Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) reviewed the safety of 30 vinylpyrrolidone polymers as used in cosmetic products; most of these ingredients have the reported cosmetic function of film former in common. The Panel reviewed data relevant to the safety of these ingredients, and determined that 27 vinylpyrrolidone polymers are safe in cosmetics in the present practices of use and concentration described in the safety assessment. The Panel also concluded that the available data are insufficient to make a determination that 3 vinylpyrrolidone polymers (all urethanes) are safe under the intended conditions of use in cosmetic formulations.
Keywords
Introduction
The safety of the following 30 vinylpyrrolidone (VP) polymer ingredients, as used in cosmetics, is reviewed in this safety assessment:
Acrylic Acid/VP Crosspolymer
Maltodextrin/VP Copolymer
PVP/Decene Copolymer
PVP/VA/Itaconic Acid Copolymer
PVP/VA/Vinyl Propionate Copolymer
Styrene/VP Copolymer*
Triacontene/VP Copolymer
VP/Eicosene Copolymer
VP/Hexadecene Copolymer
VP/VA Copolymer*
VP/Vinyl Alcohol Copolymer
Acrylates/Stearyl Methacrylate/VP Copolymer
Acrylates/VP Copolymer*
Ammonium Acryloyldimethyltaurate/VP Copolymer*
Ethylhexyl Acrylate/VP/Dimethicone Methacrylate
Copolymer
Ethylhexyl Methacrylate/Methyl Methacrylate/VP Copolymer
Methacrylic Acid/Styrene/VP Copolymer*
Vinyl Caprolactam/VP/Dimethylaminoethyl Methacrylate
Copolymer*
VP/Acrylates/Lauryl Methacrylate Copolymer
VP/Dimethylaminoethylmethacrylate Copolymer*
VP/DMAPA Acrylates Copolymer
VP/Vinyl Caprolactam/DMAPA Acrylates Copolymer
Butylated PVP
PVP*
Triacontanyl PVP
Hydrolyzed Wheat Protein/PVP Crosspolymer
Sodium Acryloyldimethyltaurate/VP Crosspolymer*
VP/Dimethiconylacrylate/Polycarbamyl/Polyglycol Ester
VP/Dimethylaminoethylmethacrylate/Polycarbamyl Polyglycol Ester
VP/Polycarbamyl Polyglycol Ester
*Previously reviewed by the Panel
Definitions, Idealized Structures, and Functions of the Ingredients in This Safety Assessment.(1; CIR Staff)*

*Please note: For the sake of simplicity, these ingredients have only been drawn as simple block co-polymers. The periodicity and pattern of interconnectivity between each monomer may vary significantly per ingredient, or even per supplier of the same ingredient.
Reports on Polymers Previously Reviewed by the Panel.

Monomer Components of Vinylpyrrolidone Polymers.

This safety assessment includes relevant published and unpublished data for each endpoint that is evaluated. Published data are identified by conducting an exhaustive search of the world’s literature. A list of the typical search engines and websites used, sources explored, and endpoints that the Panel evaluates, is available on the CIR (https://www.cir-safety.org/supplementaldoc/preliminary-search-engines-and-websites; https://www.cir-safety.org/supplementaldoc/cir-report-format-outline). Unpublished data are provided by the cosmetics industry, as well as by other interested parties.
