Abstract
Post-hematopoietic stem cell transplant secondary solid neoplasms are uncommon and usually host-derived. We describe a 6-year-old female who developed a mixed donor-recipient origin mesenchymal stromal tumor-like lesion in the liver following an unrelated hematopoietic stem cell transplant complicated by severe graft-versus-host disease. This lesion arose early post-transplant in association with hepatic graft-versus-host disease. At 12 years post-transplant, the neoplasm has progressively shrunken in size and the patient remains well with no neoplasm-associated sequelae. This report characterizes a novel lesion of mixed origin post-transplant and offers unique insights into the contribution of bone marrow-derived cells to extra-medullary tissues.
Keywords
Introduction
Second malignant neoplasms (SMN), a recognized risk after allogeneic hematopoietic stem cell transplant (alloHSCT), are predominately recipient-derived. Donor-derived SMNs are comparatively infrequent, and are usually leukaemias (representing up to 5% of post-transplant relapses). 1 Non-hematopoietic donor-derived neoplasms, thought to arise from transformation of bone marrow-derived cells, are rare; with isolated reports in adult upper gastrointestinal carcinomas and cholangiocarcinomas.2–4 We provide first evidence of a child who developed a primitive mesenchymal stromal neoplastic-like lesion of mixed donor-recipient origin in the liver post-alloHSCT.
Results
We report on a 3-year-old female with pre-B acute lymphoblastic leukaemia (ALL) who was treated with conventional chemotherapy. The leukemia phenotype was CD19+CD10+CD34+ and TdT+ with a hyperdiploid karyotype (56-57 chromosomes). Molecular analysis was negative for BCR-ABL, TEL-AML and