Abstract
Interleukin-21 (IL-21), a pleiotropic immunostimulatory type I cytokine, has anticancer effects in animal models. Preclinical studies designed to assess the safety of recombinant human IL-21 (rIL-21) for use in phase I oncology studies are described. The rIL-21 (≤3.0 mg/kg per dose) was given intravenously to cynomolgus monkeys (Macaca fascicularis) once daily for 5 days, followed by 9 nondosing days (1 cycle) for ≤4 cycles. The rIL-21 pharmacokinetics was dose-dependent. Accumulation was not observed after repeated dosing, consistent with the short elimination half-life (t 1/2,λz; 0.4-0.8 hours). Safety findings included cyclical anemia and thrombocytopenia, clinical pathology changes consistent with acute-phase response, leukocyte infiltrates in hepatic sinusoids, and sporadic serum transaminase elevations (typically <3 times upper-limit of normal); all were reversible upon cessation of treatment. Decreased pharmacodynamic responses with time corresponded to the development of anti-rIL-21 antibodies; effects varied among individuals and were dose-dependent. These studies demonstrated rIL-21 to be generally well-tolerated when administered to cynomolgus monkeys, and all adverse effects were reversible upon treatment cessation. However, development of anti-rIL-21 antibodies may limit the use of this species in long-term studies.
Introduction
Interleukin-21 (IL-21) is a type I cytokine produced by activated CD4+ T cells and natural killer (NK) T cells. 1 –3 Sequence, protein structure, and gene structure place IL-21 in the IL-2 family of cytokines, with greatest similarity to IL-2, IL-4, and IL-15. The receptor complex for IL-21 consists of the common cytokine receptor γ chain and an IL-21 specific receptor protein. Physiologic expression of the IL-21 receptor complex is limited to lymphoid organs and peripheral blood mononuclear cells; however, expression of both the receptor subunits can be acquired by such tissues as synovium and epithelium. 3 The IL-21 receptor–ligand interaction results in signaling primarily through Jak (Janus-activated kinase)/STAT (signal transducer and activator of transcription) pathways 1 and 3. This produces multiple effects, including enhanced T-cell immunity, NK cell activation, and B-cell differentiation and antibody production.
Proof of concept studies conducted in mice, using recombinant murine IL-21, demonstrate potent anti-tumor activity in a variety of syngeneic models. 4 –7 The IL-21 efficacy in these models is thought to be primarily due to the enhancement of cytotoxic T cell and NK cell function. These studies substantiate the immunomodulatory role of IL-21 and support the use of this cytokine as a potential therapeutic agent for cancer and other disease states in which enhanced immune function and surveillance may be critical.
Due to their complex structure, biopharmaceuticals often possess a high degree of target specificity, which often limits activity across animal species. Because of this, the activity of a biopharmaceutical must be demonstrated in a species prior to its use as a model system for safety evaluation of that biopharmaceutical. 8 This article summarizes results of in vitro studies, identifying cynomolgus monkeys (Macaca fascicularis) as a relevant species for testing recombinant human IL-21 (rIL 21), and describes the in vivo biological activity, pharmacokinetics, and safety of rIL-21 in these macaques. These safety studies were designed to support phases I and II clinical study treatment schedules consisting of a series of 5 consecutive daily intravenous (IV) infusions, with each 5 day series separated by a 9-day rest period. This is similar to the approved treatment schedule for IL-2. 9
Materials and Methods
Animal Care and Use
Purpose-bred cynomolgus monkeys (M. fascicularis) were imported or obtained from licensed vendors and underwent standard quarantine periods prior to study initiation. During the study periods, animals were single-housed in primary enclosures compliant with specifications set by the United States Department of Agriculture (USDA) Animal Welfare Act (9 CFR, Parts 1, 2, and 3) and as described in the Guide for the Care and Use of Laboratory Animals. 10 All experimental procedures were conducted in AAALAC accredited facilities under protocols approved by the Animal Care and Use Committee of each facility.
Regulatory Compliance
Good Laboratory Practice (GLP)-compliant studies were conducted in accordance with the Organization for Economic Co-operation and Development Principles on GLP (C(97) 186/Final, effective 1997) and The Nonclinical Laboratory Studies GLP Regulations issued by the US Food and Drug Administration (Title 21 of the Code of Federal Regulations, Part 58, effective June 29, 1979). In addition, the nonclinical safety program was conducted in accordance with the Preclinical Safety Evaluation of Biotechnology-Derived Pharmaceuticals issued by the International Conference on Harmonization (ICH S6).
Test Items and Formulation
The rIL-21 for nonclinical studies was expressed in Escherichia coli (134 amino acids with an additional N-terminal methionine, molecular weight ~15 kDa). Purity of rIL-21 was judged to be in excess of 95% by cation exchange high pressure liquid chromatography. Dosing solutions in the first dose range-finding study (see below) contained 0.2, 2.0, or 9.0 mg/mL rIL-21 in phosphate-buffered saline, pH 6.0. Dosing solutions used in the second dose range-finding and GLP-toxicology studies contained 3 or 10 mg/mL rIL-21 in aqueous-based formulations with histidine (10 mM) and mannitol (4.72%) at pH 5.3. Endotoxin content determined by limulus amebocyte lysate test was <2.14 EU/mL.
Experimental Design
Relevant species were identified via DNA sequence analysis of the IL-21 receptor and STAT3 activation of peripheral blood lymphocytes (PBLs).
Two non-GLP dose range-finding studies were conducted, followed by a GLP toxicology study. In the first dose range-finding study, which also supported the cynomolgus monkey as a relevant species, rIL-21 was given by IV bolus once daily for 3 days (3 mg/kg per dose) with animals being necropsied on the day after the last dose, or once daily by IV bolus for 5 days (0, 0.06 or 0.6 mg/kg per dose) with necropsy 3 days following the last dose (on day 8, to avoid weekend necropsy). Each treatment group contained three >2-year-old male cynomolgus monkeys weighing 3.76 to 5.15 kg.
In the second dose range-finding study, rIL-21 (0, 0.5, or 1.5 mg/kg per dose) was given by IV bolus injection once daily for 5 consecutive days. The dosing period was followed by 9 treatment-free days. A 5-day dosing period plus 9 treatment-free days was defined as 1 dosing cycle. Following 1 complete dosing cycle, 5 additional daily doses were administered. The study was terminated 6 days following the last dose. Each treatment group contained 1 male (4.6-5 years old, weighing 4.8-5.1 kg) and 1 female (4.3-5.6 years old, weighing 3-4 kg) cynomolgus monkey. After study termination, animals from the second dose range-finding study were returned to the general colony.
The GLP toxicology study was conducted in male (3.5-5.2 years old, 3.4-5.6 kg) and female (3.3-5.5 years old, 2.8-5.2 kg) cynomolgus monkeys. Three complete dosing cycles followed by 5 additional days of treatment (0, 0.01, 0.1, or 0.5 mg/kg rIL-21) were administered, with dosing occurring on study days 1 to 5, 15 to 19, 29 to 33, and 43 to 47. Subgroups of 2 male plus 2 female monkeys were euthanized 1 (all groups), 8 (0 and 0.5 mg/kg dose groups), and 28 (all groups) days following the last dose of rIL-21 (study days 48, 55, and 75, respectively).
Phosphorylation of STAT3
Whole blood of 30 mL, collected into heparin or EDTA, was obtained from the following species: human (ZymoGenetics), nonhuman primates (cynomolgous and rhesus; SNBL USA, Ltd Everett, Washington, USA), dog (beagle; Covance, Inc Madison, Wisconsin, USA), pig (Yucatan miniature swine; Sinclair Research Center, Inc Columbia, Missouri, USA), rabbit (New Zealand White; Western Oregon Rabbitry Philomath, Oregon, USA), and rat (Sprague Dawley; Charles River Laboratories Wilmington, Massachusetts, USA). Briefly, peripheral blood mononuclear cells were isolated on a Ficoll gradient (BD Pharmingen San Diego, California, USA), incubated with serial dilutions of rIL-21, stained for surface markers, fixed with paraformaldehyde, permeabilized, and stained with anti-pSTAT3 antibody (BD Pharmingen). Staining was evaluated using a FACS Calibur flow cytometer (BD Biosciences, San Jose, California, USA).
Receptor Binding
The rIL-21 was labeled with 125I using the indirect Iodogen labeling procedure of Chizzonite (Pierce Chemical, Rockford, Illinois, USA). Briefly, BAF3 cells expressing either the cynomolgus or human IL-21 receptor were incubated in 96-well microtiter plates at 4°C with 125I-rIL-21 in the absence (total ligand binding) and in the presence (nonspecific binding) of a large excess of unlabeled rIL-21. Specific binding was calculated as the difference between the total and nonspecific values. Equilibrium dissociation constants (Kd) were determined by fitting the data to a one-site model using nonlinear regression.
General Toxicology, Pharmacokinetics, and Pharmacodynamics
Clinical observations
Clinical signs of toxicity were subjectively determined following standard procedures, food consumption was qualitatively assessed, and body temperature was determined by rectal probe.
Clinical pathology
In the GLP toxicology study, blood samples for hematology, coagulation parameters, and clinical chemistry were drawn prestudy and on days 1, 6, 15, 20, 29, 34, 43, 48, 61, and 75. Comprehensive hematology evaluations included determinations of differential leukocyte count, platelet counts, indicators of erythrocyte mass (ie, red blood cell count, hemoglobin, and hematocrit), and visual evaluation of cellular morphology. Standard coagulation tests were conducted. Comprehensive serum chemistry analyses included determination of serum enzyme activity, markers of renal integrity, serum electrolytes, and a determination of C-reactive protein (CRP) concentration.
