Abstract
The advent of bone marrow transplant has opened doors to a different approach and offered a new treatment modality for various hematopoietic stem-cell-related disorders. Since the first bone marrow transplant in 1957, there has been significant progress in managing patients who undergo bone marrow transplants. Plasma-cell disorders, lymphoproliferative disorders, and myelodysplastic syndrome are the most common indications for hematopoietic stem-cell transplant. Despite the advances, invasive fungal infections remain a significant cause of morbidity and mortality in this high-risk population. The overall incidence of invasive fungal infection in patients with hematopoietic stem-cell transplant is around 4%, but the mortality in patients with allogeneic stem-cell transplant is as high as 13% in one study. Type of stem-cell transplant, conditioning regimen, and development of graft-versus-host disease are some of the risk factors that impact the risk and outcomes in patients with invasive fungal infections. Aspergillus and candida remain the two most common organisms causing invasive fungal infections. Molecular diagnostic methods have replaced some traditional methods due to their simplicity of use and rapid turnaround time. Primary prophylaxis has undoubtedly shown to improve outcomes even though breakthrough infection rates remain high. The directed treatment has seen a significant shift from amphotericin B to itraconazole, voriconazole, and echinocandins, which have shown better efficacy and fewer adverse effects. In this comprehensive review, we aim to detail epidemiology, risk factors, diagnosis, and management, including prophylaxis, empiric and directed management of invasive fungal infections in patients with hematopoietic stem-cell transplant.
Introduction
Since the first bone marrow transplant in 1957, there has been significant progress in managing patients who undergo bone marrow transplants. The Center for International Blood and Marrow Transplant Research (CIBMTR) recorded about 8000 allogeneic and 14,000 autologous bone marrow transplants in the United States (US) in 2018. Their data showed that the most common indications for autologous bone marrow transplants are myeloma and lymphomas. In contrast, most allogeneic stem-cell transplants were indicated for acute myelocytic leukemia (AML) and myelodysplastic syndrome (MDS). 1 Largely, in 2018, the number of autologous hematopoietic stem-cell transplants (HSCTs) decreased by 5%, whereas allogeneic HSCT increased by 1% when compared with 2017. Interestingly, there is a continuous increase in HSCT among older adults age >70 years. 1 Other common indications include aplastic anemia, thalassemia, and severe combined immunodeficiency.
Randomized control trials have suggested benefits for HSCT, including longer progression-free survival, as well as interval freedom from treatment failure.2,3 While this appears promising, common complications associated with HSCT include graft-versus-host disease (GVHD), infection, graft rejections, and organ failure. Beside the progression of primary disease, infections and GVHD are predominantly responsible for negatively affecting HSCT outcomes.
The HSCT treatment period has been divided into three phases: the pre-engraftment period, early post-engraftment period, and late post-engraftment period. Early in the course of transplant, infection accounts for about a third of deaths. 1 The microbiology of infections during these periods is based on the underlying defect in the immune system and host risk factors. Common early infections include bacterial infections, including Gram-negative bacilli acquired from the gastrointestinal (GI) tract during the transplantation process and Gram-positive cocci from indwelling access sites. According to one study, while 38% of the infections were viral, about a quarter of the infections occurring in HSCT were fungal, with bacterial infections accounting for a third. 4
This review addresses fungal infections in HSCT, including the epidemiology of fungal infections beside candida and aspergillus. We address risk factors predisposing to fungal infections based on the type of HSCT, the time since HSCT, and the presence of GVHD. We compare different options for diagnosing fungal infections, and finally, we review studies addressing prophylaxis and management for fungal infections.
Epidemiology
Invasive fungal infections (IFIs) cause significant morbidity and mortality in high-risk populations such as those with HSCT. The establishment of the TRANSNET in 2001 was helpful in better understanding of epidemiology of IFI on a large scale. This led to the establishment of the Transplant-Associated Infections Surveillance Network (TRANSNET) in 2001, consisting of 23 US academic transplant centers that perform HSCTs and solid organ transplant, administered by the Centers for Disease Control and Prevention (CDC) and coordinated by the University of Alabama at Birmingham. Through TRANSNET, a prospective surveillance study was conducted from 2001 to 2006 on IFI in solid organ and HSCT recipients that revealed essential data on epidemiological trends and the burden of IFI in the US. 5 In addition, several other similar studies are now available, aiming to identify the incidence and outcomes of IFI in this population. The SEIFEM-B-2004 study was a major retrospective cohort study undertaken from 1999 to 2003 in 11 academic hospitals in Italy involving HSCT patients who developed IFI. 6 Another major study was a multicenter, prospective, observational study performed on the Prospective Antifungal Therapy (PATH) Alliance registry during the period July 2004 through September 2007. 7
Based on the TRANSNET data, the incidence of IFI in HSCT patients overall is 3.4%. The SEIFEM study showed a similar incidence rate of 3.7%, where 49% were proven infections and the rest were probable. Both the studies found invasive aspergillosis (IA) to be the most common IFI encountered in this population, followed by invasive candidiasis (IC), other unspecified molds, and finally, zygomycetes. Pneumocystis, endemic fungal infection, and cryptococcus infections were rare. The IFI-attributed rate from the SEIFEM study was 2.4%, with the mortality rate from aspergillosis significantly higher than that reported for candida infection. Epidemiology and risk factors of IFI in HSCT patients are presented in Table 1.
