Abstract
Cryptosporidium is a coccidian that can lead to diarrhea, especially in immunosuppressed individuals. Retroviruses are considered a primary cause of immunosuppression in cats. Fecal specimens and blood collected from the 60 cats were evaluated for the presence of acid-fast cryptosporidia in three consecutive stool samples and for feline leukemia virus (FeLV) antigen and feline immunodeficiency virus (FIV) antibody by ELISA testing. Five animals (8.33%) shedding oocysts were found, one was both FIV- and FeLV-negative and four were FeLV-positive.
Introduction
Cryptosporidium (Phylum: Apicomplexa) is an obligate intracellular coccidian, whose trophozoites reside in a parasitophorous vacuole at the enterocyte surface. The vacuoles are formed by host cell and coccidian membranes.1,2 Cryptosporidia were first reported by Tyzzer in 1907 and, until the early 1980s, they were considered a parasite of veterinary interest only, and found uncommonly in poultry and cattle. 2 The immunosuppression associated with human immunodeficiency virus (HIV) resulted in increased interest in these organisms, as they were a cause of severe diarrhea in humans infected with HIV and were considered potentially zoonotic.3,4
Transmission occurs by the orofecal route, primarily through ingestion of contaminated water. However, contaminated food and direct contact with animals, as well as with contaminated surfaces, can be a source of infection.4–8 Cattle are considered the main reservoir,5,9 but this parasite has also been found in wild animals, dogs, cats, small mammals and avian species kept as pets.5,6 It is suggested that the zoonotic potential is minimal and human and animal transmission is more likely from contact with contaminated water and environment rather than from each other.10–13
In mammals, Cryptosporidium species encyst within vacuoles along the microvillus surface of the small intestine and both sexual and asexual stages occur within the same host. In humans and immunocompetent animals, infection can be self-limiting or even asymptomatic but, in immunodeficient patients, it can become severe and chronic.3–5,9
In felines, the species found are C parvum (Tyzzer 1912) 14 and C felis (Iseki 1979). Rare reports of infection in immunosuppressed humans and one child whose house cat was shedding oocysts can be found in the literature,14–17 but infection from other sources of contamination could not be excluded. Infection in cats may remain unnoticed18–21 and is generally subclinical in cats infected by C felis but may lead to diarrhea when cats are infected by C parvum.12,22
Young animals are more prone to Cryptosporidium species infection and although the relationship between human immune status and cryptosporidiosis has been defined and extrapolated to animals, an association between domestic cats immune status and disease course/signs still has to be established.19,23,24
This coccidian is not routinely identified during parasite fecal screening, as the small oocyst and poorly-defined internal structures lead to confusion with yeasts and other fecal debris.25–27 Concentration methods and specific stains can be used to help in oocysts identification.12,13,22,28–35 The organism is also intermittently shed, making collection of more than one stool sample mandatory.3,4,11,13,22,24,28,34–36
Retroviruses are considered a common cause of immunosuppression in the cat.37–41 It is suggested that animals infected by these retroviruses are more prone to Cryptosporidium species infection and may exhibit more severe clinical signs.23,42
Diarrhea is commonly observed in feline practice; there are many causes, including: bacterial, parasitic, viral, food intolerance or true food allergies, and neoplasia. As Cryptosporidium species could be a significant cause of diarrhea, especially in young or immunosuppressed animals, a routine, simple method to identify the parasite would be helpful. The purpose of this investigation was to evaluate stools from diarrheic domestic cats using a simple and affordable technique, and to correlate the parasite burden of these cats with clinical signs and infections with feline immunodeficiency virus (FIV) and feline leukemia virus (FeLV). 34
Materials and methods
Sixty diarrheic (acute or chronic) domestic cats, aged from 6 months to 13 years old from Rio de Janeiro, Niterói and Praia de Mauá, took part in this survey. The owners gave consent for their pets to participate and for collection of a blood sample and three stool samples from each cat. A history form was filled in by the owner, with information including outdoor access or contact with other animals.