EZA-PBX1 fusion genes. After 2 years on maintenance therapy, she had an on therapy combined bone marrow and CNS relapse. She subsequently received reinduction chemotherapy followed by an unrelated donor, sex-mismatched (HLA 10/10 match) alloHSCT using bone marrow as the source. The transplant preparative regimen was fractionated total body irradiation (1200 cGy) with CNS boost (600 cGy) and cyclophosphamide. Graft-versus-host disease (GVHD) prophylaxis included cyclosporine and short-course methotrexate. The early post-transplant period had expected mucositis and pancytopenia. She had prompt recovery of hematopoiesis with full donor blood chimerism but developed steroid-refractory stage IV intestinal GVHD managed with multiple therapies. At 4-months post-transplant she developed an intestinal GVHD flare and pneumatosis intestinalis. An abdominal ultrasound and CT performed for fungal workup revealed a new solitary hepatic lesion, measuring 4cm in maximal dimension (Figure 1(A) and (B)). A core biopsy was performed (Figure 1(C)).
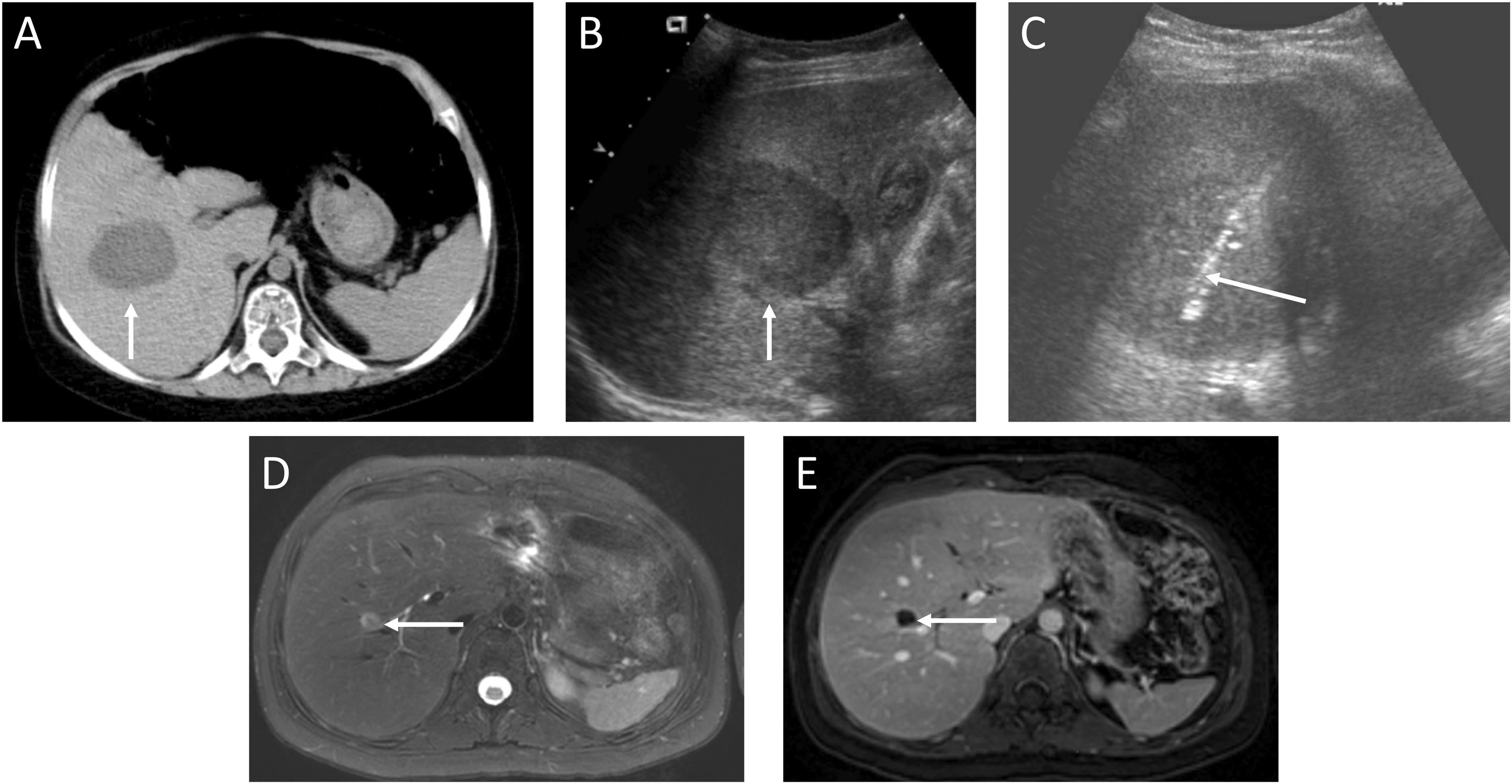
Multi-modal imaging of liver lesion at 4 weeks post-transplant and at follow-up 12 years post-transplant. A, Axial non contrast CT shows a hypodense lesion measuring approximately 4 x 4 cm in segment 5 of the liver (arrow). B, The lesion is hypoechoic on ultrasound (arrow). C, Ultrasound guided core biopsy shows biopsy tract within the lesion (arrow). D/E, Axial T2 fat-saturated (D), and contrast-enhanced T1 fat-saturated (E). MR images show the residual lesion in segment 5 of the liver, markedly reduced in size compared with baseline imaging at 4 weeks post-transplant (A). The lesion is predominantly T2 hyperintense (D, arrow) and does not enhance (E, arrow).