GVHD, graft-versus-host disease; HIV, human immunodeficiency virus; HLA, human leukocyte antigen; HSCT, hematopoietic stem-cell transplant; IA, invasive aspergillosis; IC, invasive candidiasis; IFI, invasive fungal infections; PCR, polymerase chain reaction.
Risk factors
The risk factors for the development of IFI after HSCT depend on the type of HSCT, the presence of acute or chronic GVHD, administration of steroids, the presence of cytomegalovirus (CMV) disease, and antifungal prophylaxis. 6
Type of HSCT
Allogeneic HSCT had more negative IFI-associated outcomes compared with autologous HSCT. According to the TRANSNET study, the cumulative incidence of IFI in autologous HSCT was 1.2%. Comparatively, among recipients of allogeneic HSCT, the 12-month cumulative incidence for matched related donor (MRD), an unrelated donor (URD), and mismatched-related donors (MMRs) was 5.8%, 7.7%, and 8.1%, respectively. 5 In the SEIFEM study, the mortality rate due to IFI in allogeneic HSCT versus autologous HSCT was 72% versus 35% (p = 0.001), and the incidence and mortality of aspergillosis were higher in allogeneic HSCT than autologous HSCT. There was no difference in incidence and mortality among these groups in invasive candida infection. 6 The RISK study, a 2013 multicenter retrospective observational study performed on patients undergoing allogeneic HSCT from six hospitals in Korea, revealed the cumulative incidence of IFI (proven or probable) in allogeneic HSCT to be 15.36% at 1 year, with aspergillus as the most common organism found, followed by candida, cryptococcus, and fusarium. 23 Recently, the haploidentical transplant has increased donor availability but also increases the risk of infection, especially when highly active myeloablative regimens are used to reduce GVHD. 24 In one retrospective analysis, the cumulative incidence of fungal infections was 24% at day 100, and 28% at 2 years after haploidentical stem-cell transplantation, with post-transplant cyclophosphamide. 25 In a study by Omer et al., 26 haploidentical stem-cell transplantation was one of the risk factors identified for the development of IFI [hazard ratio (HR) 3.82; 95% confidence interval (CI) 1.49–9.77; p = 0.005]. In a retrospective study including 381 patients with haploidentical stem-cell transplantation, 20% developed IFI. Invasive aspergillosis was the most common (44%), followed by invasive candidiasis (33%). 24
Time since HSCT
According to the RISK study, risk factors for IFI in allogeneic HSCT patients in the early post-engraftment period (<40 days) were unrelated or related-mismatch HSCT, underlying chronic diseases, and prolonged neutropenia (>3 weeks). In the late post-engraftment period (41–100 days), IFI risks were the prescription of immunosuppressive agents for refractory GVHD and CMV reactivation. Whereas in the very late post-engraftment phase (>100 days), the risk factors were secondary neutropenia, severe GVHD, and use of tumor necrosis factor alpha (TNF-α) inhibitors for the treatment of refractory GVHD. 23 A significantly higher mortality was seen in patients with severe GVHD requiring intensive immunosuppressive therapy. 27 According to the PATH Alliance registry study, IC tended to occur earlier after autologous HSCT (median 28 days) compared with allogeneic HSCT (median 108 days). 7 Early-onset IC is influenced by neutropenia and mucositis, whereas later-onset infection is influenced by GVHD and the presence of chronic indwelling catheters. 11 IA tends to occur more frequently, and interval from transplant to diagnosis did not appear to be a significant risk factor for IA. Late IA is associated with an increased fungal burden due to delay in diagnosis and immunosuppression. IFI due to zygomycetes and other molds occurred late after HSCT (median interval, 173 days) and tended to occur later in autologous HSCT recipients than in allogeneic HSCT recipients (median interval, 162 days). 7
Conditioning regimen
A conditioning regimen usually consists of myeloablative therapy with or without total body irradiation. It is used to either treat underlying cancer or suppress the recipient’s immune system before HSCT. Intensive regimens are associated with prolonged neutropenia increasing susceptibility to infections. Incidence of mold infection was low in patients who underwent only total body irradiation, since it favored engraftment and neutrophil recovery. 28 Currently, reduced intensity/non-myeloablative ablative regimens are increasingly used, are associated with shorter neutropenia duration and less mucosal damage, and lead to fewer adverse outcomes and lower transplant-related mortality.29,30
Type of antifungal prophylaxis
Prophylaxis consists of the administration of antifungal agents at the onset of a period of high risk of infection, usually at the start of the conditioning regimen or the beginning of neutropenia in HSCT recipients. Although prophylaxis has improved early mortality, there have been limited epidemiological studies since posaconazole use was widely accepted for primary prophylaxis. Harrison et al. 27 reported a single-center, retrospective cohort study that used primary antifungal prophylaxis with posaconazole (49.5%), voriconazole (26.7%), and fluconazole (16.2%) in 43.3% of allogeneic HSCT patients. Despite a fivefold increase in systemic antifungal prophylaxis, the incidence of IFI was not affected at 10.3% of patients at 1 year.
