Abstract
Interleukin-13 (IL-13) is a cytokine implicated in the pathophysiology of type 2 inflammatory diseases and is a clinically validated target in atopic dermatitis. APG777 is a humanized IgG1 monoclonal antibody with an optimized pharmacokinetic profile. APG777 has high affinity to IL-13 and includes a triple amino acid modification (the “YTE” modification) in its Fc region that is designed to extend its half-life. The current study examined the safety and potential toxicity of APG777 when given by once-weekly subcutaneous injection for 27 weeks to cynomolgus monkeys, and the potential reversibility of any findings following a 2-month recovery period. Toxicokinetic characteristics of APG777 were also determined. APG777 exhibited dose-proportional systemic exposure, with a half-life of approximately 28 days. No APG777-related adverse effects were noted in clinical observations, body weight, ophthalmology, electrocardiogram readings, neurologic parameters, hematology, coagulation, clinical chemistry, urinalysis, organ weights, or histopathology. Anti-drug antibodies were not detected in any APG777-exposed animals. Drug accumulation was evident over the study duration; however, there were no APG777-related adverse findings in any of the parameters analyzed. The no-observed-adverse-effect level (NOAEL) was 150 mg/kg/week. These findings provide preclinical evidence supporting continued clinical development of APG777 for IL-13-mediated diseases. The extended half-life of APG777 suggests potential benefits in reducing dosing frequency compared with existing IL-13-targeting therapies, which could improve treatment adherence and patient outcomes. The safety and efficacy of APG777 are currently being investigated in a Phase 2 clinical trial (NCT06395948) in patients with moderate-to-severe atopic dermatitis.
Keywords
Introduction
Interleukin-13 (IL-13) is a key cytokine involved in the induction and maintenance of the type 2 immune response seen in atopic dermatitis, asthma, and other inflammatory and immunologic conditions.1–3 IL-13 is primarily produced by T helper type 2 (Th2) cells and other immune cells such as mast cells, basophils, and eosinophils,4,5 and is involved in the release of other pro-inflammatory cytokines, the recruitment of inflammatory cells, and isotype switching in B-cells to immunoglobulin E (IgE).6,7 IL-13 exerts its biological effects by binding to the IL-13Rα1, which then dimerizes with the IL-4Rα to form the functional IL-13Rα1/IL-4Rα heterodimer. 8 This dimerization triggers the activation of JAK1 and JAK2, leading to the phosphorylation of signal transducer and activator of transcription 6 (STAT6) and the expression of IL-13-responsive genes such as thymus and activation-regulated chemokine (TARC).6,9–12 Clinical trials with two IL-13-targeting monoclonal antibodies, lebrikizumab and tralokinumab, have shown efficacy in reducing atopic dermatitis symptoms and improving health-related quality of life,13–16 and these therapies are now approved for use in adult and adolescent patients with moderate-to-severe atopic dermatitis.17,18
APG777 is a humanized IgG1 monoclonal antibody with an optimized pharmacokinetic (PK) profile that has been engineered for high affinity to IL-13. 19 The binding of APG777 to IL-13 prevents IL-4Rα from binding to the IL-13Rα1/IL-13 complex, which prevents formation of the active IL-13Rα1/IL-4Rα heterodimer and blocks downstream inflammatory signaling, which is similar to the mechanism of action of lebrikizumab. APG777 contains a triple amino acid modification, M253Y/S255T/T257E (referred to as a “YTE” modification) in its Fc region, that is designed to increase the binding affinity to the human neonatal Fc receptor (FcRn) under acidic pH conditions, which promotes recycling via natural mechanisms leading to a longer half-life in circulation and reduced clearance.19,20 The Fc region of APG777 also contains two additional amino acid modifications—L235A/L236A (referred to as a “LALA” modification)—designed to ablate Fc and complement effector functions. 21 An extended half-life monoclonal antibody offers potential to improve the management of atopic dermatitis and other inflammatory diseases through less frequent administration, which may be less burdensome for patients. 22
APG777 binds with high affinity to cynomolgus monkey and human IL-13 but does not bind to mouse or rat IL-13. 23 In addition, the attributes of APG777 that are designed to extend its half-life (i.e., YTE modification) in humans are also capable of extending half-life in cynomolgus monkeys. 20 In preclinical studies in female cynomolgus monkeys, a single dose of 3 mg/kg of APG777 administered intravenously and subcutaneously (SC) exhibited an average half-life of 27.6 days and a clearance rate of 1.45 mL/day/kg. APG777 showed high SC bioavailability of 81.2% and a volume of distribution at steady state of 55.7 mL/kg. In contrast, a monoclonal antibody produced based on the publicly available sequence of lebrikizumab exhibited an average half-life of 18.0 days and a clearance rate of 2.9 (mL/day/kg); its bioavailability was 75.7%, while the volume of distribution at steady state was 52.1 mL/kg. 24
The objectives of the current study were to determine the safety and potential toxicity of APG777 when given by once-weekly SC injections for 27 weeks to cynomolgus monkeys and to evaluate the potential reversibility of any findings following a 2-month recovery period. In addition, the toxicokinetic (TK) characteristics of APG777 were determined.