Histological analysis revealed diffuse proliferation of fibroblastic-appearing spindle cells containing condensed chromatin without nucleoli. The cells formed interweaving fascicles within a myxoid stroma (Figure 2(A)) containing minimal glycoprotein and collagen (Figure 2(B)), inconsistent with fibrosis. The lack of anaplasia, abnormal mitotic activity, or excessive proliferation rate (15% MIB-1 positivity) was consistent with benign mesenchymal proliferation. Aside from a few CD68+ stromal histiocytes, no liver stroma, inflammatory infiltrates, lipoblasts, lipocytes or chondroitin tissue were present, excluding a primary hepatic or hematologic neoplasm. Immunohistochemistry revealed expression of primitive mesenchymal stem cell (MSC) markers CD44 and CD133, with weak expression of CD117 (Figure 2(C)–(E)). Vascular endothelial growth factor (VEGF) and VEGF receptors were expressed. CD34 expression was limited to few single spindle cells not associated with capillaries (Figure 2(F)). There was expression of alpha smooth muscle actin (α-SMA) with occasional desmin and actin positive subsets, but other mature smooth muscle markers were absent (Figure 2(G) and (H)). There was no evidence for leukemic relapse, cytomegalovirus, adenovirus, Epstein-Barr virus (associated with smooth muscle/spindle cell tumors) or ALK rearrangements (associated with inflammatory myofibroblastic tumor). The overall impression was consistent with proliferation of stromal tissue with a primitive mesenchymal phenotype.
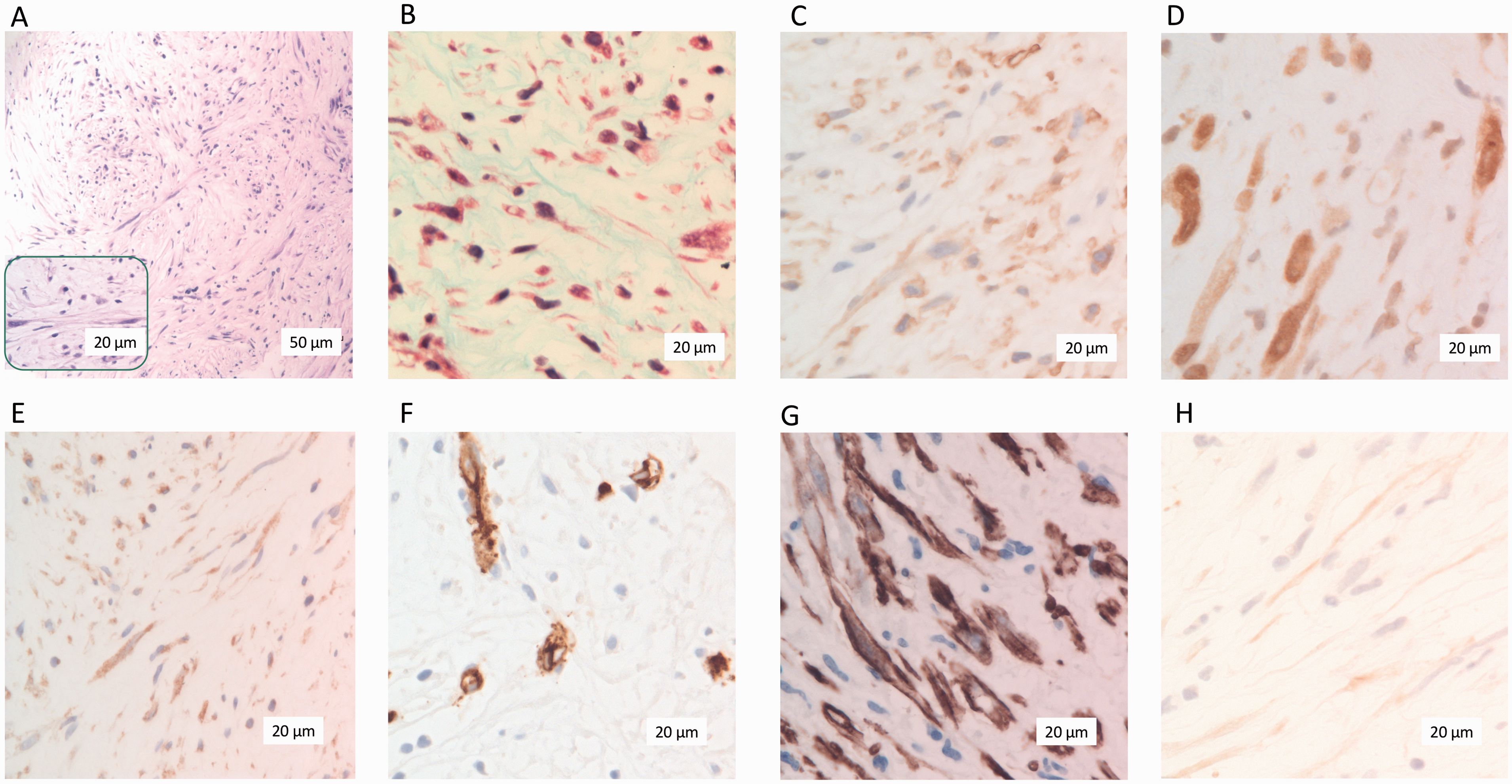
Representative immunohistochemistry staining of lesion core biopsy. A, Hematoxylin & eosin staining (x20 objective) with a high-magnification inset (x40 objective) showing long, slender fibroblast-like cells forming interweaving bundles within a myxoid stroma. B, Masson’s trichrome staining demonstrating a paucity of collagen within a myxoid stroma. C/D, CD44 and CD133-positive spindle cells present throughout tissue biopsy. E, Minimal CD117-positive spindle cells were present. F, Few scattered spindle cells, not associated with microvascular structures, were CD34-positive. G, Spindle cells were strongly positive for α-smooth muscle actin. H, Specific spindle cell subsets were positive for desmin.