Organisms causing invasive fungal infections (Table 1 and Figure 1):
Aspergillus
Aspergillus fumigatus is the most frequent species causing IFI.5,6 This is attributed to virulence factors associated with this species; ongoing research of these complexities may help in the development of new rapid diagnostic tests and antifungal agents. 8 Other species that are often detected are Aspergillus niger, Aspergillus terreus, and Aspergillus flavus. Aspergillus infection commonly presents as invasive pulmonary aspergillosis with subsequent dissemination. 9 A. terreus infection has been associated with amphotericin B resistance, a lower response rate to treatment, and a higher mortality rate. 10 According to the TRANSNET study, the 1-year survival in patients with IA is 25.4%. 5 In recipients of allogeneic HSCT, three periods of increased risk for invasive aspergillosis and other mold diseases occur: first, during neutropenia following the conditioning regimen; second, during acute GVHD; and third, during chronic GVHD. The risk is higher during GVHD than during neutropenia due to the use of chronic immunosuppressive therapy 9 (Table 1, Figure 1).

Timeline and risk factors of invasive fungal infections.
Candida
Candida species are the second most common cause of IFI in HSCT patients. 12 The epidemiology of candida infection has changed significantly in the past 2 decades after several randomized controlled trials showed that fluconazole significantly decreased the incidence of candidiasis, followed by the subsequent widespread use of fluconazole prophylaxis. 13 Candida albicans had historically been the most common pathogen; however, the routine use of azoles for prophylaxis has contributed to an overall decreased incidence of IC and an increased frequency of non-albicans candida infection. 11 Non-albicans candida accounted for almost 70% of all candida infections in the TRANSNET study. The major non-albicans species were Candida glabrata, Candida parapsilosis, Candida tropicalis, and Candida krusei, with C. glabrata now emerging as an important pathogen. According to the TRANSNET study, the 1-year survival in patients with IC is 33.6%, slightly higher than in other IFI. 5
Other molds
These include over 30 non-aspergillus hyalohyphomycetes, including Acremonium, Fusarium, Scedosporium, and Paecilomyces species. Infection with these organisms usually follows environmental exposure. 14 During their study period, SEIFEM study reported 0.11% of fungal infections resulting from Fusarium species with a mortality of 53%. Scedosporiosis causes a wide range of clinical diseases, from transient colonization of the respiratory tract to invasive localized and disseminated diseases in immunosuppressed patients and involves two medically important species: Scedosporium apiospermum and Scedosporium prolificans. They have been known to proliferate in patients receiving amphotericin B, fluconazole, or itraconazole.15,16 In a retrospective case series of 80 scedosporium patients which included 22 HSCT recipients, the overall mortality was found to be 68%, with 61.5% for patients having S. apiospermum infection and 77.8% for patients having S. prolificans infection. 16
Zygomycetes
Zygomycosis represented 8% of IFI in the TRANSNET Study, and 7% in the PATH study.5,7 According to the TRANSNET study, zygomycosis incidence increased from 1.7 to 6.2 per 1000 from 2001 to 2004. It is a devastatingly invasive disease with mortality of 60–80%. 11 The increased incidence of zygomycosis reflects the increase in predisposing factors, such as higher numbers of patients undergoing HSCT and immunosuppressive chemotherapy, with the effectiveness of antifungal therapy against common species correspondingly increasing the emergence of such rare pathogens. 16
Pneumocystis
Historically, pneumocystis pneumonia (PCP) was considered a severe life-threatening infection, especially in patients with acute lymphoblastic leukemia, and HSCT recipients. The incidence of PCP, however, dropped after the introduction of prophylaxis. Currently, only HSCT patients not on or non-compliant to prophylaxis develop PCP. Large retrospective analyses have reported an incidence rate between 0% and 2.5% in allogeneic HSCT recipients and 1.4% in autologous HSCT recipients. 18 HSCT recipients who develop PCP typically have acute or chronic graft-versus-host disease and are receiving corticosteroids or other immunosuppressive drugs more than 6 months after transplant. A large retrospective analysis reported an incidence rate between 0% and 2.5% in allogeneic HSCT recipients, and 1.4% in autologous HSCT recipients. 19
Cryptococcus
Cryptococcus neoformans and Cryptococcus gatti are the main pathogenic species in this genus. Due to effective antifungal prophylaxis, these organisms are significantly rarer among HSCT recipients, representing <1% of the total IFI in this population.5,11 The timing of cryptococcosis in HSCT recipients is not well defined. Types of infection in these patients include meningitis, fungemia, and pneumonia. The overall mortality of cryptococcosis in the non-human immunodeficiency virus (HIV) population is 30%. 11
Endemic fungi
This class includes Histoplasma capsulatum, Blastomyces dermatidis, and Coccidioides immitis. These organisms are present in the soil in certain geographic regions, and conidia inhalation leads to disseminated infections. They are rarely encountered in HSCT recipients, even in endemic areas. According to a recent literature review, histoplasmosis has been shown to occur between 5 weeks and 18 months post-transplantation and is associated with high mortality.11,21,22
Diagnosis
The rate of pre-mortem diagnosis of IFI is in the range of 12–60%. This indicates the necessity of improved diagnosis. 31 Usually, in clinically unstable patients, IFIs are treated empirically, which raises economic burden, and delay in diagnosis is associated with increased mortality. Different terminologies have been used for the diagnosis of IFI, which has now been standardized per European Organization for Research and Treatment of Cancer/National Institute of Allergy and Infectious Diseases Mycoses Study Group to streamline clinical studies. 32 A proven diagnosis requires culture or histopathological evidence from a tissue biopsy or normally sterile body fluids. Probable diagnosis requires conducive host factors such as neutropenia, immunosuppression, clinical suspicion through imaging, direct visualization, or clinical exam, and includes cytology or culture evidence of molds or indirect tests like galactomannan (GM) or β-D-glucan. Thus, a host factor, a clinical criterion, and a mycological criterion are necessary. Possible diagnoses are the cases that meet the criteria for a host factor and a clinical criterion but for which mycological criteria are absent. Conventional techniques for clinical diagnosis are limited by slow turnaround time or associated invasive procedures. Newer methods that do not require detection of the organism in culture or tissues are limited by availability and clinical performance. Thus, standardization of diagnostic tests for each clinical scenario is difficult.