Materials and Methods
Study Design
Experimental Design
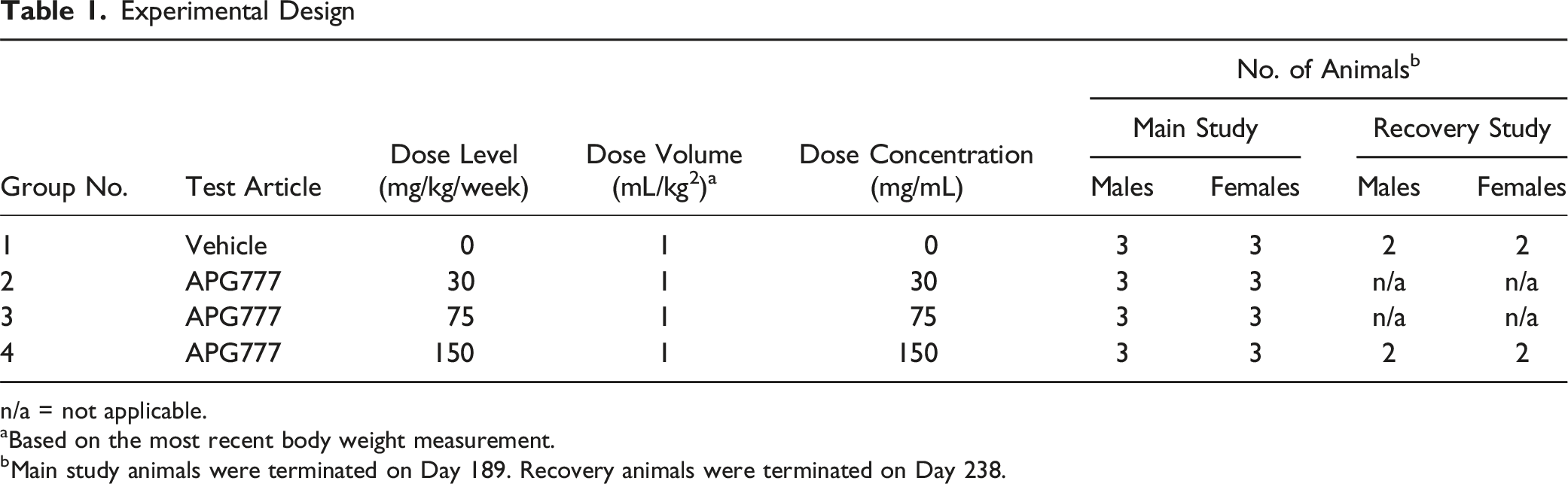
n/a = not applicable.
aBased on the most recent body weight measurement.
bMain study animals were terminated on Day 189. Recovery animals were terminated on Day 238.
The study was conducted in accordance with the U.S. FDA Good Laboratory Practice (GLP) for Nonclinical Laboratory Studies and as accepted by Regulatory Authorities throughout the European Union, Japan and other countries. The protocol and any amendments or procedures involving the care and use of animals in this study were reviewed and approved by Charles River—Nevada Institutional Animal Care and Use Committee (IACUC) before conduct.
Test Substance
The test substance, APG777, was diluted according to target concentration (30, 75, or 150 mg/mL) in buffer containing 20 mM histidine, 6% (w/v) sucrose, 0.04% (w/v) polysorbate 80, and 10 mM L-Methionine, pH 6.0. The vehicle control contained 20 mM histidine, 6% (w/v) sucrose, 0.04% (w/v) polysorbate 80, and 10 mM L-Methionine, pH 6.0.
Animals
Naïve cynomolgus monkeys of Vietnam origin between 22 and 36 months of age and weighing 2.0−3.0 kg were supplied by Charles River Laboratories (Frederick, MD, United States). The animals were housed as specified in the USDA Animal Welfare Act (Code of Federal Regulations, Title 9) and as described in the Guide for the Care and Use of Laboratory Animals. Male and female monkeys were housed separately (up to three animals of the same sex and same dosing group together) and fed with PMI Nutrition International Certified Primate Chow No. 5048 (Henderson, Colorado, US). The diet was supplemented with fruit or vegetables at least 2 to 3 times weekly. Water was available ad libitum via an automatic watering system. Animals were acclimated to laboratory housing for at least 14 days before the initiation of dosing. Veterinary exams were conducted once during the pretreatment period to confirm health status prior to the dosing period; animals in poor health or at extremes of body weight range were not assigned to groups and were replaced by alternate animals, if applicable. Animal termination was conducted using intravenous injection of a commercially available veterinary euthanasia solution, followed by exsanguination. If an animal was determined to be in overt pain/distress, or appeared moribund and was beyond the point where recovery appeared reasonable, the animal would be euthanized for humane reasons in accordance with the American Veterinary Medical Association (AVMA) Guidelines on Euthanasia.