To address the lesion’s origin, we compared short nucleotide polymorphisms (SNPs) within known highly polymorphic loci and identified this monotonous primitive stromal tumor-like lesion was 42% recipient and 58% donor origin.
At 12 years post-transplant, the liver mass has decreased in size on serial imaging, with a current MRI demonstrating a sub-centimetric lesion (Figure 1(D) and (E)), resulting in no clinical sequelae nor requiring surgical or other intervention. She remains in remission but with late effects of therapy including inflammatory polyarthritis, Crohn’s colitis, multiple endocrinopathies, and a parietotemporal cavernous malformation secondary to radiation therapy.
Discussion
To our knowledge, this is the first report describing a mixed donor-recipient secondary solid neoplasm following alloHSCT in pediatrics. Post-alloHSCT, benign hepatic neoplasms are uncommon with most being recipient-derived or uninvestigated for chimerism. Unlike this case, the majority of such lesions, including focal nodular hyperplasia, contain hyperplastic hepatic tissue.5, 6 Potential contributing causes include iatrogenic damage, GVHD-mediated inflammation, or immunosuppression.
A broad differential exists for a solitary liver mass, but the pathology was only consistent with a mesenchymal tumor-like lesion (Supplemental Table 1). Molecular chimerism analysis of this monotonous mass identified a majority donor contribution. This was likely represented by the mesenchymal tissue as recipient bile ducts and vasculature were noted and few hematopoietic cells were present. The immature fibroblast-like phenotype with expression of CD44, the stem cell marker CD133 and minimal CD117 supports a mesenchymal progenitor source. 7 Endothelial lineage markers including VEGF and VEGF receptors, co-expressed by MSCs, were identified in this neoplasm and likely supported its early proliferation. 8 Co-staining of α-SMA and desmin, despite little collagen and other mature smooth muscle markers, also suggested differentiating myofibroblast precursors. Thus, this mixed origin, immature, fibroblast-like tissue expressing markers of stemness and multiple mesodermal lineages may represent the proliferation and early differentiation of donor and host-derived mesenchymal progenitors, best described as post-transplant mesenchymal proliferation.