Culture-based detection
This remains the gold standard for diagnosis and provides critical data on organism susceptibility. However, it also leads to considerable delays in the initiation of treatment. The time to initiation of antifungal treatment of at least 12 h after the first positive blood culture sample was an independent determinant of hospital mortality in patients with invasive candidiasis [odds ratio (OR) = 2.09; p = 0.018]. 33 A prospective study in 2000 determined that fungal colonization detected by surveillance fungal cultures lacked positive predictive value for diagnosis of fungal infection in critically ill patients, and had minimal utility. 34 The sensitivity and specificity of cultures vary for organisms and type of body fluid. Cerebrospinal fluid (CSF) cultures for cryptococcus are highly sensitive and specific, whereas blood culture sensitivity in invasive candidiasis is only 50–60%, with 95% specificity. The positive predictive value of cultures depends on the prevalence of the infection and, therefore, is much higher in transplant patients and endemic areas. 35 A study looking at immunocompromised patients with positive lower respiratory-tract culture for aspergillus showed a positive predictive value of 72% in patients with hematologic malignancy, granulocytopenia, or bone-marrow transplant; 58% in those with a solid-organ transplant or using corticosteroids; and 14% in those with HIV infection. 36 Identification of yeast or molds from clinical specimens is exceptionally laborious. Microbiologists have begun using mass spectrometry identification techniques (MALDI-TOF, matrix-assisted laser desorption time of flight mass spectrometry) on specimens for rapid diagnosis. 37
Histopathology
This is another gold-standard test, especially in invasive aspergillosis, pneumocystis, and endemic fungal infections (histoplasma, blastomyces, and coccidioides), often used in conjunction with culture. Histopathology can detect an invasion of fungus, as well as a host response. For example, visualization of a biofilm can help direct therapy since this is associated with resistance to antifungal regimens. Histopathology, unfortunately, gives only a descriptive diagnosis; fungal morphology can be non-specific, and it does not allow for the identification of species, which is usually necessary to direct therapy. 35
β-D-glucan
Testing to detect the explicitly fungal cell wall component (1,3)-β-D-glucan (BDG) is commonly used as a screening and diagnostic tool in IFI. The Fungitell® assay is currently the only US Food and Drug Administration (FDA)-approved assay. 37 BDG is found in several organisms, including Candida species, pneumocystis, and molds such as Aspergillus species and Fusarium species. In a prospective study of HSCT recipients, BDG assays were found to have a specificity of 98% and a negative predictive value of 99%, making this test a useful tool to rule out IFI. 38 Similar results were found in a recent meta-analysis in patients with hematological malignancies where two consecutive BDG tests had an excellent specificity (98.9%) but a low sensitivity (49.6%) for the diagnosis of IFI. 39 This low sensitivity is likely related to the higher incidence of colonization in the HSCT population and testing on specific populations like hemodialysis patients or those with Gram-negative bacteremia.
Aspergillus galactomannan
GM is another fungal cell-wall biomarker produced by several fungi, including Aspergillus, Penicillium, and Histoplasma species. This assay is reported as an index of optical density (GM index or GMI) and is currently FDA approved for the detection of aspergillus in serum and bronchoalveolar lavage (BAL) specimens. Serum GM testing is now considered the standard of care given strong recommendations for use in immunocompromised patients by several high-quality studies (in patients not on posaconazole prophylaxis). 40 In a retrospective cohort study in allogeneic HSCT recipients with invasive pulmonary aspergillosis (IPA), the magnitude of GMI (drawn on clinical suspicion) correlated with higher all-cause mortality, likely representing increased aspergillus burden. 41 Another similar study concluded that a GMI screening strategy could lead to lower mortality by early detection of invasive aspergillosis. 42
Antigen–antibody detection
Sensitivity of antibody tests may be limited in immunocompromised patients, as there is a lack of antibody response to infections. They also have limited specificity, since they cannot differentiate between the presence of normal flora, colonization, and infections. However, several specific antibody detection tests are available.
C. albicans germ-tube antibody (CAGTA) assay
This test detects antibodies by an indirect immunofluorescence technique. It has a sensitivity of 77–89% and a specificity of 91–100%. 35 Lateral-flow devices detect a glycoprotein in the serum and BAL fluid of patients with invasive aspergillosis. It is used in combination with molecular studies.