Chemistry
Definition and General Characterization
The definitions, structures, and functions of the vinylpyrrolidone polymers that are reviewed in this safety assessment are presented in Table 1. These polymeric ingredients share in common a vinylpyrrolidone monomer (Figure 1). Vinylpyrrolidone polymer ingredients (wherein –C(R)HCH2- represents at least one co-monomer residue).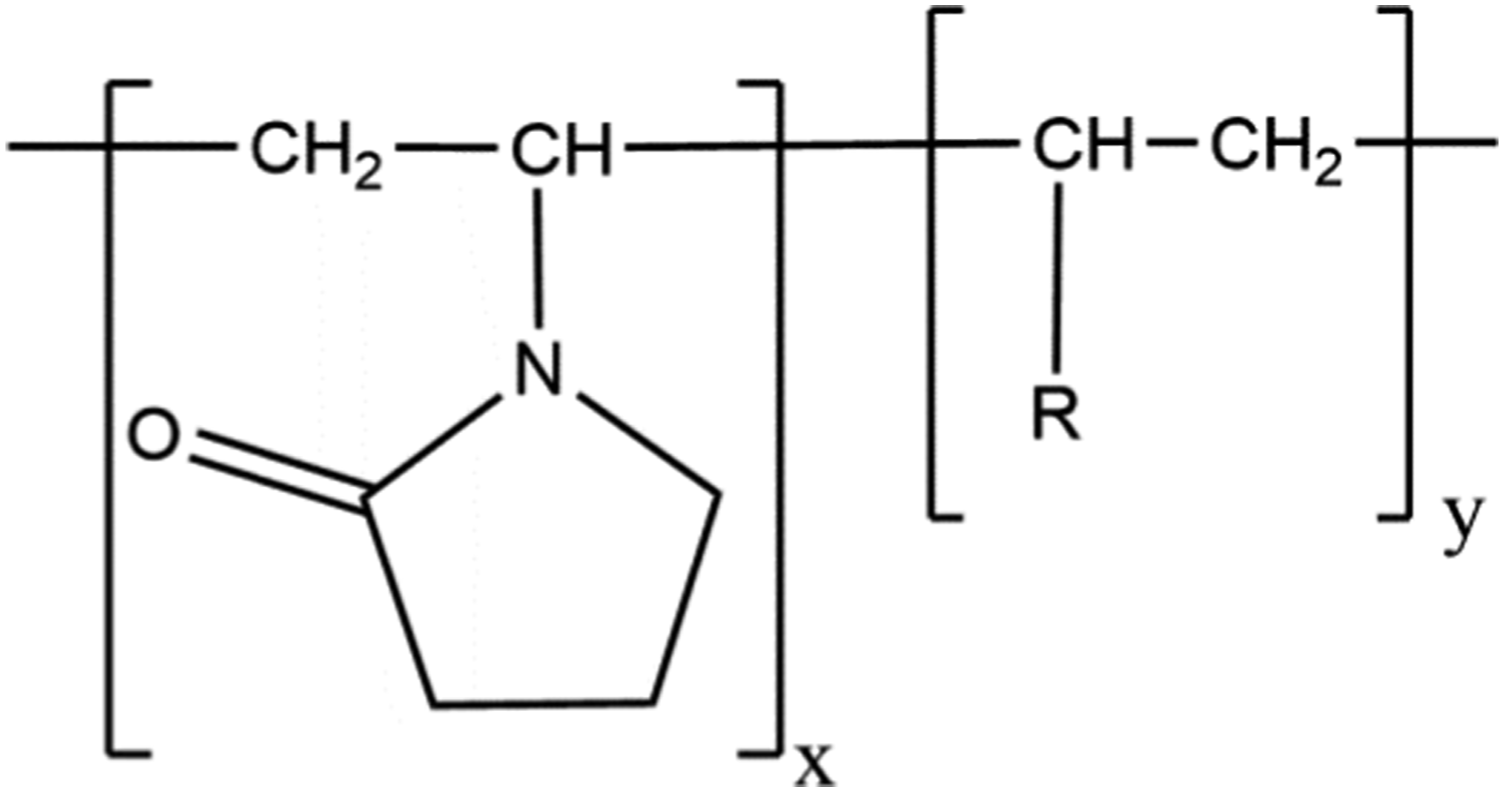
Chemical and Physical Properties
Chemical and Physical Properties of Vinylpyrrolidone Polymers.