Assessments and Schedule
General In-Life Assessments Conducted in Main Study and Recovery Animals
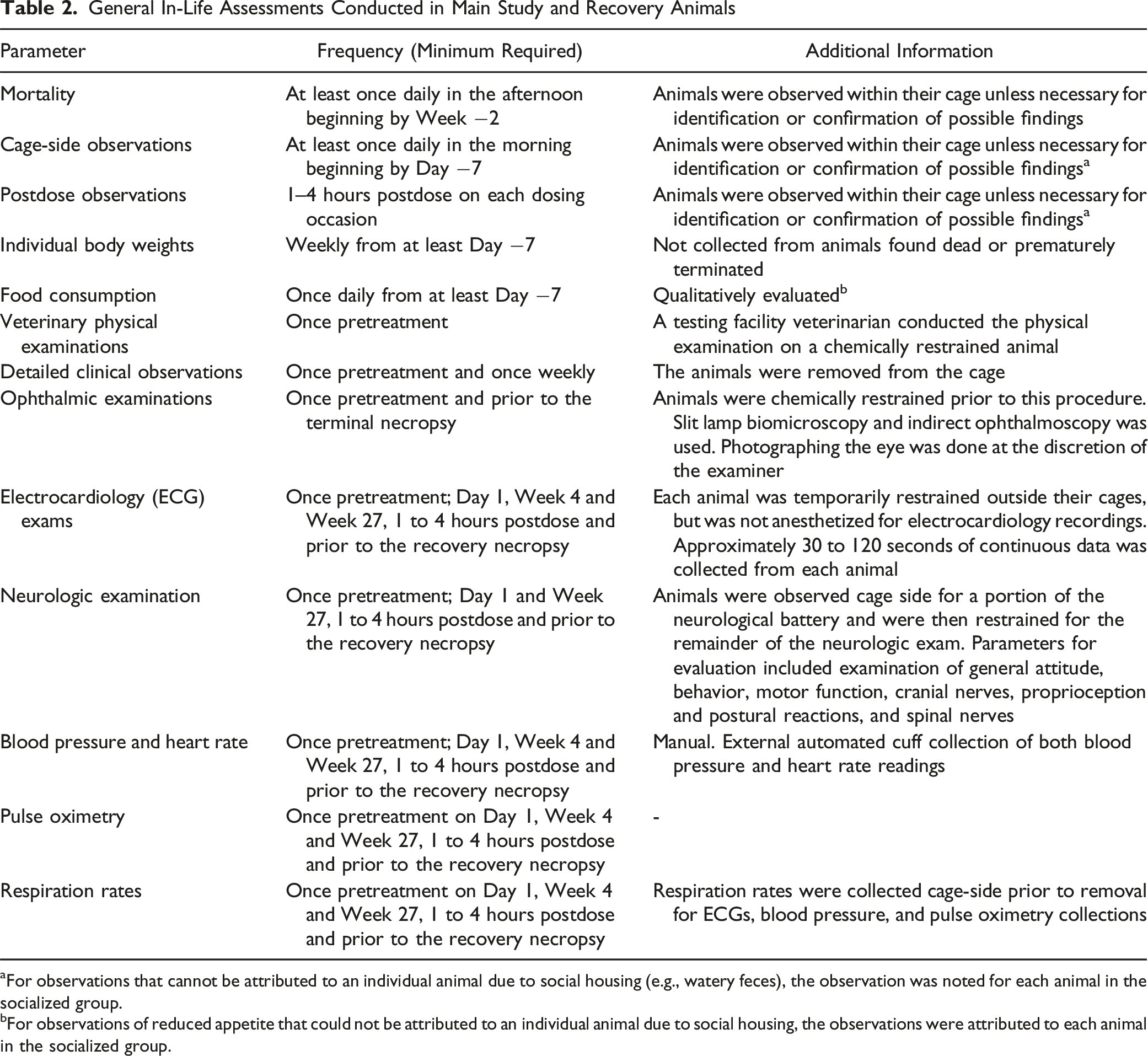
aFor observations that cannot be attributed to an individual animal due to social housing (e.g., watery feces), the observation was noted for each animal in the socialized group.
bFor observations of reduced appetite that could not be attributed to an individual animal due to social housing, the observations were attributed to each animal in the socialized group.
For clinical pathology (hematology, coagulation, clinical chemistry, and urinalysis), blood and urine samples were collected twice pretreatment (except urinalysis only once pretreatment) and on Days 30, 85, 188 (all animals), and 237 (recovery animals only).
Anti-Drug Antibodies
Blood was collected and processed to serum from all animals pretreatment and predose on Days 85 and 183. Animals designated as recovery animals from Groups 1 and 4 had an additional blood collection on Day 238. All samples were analyzed for antibodies against APG777 using an electrochemiluminescence method validated under Charles River Laboratories Testing Facility Study No. 20392617.
TK Analyses and Bioanalyses
Serum samples were collected from all animals predose and at 5 minutes, 4 and 24 hours postdose on Days 1 and 183, with further collections 48, 96, and 168 hours following dosing. Additional predose samples were collected on Days 15, 29, 57, 85, 113, 141, and 169. From recovery animals, additional samples were collected at 336, 504, 672, 840, 1008, 1176, and 1320 hours postdose on Day 183.