Cryptococcal antigen test
When performed on CSF, this test has a sensitivity of 97% and a specificity of 93–100%. It can also be used on a blood specimen, but it has a sensitivity of 87%. It has become a point-of-care screening test in the immunocompromised population.
Histoplasma and blastomyces antigen tests
These tests can be performed on urine or serum and have superior sensitivities (83.3–91.8% for histoplasmosis and 92.9% for blastomycosis) than antibody testing. A novel enzyme-linked immunosorbent assay test has been developed with high specificity for B. dermatidis.
Coccidioides antibodies
This is a highly accurate antibody test using complement fixation and tube precipitin antibody detection methods.
Molecular studies
Molecular diagnostic methods, especially polymerase chain reaction (PCR), have increased in popularity and replaced some traditional methods due to simplicity of use and rapid turnaround time. The high sensitivity of PCR allows for early detection of infection when treatment is easier and may prevent clinical manifestations. Since quantifying microbial burden in IFI is vital in terms of management, the development of real-time PCR, which can quantify the amount of DNA in real-time, has been revolutionary. Some technical difficulties associated with PCR diagnosis include cumbersome methods for cell wall lysis to expose DNA, primer standardization, lab-to-lab discrepancies, and sensitivity to contamination. 40
Aspergillus polymerase chain reaction (PCR)
There are several recent meta-analyses analyzing the performance of PCR on blood or serum samples for aspergillus detection. One meta-analysis showed the sensitivity and specificity of blood or serum PCR for two consecutive positive samples were 0.75 (95% CI 0.54–0.88) and 0.87 (95% CI 0.78–0.93), respectively, and for a single positive sample were 0.88 (95% CI 0.75–0.94) and 0.75 (95% CI 0.63–0.84), respectively. 43 In another meta-analysis that compared the performance of PCR of BAL with a BAL GM test, mean sensitivity and specificity values for diagnosis of proven or probable IPA were 90.2% (77.2–96.1%) and 96.4% (93.3–98.1%), respectively. 44 The American Thoracic Society has published a summary of recommendations based on high-quality meta-analyses for diagnosing IPA in severely immunocompromised patients, such as HSCT with high pretest probability. They recommended testing blood or serum for aspergillus using PCR. In patients with high pretest probability, a single positive PCR gives moderate-to-high sensitivity to exclude the disease, and two positive PCRs provide high specificity. In patients with high pretest probability suspected of having IPA, aspergillus PCR testing of BAL is recommended. 40 However, these recommendations have been made for adult patients only. There is a significant lack of studies in the pediatric population, as well as a lack of outcome research in PCR diagnostics.
Candida PCR
Similar studies were performed for assessing the utility of diagnostic PCR in patients suspected of invasive candidiasis. In one meta-analysis of 54 studies that included almost 5000 patients tested by blood-based PCR, pooled sensitivity and specificity for proven or probable invasive candidiasis versus at-risk controls were 0.95 (CI, 0.88–0.98) and 0.92 (CI 0.88–0.95), respectively. 45 Recently, there has been an innovative contribution to invasive candidiasis diagnostics in the form of targeted molecular direct detection using an FDA-approved candida nucleic acid test. It combines targeted PCR with T2 magnetic resonance to detect the five most common Candida species (C. albicans, C. glabrata, C. parapsilosis, C. tropicalis, and C. krusei) directly from blood specimens. The overall specificity was 99.4%, and sensitivity was 91.1% in the multicenter DIRECT trial. 46
Aspergillus PCR + galactomannan
An emerging strategy is to combine molecular and antigen testing to improve clinical utility. This was assessed in a recent meta-analysis of high-risk hematological patients, which found that the highest sensitivity (99%) was achieved when at least one positive GM or PCR was used to define a positive episode. The absence of any positive test result had a negative predictive value of 100%, averting unnecessary antifungal exposure. 47
PCR + BDG assay
A similar approach was made for the diagnosis of Pneumocystis jirovecii in a large HSCT population with clinical suspicion of P. jirovecii pneumonia (PJP). This retrospective cohort study used PJP PCR on bronchoscopy samples and serum BDG testing. In patients with a positive BAL PCR result, a positive BDG resulted in 100% specificity with a 100% positive predictive value. 48
Next-generation sequencing techniques
This includes sequencing of cell-free DNA (cfDNA) circulating in the bloodstream to identify non-human sequences, and compare them with known genomic databases of bacterial, viral, and fungal pathogens. This is a newer technique and its applicability in detection of IFI in HSCT patient is still under review.35,49
Management
Early recognition and early initiation of treatment of invasive fungal infection may improve patient outcomes. Effective management strategies include prophylaxis (primary and secondary), empiric antifungal treatment, and directed treatment of established fungal infections. An increased incidence of fungal infections in bone marrow transplant patients has been seen over time. 50 Given the high mortality related to invasive fungal infection, prophylaxis and empiric therapy should not be delayed while awaiting identification of the pathogen. 51
Primary prophylaxis
Table 2 lists major trials of primary prophylaxis agents. In the late 1900s, several trials explored the benefits of prophylactic antifungal regimens including nystatin and ketoconazole.52 –55 These agents are seldom used now. A landmark trial by Goodman et al. 56 showed reduction in the incidence of superficial and systemic fungal infections with 400 mg of fluconazole without any difference in 90-day mortality. Subsequently, Slavin et al. 13 reported a statistically significant difference in mortality at 110 days with 400 mg daily prophylactic fluconazole versus placebo. These two studies established the use of fluconazole in bone marrow transplants as the standard of care at the end of the 20th century. Long-term follow up by Marr et al. 57 showed survival benefit in the fluconazole group compared with placebo. However, this benefit was statistically significant only in the allogeneic transplant patient subgroup.