Australia’s National Industrial Chemical Notification and Assessment Scheme (NICNAS) has determined that Maltodextrin/VP Copolymer, VP/Acrylates/Lauryl Methacrylate Copolymer, and Sodium Acryloyldimethyltaurate/VP Crosspolymer are polymers of low concern (PLC).9,11,12 The following statements relate to some of the NICNAS-established characteristics of a PLC: A polymer cannot be a PLC if it is designed to or can be expected to substantially degrade, decompose, or depolymerize, including polymers that substantially degrade, decompose or depolymerize after manufacture and end use, even though they are not intended to do so. 14
VP Copolymers
VP/VA Copolymer
VP/VA Copolymer does not absorb energy over the long-wavelength ultraviolet (UVA), mid-wavelength ultraviolet (UVB), or visible light spectrum. 2
Method of Manufacture
VP Copolymers
VP/VA Copolymer
VP/VA Copolymer is prepared by free radical polymerization in ethyl alcohol. 2 Details about radical initiators, propagators, chain terminators, and solvent(s) were not provided.
VP/VA Copolymer is produced by free radical copolymerization of N-vinyl-2-pyrrolidone (NVP) and vinyl acetate (VA) in an isopropanol solution, in the presence of initiators. 15 The process is continuous and temperature controlled, and sodium bisulfite is added to the batch for color stability. Isopropanol is exchanged for deionized water by adding deionized water to the reactor and performing a solvent exchange via vacuum distillation. Sodium acetate (for pH stabilization) and a microbiological preservative (identity not specified) are added. The batch is then heated, sampled, and adjusted for solids content. The product is isolated as an aqueous solution/emulsion, or as a spray-dried solid. The specification for the maximum concentration of hydrazine that is formed in the reaction mixture is included in the ‘Composition/Impurities’ section.
A cosmetic ingredient supplier reports that radical polymerization is used to make VP/VA Copolymer from vinylpyrrolidone and vinyl acetate. 16 Details about radical initiators, propagators, chain terminators, and solvent(s), were not provided.
PVP and Modified PVP Polymers
PVP
Two cosmetic ingredient suppliers report that radical polymerization is used to make PVP.17,18 Details about radical initiators, propagators, chain terminators, and solvent(s) were not provided.
VP Crosspolymers
Hydrolyzed Wheat Protein/PVP Crosspolymer
According to one manufacturer, the method of manufacture of Hydrolyzed Wheat Protein begins with a solution of protein, water, and enzyme. 19 The pH of the mixture is adjusted and additional enzyme is added. This is accompanied by addition of a denaturant, followed by filtration using activated carbon. Filtration is followed by purification, evaporation, and preservation. The next step is the copolymerization of vinylpyrrolidone in the presence of an initiator, and this is followed by pH adjustment and preservation. The reaction mixture is then diluted in accordance with established specifications, which is followed by filtration into packs.
Composition/Impurities
VP Copolymers
Acrylic Acid/VP Crosspolymer
Acrylic Acid/VP Crosspolymer contains ≤500 ppm acrylic acid (residual monomer) and ≤100 ppm vinyl-pyrrolidone (residual monomer). 20 Heptane (<0.99%) has been identified as another impurity in Acrylic Acid/VP Crosspolymer.
Maltodextrin/VP Copolymer
Maltodextrin
Styrene/VP Copolymer
Data provided by industry indicate that styrene and vinyl-type styrene copolymer trade name materials contain styrene monomer at levels of 100 ppm or less. 5
Styrene/VP Copolymer contains <0.2% styrene (residual monomer) and <10 ppm N-vinylpyrrolidone (residual monomer). 20
VP/Eicosene Copolymer
VP/Eicosene Copolymer contains ≤100 ppm vinylpyrrolidone (residual monomer). 20 Other impurities that have been detected in VP/Eicosene Copolymer include t-butanol (<0.5%) and isopropanol (<0.5%).
VP/Hexadecene Copolymer
VP/Hexadecene Copolymer contains ≤100 ppm vinylpyrrolidone
VP/VA Copolymer
VP/VA Copolymer is supplied either in 100% concentration as a powder or as a 50% solution in alcohol. 2 VP/VA Copolymers may contain the residual monomers vinyl acetate at 1.0% (max) and vinylpyrrolidone at 0.5% (max).
