Abstract
Endocrine-disrupting chemicals (EDCs) play an important role in the incidence of type-2 diabetes. Di-2-ethyl hexyl Phthalate (DEHP) is one of the endocrine-disrupting chemicals used as a plasticizer to impart flexibility and softness to plastic-containing materials. Mono-2-ethylhexyl Phthalate (MEHP), a DEHP’s primary metabolite, is preferentially absorbed once metabolized. A previous study from our laboratory showed that DEHP and MEHP altered the key proteins such as insulin receptor (INSR) and glucose transporter-4 (GLUT4) in L6 myotubes. In a sequel to the previous study, the present study hypothesized that DEHP and its metabolite MEHP may alter the Insr and Glut4 gene expression in L6 myotubes. Therefore, to find out the molecular mechanism behind the decreased INSR and GLUT4 protein levels in the previous study, the direct effect of DEHP and its metabolite MEHP in regulating Insr and Glut4 gene transcription in L6 myotubes was studied. The L6 myotubes were exposed to 50 and 100 μM DEHP and MEHP for 24 h, followed by insulin stimulation for 20 min. We observed decreased Insr and Glut4 mRNA levels in DEHP and MEHP-treated groups. Western blot data showed decreased protein levels of MEF2A and MyoD in treated groups. ChIP assay detected a decreased association of MEF2A and MyoD to the Glut4 gene promoter and HMGA1 to the Insr gene promoter. The study revealed that DEHP and MEHP diminished the Insr and Glut4 gene expression through weakened interaction of their transcription factors on the respective promoter.
Introduction
Worldwide, it is estimated that 537 million individuals have diabetes, and the incidence of type-2 diabetes is increasing at an alarming rate. 1 Type-2 diabetes (T2D) is a chronic metabolic disorder characterized by insulin resistance (IR) and pancreatic β- cell dysfunction associated with microvascular and macrovascular complications.2–4 Apart from classical risk factors, Endocrine disrupting chemicals (EDCs) play a vital role in developing T2D. 5 Di-2-ethyl hexyl phthalate (DEHP) is an ester of phthalic acid used to impart structural flexibility and pliability to polyvinyl chloride (PVC)-containing material. They are found in cosmetics, medical devices, industrial paints, solvents, and food packaging materials.6,7 Since DEHP is not covalently bound to the matrix, it can easily leach into the air, food, and water upon repeated usage. Due to its widespread usage, humans are constantly exposed to DEHP through ingestion, inhalation, and dermal exposure.8,9 DEHP absorbed in the body is rapidly metabolized by lipases and esterases to mono-2-ethylhexyl phthalate (MEHP) and 2-ethylhexanol.10,11
Numerous epidemiological studies have reported that increased urinary DEHP metabolites were associated with the incidence of IR, T2D, and obesity in childhood and adolescent period.12–17 Consistent with the epidemiological studies, experimental studies demonstrated the negative association of DEHP in the development of glucose intolerances, IR, and T2D.18–20 It is shown that DEHP mediates its toxic effect through the primary metabolite, MEHP. The toxicity potency of MEHP is higher than DEHP.11,21,22 Studies from our laboratory have shown that DEHP significantly reduces the gene expression of Insr, INSR and GLUT4 protein levels associated with impaired glucose oxidation in Chang liver cells and L6 myotubes.23,24 DEHP exposure alters the expression of genes involved in hepatic fatty acid oxidation and disrupts lipid homeostasis in the liver.25,26 Gestational and lactational exposure to DEHP showed impaired pancreatic β-cell function, glucose and insulin intolerances, hyperglycemia, hyperinsulinemia, and altered insulin signaling molecules in F1 offspring, leading to the development of T2D.19,27 MEHP exposure increases the incidence of obesity by promoting adipocyte differentiation and increase in adipogenic markers such as adipocyte protein2 (aP2), lipoprotein lipase (LPL), and peroxisome proliferator-activated receptor gamma γ (PPARγ) were observed. 28 We analyzed the direct effect of DEHP and its primary metabolite MEHP on insulin signaling and GLUT4 translocating molecules in L6 myotubes. DEHP and MEHP altered the insulin signaling and GLUT4 translocating molecules, decreasing the cytosolic and plasma membrane GLUT4 protein. DEHP and its metabolite MEHP exert a toxic effect on GLUT4 protein and translocation of GLUT4 from the cytosol to the membrane; the toxic potency of MEHP is higher than DEHP in L6 myotubes. 29
Skeletal muscle is the primary tissue responsible for insulin-mediated glucose disposal. 30 Skeletal muscle accounts for about 70–80% of glucose disposal and helps in glucose homeostasis. 31 Insulin binds to the INSR on the surface of muscle cells, triggering the autophosphorylation of the INSR and activating the signaling cascade necessary for the translocation of GLUT4 protein from the cytosol to the membrane. 30 We observed decreased INSR and GLUT4 protein levels upon DEHP and MEHP exposure in L6 myotubes. 29 INSR belongs to the ligand-activated receptor-tyrosine kinase family and is essential in metabolic regulation. Studies have reported that defects in INSR protein leads to loss of insulin sensitivity and impaired activation of downstream signaling molecules.32–34 GLUT4 is a significant protein in skeletal muscle responsible for insulin-mediated glucose uptake. 35 Dysregulation of GLUT4 trafficking and expression is associated with the primary development of T2D. 36 The molecular mechanism by which DEHP and its metabolite MEHP decreased INSR and GLUT4 protein in L6 myotubes remains obscure. Therefore, we hypothesized that DEHP and MEHP exposure may alter the Insr and Glut4 gene expression by hampering the interaction of the corresponding transcription factors with the promoter region.
Materials and Methods
Chemicals and Reagents
All chemicals and reagents used in the present study were of molecular biology and analytical grade (AR), and they were purchased from Amersham Biosciences Ltd., UK; Sigma Aldrich, USA; Sisco Research Laboratories, Mumbai, India; and Bio-Rad Laboratories Inc., Hercules, USA. DEHP, MEHP, Dulbecco’s modified Eagle medium (DMEM), and 3-(4,5-dimethylthiazol-2yl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) were obtained from Sigma Aldrich, USA. Fetal Bovine Serum (FBS), 2% horse serum, 0.25 % Trypsin, and Phosphate-buffered Saline (PBS) were obtained from Gibco, USA. A super-signal Chemiluminescence kit was obtained from Thermo Scientific. High mobility group A1 (HMGA1) (sc-8982), MyoD (sc-377460), Myocyte enhancer factor 2A (MEF2A) (sc-293456), and TATA binding protein (TBP) (sc-204) antibodies were purchased from Santa Cruz Biotechnology Inc. (Dallas, Texas, USA). Horseradish peroxidase (HRP)-conjugated secondary antibodies (goat anti-rabbit IgG (sc-2004) and rabbit anti-mouse IgG (sc-358914) were obtained from Santa Cruz Biotechnology (Dallas, Texas, USA).
