed in the same solution were it was kept for 24 h. The testicles were then sliced into average thickness cuts of 0.5 cm and processed for routine histological techniques for the preparation of paraffin blocks.
Slides were stained with hematoxylin and eosin (HE) and finally mounted using Canada’s balm, identified with the animal’s name and group.
Morphometric procedures
The thickness of the tunica albuginea, height of the epithelium of seminiferous tubules, perimeter, area, maximum and minimum diameters, and form factor of the nucleus and cytoplasm of spermatogonia and Leydig cells, were measured.
Uniform images were selected to measure the thickness of the tunica albuginea. Averages were calculated using two equidistant points in four histological slides per animal.
To measure the height of the epithelium of seminiferous tubules, sections with the most uniform circular outline were selected. Four equidistant measurements were taken and averages were obtained from 10 structures per sample.
Average values for perimeter and area were obtained through analysis of 10 cells of each type per group. The boundaries of the cells were drawn by hand in a computer using a mouse. A calibrated analysis software automatically calculated the parameters.
For calculation of the diameters, 10 cells of each type were visualized and the averages were recorded using two equidistant points.
The form factor was mathematically defined by the expression:
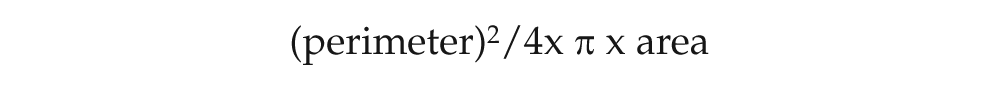
The smallest value for this factor is one, which means that the shape of the cytoplasm is similar to a circle whose area is defined by π × R2 and whose perimeter is defined by 2 × π × R. By replacing area and perimeter values, there is:
Form factor:
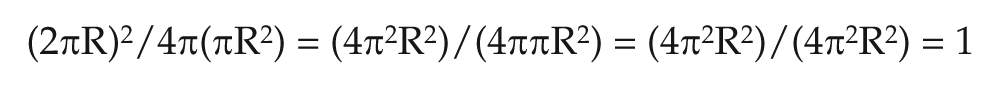
Where π=3.14 and R=radius
If the value of the shape factor was higher than one (> 1), it would mean that the shape of the cytoplasm or nucleus is irregular.
Histological slides were evaluated using an Olympus (Center Valley, PA, USA) BX 50 CCD light microscope and 4x and 40x objectives according to the structure being analyzed, connected to an imaging system (Image Pro plus, version 3.0, Silver Spring, MD, USA) to take pictures of the structures of interest.
Statistical procedures
Data were analyzed using the general linear model (GLM) procedure system by Statistical Analysis System [(SAS) 9.1; SAS Institute, Cary, NC, USA].
Results obtained for cellular morphometry of groups were subjected to single factor (age) analysis of variance (ANOVA) by the least squares method and the averages were compared using the Tukey’s test at 5% level of significance. 3
Linear regressions of the observed values for each characteristic were performed as a function of the age of the animals. The Student’s t-test was used to verify the hypothesis that the regression coefficient of each equation is zero.
Results and discussion
There were no complications regarding the surgical procedures and all animals were released after recovery from anesthesia on the same day of the surgery.
Tunica albuginea and seminiferous epithelium
In all testicles analyzed, the tunica albuginea was thick, formed by a dense connective tissue rich in collagen fibers (Figure 1).