Three stool samples were stored together in a container with a 10% formalin solution present in a 1:2 ratio. The material was passed through gauze mesh and a centrifugal sedimentation concentration method with formalin-ethyl acetate solution was performed (Table 1). After detaching the debris at the surface, the supernatant was discarded and 10 µl of the sediment used to make a smear. The smear was air-dried heat fixed and stained by the Kinyoun method (Table 2). The stained smear was systematically evaluated under light microscopy (100 x objective, immersion). Oocysts are acid, alcohol-resistant, staining red/purple, round and measure 4–6 µm in diameter, with four sporozoites inside. The other organisms and structures that are non-acid-alcohol resistant are stained by the counterstain, blue in this case.
Description of centrifugal sedimentation concentration method using formalin-ethyl acetate solution

Description of Kinyoun staining technique

Sera collected from the animals were used to simultaneously detect FeLV antigens/FIV antibodies (Snap Combo; Idexx Laboratories, Westbrook, ME, USA). Blood was collected in the same week that the stool samples were obtained and tests were carried out on the same day. The tests were run and interpreted according to the manufacturer’s instructions.
Results
Parasite results of the fecal samples are summarized in Table 3. Stained smears identified Cryptosporidium species oocysts in five cats (8.33%) (Figure 1). The Kinyoun method stained yeasts and spores (Figure 2), which could be distinguished from the oocysts by size, staining pattern and absence of internal sporozoites.
Occurrence, by species, of endoparasites observed in stool samples from 60 cats presented with diarrhea and technique by which they were found.

FEA = centrifugal sedimentation with formalin-ethyl acetate solution
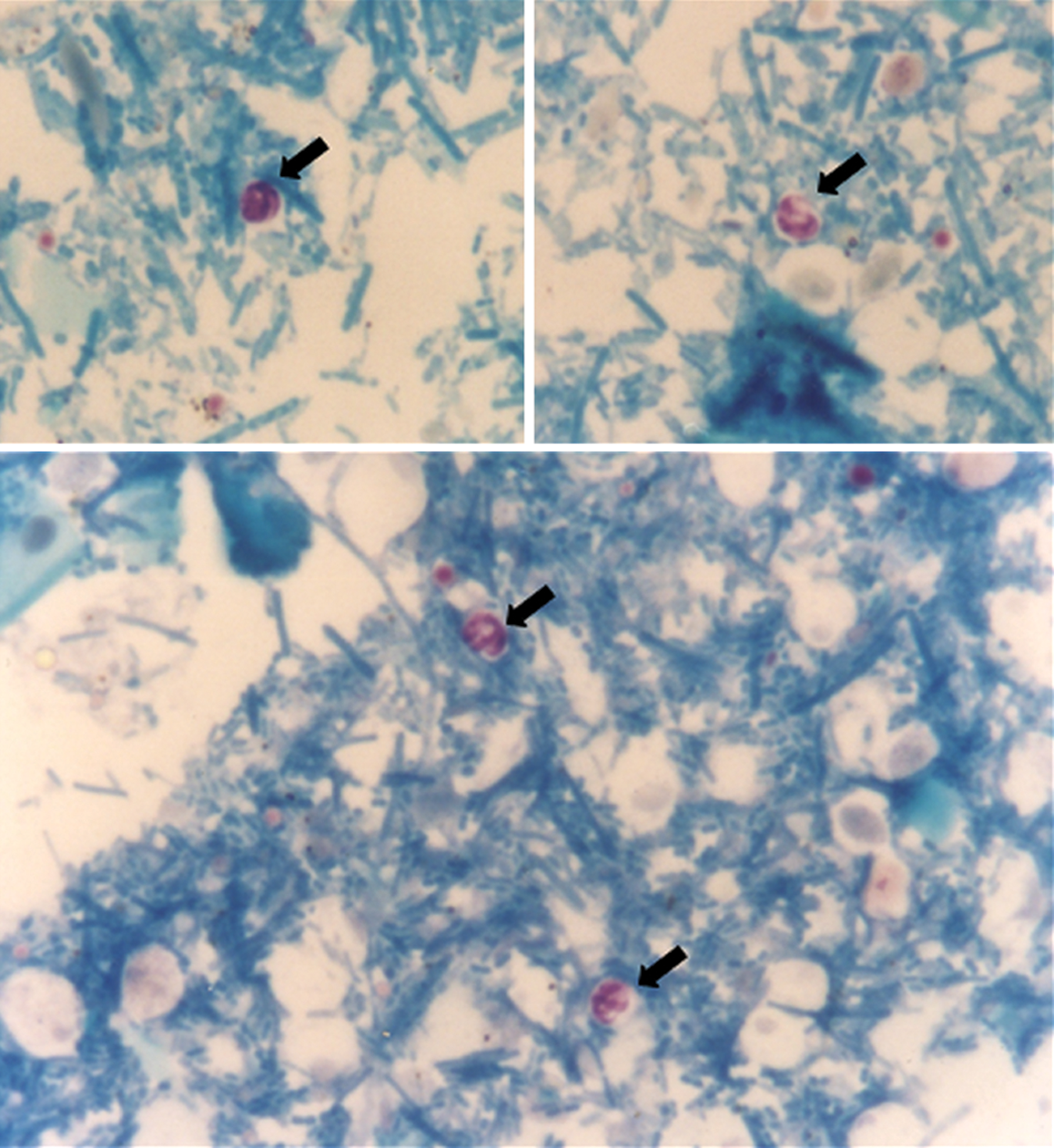
Cryptosporidium species oocysts (arrows). Kinyoun staining, 100 × objective (immersion), light microscopy

Non-Cryptosporidium species structures (arrows). Kinyoun staining, 100 × objective (immersion), light microscopy
Among the 60 animals tested, nine had positive results for FIV antibodies (15%), 15 were positive for FeLV antigen (25%) and two were positive for both FIV antibodies and FeLV antigen (3.33%). Thirty-four animals (55.66%) had negative results for both.