For bioanalytical sample analysis, serum was analyzed for the concentration of APG777 using a validated analytical procedure. Statistical analyses including regression analysis and descriptive statistics (arithmetic means and standard deviations, accuracy and precision) were performed.
TK analyses were performed on serum concentration-versus-time data using Phoenix WinNonlin (v 8.44; Certara; Radnor, PA, USA) with the non-compartmental analysis function (linear trapezoidal rule for area under the serum concentration-time curve [AUC] calculations). Nominal dose values and sampling times were used for calculations. For TK calculations and serum concentration descriptive statistics, any concentration reported as below the limit of quantification (BLQ) was set equal to zero. The Cmax, Tmax, and Tlast values were determined by direct assessment of the concentration-versus-time data. All AUC calculations were performed using the linear trapezoidal linear interpolation method. In the Group 4 recovery animals only, the terminal rate constant (lambda z, λz) was calculated using the concentration values between Day 2 and Day 55 providing a correlation coefficient greater than 0.9. Individual animal serum concentration-versus-time data and TK parameters were summarized by arithmetic mean, standard deviation (SD), and percent coefficient of variation (CV%) and reported to three significant figures, with the exception of N and individual animal Tlast, Tmax, and λz upper / lower values, which were reported as whole numbers, and individual animal ratio, half-life (t1/2), and λz span values, which were reported to two significant figures.
Results
Summary of Animal Dosing
Thirty-two cynomolgus monkeys, including 16 males and 16 females, were administered 0 (vehicle), 30, 75, or 150 mg/kg/week of APG777 by subcutaneous injection and were necropsied 27 weeks later or survived for an additional 2-month recovery period as outlined in the experimental design (Table 1). All APG777 concentrations used for animal dosing met the acceptance criteria (± 10% of theoretical concentration).
Safety/Toxicity
There were no APG777-related effects on any of the safety or toxicity parameters evaluated. There were no early deaths or unscheduled terminations during the study duration. There were no APG777-related clinical observations or changes in qualitative food consumption. All clinical observations that occurred were considered unrelated to APG777 because they were dose-independent, transient, occurred exclusively during the pretreatment period, and/or were considered commonly observed clinical signs in cynomolgus monkeys under similar study conditions. These clinical signs included localized alopecia, abrasions, lacerations, bruising (procedural related), red skin, petechia (prestudy only), emesis, liquid feces, and red liquid material on cage floor (likely associated with menses in the females). Differences in body weights, none of which attained statistical significance, were attributed to biologic variation because they were similar to fluctuations in control and/or were of a magnitude of change commonly observed in cynomolgus monkeys under similar study conditions. There were no macroscopic observations at the injection site in any animal. Infrequent mild microscopic findings observed at subcutaneous injection sites were considered procedurally-induced and unrelated to administration of APG777. There were no APG777-related changes in ophthalmology parameters during the study duration. The administration of APG777 did not result in any changes of the measured electrocardiogram intervals, which included the RR-derived heart rate, RR interval, PR interval, QRS interval, QT interval, and the rate corrected QTc interval. There were no APG777-related changes in qualitative electrocardiograms, neurologic parameters, blood pressure or heart rate, pulse oximetry values, respiration rate, hematology parameters, coagulation parameters, clinical chemistry parameters, or urinalysis parameters. There were no APG777-related macroscopic observations and no APG777-related organ weight changes. Microscopic findings observed in the main and recovery studies were considered incidental, of the nature commonly observed in cynomolgus macaques of this age, and/or were of similar incidence and severity in control and treated animals and, therefore, were considered unrelated to administration of APG777.
Bioanalysis, TK, and Anti-Drug Antibody Evaluations
Evidence of systemic exposure to APG777 was observed in all treated monkeys following weekly SC administration. All control animal samples were below the lower limit of quantification (LLOQ < 0.0500 μg/mL), except for one animal sample that showed a quantifiable concentration (1.21 μg/mL) on Day 183 at 4 hours postdose. This animal’s positive concentration value was over 1000-fold lower than the mean concentration value at the same time point in the low dose (30 mg/kg/week) group. As such, this value is considered an anomaly and has no impact on TK interpretation.
There were no anti-drug antibodies detected in any APG777-exposed animals.