Cell Culture
L6 myoblast cells were procured from the National Centre for Cell Science (NCCS), Pune. Cells were grown in DMEM containing 2% horse serum for seven days at 37°C in a humidified 5% CO2 atmosphere to complete the conversion of myoblasts to myotubes. The following experimental design was framed after ensuring the complete conversion of myoblasts (mononucleated) to myotubes (multinucleated) through Giemsa staining and probing the myogenin level in myotubes at day 7 through immunoblot analysis.
DEHP and MEHP Treatment
L6 myotubes were serum-starved for 4 hr and treated with DEHP and MEHP as mentioned below. Group I serves as a control or basal. Group II—L6 myotubes were exposed to 100 nM insulin alone for 20 min at the terminal. Both DEHP (Group III and IV) and MEHP (Group V and VI) were dissolved in Dimethyl Sulfoxide (DMSO), exposed at 50 and 100 μM concentrations for 24 h, and at the terminal end, L6 myotubes were stimulated with 100 nM insulin for 20 mins (i.e., at the 24th hour). After treatment, samples were collected and stored at −80°C for further analysis.
Justification of DEHP and MEHP Concentrations
The estimated daily exposure of the U.S. population to DEHP ranges from 5.8 to 19 μg/kg/day, whereas exposure through medical settings may exceed 167.9 mg/day. 37 MEHP concentration in infant plasma is 15.1 μg/ml (equal to 54.25 μM) and in maternal and umbilical cord blood samples (11.87 μg/ml (42.6 μM) and 9.94 μg/ml (35.7 μM), respectively). 38 In blood transfusions, the replacement blood DEHP levels have been detected to range from 10 to 650 μg/ml (about 25–1600 μM). Hemodialysis averaged over a year results in an average of 640 μg/kg bw/day DEHP exposure. 37 In patients with long-term exposure to DEHP-containing devices, the serum level of DEHP ranges from 70 to 80 μg/ml. In neonates, DEHP concentrations range from 13.2 to 84.9 μg/ml following PVC catheter exchange transfusions. In addition, patients undergoing intensive medical care usually have markedly higher MEHP levels than healthy people. Individuals undergoing specific medical procedures may be exposed to even higher levels (more than 100-fold) via plastic medical devices. 37 DEHP and MEHP concentrations (50 and 100 μM) were selected based on the inhibitory concentration (IC50) value through MTT assay and general, occupational, and medical exposure.
Cell Viability Assay
Cell viability was determined by the 3-(4,5-dimethylthiazol-2yl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) assay. Cells seeded in a 96-well plate were treated with DEHP and MEHP at different concentrations (0, 25, 50, 100, 150, and 200 μM) for 24 h. Next, 10 μL MTT stock solution (5 mg/ml in PBS) was added to each well and then incubated for 4 h. DMSO was added to dissolve any deposited formazan. The optical density was measured at 570 nm with a spectrophotometer.
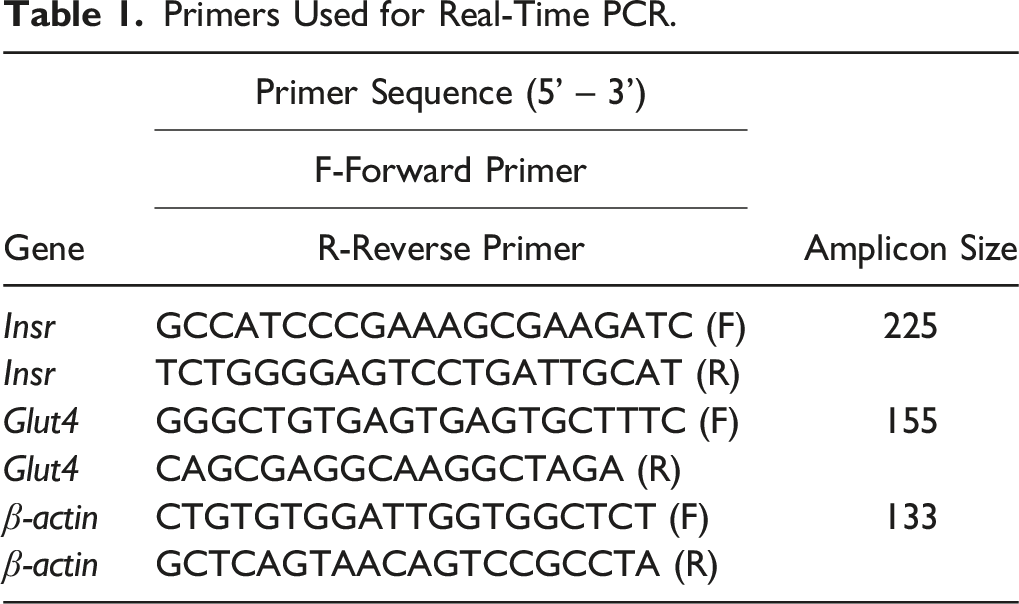
Real-Time Polymerase Chain Reaction (RT-PCR)
Primers Used for Real-Time PCR.
Nuclear Lysate Preparation
Nuclear lysate fractions from control and treated cells were prepared as Im et al 39 described. Briefly, cells were scraped with buffer (1 ml) containing 20 mM Tris/HCl (pH7.4), 10 mM EDTA, 10 mM EGTA, 0.25 M sucrose, proteases (1 M dithiothreitol and 0.1 M PMSF), 2 mM MgCl2 and 1x protease inhibitor cocktail (5 mM leupeptin, 5 mM pepstatin, 5 mM chymostatin and 5 mM aprotinin). The cell suspension was kept on ice for 10 min and centrifuged for 5 min at 2500× g at 4°C. The pellet was resuspended in 1 ml buffer, including protease inhibitor cocktail, and centrifuged at 1000× g for 5 min at 4°C. The pellet was again resuspended with buffer C (300 μl) [20 mM Hepes (pH 7.6), 1.5 mM MgCl2, 0.42 M NaCl, 2.5% glycerol, 1 mM EDTA, 1 mM EGTA, and protease inhibitor cocktail] and sonicated (20 pulse) and stored at −80°C for future use. MEF2A, MyoD, and HMGA1 levels were estimated by Western blot analysis, and TBP was a loading control in the nuclear lysate.