Of the five animals shedding Cryptosporidium species oocysts, four tested positive for FeLV and one tested negative for FIV and FeLV.
Discussion and conclusion
The ratio of animals shedding oocysts was compatible to previous reports.19,43–45 The oocysts visualization is difficult and experience is needed to make an accurate diagnosis as it can be confused with yeasts and other fecal debris. Staining makes oocyst identification easier, but sporozoites inside the oocysts must be observed to avoid a misdiagnosis. False-negative results are also possible as, in some cases, oocysts may fail to be stained (ghost-like appearance) so they can not be identified. Oocysts are shed intermittently so they may still be missed even when three samples are collected.
The number of observed fields should be standardized. This can be done by regulating the drop of sediment used for staining, and by scanning the entire slide. The number of oocysts shed is variable and can influence the ability to detect positive animals. The concentration method followed by this staining procedure increases the chances of finding oocysts 22 and is affordable and sensitive enough to be routinely used.34,35
Parasite identification of the species level is not possible with this method, 46 but it can be used as an early screening procedure.
The five cats positive for cryptosporidiosis included one with outdoor access and four with no outdoor access; all of them lived with other cats. These data are similar to those reported by Ballweber et al. 22
Although a correlation between Cryptosporidium species and FIV and/or FeLV could not be established, it was noted that the animals that were FeLV positive and were shedding oocysts had more severe and chronic diarrhea with a higher number of oocysts per field. The FIV and FeLV negative animal had a self-limiting diarrhea.
Footnotes
Acknowledgements
We would like to acknowledge Dr Teresa Cristina Bergamo do Bomfim for the identification of the Cryptosporidium species oocysts.
Funding
This research was generously funded by the Waltham Foundation.
Conflict of interest
The authors declare that there is no conflict of interest.