Flow cytometry
In the GLP toxicology study, blood samples for flow cytometry were drawn prestudy and on days 1, 6, 15, 20, 29, 34, 43, 48, 61, and 75. Peripheral blood leukocytes were immunophenotyped for the identification of the following cell types: T cells—total (CD3+ [clone SP34, BD Pharmingen, San Diego, California, USA]), helper (CD3+/CD4+ [clone L200, BD Pharmingen]), and suppressor/cytotoxic (CD3+/CD8+ [clone B9.11, Beckman Coulter, Fullerton, California, USA]); B cells—resting [CD20+ clone B9E9, Beckman Coulter]/CD86− [clone IT2.2, BD Pharmingen]), and activated (CD20+/CD54+ [clone LB-2, BD Pharmingen] or CD86+); and monocytes—naive (CD14+ (clone M5E3, BD Pharmingen)/CD64 (clone 10.1, BD Pharmingen)), and activated (CD14+/CD64+). Human antigen-specific fluorochrome-labeled antibodies were determined to be cross-reactive in the cynomolgus prior to study sample evaluation.
Flow-count fluorospheres (Beckman Coulter) were included to allow for leukocyte quantification. Four-color, multiparameter data acquisition was conducted on a Coulter Epics XL-MCL flow cytometer using System IITM software (Beckman Coulter).
Serum cytokines
In the GLP toxicology study, blood samples for serum cytokines were drawn prestudy; predose and 4 hours postdose on day 1; and 4 hours postdose on days 5, 19, 33, 47, and 75. Sera were evaluated for IL-1β, IL-6, tumor necrosis factor (TNF)-α, and interferon (INF)-γ using validated enzyme-linked immunosorbent assay (ELISA) methods developed at Sierra Biomedical Inc. (Sparks, Nevada, USA).
Physiologic safety evaluations
Body temperature, heart rate, electrocardiograms (ECG), and blood pressure measurements were conducted on nonsedated cynomolgus monkeys on study days 1, 5, 19, and 47 of the GLP toxicology study. The ECG tracings were qualitatively evaluated by a veterinary cardiologist and physical exams were conducted by a staff veterinarian.
Ophthalmology
Animals were lightly sedated with ketamine prior to examination of the anterior and posterior chambers of the eye by direct ophthalmoscope. One to two drops of a mydriatic solution (1% tropicamide) was instilled into each eye to facilitate the evaluation. Ophthalmology examinations were conducted predose and during the dosing and recovery phases of the GLP toxicology study.
Serum IL-21 concentration
An ELISA method was developed and validated by ZymoGenetics for the measurement of rIL-21 in cynomolgus monkey serum. The assay used murine monoclonal anti-rIL-21 capture and detection antibodies. The assay had a lower limit of quantitation of 7.81 ng/mL rIL-21.
Anti-rIL-21 antibodies
An ELISA for the determination of anti-rIL-21 immunoglobulin G (IgG) antibodies in serum from cynomolgus monkeys was developed and validated at ZymoGenetics. Anti-rIL-21 antibodies were captured with rIL-21 (coated to plate) and detected with a protein G-HRP (horse radish peroxidase) conjugate that recognized cynomolgus IgG. The assay format was titer-based and semiquantitative; titer measurement was based on the log10 of the dilution necessary to cross a defined cut point. In the GLP toxicology study, blood was drawn for serum antibody determination on days 1, 11, 19, 25, 39, 47, 61, and 75.
The presence of neutralizing antibodies was established using a cell-based bioassay to monitor for neutralizing activity in serum. This assay was based on growth inhibition in an IL-21-dependent cell line exposed to assay control serum or serum from antibody positive rIL-21-treated study animals. Antibody-positive sera were reported as “neutralizing” if the calculated percentage neutralization exceeded 30%.
Anatomic pathology
Tissues and organs were fixed in 10% neutral-buffered formalin, routinely processed into paraffin blocks, sectioned, and stained with hematoxylin and eosin for examination by light microscopy. Immunohistochemical evaluations were conducted on formalin-fixed tissues or on tissues snap-frozen in liquid nitrogen at the time of necropsy.
Immunohistochemistry
Sections of spleen, liver, thymus, and lymph nodes (mesenteric and inguinal) were immunohistochemically stained for surface markers on T-helper cells (CD4+), B cells (CD20+), plasma cells (plasma cell antigen+), and cells of the monocyte/macrophage/dendritic cell lineage (CD11c+).
Pharmacokinetics
Pharmacokinetic analysis of serum rIL-21 concentration versus time data was performed using WinNonlin Professional 4.0 software (Pharsight, Inc, Cary, North Carolina, USA). Noncompartmental methods were used to determine pharmacokinetic parameters for each individual animal predose and 5, 15, 60, and 240 minutes postdose on days 1, 19, and 47 of the GLP toxicology study. The elimination half-life, t 1/2,λz, was calculated from the slope of the regression line that best fit the terminal portion of the log-linear concentration versus time curve (λz). The area under the concentration versus time curve (AUC) was calculated using the trapezoidal rule and extrapolated to infinity based on the equation: AUCinf = AUC + C n/λz, where C n is the last measurable time point. The back extrapolated concentration at time 0 (C 0), total body clearance (CL), and volume of distribution at steady state (V ss) were estimated using standard noncompartmental methods.
Statistical analyses
In the GLP toxicology study, descriptive statistical values (eg, group means and standard deviations) were calculated for numerical data using Excel (Microsoft Corporation, Redmond, Washington, USA). Further statistical analyses were performed with the SAS System, Version 8.1 (SAS Institute Inc, Cary, North Carolina, USA). Significant intergroup differences were evaluated using analysis of variance (ANOVA), followed by a multiple comparison test. The assumptions that permit the use of a parametric ANOVA were verified using the Shapiro Wilk test for normality of the data and Levene test for homogeneity of variance, with P ≤ .001 level of significance required for either test to reject the assumptions. If both assumptions were fulfilled, a single-factor ANOVA was applied, with animal grouping as the factor, utilizing a P ≤ 0.05 level of significance. If the parametric ANOVA was significant at P ≤ 0.05, Dunnett test was used to identify statistically significant differences between the control group and each test-article-treated group at the 0.05 level of significance. If either of the parametric assumptions was not satisfied, the Kruskal-Wallis nonparametric ANOVA procedure was used to evaluate intergroup differences (P ≤ 0.05). The Dunnett multiple comparison test was applied when this ANOVA was significant, again utilizing a significance level of P ≤ 0.05.
Results
Species Justification
Previous DNA sequence analysis of the IL-21 receptor from humans and mice showed 62% homology. 1 In contrast, DNA sequence analysis of the IL-21 receptor from humans and cynomolgus monkeys showed approximately 97% homology with only minor, mostly conservative changes in sequences (data not shown). Ex vivo studies with rIL-21 demonstrated dose-responsive and lineage-specific STAT3 phosphorylation in isolated PBLs from cynomolgus monkeys comparable with that seen with human PBLs (Figure 1). Similar responses in STAT3 phosporylation were also found in PBLs from rhesus monkeys and rabbits. No-to-minimal STAT3 phosphorylation was observed in PBLs from rodents or dogs. Binding affinities of rIL-21 to cloned cynomolgus IL-21R and human IL-21R were also found to be comparable (Kd = 68 ± 19 pM and 18 ± 6 pM, respectively). Because of the similarities in receptor sequences and of the biological activities between human and cynomolgus PBLs, cynomolgus monkeys were selected as the model species for safety studies prior to the initiation of clinical investigations of rIL-21.
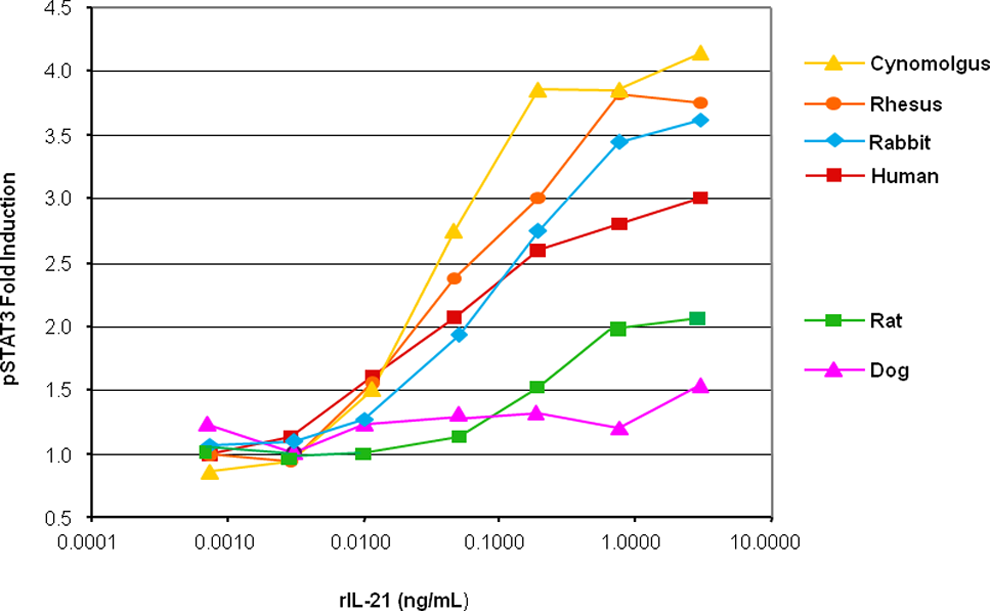
STAT3 phosphorylation (pSTAT3) in cynomolgus monkey, rhesus monkey, rabbit, human, rat, and dog peripheral blood lymphocytes in response to rIL-21. Y-Axis indicates fold induction of pSTAT3 from baseline and x-axis indicates rIL-21 concentration in nanogram per milliliter. rIL-21 indicates recombinant human interleukin-21; STAT3, signal transducer and activator of transcription 3.