Western Blotting
Protein concentration was estimated using the method of Lowry et al, 40 using bovine serum albumin (BSA) as a standard. The lysate proteins (50 μg/lane) were separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (10% gel) and transferred by electroblotting to polyvinylidene difluoride (PVDF) membrane (Bio-Rad Laboratories Inc.). The membranes were blocked with 5% non-fat dry milk and probed with the primary antibodies [HMGA1, MEF2A, MyoD] (1:1000). Following incubation, the blot was washed three times (5 min each with Tris-buffered saline containing Tween-20 (TBS-T)). After washing with TBS-T, membranes were incubated for 1 h with horseradish peroxidase-conjugated rabbit-anti-mouse or goat-anti-rabbit antibodies (1:5000) (GeNei, Bangalore, India). The specific signals and protein bands were captured using an enhanced chemiluminescence (ECL) detection system (Gel DocTM XR + System, Bio-Rad Laboratories, CA) and quantified using Quantity One image analysis software by Bio-Rad Laboratories, CA. Later, the membranes were incubated in a stripping buffer at 50°C for 30 min. After this, the membrane was reprobed using a TBP antibody (1:1000).
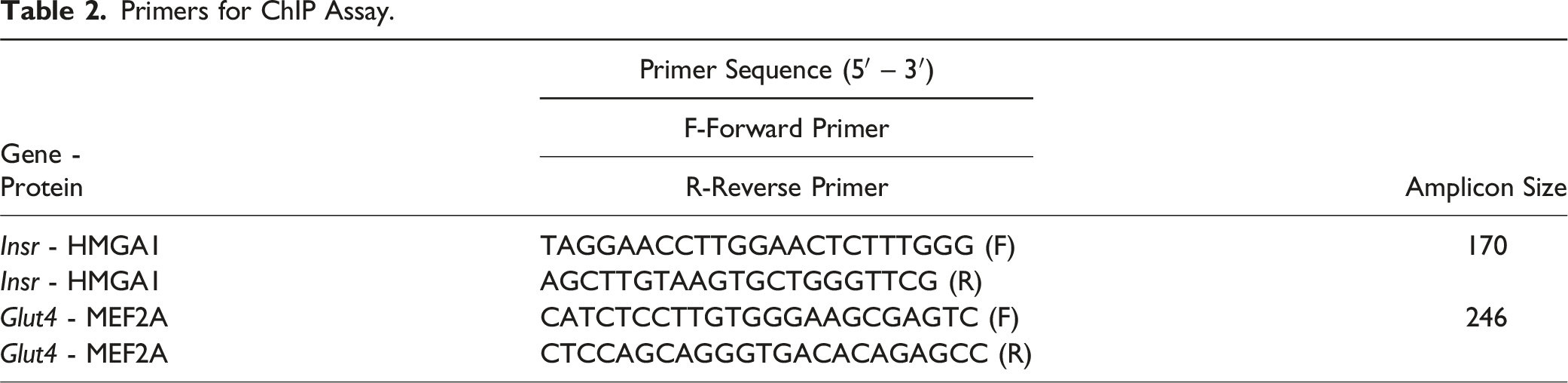
Chromatin Immunoprecipitation (ChIP) Assay
Primers for ChIP Assay.
Statistical Analysis
For each set of results, independent trials were repeated at least three times. The data were subjected to a one-way Analysis of Variance (ANOVA) followed by Students Newman–Keul’s (SNK) test to assess the significance between mean values of the control and experimental groups, using computer-based software Graph Pad Prism 5. The values are expressed as mean ± standard error of the mean (SEM), and P < 0.05 is considered statistically significant.
Results
The cell viability of L6 myotubes upon DEHP and MEHP exposure was determined by MTT assay. L6 myotubes were exposed to different concentrations of DEHP and MEHP (0, 25, 50, 100, 150, 200 μM). We observed that cell viability was significantly decreased below 50 % in 150 and 200 μM DEHP and MEHP concentrations. So, in the current study, 50 and 100 μM DEHP and MEHP concentrations were considered for further experimental analysis (Figure 1). Effect of DEHP and MEHP on the cell viability in L6 myotubes. Viability of the cells was measured by MTT assay. Each bar represents mean ± SEM of 3 separate experiments. Significance at P < .05. a: compared to Control.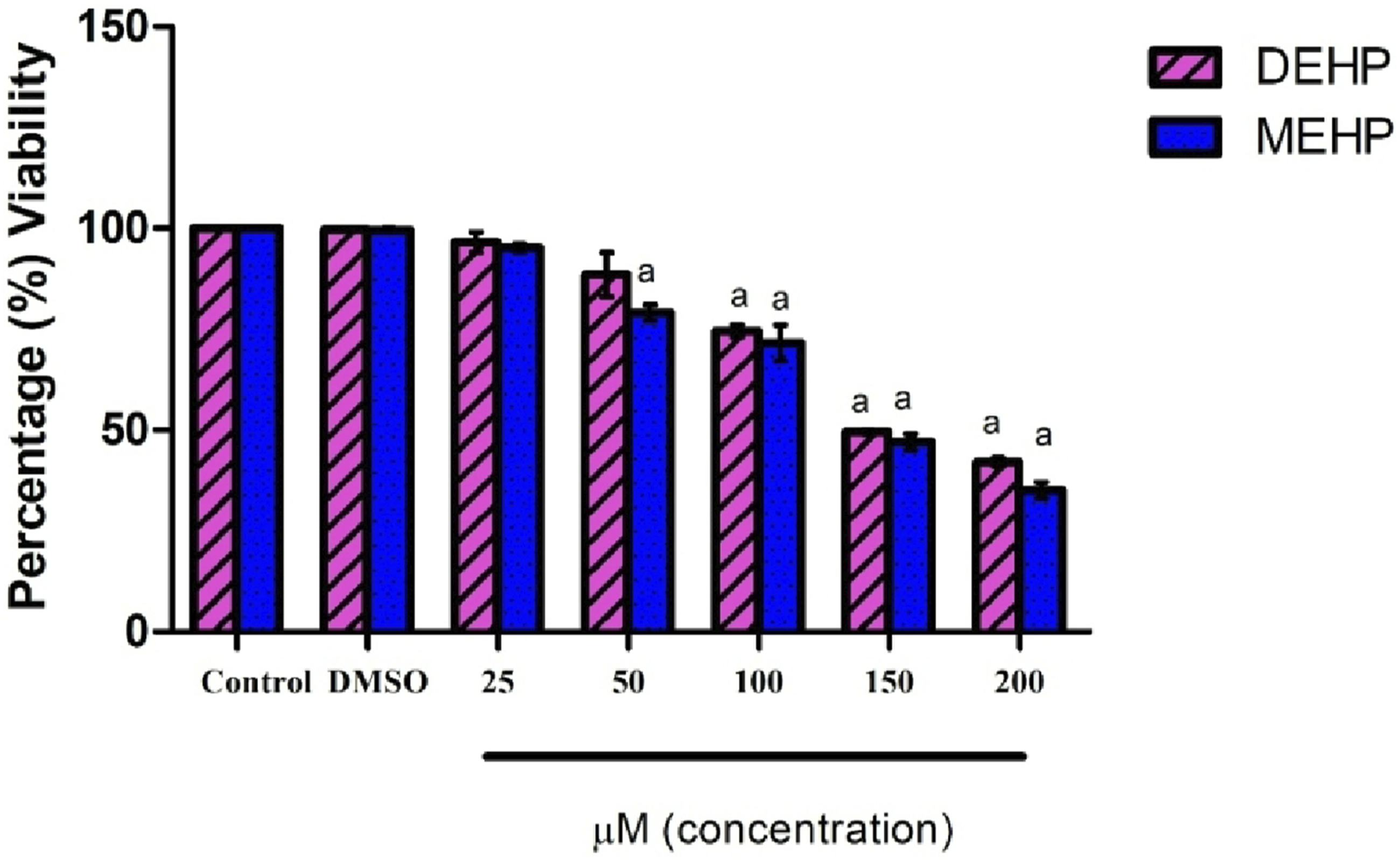