For VP/VA Copolymers with molecular weights of approximately 12,000 and greater, the level of vinyl acetate is smaller than or equal to 300 ppm as measured using HPLC.3,21 Another source reported vinyl acetate levels of less than 100 ppm for copolymers of molecular weights of 12,700 to approximately 30,000 Da, and levels of less than 1000 ppm for a copolymer of a molecular weight of approximately 51,000 Da.
Specifications for VP/VA Copolymer. 15

Regarding the production process for VP/VA Copolymer, free radical copolymerization of NVP and vinyl acetate, specifications limit the concentration of hydrazine that is formed from amines present in the reaction mixture to a maximum of 1 mg/kg. 15 Furthermore, due to the method of production (radical polymerization from vinylpyrrolidone and vinyl acetate) as described by a supplier, residual monomers may be present in VP/VA Copolymer at a maximum of 50 ppm vinylpyrrolidone and a maximum of 100 ppm vinyl acetate. 16 Data received from the cosmetics industry also indicate that VP/VA Copolymer contains ≤ 1000 ppm vinylpyrrolidone (residual monomer) and ≤1000 ppm vinyl acetate (residual monomer). 20
VP Acrylate Copolymers
Vinyl Caprolactam/VP/Dimethylaminoethyl Methacrylate Copolymer
Vinyl Caprolactam/VP/Dimethylaminoethyl Methacrylate Copolymer contains <0.1% vinylpyrrolidone (residual monomer) and ≤1% vinylcaprolactam (residual monomer). 20
VP Acrylates/Lauryl Methacrylate Copolymer
The residual monomer content of VP Acrylates/Lauryl Methacrylate Copolymer has been described as follows: ≤1000 ppm lauryl methacrylate, ≤1000 ppm vinylpyrrolidone, and ≤2000 ppm acrylic acid. 20 Heptane (<10,000 ppm) is another impurity that has been identified in VP Acrylates/Lauryl Methacrylate Copolymer.
VP Dimethylaminoethylmethacrylate Copolymer
VP Dimethylaminoethylmethacrylate Copolymer contains ≤1000 ppm vinylpyrrolidone. 20
VP/DMAPA Acrylates Copolymer
The residual monomer content of VP/DMAPA Acrylates Copolymer has been identified as ≤100 ppm vinyl-pyrrolidone. 20
VP/Vinyl Caprolactam/DMAPA Acrylates Copolymer
VP/Vinyl Caprolactam/DMAPA Acrylates Copolymer contains ≤100 ppm vinylpyrrolidone (residual monomer) and ≤100 ppm vinyl caprolactam (residual monomer). 20 Water is another impurity that has been identified in VP/Vinyl Caprolactam/DMAPA Acrylates Copolymer.
PVP and Modified PVP Polymers
Butylated PVP
Butylated PVP contains ≤100 ppm (residual monomer). 20
PVP
According to the Food Chemicals Codex, the acceptance criteria (i.e., limitations on PVP impurities) relating to the use of PVP in food are as follows: lead (not more than 2 mg/kg), aldehydes (as acetaldehyde, not more than 0.05%), nitrogen (not less than 11.5% and not more than 12.8%), and hydrazine (not more than 1 mg/kg). 22
PVP, an NVP-containing polymer, is imported into Australia for industrial uses, and the residual NVP monomer levels in PVP were obtained from a few (number not stated) major importers of PVP. 23 It was noted that it appears that there are different grades of PVP imported into Australia, depending on the end use (i.e., pharmaceutical, cosmetic, or industrial grade). The residual NVP monomer content in the PVP imported into Australia varies and ranges from 10 ppm to 2000 ppm. In Europe (countries not specified), NVP residues in PVP are generally below 100 ppm.