Sex Combined Summary of TK Parameters Following Weekly APG777 Subcutaneous Administration During the Main Study
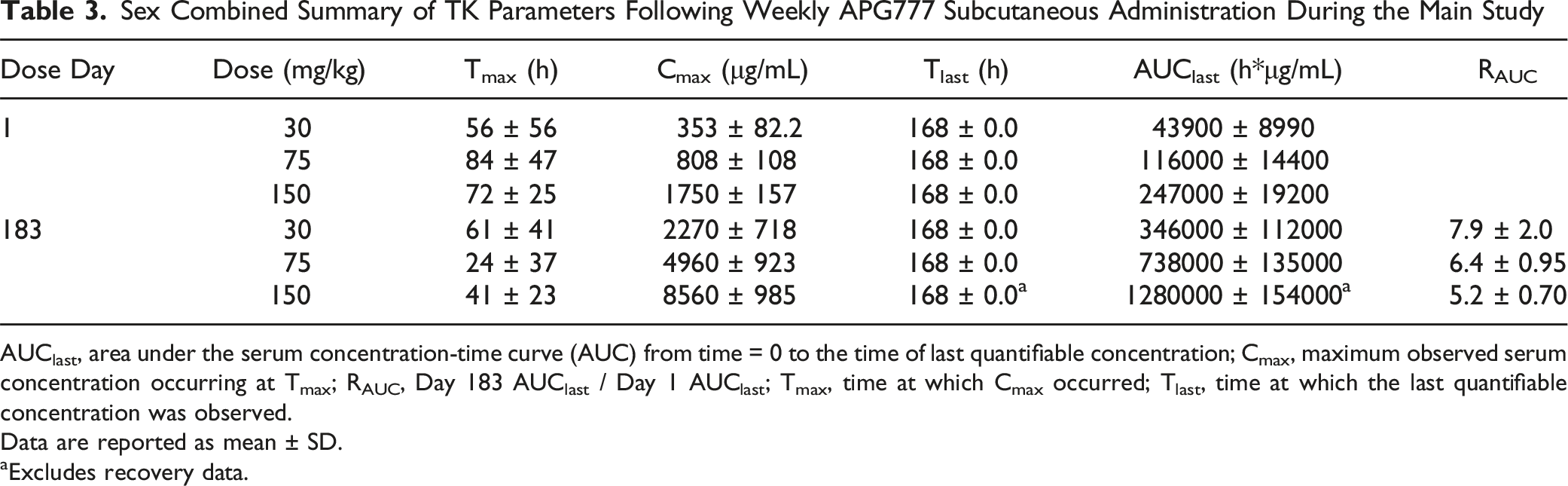
AUClast, area under the serum concentration-time curve (AUC) from time = 0 to the time of last quantifiable concentration; Cmax, maximum observed serum concentration occurring at Tmax; RAUC, Day 183 AUClast / Day 1 AUClast; Tmax, time at which Cmax occurred; Tlast, time at which the last quantifiable concentration was observed.
Data are reported as mean ± SD.
aExcludes recovery data.
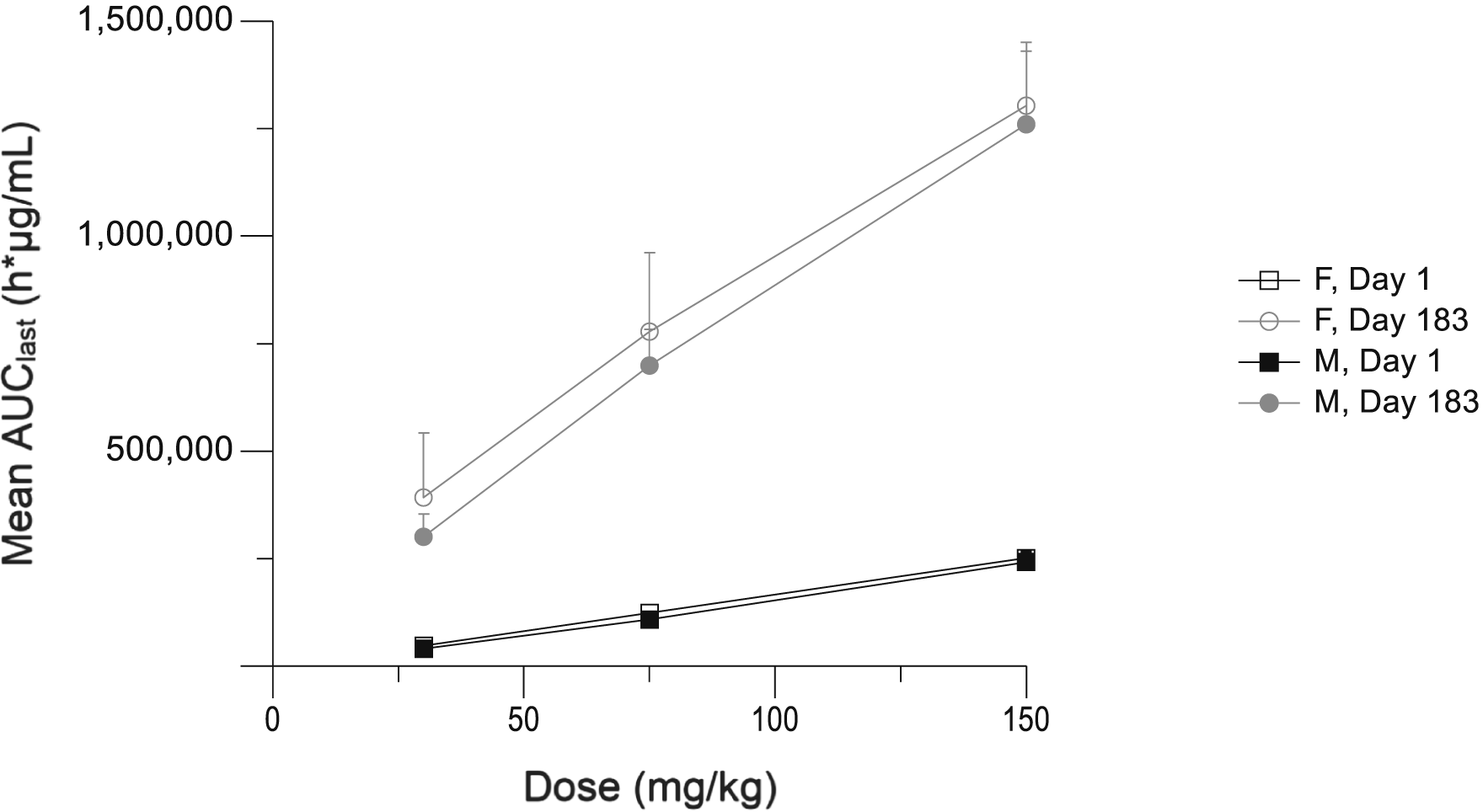
Mean AUClast versus dose following weekly APG777 subcutaneous administration to female and male monkeys. AUClast, area under the serum concentration-time curve (AUC) from time = 0 to the time of last quantifiable concentration; F, female; M, male. Sex, Day; error bars represent standard deviation. For recovery animals, Day 183 data was truncated to 168 hours.