DEHP and MEHP Decreased the Insr mRNA Level
The IR mRNA level is shown in Figure 1. Insr mRNA was significantly decreased in 100 μM DEHP and at both concentrations of MEHP compared to the control and insulin-alone treated groups. There was no change observed between the control and insulin-alone treated group (Figure 2). Effect of DEHP and MEHP on Insr mRNA in L6 myotubes. Each bar represents mean ± SEM of 3 separate experiments. Significance at P < .05. a: compared to Control; b: compared to Control + 100 nM insulin; c: compared to 50 μM DEHP; d: compared to 100 μM DEHP. Results are expressed as fold change from control.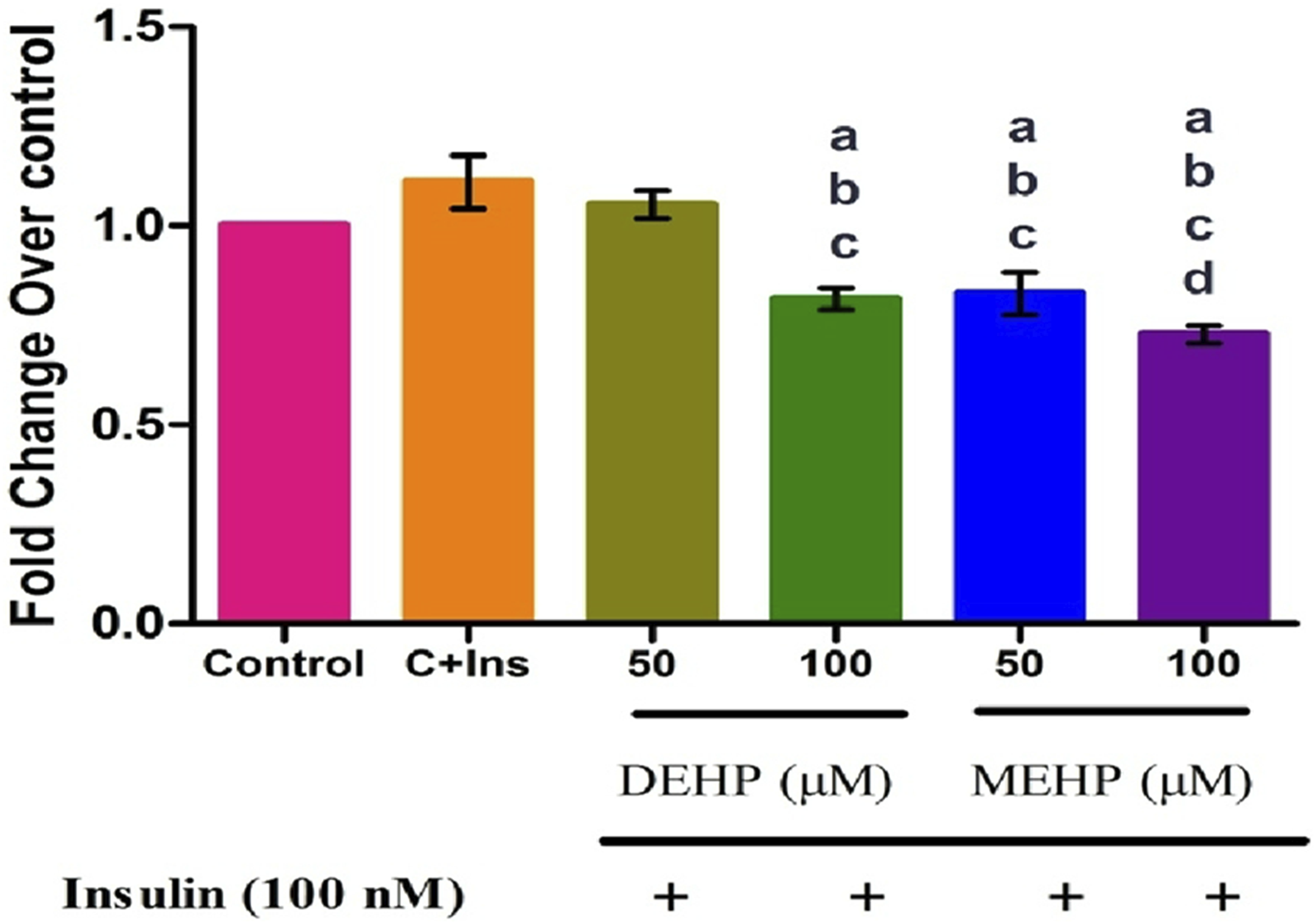
DEHP and MEHP on HMGA1 Protein Level
Immunoblot detection of proteins in the nuclear fraction of L6 myotubes revealed unaltered protein levels of HMGA1 transcription factor controlling Insr expression in DEHP and MEHP-treated groups compared to the control (Figure 3). Effect of DEHP and MEHP on HMGA1 protein in L6 myotubes. Each bar represents mean ± SEM of 3 separate experiments.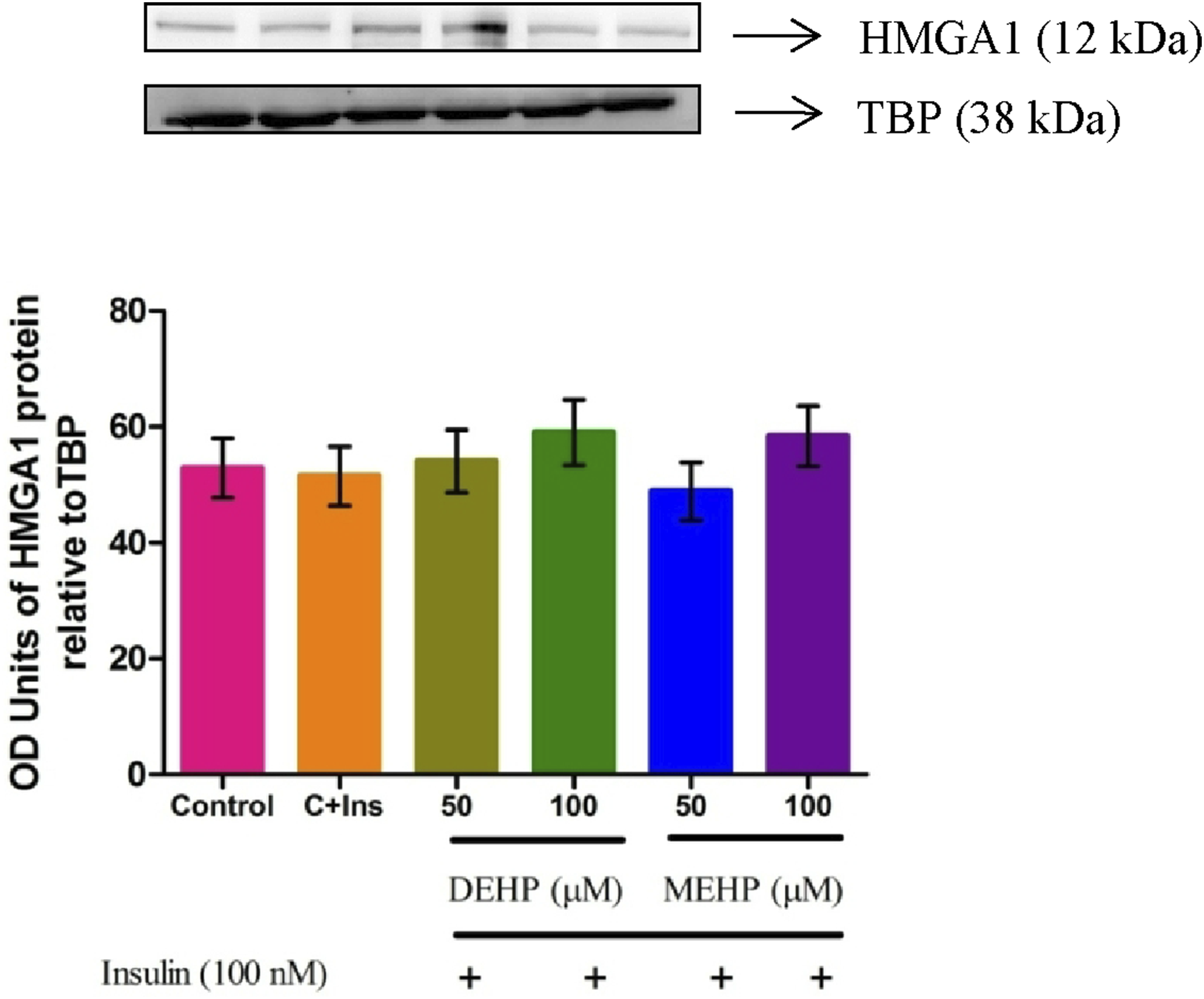
DEHP and MEHP Hampered the Interaction Between the HMGA1-Insr Gene in L6 Myotubes
Through ChIP analysis, the interaction of HMGA-1 on the Insr promoter was studied. Compared to the control, the binding of HMGA1 on the Insr promoter was significantly decreased in DEHP and MEHP-treated groups at both concentrations (Figure 4). Effect of DEHP and MEHP on chromatin immunoprecipitation (ChIP) of HMGA1-Insr gene in L6 myotubes. The quantity of precipitated DNA was normalized to 1% of the input and each bar represents mean ± SEM of 3 separate experiments. Significance at P < .05. a: compared to Control; b: compared to Control + 100 nM insulin; c: compared to 50 μM DEHP.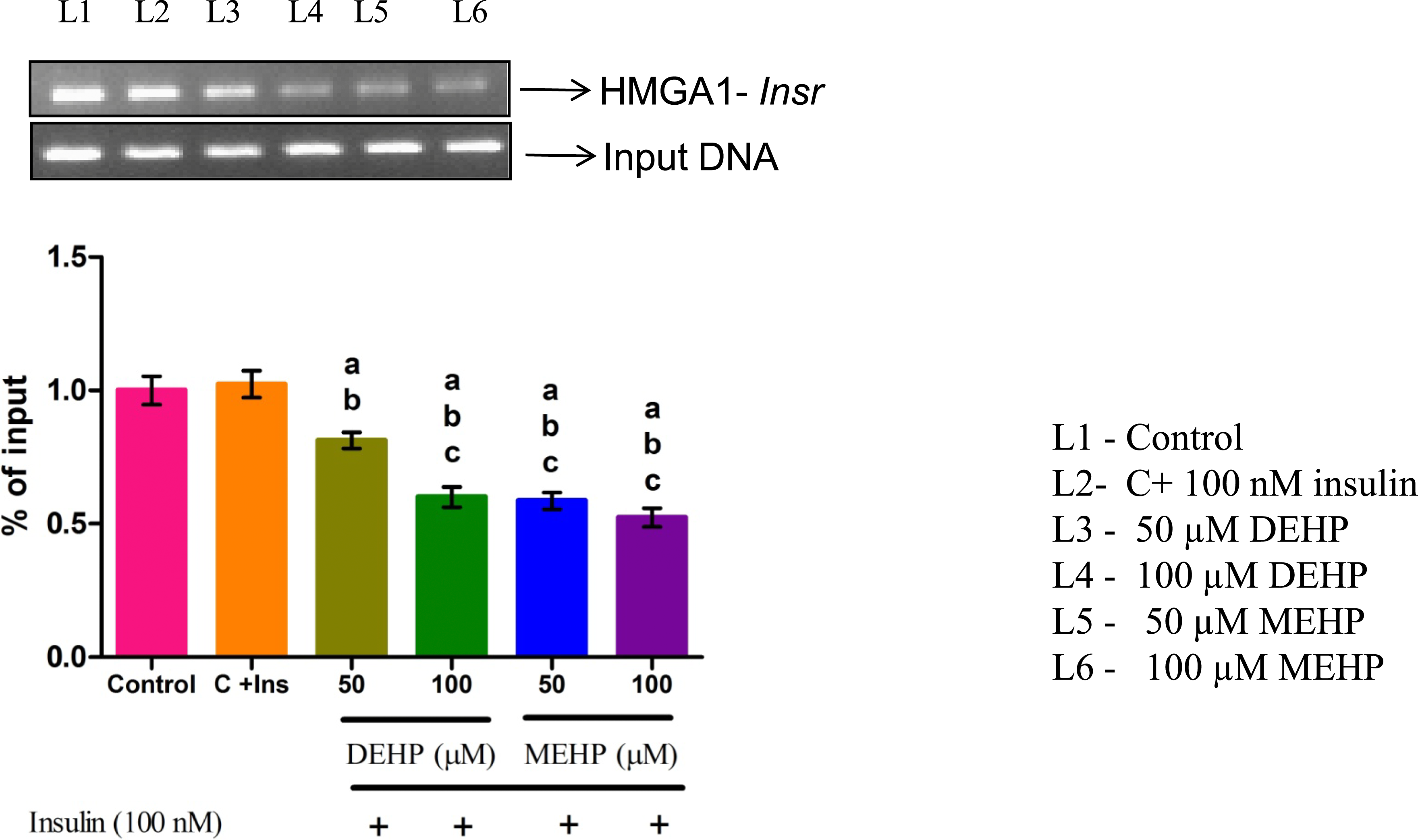
DEHP and MEHP Debilitated Glut4 mRNA Level