Recent trials of primary prophylaxis in invasive fungal infections.

GI, gastrointestinal; GVHD, graft-versus-host disease.
Consequently, itraconazole was introduced as a potential prophylactic with some evidence that itraconazole may be a superior antifungal for prophylaxis than fluconazole.62,63 However, there also was concern regarding its GI side effects. 63 In 2004, Marr published a prospective comparative study between fluconazole and itraconazole, which reiterated the abovementioned aspects, including reduced IFI with itraconazole compared with fluconazole. 58 The study showed no difference in the incidence of candidemia between the two groups. However, fluconazole prophylaxis was recommended, since there was no difference in fungal-free survival between the two groups, but increased side effects associated with itraconazole, especially in the GI tract, leading to frequent discontinuation of the drug. Fluconazole and voriconazole had similar fungal-free survival and overall survival in patients with allogeneic HSCT. 61
With the introduction of echinocandins, the prophylactic potential of micafungin was studied, given its anti-mold effects. There was some concern that fluconazole was not as effective against mold species as it was against yeast species, based on a single-center autopsy study in pediatric patients, which defined fluconazole prophylaxis as a minimum of five consecutive doses of fluconazole. In approximately 300 autopsies, they reported a decreased incidence of candida but increased incidence of aspergillus via histopathology or culture. 64 In 2004, a randomized controlled trial compared micafungin with fluconazole as prophylactic medication in bone marrow transplant patients with a non-inferiority margin of 10%. The study reported micafungin as superior to fluconazole with a statistically significantly higher success rate, the primary outcome defined as the absence of proven, suspected, or probable IFI. While there was no difference in mortality, candidemia, or colonization rates reported in the two groups, the study reported a non-significant but higher incidence of aspergillosis in the fluconazole group (seven breakthrough aspergillus infections) compared with micafungin (one breakthrough aspergillus infection). There were no significant differences in the adverse effect profiles of the two drugs, which involved approximately 400 patients in each group. 59 This has provided a different drug-class option for fungal prophylaxis.
A study was published in the New England Journal of Medicine in 2007 that compared the newer drug posaconazole with fluconazole in a randomized trial, based on evidence of better in vitro efficacy of posaconazole against aspergillus. The study was conducted in patients who had acute GVHD grade 2–4 or chronic GVHD. Approximately 300 patients in each GVHD category were randomized, and no differences were found in the total incidence of fungal infections. More importantly, the study concluded that posaconazole administration was superior in reducing the incidence of aspergillosis and resulted in statistically significantly fewer deaths related to fungal infections, raising the possibility of improved protection with posaconazole in GVHD patients. 60 The side-effect profiles were similar for the two drugs.
Other medications evaluated for prophylaxis have included liposomal amphotericin B and voriconazole.65,66 Once-weekly liposomal amphotericin was evaluated from a safety standpoint, as it is not affected by GI absorption variability, which is a particular concern in GVHD patients. Although it was known to have effective anti-mold activity, studies found it to be frequently discontinued in SCT patients due to infusion-related adverse events.67,68 In one study, voriconazole-treated patients trended towards fewer IFI and aspergillus infections than those treated with fluconazole, but the difference remained statistically insignificant. 63
Over time, primary prophylaxis has developed for IFI in the bone marrow transplant population (Table 2). Fluconazole has demonstrated prophylaxis against candida infection, while newer azoles and echinocandins show trends towards more anti-mold activity, which fluconazole lacked, providing better options for primary prophylaxis. Recently, robust data for voriconazole and posaconazole have made them favorable options for prophylaxis against IFI in bone marrow transplant patients.
Secondary prophylaxis
While there have been considerable advances in primary prophylaxis, the IFI relapse rate remains at 30–40%. 69 Hence, secondary, or continued prophylaxis is an area that remains to be satisfactorily addressed. A small case series reported outcomes of 11 patients who had undergone HSCT with voriconazole as secondary prophylaxis. None of them went on to develop IFI during the chemotherapy-induced neutropenia phase. 70 The best evidence for secondary prophylaxis came from a prospective study in 45 patients, of which 31 had aspergillosis, and 5 had candidiasis. Voriconazole was used for prophylaxis, and at the end of 12 months, the cumulative incidence of IFI was 6% compared with 30% previously reported otherwise. 71 Both studies reported hepatotoxicity as an adverse event. Although neither was a randomized trial and both were conducted in a small number of patients, they provide some evidence to consider for secondary prophylaxis in patients, especially given the known relapse rates and the significant morbidity and mortality burden. However, the optimum antifungal, dose, and duration remain to be addressed.