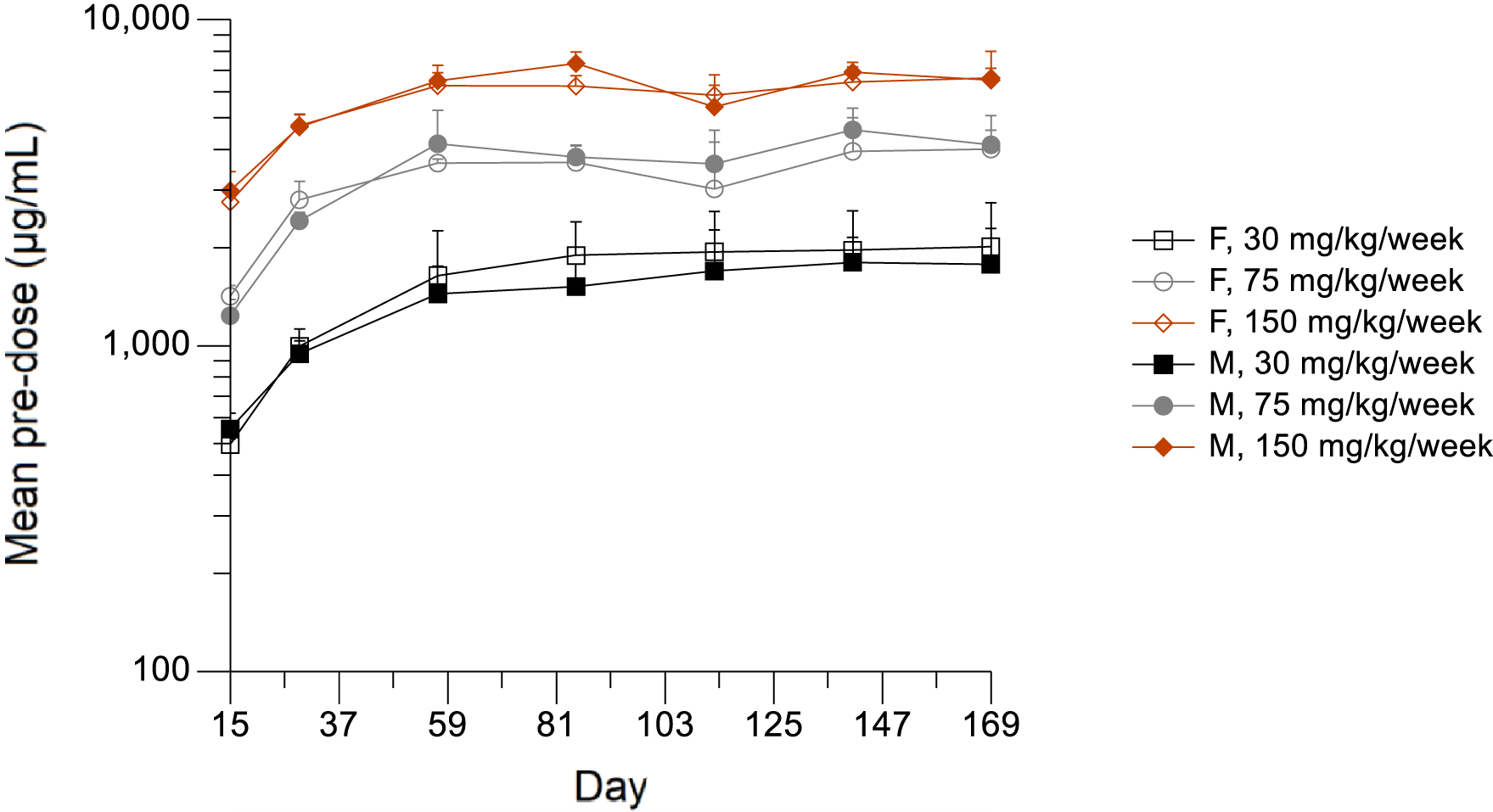
Mean APG777 serum predose concentrations by day in female and male monkeys. Error bars represent standard deviation.