Glut4 mRNA was significantly decreased in 100 μM DEHP and at both concentrations of MEHP compared to control and insulin-alone treated groups. No significant change was observed between the control and insulin-alone treated group (Figure 5). Effect of DEHP and MEHP on Glut4 mRNA in L6 myotubes. Each bar represents mean ± SEM of 3 separate experiments. Significance at P < .05. a: compared to Control; b: compared to Control + 100 nM insulin; c: compared to 50 μM DEHP; d: compared to 100 μM DEHP. Results are expressed as fold change from control.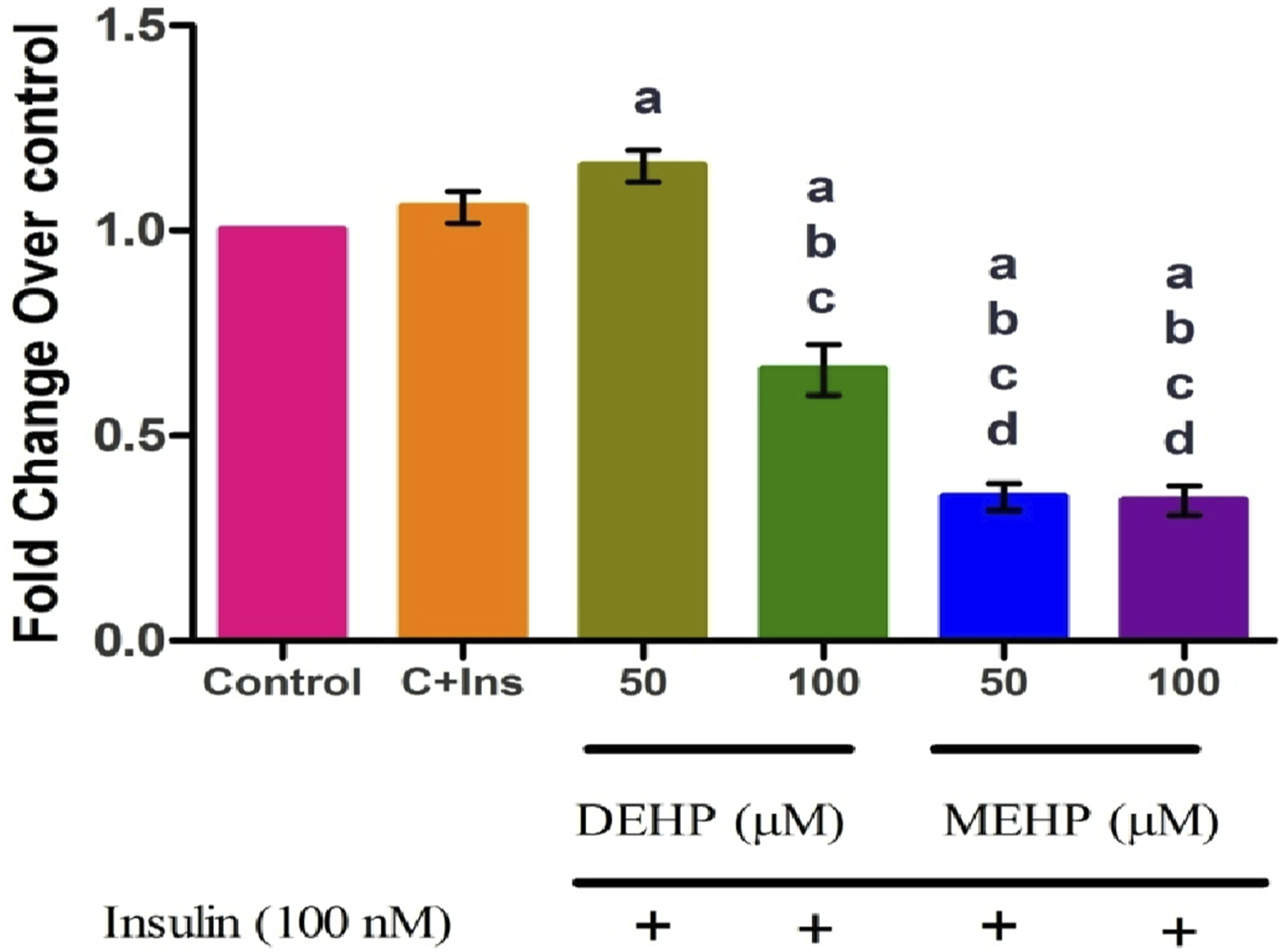
Effect of DEHP and MEHP on MEF2A and MyoD Transcription Factors Regulating Glut4 gene Expression in L6 Myotubes
Immunoblot detection of proteins in the nuclear fraction showed decreased protein levels of MEF2A transcription factor at both concentrations of MEHP compared to the control. No significant change was observed in DEHP-treated groups compared to the control group. MyoD protein level significantly decreased in the 100 μM MEHP-treated group compared to the control group. There was no change in DEHP-treated groups compared to the control (Figure 6). Effect of DEHP and MEHP on MEF2A and MyoD proteins in L6 myotubes. Each bar represents mean ± SEM of 3 separate experiments. Significance at P < .05. a: compared to Control; b: compared to Control + 100 nM insulin; c: compared to 50 μM DEHP; d: compared to 100 μM DEHP; e: compared to 50 μM MEHP.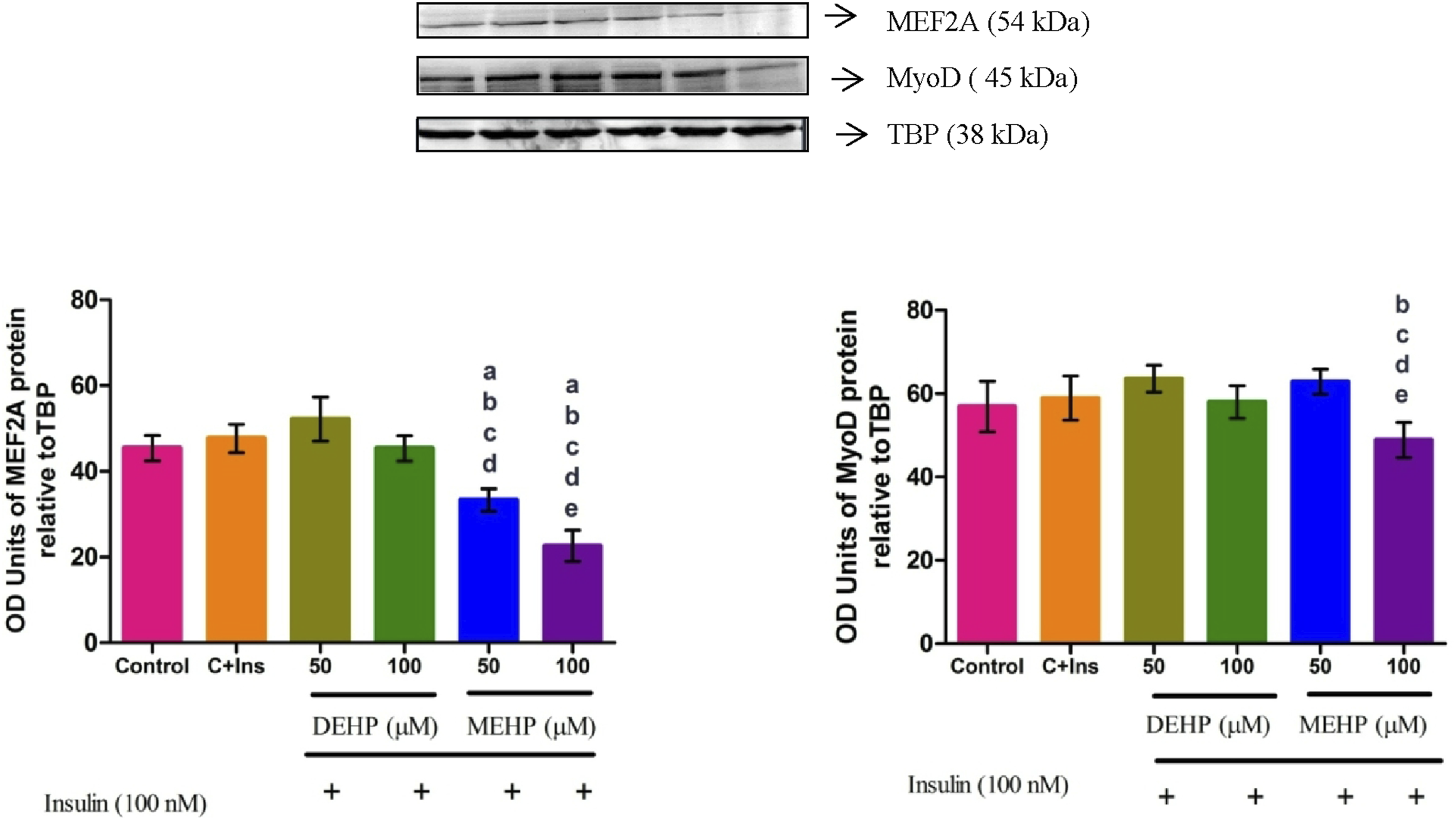
Effect of DEHP and MEHP on MEF2A- Glut4 Gene Interaction in L6 Myotubes
The binding of MEF2A on the Glut4 promoter region was significantly decreased in DEHP and MEHP-treated groups compared to the control (Figure 7). Effect of DEHP & MEHP on chromatin immunoprecipitation (ChIP) of MEF2A-Glut4 gene in L6 myotubes. The quantity of precipitated DNA was normalized to 1% of the input and each bar represents mean ± SEM of 3 separate experiments. Significance at P < .05. a: compared to Control; b: compared to Control + 100 nM insulin; c: compared to 50 μM DEHP; d: compared to 100 μM DEHP; e: compared to 50 μM MEHP.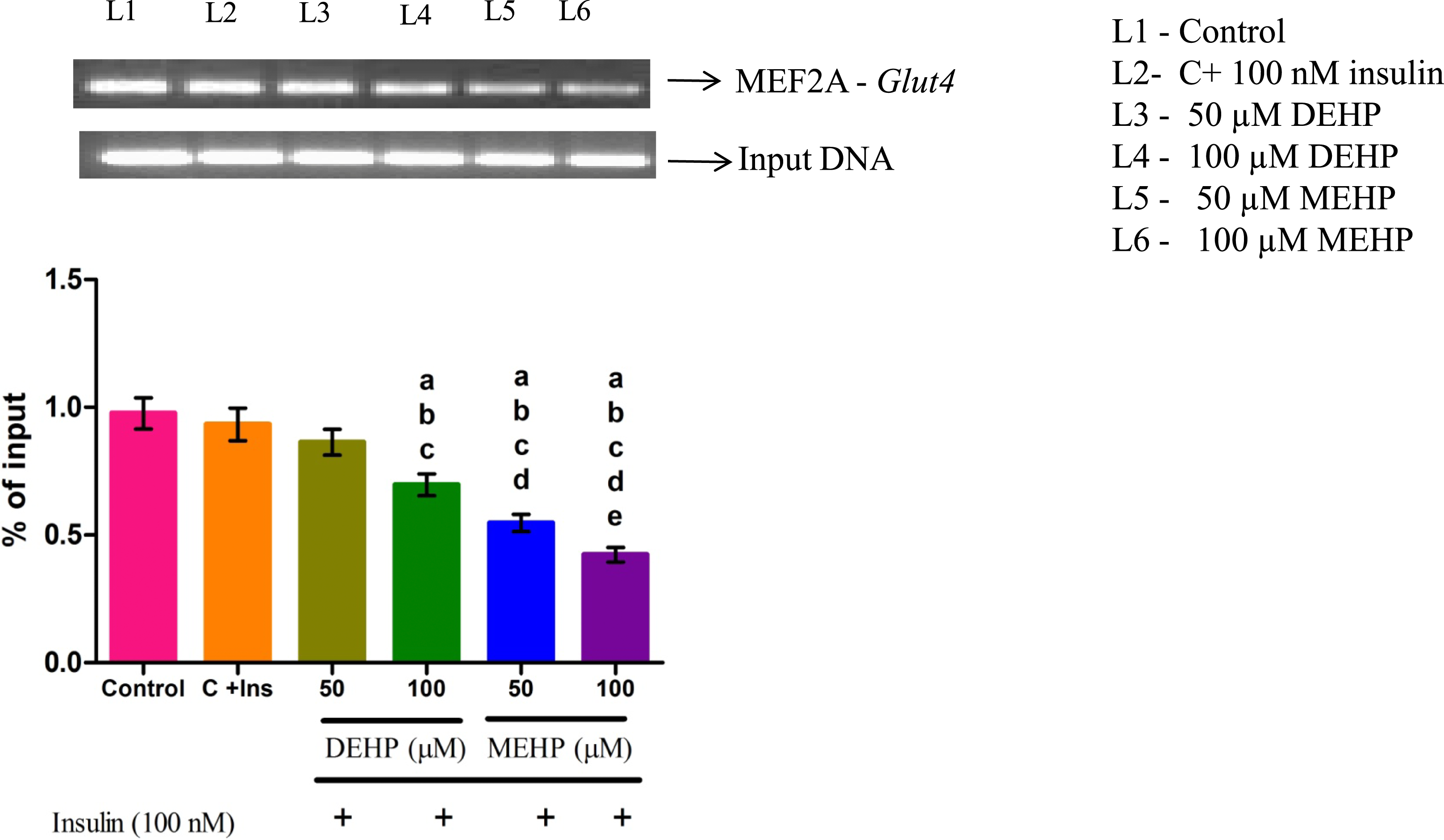
Discussion