Empiric treatment
In patients with a neutropenic fever after HSCT, empiric antifungal therapy is considered after 3–5 days of broad-spectrum antibiotics and persistent or recurrent fever, 72 although neutrophil recovery generally occurs quickly in this subset of patients. Liposomal amphotericin B, which is less toxic and similar in efficacy to amphotericin B, is used for empiric therapy for IFI.73,74 Azoles and echinocandins are favorable for empiric treatment as well, except for fluconazole as it is frequently used in prophylaxis. 75 Boogaerts et al. 76 compared itraconazole and amphotericin B for empiric antifungal therapy in patients with persistent neutropenic fever. Both drugs had similar efficacy, but itraconazole had significantly fewer adverse events or withdrawal of therapy, and more renal toxicity was seen in the amphotericin group. Similar safety outcomes were seen in a more recent study, where the itraconazole group demonstrated a higher success rate than the amphotericin B group. 77 Caspofungin had similar efficacy and less adverse events compared with liposomal amphotericin B when studied in a randomized controlled trial involving over 1000 patients. 78 Voriconazole may not be a preferred agent for empiric treatment as it resulted in a lower success rate when compared with liposomal amphotericin B, even though voriconazole had fewer infusion-related adverse effects and less renal toxicity. 79
Pre-emptive treatment
It has long been debated whether using fever as the sole criterion for initiation of antimicrobial treatment is appropriate, especially with the advent of imaging and biomarker assays. It was argued that reserving antifungal treatment for patients with strong evidence of infection in clinical evaluation and non-invasive tests may help bring down cost and drug toxicity, and thereby may have effect on overall mortality. Cordonnier et al. 80 conducted a randomized clinical trial to look for survival benefit in empirical versus pre-emptive treatment and concluded that although there was no difference in mortality or nephrotoxicity, pre-emptive treatment was more cost-effective. Thereafter, several randomized control trails and meta-analyses have been published comparing empirical versus pre-emptive use of antimicrobials in high-risk neutropenic patients and although there was no survival benefit, there is no strong evidence supporting a significant difference in cost effectiveness as well, and therefore, guidelines advise caution in using a pre-emptive approach, due to lack of strong evidence.81 –84
Recently, there has been a shift from empirical to pre-emptive treatment strategies in clinically stable patients to support resource reduction and a diagnostic-driven approach which can be seen in newer updated guidelines.85,86
Directed treatment
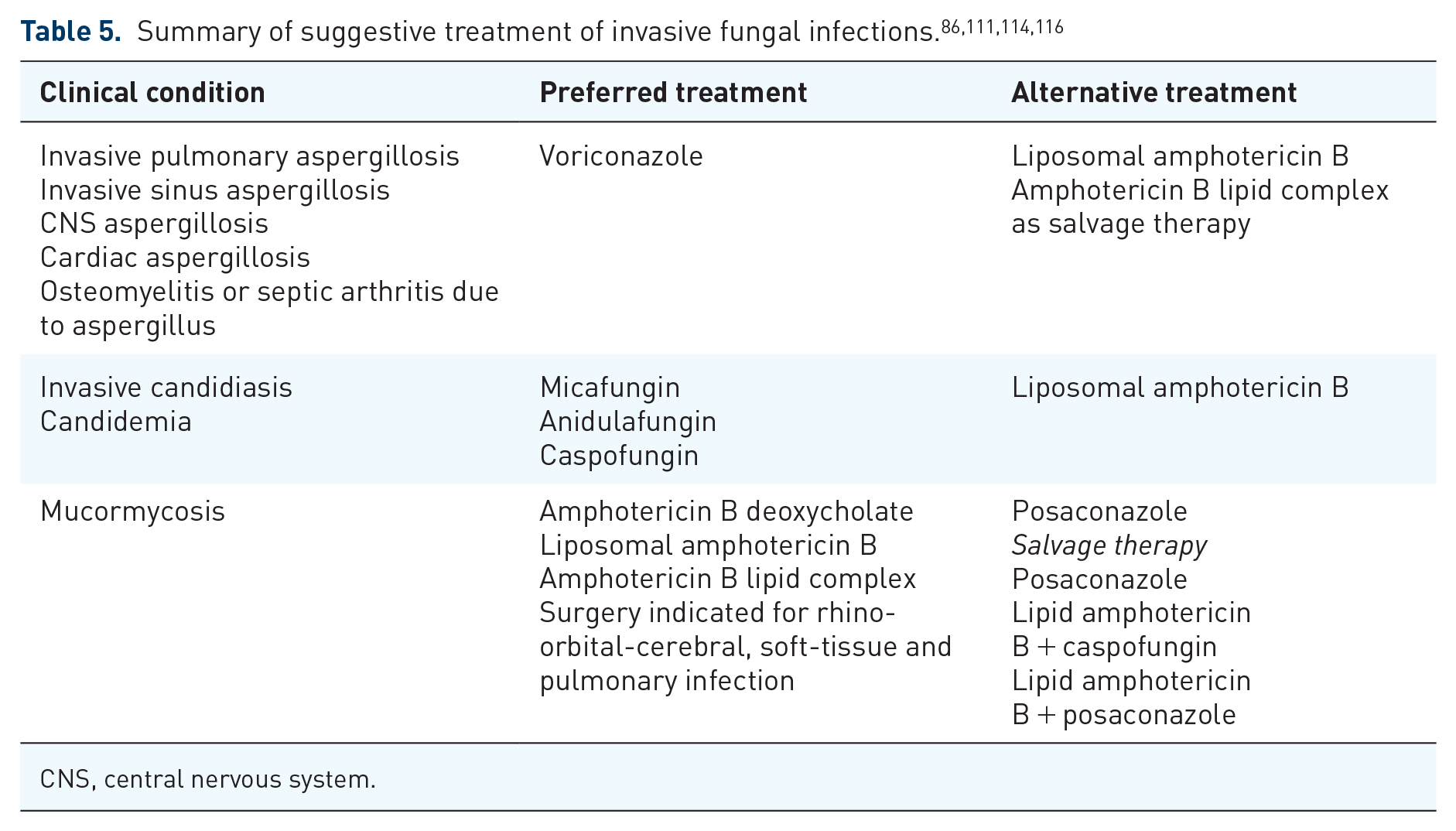
Historically, amphotericin B has been the gold standard for the treatment of IFI. However, infusion-related adverse effects and renal toxicity limit its use, and newer antifungal agents are preferred. Tables 3 and 4 detail the major clinical trials of various antifungal agents used for the treatment of invasive fungal infections.51,75,87 –110 For invasive aspergillosis, voriconazole and isavuconazole are the preferred agents with the highest quality of evidence. Voriconazole requires monitoring of serum levels.86,111 Caspofungin and itraconazole have been used, but their efficacy is unproven as data is limited. Liposomal amphotericin B remains efficacious and is used as salvage therapy.112,113 For invasive candidiasis in hematological patients, echinocandins and liposomal amphotericin B are preferred, as is voriconazole, as there is increased innate resistance to fluconazole.111,114 Mucormycosis carries high mortality, ranging from 40% to 80%. 115 Management of mucormycosis is challenging and involves a multidisciplinary approach among infectious disease, oncology, and surgical specialists. Liposomal amphotericin B with early surgery is recommended. Posaconazole has shown similar efficacy to liposomal amphotericin B.111,115 Table 5 summarizes suggestive treatment of IFI.