As a result of the production method described by two cosmetic-ingredient suppliers of PVP, residual monomers may be present in PVP at a maximum of 100 ppm vinylpyrrolidone and a maximum of 100 ppm vinyl acetate.17,18 Also, according to one of the suppliers, other impurities that may be present are acetaldehyde at a maximization concentration of 100 ppm and heavy metals in sum (as lead) at a maximum of 10 ppm. 18 Other data received from the cosmetics industry indicate that PVP contains ≤1000 ppm vinylpyrrolidone (residual monomer). 20
Triacontanyl PVP
According to one source, Triacontanyl PVP has a purity of >97% and consists of <2% water. 10 Data received from the cosmetics industry indicate that Triacontanyl PVP contains ≤20 ppm vinylpyrrolidone (residual monomer). 20 Hexanol (≤500 ppm) is another impurity that has been detected in Triacontanyl PVP.
VP Crosspolymers
Hydrolyzed Wheat Protein/PVP Crosspolymer
According to a chemical supplier, the main impurity in Hydrolyzed Wheat Protein/PVP Crosspolymer is ash, up to a maximum of 2%. 19 Furthermore, an internal specification of 0.2% maximum has been established for residual N-vinylpyrrolidone monomer.
Sodium Acryloyldimethyltaurate/VP Crosspolymer
NICNAS has noted that Sodium Acryloyldimethyltaurate/VP Crosspolymer contains residual monomers and/or impurities (identities and concentrations not stated) that are classified as hazardous according to the Globally Harmonized System of Classification and Labeling of Chemicals (GHS), as adopted for industrial chemicals in Australia. 12 “These are not present in the notified polymer as introduced above the cut off concentration for classification.” Sodium polyacryloyldimethyl taurate, a chemical that is similar to a component of Sodium Acryloyldimethyltaurate/VP Crosspolymer, is reported to contain <2000 ppm 2-acrylamido-2-methyl-1-propane sulfonic acid and <10 ppm acrylamide. 6
Use
Cosmetic
The safety of vinylpyrrolidone polymers is evaluated based on data received from the US Food and Drug Administration (FDA) and the cosmetics industry on the expected use of these ingredients in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in FDA’s Voluntary Cosmetic Registration Program (VCRP) database. 24 Use concentration data are submitted by the cosmetics industry in response to surveys, conducted by the Council, of maximum reported use concentrations by product. 25
According to 2018 VCRP data, the greatest use frequency is reported for PVP, which is being used in 900 cosmetic products (798 leave-on products + 101 rinse-off products + 1 product diluted for bath use). 24 The second highest use frequency is being reported for Ammonium Acryloyldimethyltaurate/VP Copolymer (597 cosmetic products: 525 leave-on products + 62 rinse-off products). In general, the differences in current use frequencies of vinylpyrrolidone polymers in cosmetics versus those reported in previous years are unremarkable.2-8,24
Frequency and Concentration of Vinylpyrrolidone Polymers According to Duration and Exposure.
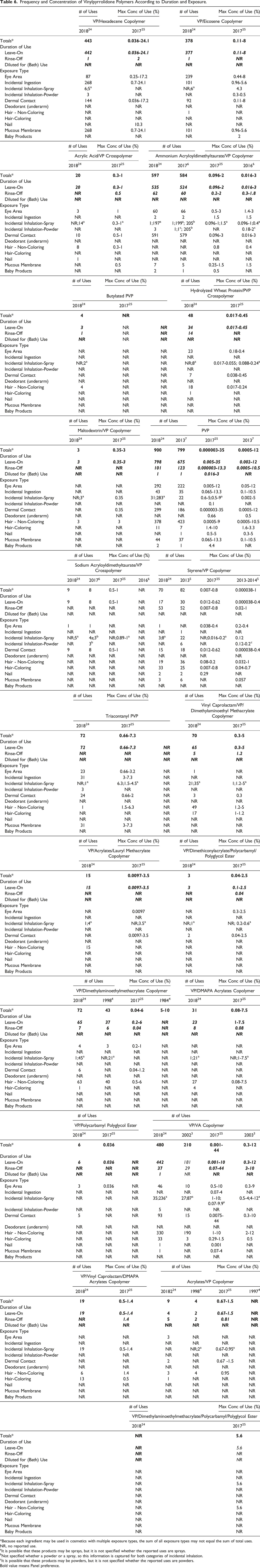
*Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
NR, no reported use.
aIt is possible that these products may be sprays, but it is not specified whether the reported uses are sprays.
bNot specified whether a powder or a spray, so this information is captured for both categories of incidental inhalation.
cIt is possible that these products may be powders, but it is not specified whether the reported uses are powders.