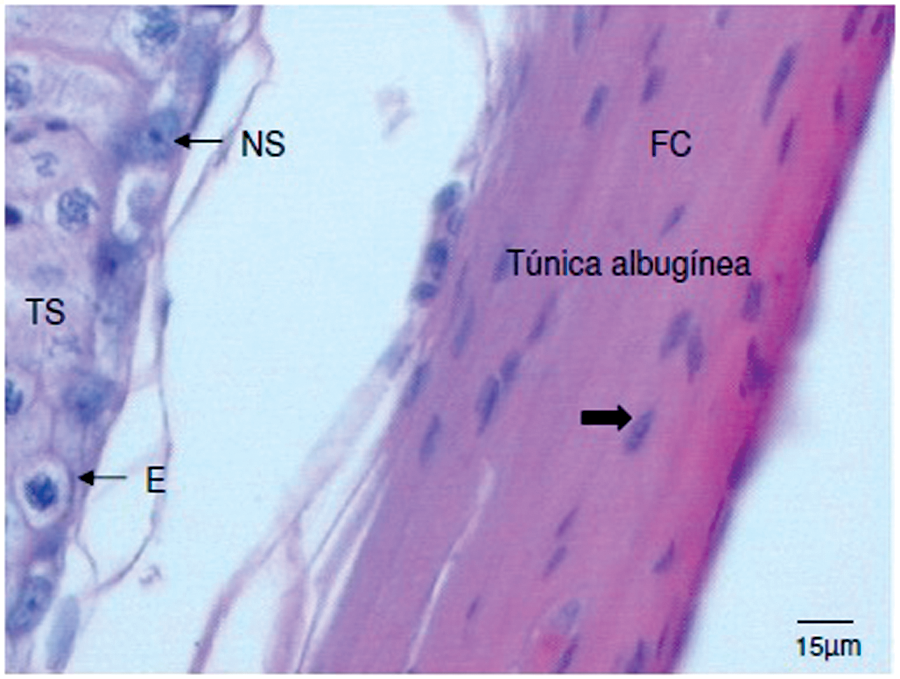
Photomicrograph of transverse cross-section of the testicle of a domestic cat. Note the tunica albuginea, collagen fibers (FC), nucleus of connective tissue cells (large arrow), seminiferous tubule (TS), spermatogonium (E) and nucleus of a Sertoli cell (NS). Stained with hematoxylin and eosin, 20x
Table 2 shows the thickness of tunica albuginea and height of the germinal epithelium of testicles of cats of different ages. None of the two parameters altered significantly between groups. Likewise, the data obtained confirms that there is no tendency for the values to increase as cats grow.
Thickness of the tunica albuginea and seminiferous epithelium in the testicles of domestic cats by age group. Results are expressed in mean ± standard deviation

The tunica albuginea mean values in 4- to 6-year-old cats were similar to those found by França and Godinho and Silva et al,4,5 who studied the structure and function of testicles. They studied the seminiferous epithelium cycle in adult animals and found that cats aged over 8 years exhibited thicker tunica albuginea compared with other mammals and that it was formed by collagen fibers.
On the other hand, relying on statistical results, that did not show differences between experimental groups, there is a hypothesis that the histological constitution of this tunica is not influenced by age. This hypothesis is based on reports by those same authors, which affirm that 1-year-old cats already have a thick tunica albuginea, formed by a dense, shaped connective tissue.
Figure 2 shows the interstitial tissue around the seminiferous tubules, including a large number of Leydig cells and, internally, the lining by a stratified epithelium composed of two types of cells: support cells (Sertoli) and spermatogenic cells — spermatogonia, primary spermatocytes and spermatids.