Range-finding and GLP Toxicology Studies
Results of the GLP toxicology study are also the representatives of those from the dose range-finding studies; thus results from the GLP toxicology study are detailed first, followed by additional findings from the dose range-finding studies.
Clinical Observations
Slight elevations (1.1°C > prestudy values on average) in body temperatures were noted in cynomolgus monkeys receiving the high dose of 0.5 mg/kg rIL-21. Elevations in body temperature were most prevalent following the first dosing period and progressively diminished thereafter. No elevations in body temperature occurred during the fourth dosing period.
At the end of each dosing period, food consumption tended to be decreased for cynomolgus monkeys that received doses of 0.5 mg/kg rIL-21. Body weights were decreased by up to 14% from prestudy values in 4 of the 12 monkeys administered 0.5 mg/kg IL-21 for 4 cycles over 7 weeks. The nadir in body weight occurred at the end of dosing for the third treatment cycle (day 34); body weight for these animals began to increase during the fourth treatment cycle and continued toward baseline through the recovery period. Remaining animals treated with 0.5 mg/kg rIL-21 maintained a body weight that was ±5% of their prestudy value, which was similar to body weight fluctuations in animals in the lower dose groups and controls.
Clinical Pathology
Hematology
Dose- and time-dependent decreases in erythrocyte indices (ie, cell count, hemoglobin concentration, and hematocrit) and platelet counts were observed (Figure 2). Although an aggressive blood draw schedule resulted in a noticeable decrease in erythrocyte numbers in all animals, the decrease was greater in monkeys given doses of ≥0.1 mg/kg compared with controls.
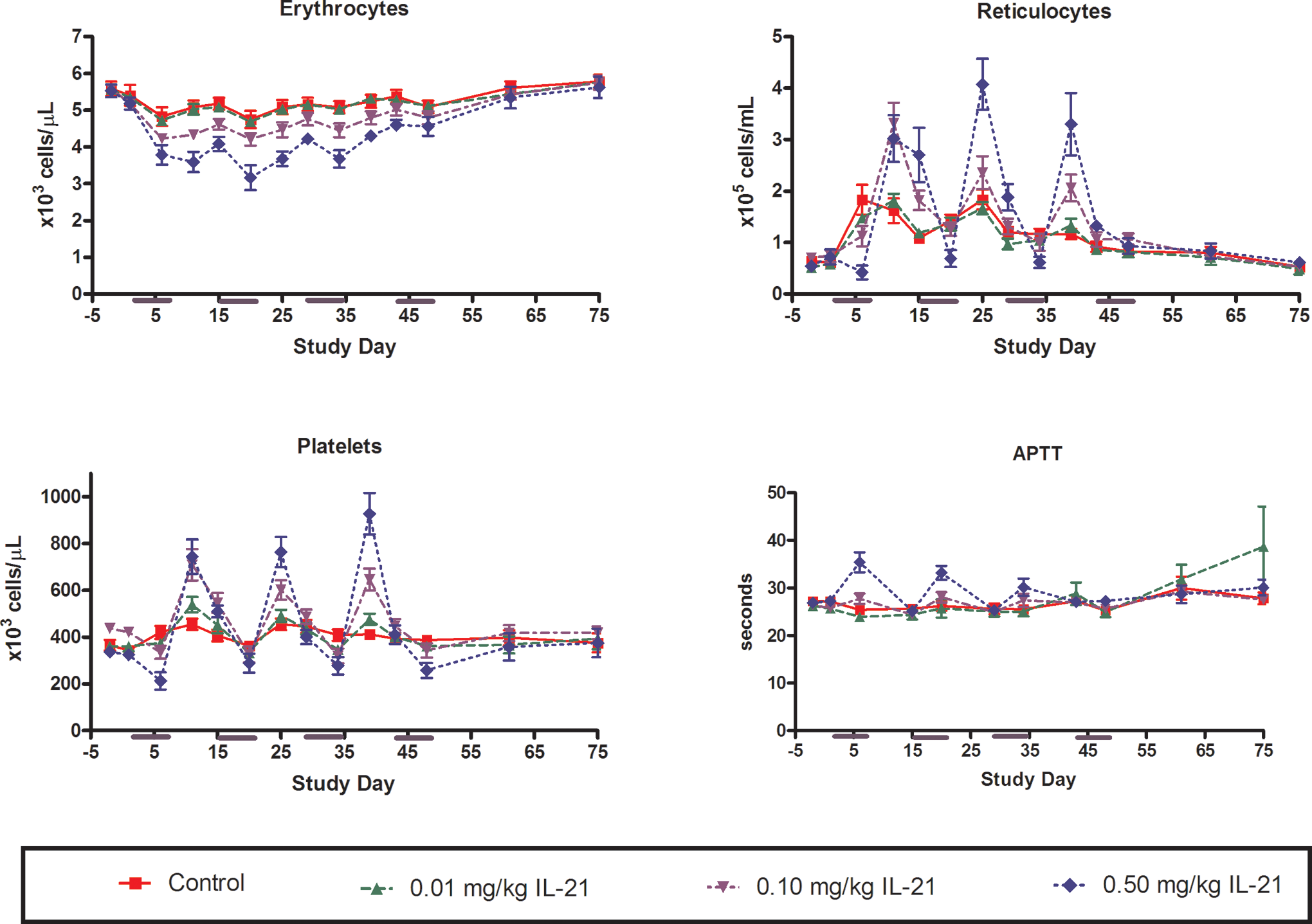
Changes in erythrocyte, reticulocyte, and platelet numbers and in plasma APTT in response to periodic dosing with rIL-21 in cynomolgus monkey. The rIL-21 was given at 0 (red), 0.01 (green), 0.10 (lavender), or 0.50 (blue) mg/kg once daily on study days 1 to 5, 15 to 19, 29 to 33, and 43 to 47. Subgroups of monkeys were euthanized on study days 48, 55, and 75. Y-Axis indicates number of cells per microliter (erythrocytes, reticulocytes, and platelets) or seconds (APTT). X-Axis indicates study day. rIL-21 indicates recombinant human interleukin-21; APTT, activated partial thromboplastin time
A regenerative reticulocytosis and thrombocytosis occurred during the nondosing period of each cycle. Platelet counts generally exceeded baseline levels 6 days after the end of dosing and were near baseline levels 9 days after the completion of dosing (Figure 2). The most profound changes in erythrocyte indices occurred in the second treatment cycle and, for platelets, in the third cycle. Thereafter, the magnitude of change decreased, and by the fourth treatment cycle, erythrocyte indices and platelet counts for all animals, regardless of group, had recovered to near baseline levels.
An increased incidence of rouleaux formation was noted in blood smears obtained from animals given ≥0.1 mg/kg rIL-21, particularly at the end of the dosing period for each cycle. Erythrophagocytosis by monocytes was also observed on day 34 (24 hours after the completion of a third cycle) in a blood smear from 1 cynomolgus monkey given 0.5 mg/kg rIL-21.
A dose-related increase in monocyte number was also observed (see Flow Cytometry section below). Monocyte counts progressively increased during the dosing periods and returned to baseline levels during the nondosing periods.
Coagulation parameters
Fibrinogen concentrations were elevated in cynomolgus monkeys given ≥0.1 mg/kg rIL-21 during the first, second, and third dosing periods (Figure 3). Fibrinogen levels generally returned to baseline during the 9 nondosing days. Minimal fibrinogen response was noted during the fourth dosing period. Minor rIL-21-related prolongations in activated partial thromboplastin time (APTT) were also noted in cynomolgus monkeys given ≥0.1 mg/kg rIL-21 (Figure 2). The increase was most pronounced during the first and second dosing cycles and was diminished thereafter.