Bold value means Panel preference.
According to VCRP and Council survey data, the following 9 vinylpyrrolidone polymers are not currently used in cosmetic products:
Acrylates/Stearyl Methacrylate/VP Copolymer
Ethylhexyl Acrylate/VP/Dimethicone Methacrylate Copolymer
Ethylhexyl Methacrylate/Methyl Methacrylate/VP Copolymer
Methacrylic Acid/Styrene/VP Copolymer
PVP/Decene Copolymer
PVP/VA/Itaconic Acid Copolymer
PVP/VA/Vinyl Propionate Copolymer
Triacontene/VP Copolymer
VP/Vinyl Alcohol Copolymer
Cosmetic products containing vinylpyrrolidone polymers may be applied to the skin and hair or, incidentally, may come in contact with the eyes (e.g., at maximum use concentrations up to 17.2% VP/Hexadecene Copolymer [in eye shadows]) and mucous membranes (e.g., at maximum use concentrations up to 24.1% VP/Hexadecene Copolymer [in lipstick]). Incidental ingestion of ingredients may result from the use of lipstick products. Products containing vinylpyrrolidone polymers may be applied as frequently as several times per day and may come in contact with the skin or hair for variable periods following application. Daily or occasional use may extend over many years.
VP/VA Copolymer is being used in both pump hair sprays (maximum use concentrations up to 9%) and aerosol hair sprays (maximum use concentrations up to 10%), which may result in incidental inhalation exposure. These 2 concentrations are the highest maximum cosmetic use concentrations that are being reported for vinylpyrrolidone polymers in cosmetic products that are sprayed In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles below 10 µm, compared with pump sprays.26-29 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (i.e., they would not enter the lungs) to any appreciable amount.26,27
VP/Eicosene Copolymer is being used in face powders at concentrations up to 0.5% (highest maximum use concentration). Conservative estimates of inhalation exposures to respirable particles during the use of loose powder cosmetic products are 400-fold to 1000-fold less than protective regulatory and guidance limits for inert airborne respirable particles in the workplace.30-32
The ingredients reviewed in this safety assessment are not restricted from use in any way under the rules governing cosmetic products in the European Union. 33
Non-Cosmetic
VP Copolymers
VP/VA Copolymer
The EFSA Panel on Food Additives and Nutrient Sources added to Food has provided a scientific opinion on the use of VP/VA Copolymer in food supplements. 15 This opinion addresses the safety of VP/VA Copolymer for use in food supplements, in tablet form as a binding/coating agent in an amount of up to 10% of weight per tablet, for a tablet weight of 1000 mg. Overall, the EFSA Panel concluded that the use of VP/VA Copolymer in solid food supplements as a binding/coating agent is unlikely to be a safety concern at the proposed uses and use levels provided. The EFSA Panel also concluded that the residual level of hydrazine, proposed at a maximum of 1.0 mg/kg in the final product, is unlikely to be of safety concern. However, the EFSA Panel noted that it would be prudent to lower the level of hydrazine as far as reasonably achievable.