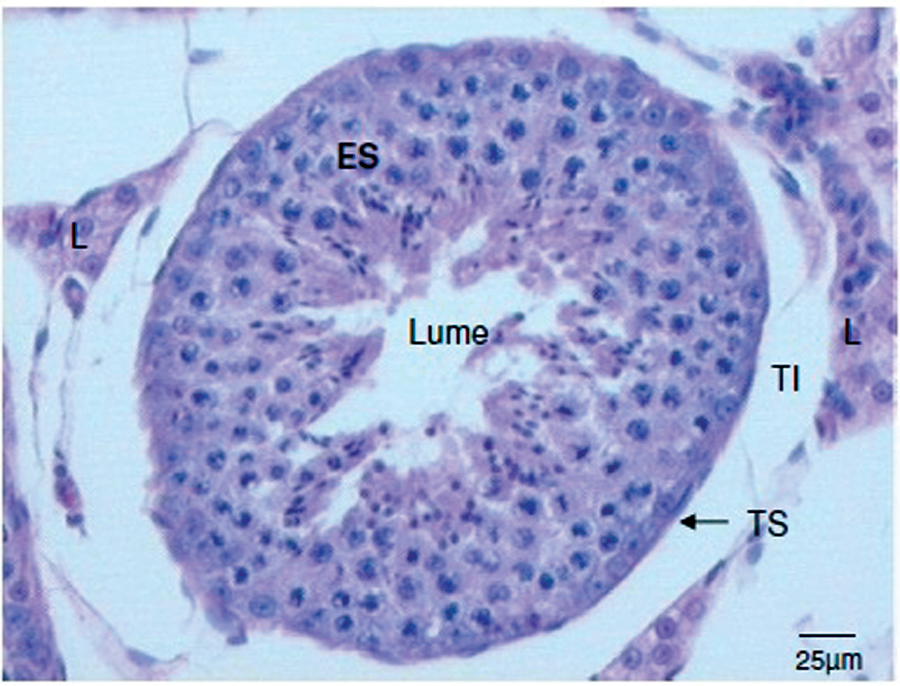
Photomicrograph of transverse cross-section of the testicle of a domestic cat. Note the seminiferous epithelium (ES), Leydig cells (L), and interstitial tissue (TI) of the seminiferous tubule (TS). Stained with hematoxylin and eosin, 10x
The height of the seminiferous epithelium exhibited mean values that are in agreement with a report by Siemieniuch and Woclawek-Potoca, 6 who examined 118 pairs of testicles in 4-month-old to 10-year-old cats and found the following mean values for diameter of seminiferous tubules: 191.83 μm in 8- to 12-month-old cats; 202.61 μm in 1- to 3-year-old cats; 193.38 μm in 3- to 6-year-old cats; and 191.84 μm for cats over 6 years old.The averages found in this study are in agreement with those reported by Hoshino et al, 7 who found mean values of 54.52 μm when researching this structure in 1- to 5-year-old cats, and Neubauer et al 8 who found a minimal tubular diameter of 195.2 μm and height of epithelium of 59.4 μmin 10 1- to 10-year-old cats, and Silva et al, 5 who compared the height of seminiferous epithelium and tubular diameter in 10 young cats (up to 1 year old) and 10 adult cats (over 1 year old) and found mean values of 49.51 μm and 63.29 μm for young and adult cats, respectively. Regarding diameter, they found mean values of 160.58 μm and 185.94 μm in young and adult cats, respectively. When performing histomorphometric measures on the testicles of cats, Soares et al 9 found the mean tubular diameter to be 148.4 μm and the mean height of epithelium to be 56.4 μm.
In 10 1- to 10-year-old cats, the mean values for height of the tubular epithelium were larger (77.19 μm) when compared with the three last studies mentioned above; however, the number of animals analyzed was also higher in this study (n = 30) and the age interval in the groups was more homogeneous, which could explain the difference.
Still regarding the height of epithelium, França and Godinho 4 evaluated adult cats and obtained mean values of 78 μm. While studying seminiferous tubules in cats, Costa et al 10 found the height of epithelium to be 81 μm. Both research groups obtained similar values to those inour study.
These statements are based on the concept that the height of epithelium is basically the radius of the tubule which, in turn, is half of the diameter. Those authors found similar mean values and very close values for the different ages studied; this fact also occurred in the current study, as the height of epithelium was: 78.02 μm in 1- to 2-year-old cats; 83.57 μm in 2- to 4-year-old cats and 66.90 μm in 4- to 6-year-old cats. These findings make it reasonable to assume that the height of seminiferous epithelium is not age dependent in the age interval proposed in this study.