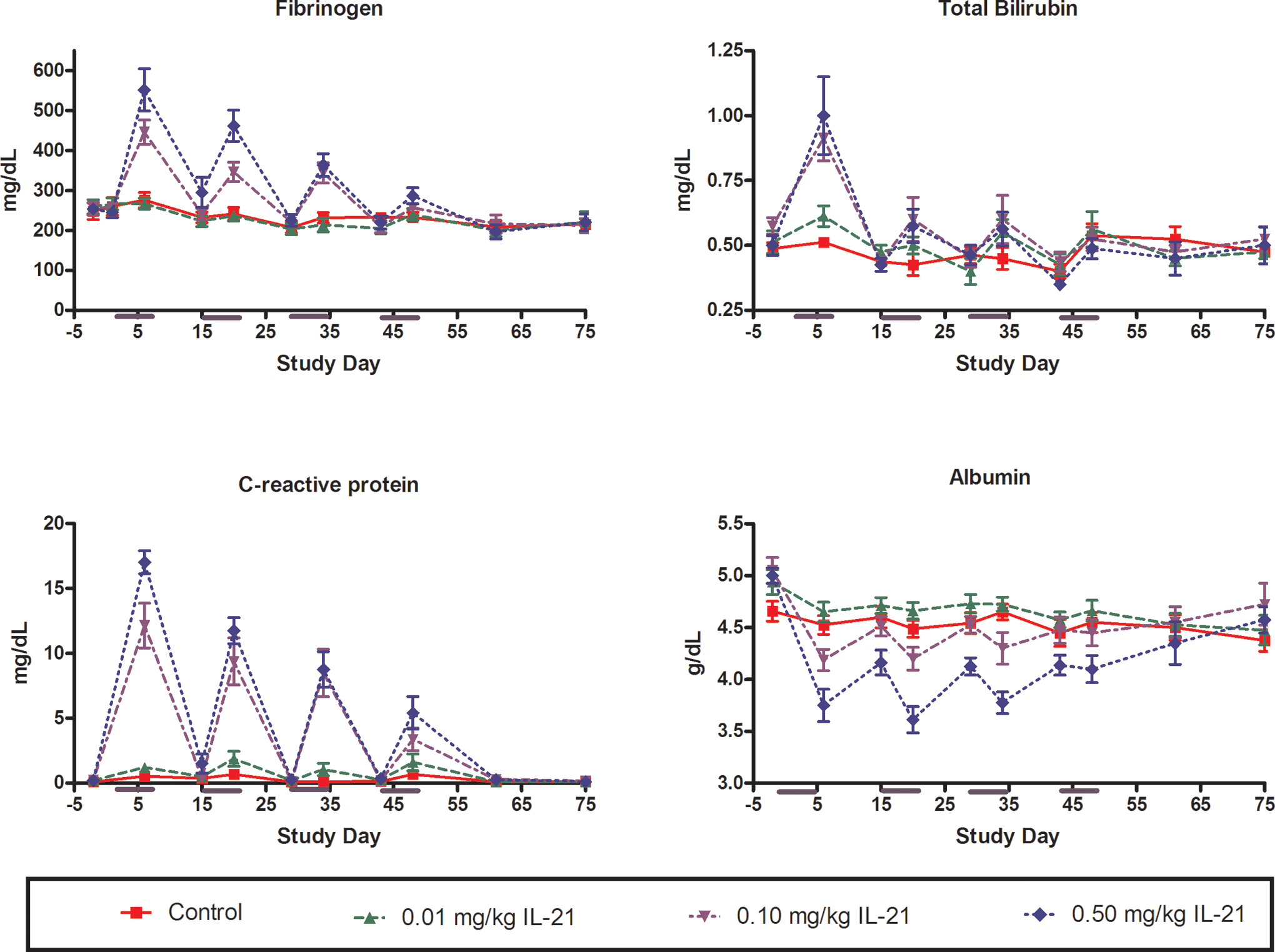
Changes in serum fibrinogen, total bilirubin, CRP, and albumin in response to periodic dosing with rIL-21 in cynomolgus monkey. The rIL-21 was given at 0 (red), 0.01 (green), 0.10 (lavender), or 0.50 (blue) mg/kg once daily on study days 1 to 5, 15 to 19, 29 to 33, and 43 to 47. Subgroups of monkeys were euthanized on study days 48, 55, and 75. Y-Axis indicates concentration in milligrams per deciliter (fibrinogen, total bilirubin, and CRP) or gram per deciliter (albumin). X-Axis indicates study day. rIL-21 indicates recombinant human interleukin-21; CRP, C-reactive protein.
Serum chemistry
Total bilirubin levels were elevated after the first dosing period in cynomolgus monkeys receiving ≥0.1 mg/kg rIL-21 (Figure 3). Slight elevations were also noted after the second and third dosing periods. Return to baseline levels occurred during the 9 nondosing days.
A dose-dependent elevation in CRP occurred in cynomolgus monkeys receiving ≥0.1 mg/kg rIL-21 (Figure 3). Peak elevation was noted 24 hours after completion of the 5-day dosing period. CRP levels returned to near baseline within the 9 nondosing days of each cycle. The greatest elevation in CRP levels occurred during the first cycle and elevations decreased in magnitude after each successive cycle.
Mild decreases in serum albumin (Figure 3) and calcium (Figure 4) occurred concurrently with alterations in CRP, and values returned to near-baseline within the nondosing period of each cycle. Serum globulin also slightly increased in a cyclic pattern; however, globulin levels did not return to baseline during nondosing periods (Figure 4).
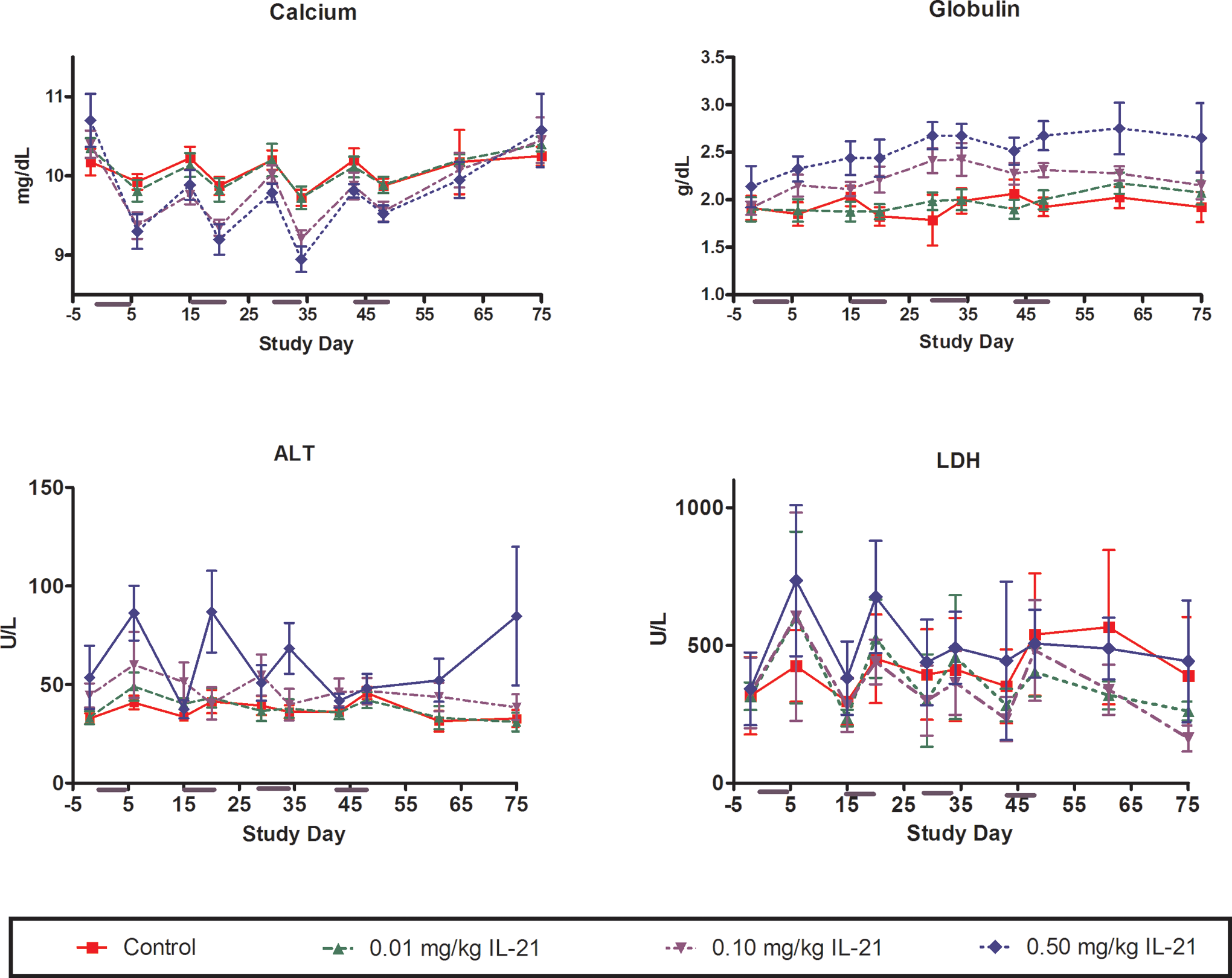
Changes in serum calcium, globulin, ALT, and LDH in response to periodic dosing with rIL-21 in cynomolgus monkey. The rIL-21 was given at 0 (red), 0.01 (green), 0.10 (lavender), or 0.50 (blue) mg/kg once daily on study days 1 to 5, 15 to 19, 29 to 33, and 43 to 47. Subgroups of monkeys were euthanized on study days 48, 55, and 75. Y-Axis indicates concentration in milligrams per decilieter (calcium), grams per deciliter (globulin) or Units per liter (ALT and LDH). X-Axis indicates study day. rIL-21 indicates recombinant human interleukin-21; LDH, lactate dehydrogenase; ALT, alanine transaminase.
Minimal and transient increases in serum alanine transaminase and lactate dehydrogenase were observed after dosing periods in approximately half of the cynomolgus monkeys receiving 0.5 mg/kg rIL-21 (Figure 4). Increases were generally 2 to 3 times the baseline values, but were rarely outside the normal range established by the laboratory performing the assays. The response was greatest during the first and second dosing cycles and progressively decreased thereafter. Serum levels returned to baseline during the nondosing periods.
Serum cytokines
Changes in serum levels of IL-1β, IL-6, TNF-α, or IFN-γ were not observed in response to rIL-21 administration.