PVP and Modified PVP Polymers
PVP
According to the Food Chemicals Codex, PVP has the following uses: clarifying agent, separation/filtration aid, stabilizer, bodying agent, tableting aid, dispersant, and coating on fresh fruit. 22
PVP is used widely in industries such as pharmaceuticals, adhesives, agriculture, and surface coating. 23 It is used in medicine and in the pharmaceutical industry as a blood plasma expander, and it is a common ingredient in drug manufacture. 34
Toxicokinetic Studies
Absorption, Distribution, Metabolism, and Excretion
PVP and Modified PVP Polymers
Animal: PVP
The absorption, distribution, metabolism, and excretion of PVP is dependent on molecular weight, amount and frequency of dosing, and route of administration. 8 Polymers with a weight <25,000 are eliminated through the kidneys. An oral dosing study using 0.9 mg per rat of a PVP trade name material found no significant absorption.
New data on the absorption, distribution, metabolism and excretion of PVP or other vinylpyrrolidone polymers were not discovered in the published literature, and unpublished data were not submitted.
Toxicological Studies
Acute Toxicity Studies
Animal
Dermal: PVP and Modified PVP Polymers: Triacontanyl PVP
A single dose of a Triacontanyl PVP trade name mixture (moistened with water, dose = 2 g/kg) was applied, under an occlusive wrap, for 24 h to the backs of 10 New Zealand white rabbits. 10 The animals were observed for up to 14 days after test substance application, and all gained weight during the study. None of the animals died, and no abnormal clinical signs were observed. The acute dermal LD50 was > 2 g/kg.
Oral: VP Copolymers: VP/VA Copolymer
Acute oral toxicity studies were performed with VP/VA Copolymer in formulation and in solutions of the raw ingredient. Tests on mice and rats showed low to no toxicity on more than 76 animals. Two animals died from administration of a formulation containing other, unidentified ingredients. The surviving animals showed, at most, decreased activity and ataxia at maximum doses of 5 g/kg of a solution containing 12.5% VP/VA Copolymer. 2
Oral: VP Acrylate Copolymers: Ammonium Acryloyldimethyltaurate/VP Copolymer
The acute oral LD50 for Ammonium Acryloyldimethyltaurate/VP Copolymer was reported to be >2 g/kg in rats. 6
Oral: VP Acrylate Copolymers: VP/Acrylates/Lauryl Methacrylate Copolymer
An oral LD50 of >5 g/kg (rats) has been reported for undiluted VP/Acrylates/Lauryl Methacrylate Copolymer. 11 Dyspnea was observed in 1 animal. The number of animals tested and details relating to the test protocol and study results were not specified.
Oral: PVP and Modified PVP Polymers: PVP
The oral LD50 of PVP (avg. MW of 40,000) is >100 g/kg body weight for both rats and guinea pigs. 8
Oral: PVP and Modified PVP Polymers: Triacontanyl PVP
The acute oral toxicity of a Triacontanyl PVP trade name material was evaluated using 10 Sprague-Dawley rats (5 males, 5 females). 10 A single 5 g/kg oral dose of the test substance (ground into a powder and mixed with peanut butter and honey) was fed to the animals. The test substance was consumed within 18 h to 24 h. Dosing was followed by a 14-day observation period. None of the animals died and no gross organ changes were observed at necropsy. The LD50 was > 5 g/kg.
Short-Term Toxicity Studies
Dermal
VP Copolymers: VP/VA Copolymer
A hair product containing 1% VP/VA Copolymer was tested in a 6-week dermal toxicity study on 50 albino rats. Volumes of 2.0 ml/kg of the product were applied 5 days a week for 6 weeks for a total of 30 applications to the clipped skin of the animals. All rats survived, and their body weight, physical appearance, behavior, and gross and microscopic anatomy were normal. No systemic toxic effects could be attributed to the test material. 2
Oral
VP Copolymers: VP/VA Copolymer
VP/VA Copolymer was administered in the diet of 3 groups of male and female Sprague-Dawley rats (5 animals/sex/group; control group: 5 animals/sex) for 28 days at doses of 0 (control), 100, 300, and 1000 mg/kg/day, respectively. 15 The control group was fed the basal diet only. All animals survived to the scheduled necropsy. There were no clinical signs of toxicity, and there were no effects on the following: body weight gain, feed consumption, hematology parameters, serum chemistry, and urinalyses. There also were no test substance-related effects on organ weights, macroscopic and microscopic evaluations. The authors concluded that the no-observed-adverse-effect level (NOAEL) was 1000 mg/kg/day.