Sex Combined Summary TK Parameters Following Weekly APG777 Subcutaneous Administration During the Recovery Period

AUClast, area under the serum concentration-time curve (AUC) from time = 0 to the time of last quantifiable concentration for recovery animals only (post the last dose on Day 183 only data out to 168 hours was used); t1/2, terminal half-life.
Data are reported as mean ± SD.
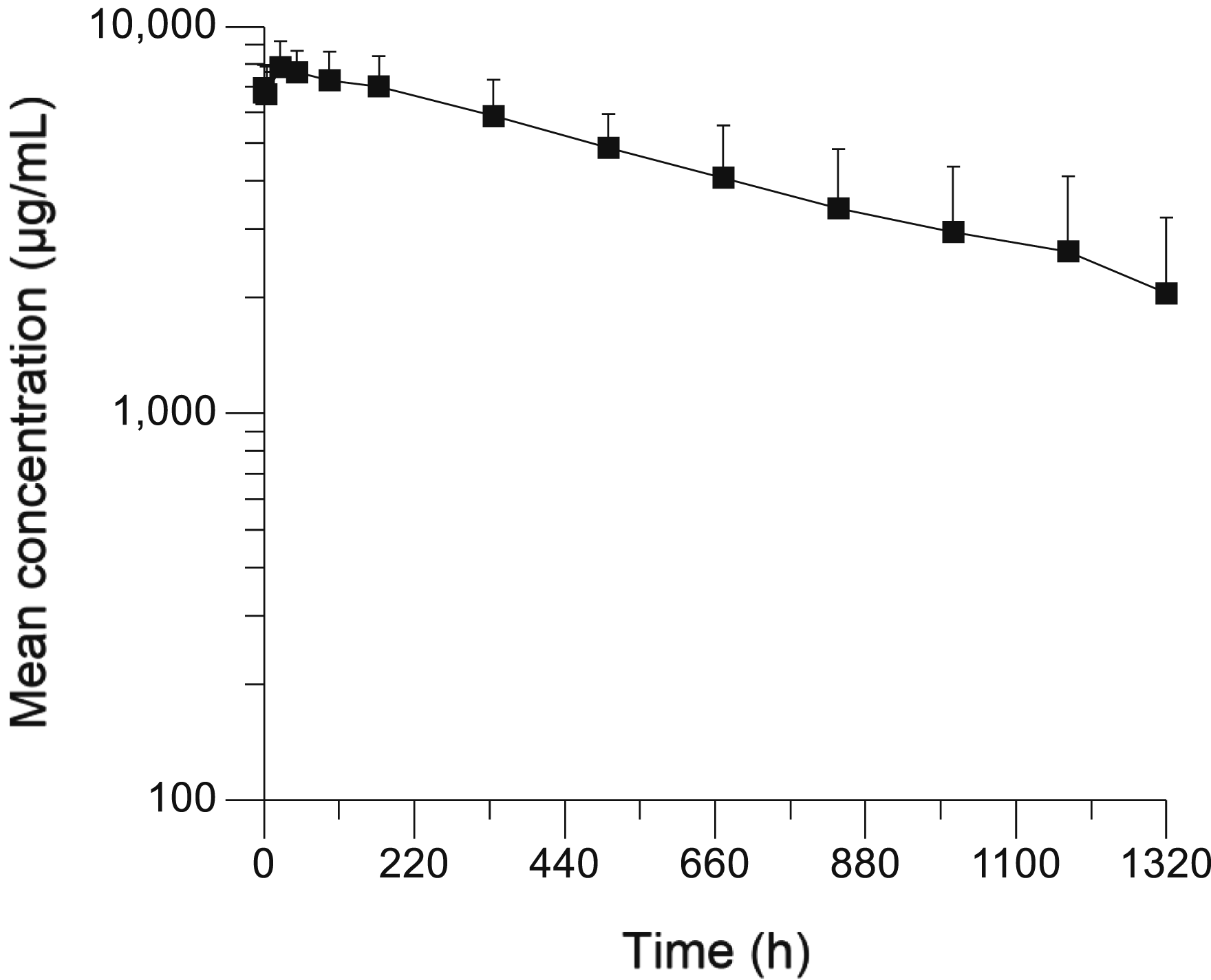
Mean APG777 serum concentration by time profile following weekly subcutaneous administration to female and male monkeys (study Day 183, recovery animals only, 150 mg/kg/week). Error bars represent standard deviation.