Flow cytometry
Immunophenotyping analyses revealed rIL-21-associated changes in activated B lymphocytes (CD20+/CD86+), naive monocytes (CD14+/CD64−), and activated monocytes (CD14+/CD64+) (Figure 5). No consistent rIL-21-associated changes were observed in the number of total T-lymphocytes, T-helper lymphocytes, or T-cytotoxic/suppressor lymphocytes when compared with baseline values.
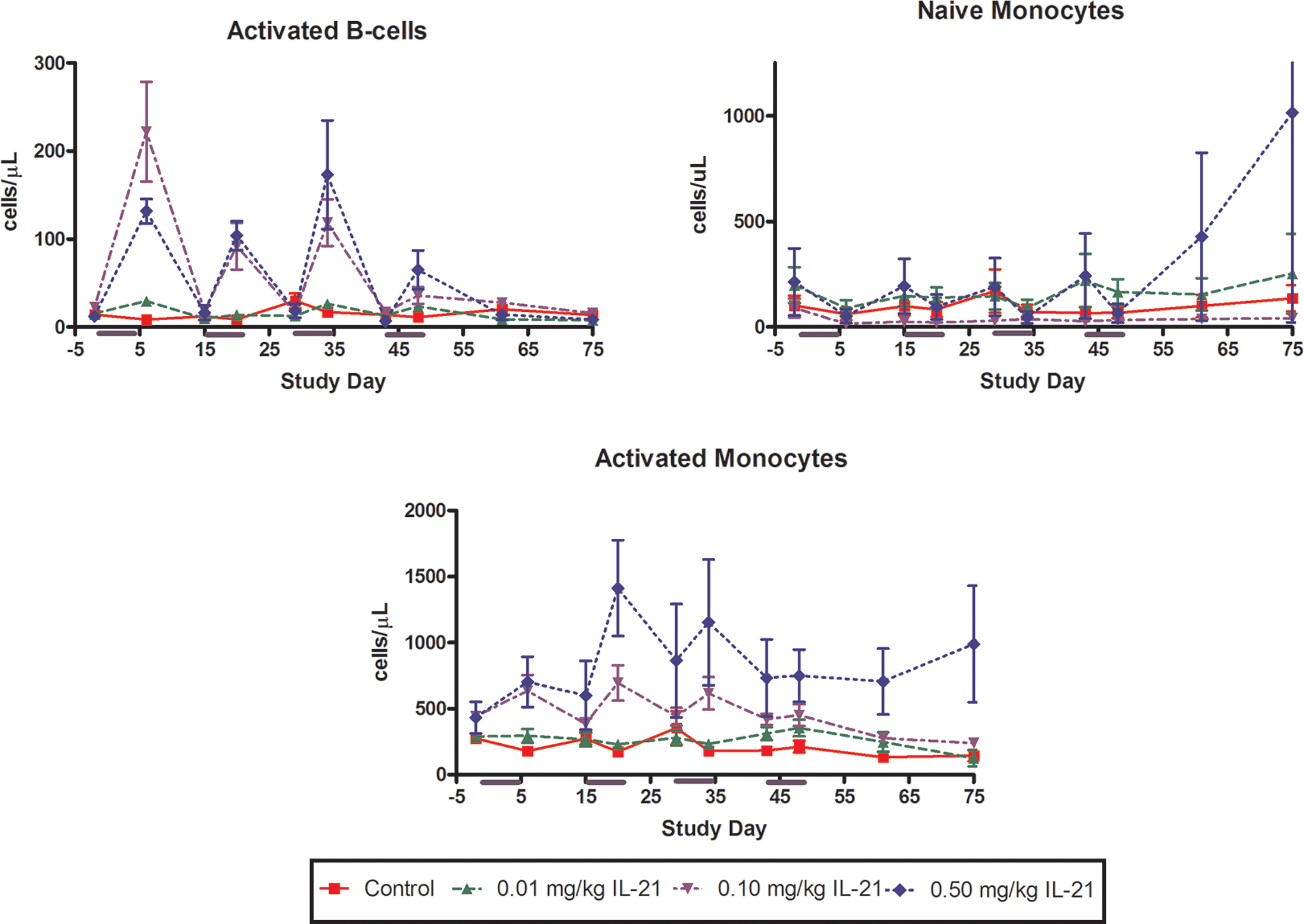
Changes in the number of cynomolgus monkey activated B cells and naive and activated monocytes in response to periodic dosing with rIL-21. The rIL-21 was given at 0 (red), 0.01 (green), 0.10 (lavender), or 0.50 (blue) mg/kg once daily on study days 1 to 5, 15 to 19, 29 to 33, and 43 to 47. Subgroups of monkeys were euthanized on study days 48, 55, and 75. Y-Axis indicates numbers of cells per microliter and x-axis indicates study day. rIL-21 indicates recombinant human interleukin-21.
Activated B lymphocyte numbers were increased at the end of the first 3 rIL-21 dosing periods (Figure 5). The magnitude of increase was greater after the first 3 dosing periods than after the fourth dosing period, and was greater in the 0.1 mg/kg dose group than the 0.5 mg/kg dose group after the first treatment period. Activated B cell numbers returned to or slightly below baseline values after completion of each nondosing period.
Numbers of naive monocytes were minimally decreased in all dose groups at the end of each treatment cycle (Figure 5). However, activated monocyte numbers were increased at the end of the second, third, and fourth treatment cycles in cynomolgus monkeys given 0.1 or 0.5 mg/kg rIL-21. Naive monocyte numbers returned to near baseline values during the nondosing interval in all rIL-21-treated groups. Activated monocyte numbers remained slightly increased during the second and third nondosing intervals in animals given 0.1 and 0.5 mg/kg rIL-21.
Anatomic Pathology
Gross and microscopic findings were identified in lymphoid tissues and bone marrow of monkeys necropsied at the end of the treatment period (day 48). At necropsy, the spleens of 3 of the 4 animals given 0.5 mg/kg rIL-21 were noted to be enlarged. Microscopic evaluation of these spleens revealed mild or marked hemosiderin accumulation in the red pulp and/or mild or moderate plasmacytosis. Hemosiderin was observed in the spleen of 2 control animals but was graded minimal in severity. Hemosiderin accumulation was also noted in the cytoplasm of histiocytes in lymph nodes from the majority of animals treated with rIL-21, but not in lymph nodes from control animals. The rIL-21 dose-dependent minimal-to-moderate germinal center atrophy and plasmacytosis was observed in one or more lymph nodes of animals treated with rIL-21 (Figure 6A and B; Table 1). Minimal-to-mild lymphoid depletion in the paracortex of lymph nodes was observed in 3 of the 4 animals given 0.5 mg/kg rIL-21, and mild-to-moderate lymphoid depletion was observed in the periarteriolar lymphoid sheath in the spleen of 1 animal given 0.1 and 1 animal given 0.5 mg/kg rIL-21. Thymic involution was observed in animals from all groups (Table 1), and a dose-related increase in severity (from minimal to mild in controls to mild-to-moderate in animals given 0.5 mg/kg rIL-21) of involution was noted. Minimal extramedullary hematopoiesis (EMH) was observed in a single lymph node from each of 2 control animals, while minimal-to-moderate EMH was noted in one or more lymph nodes from each of 2 animals given 0.1 mg/kg and 1 animal given 0.5 mg/kg rIL-21. Erythroid hypercellularity was noted in the bone marrow in 1 animal given 0.1 and 2 animals given 0.5 mg/kg rIL-21.