The current study aims to identify the reason behind the decreased INSR and GLUT4 protein levels upon DEHP and MEHP exposure in L6 myotubes. Insr mRNA was significantly reduced at 100 μM DEHP and at both concentrations of MEHP (50 and 100 μM). This may be one of the reasons for the decreased INSR protein level observed in our previous study. 29 To gain further insight into the reduced levels of Insr mRNA, we studied the transcription factor that regulates Insr. HMGA1 is one of the architectural transcription factors that bind to adenine-thymine (A-T) rich regions of DNA. 41
The HMGA1 protein level in the nuclear fraction was unaltered in treated groups compared to the control. Though the HMGA1 protein levels were unaltered, DEHP and MEHP diminished the interaction of the HMGA1 transcription factor on the Insr promoter. The decreased interaction might be due to increased free fatty acid levels following exposure to DEHP and MEHP, as observed in trophoblast cells. 42 Elevated free fatty acid increases the levels of PKCε, then the phosphorylated PKCε translocated to the nucleus, further it phosphorylates HMGA1, thereby inhibiting the binding of HMGA1 to the insulin promoter.43,44 Phosphorylated HMGA1 has reduced DNA binding activity.45–47 Based on the literature, it is proposed that increased free fatty acid production upon DEHP and MEHP exposure may have contributed to the impaired binding of HMGA1 to the Insr gene promoter. It is emphatic from the previous studies that impaired interaction of HMGA1 with Insr gene promoter region has resulted in a drastic reduction of Insr gene expression since HMGA1 could not transactivate and recruit transcription factors like Specificity protein1 (Sp1) and CCAAT-enhancer binding protein beta (cEBPβ) to initiate Insr gene expression.48,49 To be concise, DEHP and MEHP do not impact the HMGA1 protein level; irrespective of the level of the HMGA1 transcription factor, the binding of HMGA1 on the Insr promoter is crucial for initiating Insr transcription.
Glut4 mRNA was significantly decreased in DEHP (100 μM) and MEHP (50 and 100 μM)-treated groups compared to the control. The decreased Glut4 mRNA may be one of the reasons for the decreased GLUT4 protein in the cytosol. 