Spermatogonia and Leydig cells
The spermatogonia were located on the basal part of the seminiferous epithelium and had an oval nucleus with one or two nucleoli close to the nuclear envelope. Spermatogonia, as stem cells, originate another lineage of cells necessary for spermatogenesis. They exhibited a similar and normal histophysiological behavior for all ages, which is shown by the similar seminiferous epithelium height in the different age groups evaluated. In humans, these cells are considered small and measure 12 μm in diameter. 11 In 1- to 6-year-old cats, the mean value for this parameter was 28.02 μm.
There was loose connective tissue with Leydig cells located close to blood capillaries, exhibiting polygonal shape with spherical nucleus between seminiferous tubules (Figure 3).

Photomicrograph of transverse cross-section of the testicle of a domestic cat showing Leydig cells (L) in the interstitium (I), germinal epithelium with spermatogonium (E) and spermatocyte I (ES). Stained with hematoxylin and eosin, 20x
Tables 3, 4, 5, 6 and 7 show perimeter, area, maximum diameter, minimum diameter and form factor, respectively, of spermatogonia and Leydig cells of cats in different age groups. None of the parameters analyzed had significant difference when comparing the experimental groups.
Nuclear perimeter (NP) and cytoplasm perimeter (CP) of spermatogonia and Leydig cells in the testicles of domestic cats by age group. Results are expressed in mean ± standard deviation

Nuclear area (NA) and cytoplasm area (CA) of spermatogonia and Leydig cells in the testicles of domestic cats by age group. Results are expressed in mean ± standard deviation

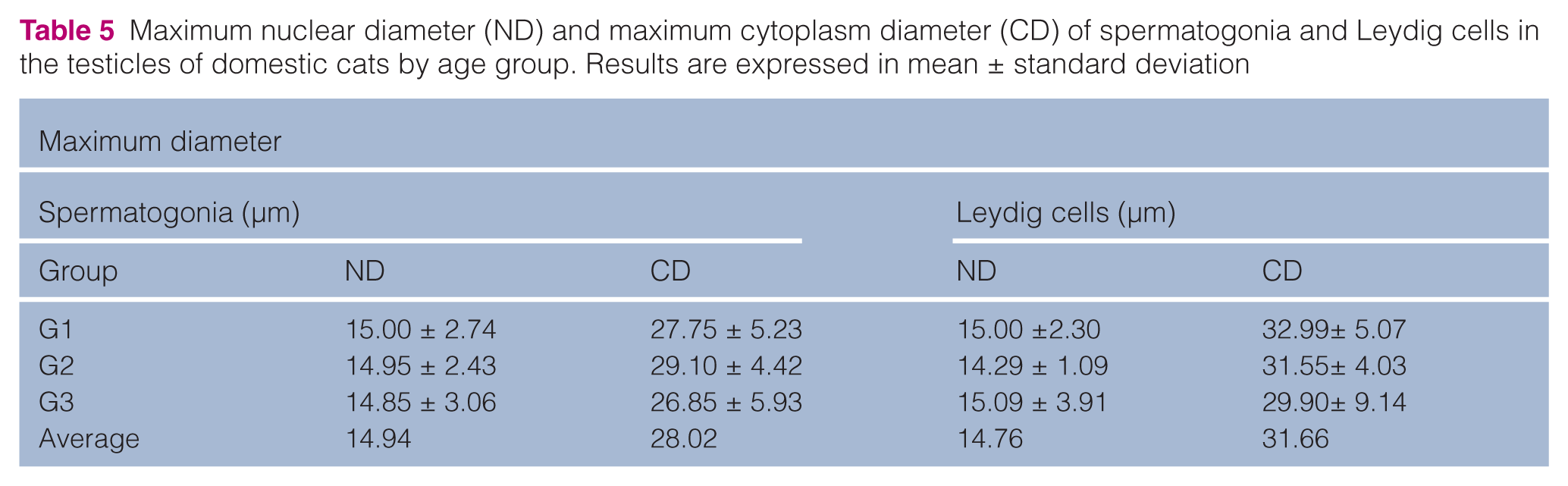
Maximum nuclear diameter (ND) and maximum cytoplasm diameter (CD) of spermatogonia and Leydig cells in the testicles of domestic cats by age group. Results are expressed in mean ± standard deviation
Minimum nuclear diameter (ND) and minimum cytoplasm diameter (CD) of spermatogonia and Leydig cells in the testicles of domestic cats by age group. Results are expressed in mean ± standard deviation

Nucleus form factor (NFF) and cytoplasm form factor (CFF) of spermatogonia and Leydig cells in the testicles of domestic cats by age group. Results are expressed in mean ± standard deviation

Recalling the classic literature, spermatogenesis is influenced by follicle stimulating hormone (FSH), which also stimulates Sertoli cells to produce androgen binding protein (ABP). The latter is associated with testosterone and increases concentration in the seminiferous tubule. Thus, it can be concluded that different age groups in cats did not influence maximum and minimum diameters of nucleus and cytoplasm of Leydig cells, which produce testosterone, and this was reflected in the similar values found for the groups, namely: maximum and minimum diameters of nucleus, respectively, 15.00 μm and 13.37 μm (group 1); 14.29 μm and 12.78 μm (group 2) and 15.09 μm and 13.93 μm (group 3). In the same manner, maximum and minimum diameters of cytoplasm were, respectively: 32.99 μm and 24.09 μm (group 1); 31.55 μm and 26.96 μm (group 2) and 29.90 μm and 21.66 μm (group 3).