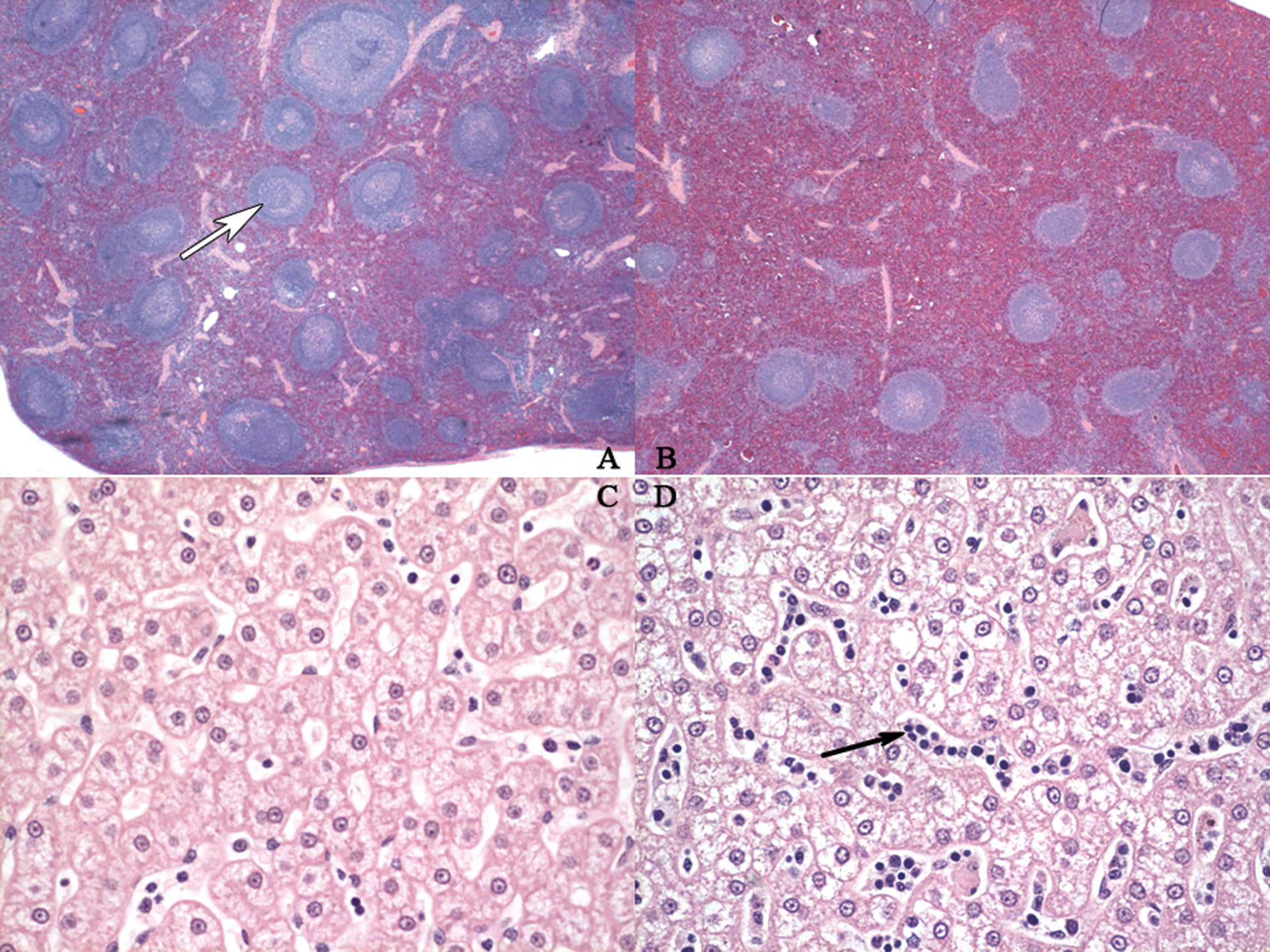
Microscopic findings in spleen and liver from control and rIL-21-treated cynomolgus monkeys. A. Control cynomolgus spleen. Numerous germinal centers (white arrow) are present. B. Spleen from cynomolgus monkey given with 0.5 mg/kg IL-21 for four 5-day dosing cycles; the first 3 cycles were separated by 9 nondosing days and necropsied 1 day following the final rIL-21 dose. Germinal centers are reduced in size or absent from follicles. C. Liver from control cynomolgus monkey. Lower numbers of mononuclear cells are present in the hepatic sinusoids. D. Liver from cynomolgus monkey given 5 consecutive daily doses of 0.06 mg/kg rIL-21 and necropsied 3 days following final rIL-21 dose. Infiltrates, predominately mononuclear cell, are present in the sinusoids (black arrow). All images were hematoxylin and eosin stained with a magnification of ×2 (A, B) and ×40 (C, D). rIL-21 indicates recombinant human interleukin-21.
Histopathology Findings, Day 48
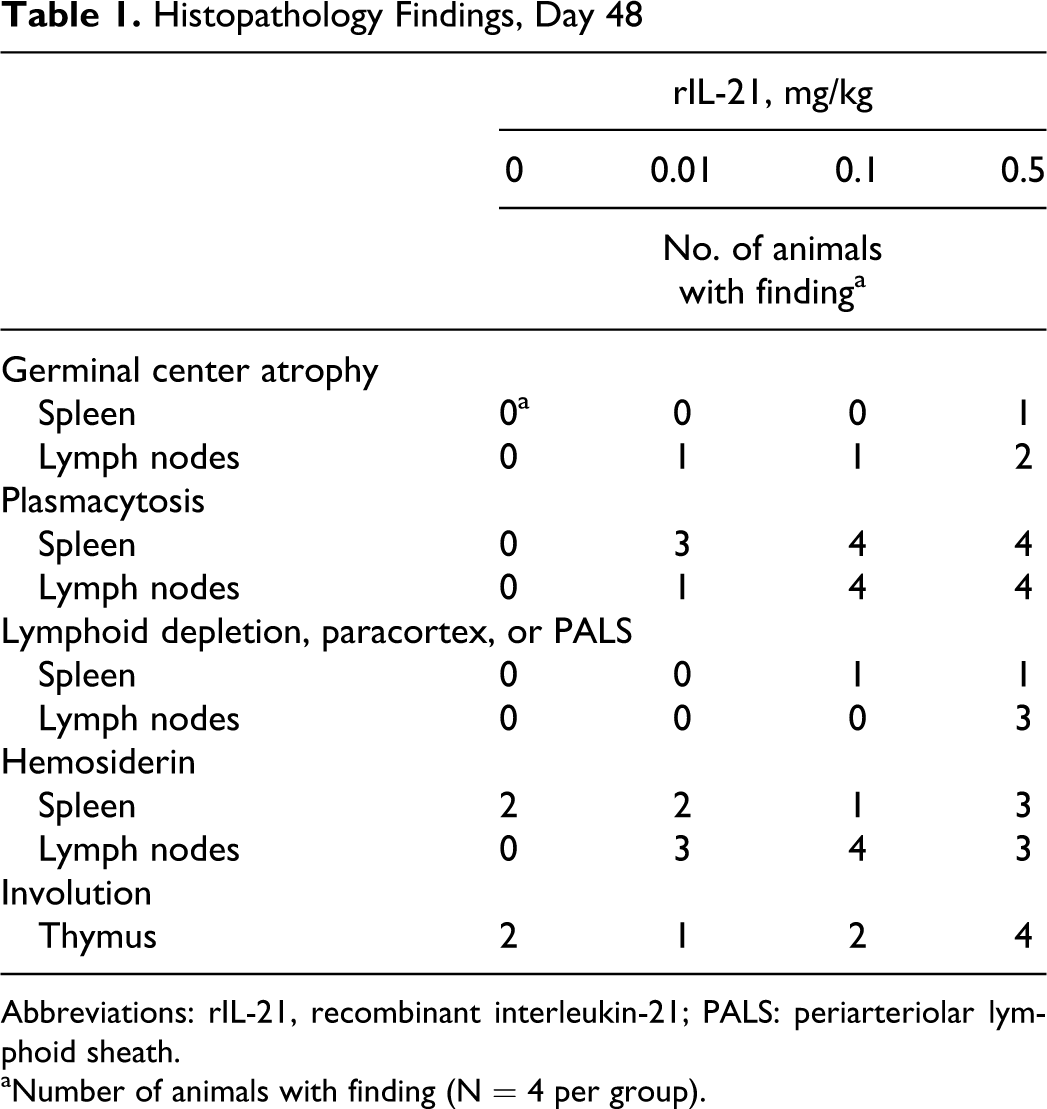
Abbreviations: rIL-21, recombinant interleukin-21; PALS: periarteriolar lymphoid sheath.
aNumber of animals with finding (N = 4 per group).
By the end of the recovery period on day 75, lymphoid alterations in spleen and lymph nodes were not observed in animals treated with 0.1 mg/kg rIL-21, but residual lymphoid atrophy was observed in the lymph nodes of 2 animals treated with 0.01 mg/kg rIL-21 and 3 animals given 0.5 mg/kg IL-21. Plasmacytosis in the spleen had largely resolved by the end of the 4 nondosing weeks. Individual animals within each of the groups, including controls, had minimal-to-moderate plasmacytosis in lymph nodes at the end of recovery. Hemosiderin accumulation was found in lymph nodes and/or spleen from one or more monkeys from all groups.
Immunohistochemical staining confirmed a dose-dependent increase in plasma cells in the marginal zone and periarteriolar sheath of the spleen and in the medullary cords of the mesenteric and inguinal lymph nodes on day 48. Staining was similar among the groups for CD20+ (B lymphocytes), CD4+ (T-helper lymphocytes), and CD11c+ (NK cells, monocytes, cytotoxic T lymphocyte subset, and lymphoid dendritic cells). On day 75, the staining patterns for all markers were similar between control and IL-21-treated animals.
Additional Findings From Dose Range-Finding Studies
During the GLP toxicology study described above, blood samples for clinical pathology were collected prior to the first dose of each 5-day dosing period and 1 day after the fifth dose. In contrast, blood was drawn more frequently during the dosing period in the dose range-finding studies. Overall findings in the dose range-finding studies were similar to those in the GLP toxicology study, including the finding of hemophagocytosis by monocytes in a blood smear from an animal given 1.5 mg/kg rIL-21 IV. Hematology from the dose range-finding studies showed a 56% decrease in lymphocyte numbers 6 hours following the initial dose of 0.5 or 1.5 mg/kg rIL-21, when compared with a 15% decrease in controls. Lymphocyte numbers returned to near baseline between days 5 and 11. On flow cytometry, this corresponded to decreases in the numbers of total T lymphocytes (CD3+), T-helper lymphocytes (CD3+CD4+), and T-cytotoxic/suppressor lymphocytes (CD3+/CD8+). Inflammatory cell infiltrates were noted in sinusoids in the livers from all monkeys treated with rIL-21 for 3 to 5 days and necropsied 1 to 3 days following the final dose. The infiltrates in the liver sinusoids were comprised of mononuclear cells accompanied by lesser numbers of polymorphonuclear cells (Figure 6C and D). Infiltrates were not noted in the sinusoids of livers of 3 animals treated with 3 mg/kg rIL-21 for 3 days, and then given a 14-day recovery period.