29 A series of transcription factors, such as MEF2A, MEF2D, and MyoD, regulate Glut4 gene transcription. The nuclear fraction of MEF2A protein level was significantly decreased at both concentrations of MEHP-treated groups compared to the control. The MyoD transcription factor decreased in the 100 μM MEHP-treated group compared to the control group. Loss of MEF2A isoform and reduced binding activity is associated with decreased Glut4 gene expression in skeletal muscle and heart of diabetic mice. 50 MEF2A and MyoD proteins decreased upon MEHP exposure, which may be the result of the direct effect of MEHP on transcription factors. Though DEHP does not affect the MEF2A and MyoD transcription factors, the Glut4 mRNA was decreased upon DEHP exposure. Therefore, to elucidate the molecular mechanism behind decreased Glut4 mRNA upon DEHP and MEHP exposure, a ChIP assay was performed to check the binding of transcription factors MEF2A and MyoD on the Glut4 promoter region.
MEF2A response element of the Glut4 gene promoter contains an AT-rich consensus sequence that initiates transcription. 51 Interestingly, both DEHP and MEHP-treated groups registered a significant decrease in the binding of MEF2A with the Glut4 promoter. The previous study has suggested that DEHP exposure during gestation increased the HDAC2 expression in the gastrocnemius muscle of F1 offspring, which ultimately resulted in a tight chromatin structure where the chromatin is inaccessible to transcription factors. 19 Histone deacetylation removes the acetyl groups and causes chromatin remodeling, resulting in a heterochromatin structure that recruits repressor protein (MEF2D) towards the promoter complex and hinders MEF2A interaction in human embryonic kidney and fibroblast cells.52,53 In addition, the DNA methyltransferases (DnMTs) level was increased upon DEHP exposure, which silenced the Glut4 gene expression in F1 offspring. 19 Despite the unaltered level of MEF2A, DEHP exposure impaired the interaction between MEF2A and the Glut4 gene promoter. A study has reported that DEHP binds to the AT-rich region of the calf thymus DNA. 54 Since the MEF2A response element has an AT-rich region in the Glut4 gene promoter, the binding of DEHP is likely to hamper the interaction between MEF2A and the Glut4 gene promoter. The MEF2A and MyoD protein levels were reduced upon MEHP exposure, which would have contributed to the decreased percentage of the interaction of the transcription factor MEF2A with the Glut4 gene promoter compared to DEHP. A study has shown the interaction of phthalic acid ester (DEHP) predominantly with thymine in DNA minor grooves. 55 However, further study is required to understand better the differential toxic effect of DEHP and its primary metabolite MEHP on Insr and Glut4 gene expression.