França and Godinho 4 reported a mean value of 7.9 μm for nucleus diameter of Leydig cells in 13 testicles of adult cats. The value found by them is lower than the 14.76 μm found in our research; this difference could be explained by the larger number of testicles (30) studied here.
The regression coefficient of the cytoplasm form factor of spermatogonia was significant and the tendency of these values as a function of age of the animals can be observed in Figure 4 (line equation y = a + bx/FF cytoplasm = x-intercept + regression coefficient x ages).

Graphic representation of averages of cytoplasm form factor of spermatogonia of cats classified into groups according to age, group 1 (1–2 years old), group 2 (over 2 and up to 4 years old) and group 3 (over 4 and up to 6 years old)
The nucleus and cytoplasm form factors of spermatogonia and Leydig cells exhibited mean values higher than one, even though they were relatively close to one. This suggests that the nucleus and cytoplasm of these cells have a shape that is similar to a circle.
On Figure 4, note that the cytoplasm of spermatogonia get slightly more irregular as cats get older. We believe these data can aid more detailed oncology studies in animals with suspected tumors, making an analogy to data reported by Watanabe et al, 12 which found nuclear heterogeneity in ovarian epithelial cells, characterized by high form factor values and giant nuclei. They affirmed that this characteristic can be related to recurrence of ovarian adenocarcinoma in women.
Following the same train of thought, Yin and Gu 13 calculated nuclear form factor, and area and perimeter of epithelial cells, when studying ovarian epithelial tumors in women, and reported that those values aided in triage of patients, diagnosis and prognosis of neoplasia.
Given the above, these data will contribute to the areas of histology, morphology and reproduction of domestic cats, as well as work as experimental models for other felines. However, further studies on this subject should be explored to increase qualification of these parameters and contribute directly to the health of domestic cats.
Conclusions
It can be concluded that the thickness of the tunica albuginea, height of the seminiferous epithelium, mean values of perimeter, area, maximum and minimum diameters of nucleus and cytoplasm of spermatogonia and Leydig cells are not age-dependent between 1 and 6 years of age.
It was observed that older animals exhibit spermatogonia irregularity and that cell size in animals aged 2–4 years was always larger when compared with other age groups, which could possibly explain the better reproductive performance in this age group.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
The authors declare that there is no conflict of interest.