Pharmacokinetics and Antigenicity
The GLP toxicology study in cynomolgus monkeys provided the most robust data set for the evaluation of IV rIL-21 pharmacokinetics and antigenicity. At the 0.01 mg/kg dose level, there were only a limited number of samples with quantifiable rIL-21 concentrations and further pharmacokinetic evaluation could not be conducted in this dose group. A larger data set was available from 0.1 and 0.5 mg/kg dose groups, and this allowed in the determination of pharmacokinetic parameters.
Pharmacokinetic analyses of day 1 data resulted in the following mean estimates for the 0.1 and 0.5 mg/kg rIL-21dose groups, respectively: C 0 of 0.22 and 0.84 µg/mL, t 1/2,λz of 0.43 and 0.81 hours, CL of 1257 and 1205 mL/h per kg, and a V ss of 684 and 1530 mL/kg (Table 2). The dose-normalized AUCinf was 0.90 and 1.4 (h/μg per mL)/(mg/kg), at the 0.1 and 0.5 mg/kg dose level, respectively. The pharmacokinetics of rIL-21 in monkeys appeared to be dose-dependent in the evaluated dose range. Although rIL-21 pharmacokinetic parameters were not generated at the end of cycle 1 on day 5, they were generated after the 5th dose of cycle 2 (day 19). There was no appreciable increase in exposure on day 19 relative to day 1 (Table 2). While the development of anti-rIL-21 antibodies (Table 3) complicates interpretation, these data indicate that there was no accumulation upon repeat dosing of rIL-21. This is consistent with the rapid clearance of rIL-21 from the circulation. All of the animals that received 0.1 mg/kg and 3 of the 8 animals that received 0.5 mg/kg rIL-21 had a marked decrease in exposure by day 47.
Pharmacokinetic Parameters
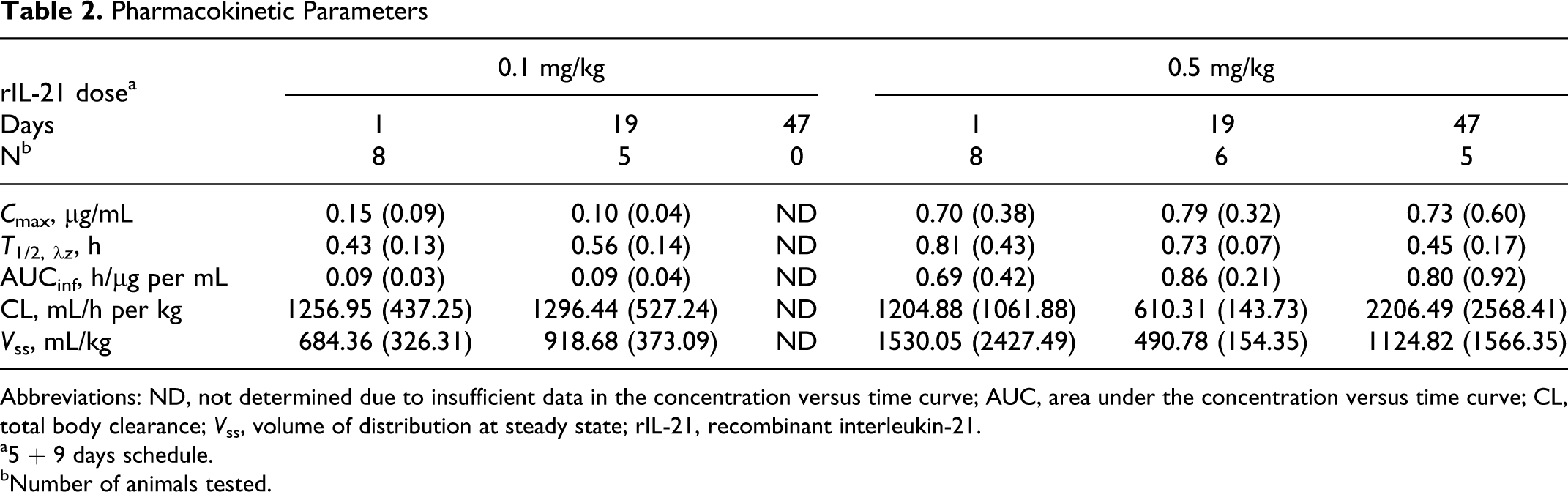
Abbreviations: ND, not determined due to insufficient data in the concentration versus time curve; AUC, area under the concentration versus time curve; CL, total body clearance; V ss, volume of distribution at steady state; rIL-21, recombinant interleukin-21.
a5 + 9 days schedule.
bNumber of animals tested.
Prevalence of Anti-rIL-21 Antibodies
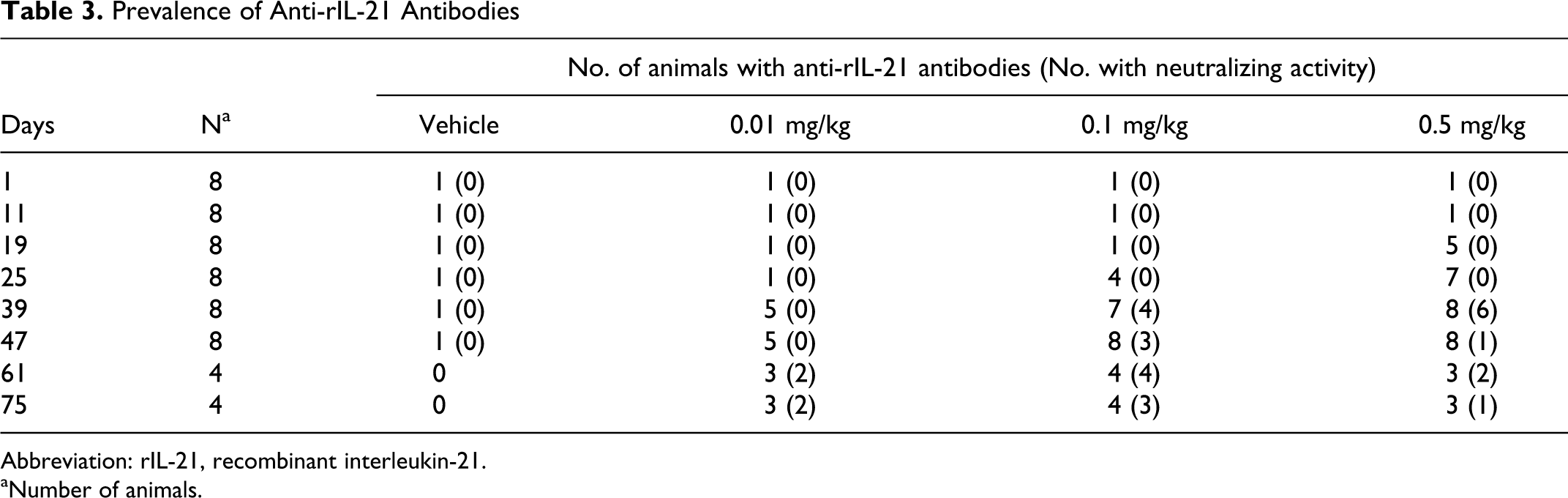
Abbreviation: rIL-21, recombinant interleukin-21.
aNumber of animals.
The prevalence of anti-rIL-21 antibodies appeared to be influenced by dose. Animals that received higher doses of rIL-21 developed positive titers earlier. Half of the animals that received doses of 0.01, 0.1, or 0.5 mg/kg developed anti-rIL-21 antibodies by days 39, 25, and 19, respectively (Table 3). By day 47 (the final dose of the fourth cycle), 5 of the 8 animals dosed with 0.01 mg/kg had positive titers and all 8 animals that received 0.1 or 0.5 mg/kg rIL-21 were positive for antibodies. Neutralizing activity was found by day 39 in over half of the antibody positive animals dosed with either 0.1 or 0.5 mg/kg rIL-21.
Safety Pharmacology
Safety pharmacology evaluations, including ECG evaluation and heart rate and blood pressure measurements, were incorporated into the GLP toxicology study. The overall status of the gastrointestinal tract, kidneys, respiratory system, and central nervous system were monitored via clinical signs, serum chemistry, pulse oximetry, and pathology (see above). The cardiovascular evaluations, conducted on week 1 and days 1, 5, 19, and 47 showed rIL-21 to be well-tolerated and generally without effect on arterial blood pressure, heart rate, lead II ECG parameters, or core body temperature.
Discussion
The cynomolgus monkey was chosen as a relevant species for preclinical studies based on human-cynomolgus IL-21 receptor homology and similar STAT3 phosphorylation responses in human and cynomolgus lymphocytes following exposure to rIL-21. The treatment schedule in dose range-finding and GLP toxicology studies was designed to support phases I and II clinical trials and was patterned after that used for IL-2 (5 consecutive days of treatment followed by a 9-day rest period). In preclinical studies, rIL-21-related effects were observed following IV doses ≥0.01 mg/kg, and potentially adverse effects occurred at doses ≥0.1 mg/kg. The rIL-21-related effects included changes indicative of an acute phase response (eg, increased body temperature, serum CRP, and fibrinogen; decreased albumin and calcium) and myeloid and erythroid alterations in the blood and tissues. The effects were most pronounced after the first dosing period and by the fourth dosing period were diminished or absent, most likely due to the formation of anti-rIL-21 antibodies. Evidence of effect reversability was observed during nondosing periods.