Bone marrow-derived MSCs remain of host origin post-alloHSCT, 9 however, the extramedullary engraftment potential of donor MSCs is inconclusive. Murine cell tracing studies showed infused MSCs preferentially home to the liver, lungs and spleen with negligible distribution to the bone marrow. 10 Human trials of HLA-unmatched donor MSC infusion identified no ectopic tissue formation and minimal MSC persistence. 11 However, the post-transplantation fate of MSCs is immune-regulated, 12 and thus, the profound early immunosuppression here may have allowed donor mesenchymal progenitor survival and homing to the liver to participate in neoplasia. Post-alloHSCT donor-derived neoplasms are theorized to be contributed to by bone marrow-derived cells, specifically MSCs.2–4 In a murine model, donor bone marrow-derived MSCs were detected in epithelial neoplasms of inflamed gastric mucosa. 13 Beyond malignant processes, donor mesenchymal and endothelial progenitors are recruited to sites of tissue injury such as in GVHD-induced inflammation in humans. 14 It is conceivable that donor-derived mesenchymal progenitors migrated into the liver to bolster the regenerative response to injury induced by the preparative regimen and/or GVHD, resulting in a mixed donor-recipient neoplasm.
This lesion also displayed characteristics of differentiating myofibroblast precursors. Myofibroblasts are integral to tissue repair and arise from direct differentiation of MSCs or through a haematopoietically-derived monocyte derivate called a fibrocyte. 15 In support, is the observation of donor marrow-derived human MSCs engrafting in a xenogeneic model of liver injury in immunodeficient mice that adopted a myofibroblast-like phenotype with strong α-SMA positivity. 16 , 17 Pericytes, myofibroblasts and other mesenchymal cells have been shown to arise from HSCs through a fibrocyte intermediate post-alloHSCT in animal models. 18 , 19 This is supported by clinical evidence of donor myofibroblasts, originating from MSCs or circulating fibrocytes, detected in peripheral tissues post-alloHSCT in the context of GVHD. 20 This myofibroblastic phenotype in liver and other tissues following HSCT and tissue injury, supports our observation of early myofibroblast commitment.
An alternative explanation for this tumor-like lesion could be that the cell of origin came from donor-derived hepatic stellate cells, liver-specific mesenchymal cells, which acquire a myofibroblast-like phenotype following liver damage and express the mesenchymal markers desmin and α-SMA. In a murine model of HSCT, labelled HSCs were observed to replenish hepatic stellate cells in the damaged livers of lethally irradiated recipients. 21 Thus, an interpretation may be that this mesenchymal lesion is comprised of activated hepatic stellate cells of donor hematopoietic origin. There is also evidence to suggest that recruited donor cells could fuse with differentiated cells, including myofibroblasts, to form hybrids. 22 In our study, it was not possible to determine which of these mechanisms principally contributed.
In conclusion, this novel mixed donor-recipient origin, mesenchymal stromal liver tumor-like lesion following alloHSCT is hypothesized to result from post-transplant proliferation of mesenchymal progenitors, potentially in response to severe cytotoxicity or GVHD-induced inflammation. The mixed origin of this mesenchymal lesion supports the potential for donor-derived bone marrow cells to contribute to non-hematopoietic tissue.
Supplemental Material
sj-pdf-1-pdp-10.1177_10935266211001656 - Supplemental material for A Novel Secondary Neoplasm Following Allogeneic Hematopoietic Stem Cell Transplant: Mixed Donor-Recipient Primitive Mesenchymal Proliferation of the Liver
Supplemental material, sj-pdf-1-pdp-10.1177_10935266211001656 for A Novel Secondary Neoplasm Following Allogeneic Hematopoietic Stem Cell Transplant: Mixed Donor-Recipient Primitive Mesenchymal Proliferation of the Liver by Brian Earl Zi Fan Yang, Harini Rao, Grace Cheng, Donna Wall and Bo-Yee Ngan in Pediatric and Developmental Pathology
Footnotes
Acknowledgments
We are grateful to the patient and family for their voluntary cooperation and participation in this study. We thank Drs. Tal Schechter-Finkelstein and Muhamad Ali for their helpful review of this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