Discussion
Monoclonal antibodies targeting IL-13 reduce symptoms and improve health-related quality of life for patients with atopic dermatitis, and have been shown to be a safe and effective treatment option for this disease13–16. Currently approved IL-13 inhibitors are administered every 2–4 weeks via the SC route to treat patients with moderate-to-severe atopic dermatitis17,18; however, there is a high unmet need for therapies which may reduce dosing frequency and injection burden for patients with atopic dermatitis and other chronic inflammatory diseases. 22
APG777 is a monoclonal antibody with an optimized PK profile that targets IL-13; it is currently in Phase 2 clinical development for the treatment of atopic dermatitis. 25 Compared with monoclonal antibodies produced based on the publicly available sequence of lebrikizumab (anti-IL-13), APG777 demonstrates similar affinity for IL-13 and similar potency in multiple assays.19,23 In the current study, we examined the safety and potential toxicity of APG777 and showed that once-weekly SC administration at dose levels of 30, 75, or 150 mg/kg/week for 27 weeks was well tolerated in cynomolgus monkeys. Despite administering high doses of APG777, which resulted in drug accumulation in the animals over the study duration, and despite the YTE modification included in APG777 that extends its half-life beyond currently approved monoclonal antibodies targeting IL-13, there were no APG777-related adverse findings in any of the parameters analyzed. Based on the results under the conditions of this study, the no-observed-adverse-effect level (NOAEL) was 150 mg/kg/week. At the NOAEL, the Day 183 mean Cmax and AUClast values for combined genders were 8,560 ± 985 μg/mL and 1,280,000 ± 154,000 h*μg/mL. Based on the predicted exposure for the highest dose being investigated in the ongoing Phase 2 study, exposures in monkeys at the NOAEL provide > 30-fold safety margin.
The favorable safety and toxicity data reported here in cynomologus monkeys build upon analyses of the in vitro potency, pharmacokinetics, and pharmacodynamics of APG777 and support progression to clinical development. In a Phase 1 first-in-human trial, APG777 was well tolerated at doses up to 1200 mg, showing a half-life of 77 days in healthy subjects. Furthermore, SC administration of APG777 in healthy subjects resulted in sustained inhibition of pSTAT6 and TARC, key biomarkers for IL-13 targeting and atopic dermatitis, with inhibition of pSTAT6 observed through 12 months of follow-up. 26
The authors are unaware of any published comparative toxicology data on extended half-life monoclonal antibodies compared to their standard half-life counterparts; however, some clinical safety data are available. Nirsevimab, for example, a YTE-modified, extended half-life monoclonal antibody for the prevention of respiratory syncytial virus-associated lower respiratory tract disease has a similar safety and tolerability profile to standard half-life palivizumab. 27 Similarly, ravulizumab, an Fc-engineered extended half-life monoclonal antibody for the treatment of paroxysmal nocturnal hemoglobinuria, showed a similar safety profile to standard half-life eculizumab. 28 While these data suggest that extending the half-life of monoclonal antibodies through Fc modifications does not necessarily result in altered safety profiles, the safety of all modified antibodies will need individual evaluation, consistent with requirements of any new molecular entity intended to ultimately serve as an approved human therapy. The development of extended half-life monoclonal antibodies is, however, gaining significant traction, with several already approved for medical use and numerous others in clinical development. 29
The results of this study demonstrate that the increased half-life of APG777 in cynomolgus monkeys did not lead to adverse safety or toxicological findings, which supports the ongoing development of APG777 for atopic dermatitis and other IL-13-mediated chronic, type 2 inflammatory diseases. The efficacy and safety of APG777 is currently being investigated in the randomized, double-blind, placebo-controlled Phase 2 APEX study (NCT06395948) in adults with moderate-to-severe atopic dermatitis, where every 3- and every 6-month maintenance dosing will be evaluated. 25 Future research may explore the potential for APG777 in other chronic type 2 inflammatory diseases.
Footnotes
Author’s Note
The study was conducted in accordance with the U.S. FDA Good Laboratory Practice (GLP) for Nonclinical Laboratory Studies as well as applicable ICH Harmonised Tripartite Guidelines which are collectively accepted by Regulatory Authorities throughout the European Union, Japan, and other countries. Qualified researchers may reach out to the corresponding author to request data access.
Acknowledgments
We would like to thank Lee Miller and Lucy Kanan (Miller Medical Communications, on behalf of Apogee Therapeutics, Inc.), and Lynnea Waters (Apogee Therapeutics, Inc.), for their editorial support.
Author Contributions
Rubio, D. contributed to analysis and interpretation, drafted manuscript, and critically revised manuscript; Zhu, E. contributed to acquisition, analysis, and interpretation and critically revised manuscript; Thurston, A. and Funk, K.A. contributed to design, contributed to analysis and interpretation, and drafted manuscript; York, K.A. contributed to design, contributed to acquisition, analysis, and interpretation, and drafted manuscript; Dambkowski, C. contributed to conception, contributed to interpretation, and critically revised manuscript; Badger, D. contributed to conception and design, contributed to interpretation, drafted manuscript, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: C. Dambkowski and D. Badger are employees of Apogee Therapeutics Inc. D. Rubio was employed by Apogee Therapeutics at the time the study was conducted. C. Dambkowski, D. Badger, D. Rubio, and E. Zhu hold stock/stock options in Apogee Therapeutics. A. Thurston is an independent consultant (Toxicology Solutions, Inc.) providing ADME support. K.A. Funk is employed at EPL, Inc. K.A. York is employed by Charles River Laboratories.
Funding
The author(s) disclosed receipt of the following financial support for the research, and/or publication of this article: The research reported in this manuscript and medical writing support was funded by Apogee Therapeutics, Inc.