Timing of sample collection was critical for detecting changes in blood and tissues. Blood samples were collected prior to the first and the day after the last rIL-21 dose in each cycle of the GLP toxicology study, and significant changes in PBL numbers were not observed. However, an approximately 50% decrease in the numbers of circulating lymphocytes was observed 6 hours after the initial dose of rIL-21 in the second dose range-finding study, with a gradual return to near baseline by the end of the dosing period. This rapid drop and recovery of lymphocyte numbers is suggestive of redistribution of lymphocytes between the blood and tissues, rather than actual destruction of lymphocytes. Inflammatory cell infiltrates primarily comprised of mononuclear cells were observed in hepatic sinusoids of rIL-21-treated animals 1 day following the third or fifth dose of rIL-21 in the first dose range-finding study. However, similar infiltrates were not observed in the livers from animals necropsied 1 day following the final rIL 21 dose of the fourth dosing cycle of the GLP toxicology study. The lack of infiltrates in the toxicology study may be related to the overall decrease in response to rIL-21 (most likely associated with neutralizing antibodies) observed in the fourth treatment cycle. Timing of sampling may also have played a role in the lack of detectable serum cytokine levels in the GLP toxicology study. The time points selected for blood sample collection were based upon changes in peripheral leukocytes observed in the dose range-finding studies, and these time points may not have been optimal for cytokine detection.
Fever and increased acute phase proteins have previously been noted in preclinical studies of cytokine-based therapies. 11 Activation of monocytes, B cells, and possibly T cells by rIL-21, in association with an influx of these cells into liver, is a probable factor in the acute phase reaction observed with rIL-21. Decreases in red blood cell and platelet numbers are also most likely due to an immune-mediated mechanism. To our knowledge, IL-21 receptor expression on erythrocytes or platelets has not been reported in the literature, and in-house tissue binding studies have not identified bone marrow precursor cells as targets for IL-21 (data not shown). This suggests that the observed decrease in erythrocyte and platelet numbers is unlikely to be a direct effect of rIL-21. However, erythrocytes and hemosiderin were observed within phagocytic cells in the spleen and lymph nodes of rIL-21-treated monkeys as well as in the cytoplasm of an occasional circulating monocyte. Therefore, it is possible that loss of red cells was secondary to macrophage activation and erythrophagocytosis. Destruction of red cells via erythrophagocytosis is also a likely cause of the observed increase in serum bilirubin. Changes in the red cell membrane or adhesion of proteins such as fibrinogen to the red cell surface may have made the cells more susceptible to phagocytosis. Macrophage activation with erythrophagocytosis has been previously proposed as the mechanism of action for the decrease in erythrocytes observed after the administration of IL-3 and IL-18. 12 –15
Decreases in erythrocyte numbers have been reported after treatment with other cytokines. Antierythrocyte antibodies, bone marrow suppression secondary to IFN-γ induction, and hemodilution secondary to increased plasma volume were proposed as causes of anemias associated with IL-2, IL-12, and IL-6 administration. 16 –19 Formation of antierythrocyte antibodies would be expected to develop later in the treatment period and result in a sustained decrease in red cells, whereas decreased erythrocyte numbers were in evidence by day 5 dosing of rIL-21 and increased erythrocyte numbers were observed after the cessation of treatment. Increased reticulocyte counts in rIL-21-treated animals during each rest phase and the presence of erythroid hypercellularity in the bone marrow and EMH in lymph nodes suggested ample capacity for a regenerative response. Finally, there were no indications of hemodilution or plasma volume expansion with rIL-21.
IL-21 has been reported to play a role in B cell differentiation. Specifically, IL-21 induces the expression of Blimp-1, a master regulator of plasma cell differentiation, and Bcl-6, which may play a role in the differentiation of germinal center cells. 20 It is, therefore, possible that the germinal center atrophy observed in the current studies is a consequence of rIL 21-driven B cell differentiation. Lymphoid depletion was also noted in T-cell zones of the spleens (periarteriolar lymphoid sheath) and lymph nodes (paracortex). This may be due to continued T-cell activation and migration from the T-cell areas to lymphoid follicles or other tissues. 21,22 Thymic involution was observed in both control and rIL-21-treated monkeys. Involution is a normal age-related change; however, a dose-related increase in severity was noted. Because the thymus is sensitive to stress-induced lymphoid depletion, it is possible that the involution in rIL-21-treated animals is, at least in part, related to the treatment. 23
An unexpected finding was an increase in activated B cell numbers in the 0.1 mg/kg rIL-21 treatment group that was greater than or equal to the response observed in the 0.5 mg/kg group after the first and second treatment cycles, respectively. This might be an example of the response expected with receptor dimerization and self-antagonism at a high ligand concentration. 24 This phenomenon was not confirmed, as activated B cells were not evaluated in the rIL-21 dose range-finding studies.
The rIL-21 demonstrated dose-dependent pharmacokinetics following IV dosing. As expected, based on the short t 1/2,λz (0.4-0.8 hours), no accumulation was seen with repeat daily dosing.
Cynomolgus monkeys in these studies produced antidrug antibodies following treatment with rIL-21, including antibodies with neutralizing activity. Development of anti-rIL-21 antibodies corresponded to a decrease in pharmacodynamic response; however, the effect of anti-rIL-21 antibodies on rIL-21 exposure varied with individuals and was dose-dependent. After 3 or 4 treatment cycles, antibody formation and decreased exposure for many animals were associated with diminished modulation of peripheral leukocytes, a decrease in the magnitude of the induced acute phase response, and less impact on erythrocyte and platelet counts. Formation of antibodies is a common issue in evaluating human-specific proteins in nonhuman primates. 13,25,26 However, antigenicity of human proteins in monkeys has rarely been predictive of antigenicity in humans. 27
Because IL-21 is in the same cytokine family as IL-2, the potential for vascular leak was of particular concern in the preclinical studies. Vascular leak syndrome has been a primary cause of toxicity and mortality with IL-2 in both nonclinical and clinical trials. 28,29 Pleural effusions, ascites, and increased organ weights were found at necropsy of cynomolgus monkeys treated with IL-2. Histopathology of tissues from these animals revealed lymphocytic and eosinophilic infiltrates in multiple tissues, thickening of alveolar septa in the lungs, and alveolar edema. In humans, signs of vascular leak appear 3 to 4 days after the initiation of IL-2 administration and include decreased serum albumin, interstitial edema, and modest weight gain. Cardiovascular changes, such as tachycardia and hypotension, may also result from intravascular hypovolemia secondary to fluid leakage. 30 In the current studies, there were no indications of fluid extravasation, eosinophilic infiltrates, or increased organ weights as described in cynomolgus monkeys treated with rIL-2 nor were there cardiovascular changes as noted in humans with vascular leak. Vascular leak syndrome has also not been reported to date in human patients receiving rIL-21 in clinical trials. 31 –33
Clinical trials have shown rIL-21 to be generally well-tolerated in humans. 31 –33 Commonly reported adverse effects have included influenza-like symptoms, fever, pruritis, and skin rash. The most frequently reported dose-limiting toxicities included grade 3 neutropenia, hepatic enzyme elevations, and hyponatremia. Less frequently reported were thrombocytopenia, leukopenia, hypophosphatemia, hyperkalemia, elevated bilirubin, and increased APTT. Changes were transient and values returned to baseline within 3 weeks after cessation of rIL-21 dosing.
Responses to rIL-21 by humans and cynomolgus monkeys were similar but not identical. Both species showed signs consistent with an acute phase response. These included elevations in body temperature and changes in acute phase proteins. 34,35 In both the species, lymphocyte counts decreased to approximately 50% of baseline during dosing, followed by recovery to numbers near or above baseline. 31 Increased APTT was occasionally reported in human trials, while minimally increased times were observed in animals from the GLP toxicology study. Anemia, one of the most significant features of the cynomolgus monkey response to rIL-21, was not a common finding in clinical trials. In cynomolgus monkeys, minimal (2-3 times baseline) elevations were observed in hepatic enzymes; these elevations were rarely, and barely, outside the upper limit of normal (ULN). However, transient grade 3 (>5-20 times ULN) hepatic enzyme elevations were observed in approximately 20% of patients receiving 30 µg/kg rIL-21 in phases I and II clinical trials. This finding emphasizes the need to keep minor changes in mind when evaluating preclinical studies conducted with nonhuman primates for clues to possible toxicities in humans.
In summary, cynomolgus monkey and human IL-21 receptors share high sequence homology and comparable binding affinity, and STAT3 responses were comparable between cynomolgus and human lymphocytes upon exposure to rIL-21. The biologic responses of cynomolgus monkeys to rIL-21 administration were similar, although not identical, to responses reported in humans. 31 –33,35 Significantly, anemia, a consistent feature of the macaque response to rIL-21, was uncommon in clinical trials. The effects of rIL-21 were readily reversible during nondosing periods or upon completion of dosing in cynomolgus monkeys, as has also been reported in rIL-21 clinical trials. Therefore, these studies demonstrate that the cynomolgus monkey is a suitable species for exploring rIL-21 biology. However, development of anti-rIL-21 antibodies may limit the usefulness of cynomolgus monkeys in long-term studies.
Footnotes
Acknowledgments
We gratefully acknowledge Steve Levine, Lucinda Yen, Jeff Ellsworth, Kien Khuu, Mike Templin, Kirk Van Ness, Jennifer Visich, Linda Zuckerman, Jeremy Freeman, HongPing Ren, Julie Hill, Jane Gunther, Richard J. Early, and Darren Warren for their contributions to the design and conduct of these studies. We also thank Kate Loughney for editorial assistance.
Kimberly S. Waggie and Matthew S. Holdren contributed equally to this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: supported by ZymoGenetics, Inc, a Bristol-Meyers Squibb Company.