Major clinical trials of antifungal treatment in patients with invasive fungal infections.

AmB, amphotericin-B; CI, confidence interval; BSI, bloodstream infection; HR, hazard ratio; HSCT, hematopoietic stem-cell transplant; IV, intravenous; MITT, modified intention-to-treat analysis; NA, not available; OR, odds ratio; POS, posaconazole; POSnew, first-line posaconazole new formulation.
AKI, acute kidney injury; BA, bioavailability; CNS, central nervous system; CSF, cerebrospinal fluid; DOACs, direct oral anticoagulants; HSCT, hematopoietic stem-cell transplant; IA, invasive aspergillosis; IC, invasive candidiasis; IFI, invasive fungal infection; IV, intravenous; UTI, urinary tract infection.
CNS, central nervous system.
Persistent fever that does not resolve after 5 days of broad-spectrum antibacterial agents during neutropenia requires extensive reassessment into cause, with due consideration given to IFI. The Infectious Diseases Society of America (IDSA) guideline recommends a set of cultures and diagnostic imaging workup (chest radiograph, chest computed tomography) to rule out fungal infections in patients with neutropenia expected to last >7 days and persistent fever. For persistently febrile patients with pulmonary nodules or nodular pulmonary infiltrates, invasive mold infection should be strongly suspected, and prompt assessment frequently requires bronchoscopy with BAL with cultures, stains, and aspergillus GM antigen testing. The guidelines recommend adding an empiric antifungal agent after 4–7 days in high-risk neutropenic patients in whom total duration of neutropenia exceeds 7 days and who have persistent or recurrent fever without isolation of a clear causative agent. Based on clinical acumen, empirical agents should be considered even earlier in clinically unstable patients. For a patient already receiving anti-mold prophylaxis, switching to a different class of anti-mold antifungal given intravenously should be considered. The choice of agent for empiric antifungal therapy depends upon which fungi are most likely to be causing infection, as well as the toxicity profiles and cost. The treatment and diagnostic algorithms are extensively explained in the IDSA guideline.117,118
Conclusion
Despite advances in the use of HSCT treatment for hematopoietic stem-cell-related disorders, fungal infections continue to cause significant morbidity and mortality. Patient risk factors and the type of conditioning regimen impact the development of IFI. Close initial follow up, early diagnosis, and prophylactic treatment with antifungal agents are important management components. Newer antifungal agents have been developed, which have at least similar efficacy when compared with amphotericin B and a better safety profile. Combination therapy is considered for salvage. Therapeutic drug monitoring and checking for drug interactions is critical. Further research to improve diagnostics and therapeutics is required to improve outcomes in patients with invasive fungal infections.
Footnotes
Author contributions
All authors made substantial contributions to the conception or design of the work, the acquisition, analysis or interpretation of data for the work, drafted and assisted in critical revisions to work for important intellectual content, provided final approval of the version to be published, and are in agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Ethics statement
Our study did not require an ethical board approval because it did not contain human or animal trials.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.