Limitations of the Present Study
We studied the basal Insr and Glut4 transcriptional activity after exposing L6 myotubes to DEHP and MEHP, followed by insulin stimulation for 20 min. Further experiments should focus on exposing L6 myotubes to insulin for 4 to 5 h to study the insulin-mediated Insr and Glut4 transcription upon DEHP and MEHP exposure. Future studies must elucidate the molecular mechanism behind the decreased MEF2A and HMGA-1 transcription factors upon DEHP and MEHP exposure. We did not include the non-insulin-stimulated DEHP and MEHP control groups in the current study due to many (six) groups and a previous study from our laboratory has shown that DEHP (without insulin) exposure (50 and 100 μM) concentrations significantly decreased the INSR and GLUT4 proteins compared to the control. Future studies are warranted to study the effect of DEHP and MEHP during non-insulin-stimulated conditions.
Conclusion
DEHP and its metabolite MEHP exposure decreased the Insr and Glut4 mRNA, which is mediated by the diminished interaction of HMGA1 and MEF2A transcription factors with the promoter region of the Insr and Glut4 genes, respectively. Despite DEHP and MEHP exerting their toxic effect on genes regulating glucose disposal, the toxicity potential of MEHP is greater than that of DEHP.
Footnotes
Author Contributions
Viswanathan, Mangala Priya contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Mullainadhan, Vigneswari contributed to analysis and interpretation and drafted manuscript; Karundevi, Balasubramanian contributed to conception and design, contributed to analysis and interpretation, drafted manuscript, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the DST PURSE Phase II Programme, which provided Ms. V. Mangala Priya with a Junior Research Fellowship (JRF) and financial assistance to the Department of Endocrinology through the UGC-SAP-DSA program in the form of infrastructural facilities.