Oral: PVP and Modified PVP Polymers: PVP
The short-term oral toxicity of a PVP tradename material (5% w/v in water) was evaluated using 2 groups of 6 HanWistar rats (RccHan:WIST; 3 males, 3 females/group). 35 The test animals received oral doses (dose volume of 10 ml/kg, by gavage) daily for 28 consecutive days. The control group received water. One day after the final dose, the animals were killed and scheduled for necropsy. The following tissues were examined microscopically: eyes, liver, kidneys, urinary bladder, lungs, heart, thymus, sternum, upper jaw (with nares and nasal turbinates), lower jaw with skin, stomach/duodenum, intestine (jejunum, ileum, cecum, and rectum), mesenteric lymph node, and the tongue. All hematology findings were within the normal background range for the rat strain that was tested, and there was no induction of cytochrome P450 protein (CYP1A1/2, CYP2B1, CYP3A, and CYP4A) levels. There were no toxicologically relevant effects on body weight gain, food consumption, or water consumption, and there were no treatment-related microscopic changes.
Inhalation
PVP and Modified PVP Polymers: PVP
In two short-term inhalation studies using rats, PVP was detected in lung samples but no inflammatory response was noted. Mild lymphoid hyperplasia and fibroplasia were noted in the subpleural, perivascular, and peribronchial lymphatics. 8 The animals were exposed to an average PVP concentration of 118 or 146 mg/m3 5 days per week (8 hours per day) for 6 weeks (a total of 30 exposures).
Subchronic Toxicity Studies
Oral
VP Copolymers: VP/VA Copolymer
VP/VA Copolymer was administered in the diet of 3 groups of male and female Sprague-Dawley rats (10 animals/sex/group; control group of 10 animals/sex) for 90 days at doses of 0 (control), 100, 300, and 1000 mg/kg/day VP/VA Copolymer. 15 The control group was fed the basal diet only. All animals survived to the scheduled necropsy. There were no clinical signs of toxicity, and there were no effects on the following: body weight gain, feed consumption, functional observational battery, and locomotor activity evaluations. Furthermore, there were no ophthalmic lesions indicative of toxicity, and no test-substance-related effects on hematology parameters, serum chemistries, and urinalyses. No test substance-related effects on organ weights, macroscopic, and microscopic evaluations were observed. The authors concluded that the NOAEL was 1000 mg/kg/day.
Inhalation
VP Copolymers: VP/VA Copolymer
Rats and hamsters were exposed for 13 weeks to a spray containing 4.0% VP/VA Copolymer. 2 Each of three groups comprised of 12 rats and 12 hamsters per group inhaled the spray for 4 hours per day, 5 days per week for 13 weeks at a concentration of 5.4 mg/m3 (calculated to be the equivalent of 100 times the normal human use level of the product). No gross or microscopic changes occurred that could be attributed to the test material. Lungs and other tissues were similar in control and tested animals. Subchronic inhalation of a spray formulation containing 1.72% VP/VA Copolymer for 90 days produced no effects in rabbits. On each day of this 90-day study, the animals received one 30-second exposure each morning and afternoon and were left in the spray atmosphere for 15 minutes.
Chronic Toxicity Studies
Animal
Oral: VP Copolymers: VP/VA Copolymer
A chronic (1 year) oral ingestion study of a solution containing 10.2 mg/L of VP/VA Copolymer was conducted. These exposures produced no effects in mice or rats. 2
In a 52-week dietary study, the chronic oral toxicity of VP/VA Copolymer was evaluated using the following groups of male and female pure-bred Beagle dogs: group 1 (4 males, 4 females: 510 mg/kg/day), group 2 (4 males, 4 females: 1518 mg/kg/day), and group 3 (6 males, 6 females: 2522 mg/kg/day). 36 The control group (6 males, 6 females) was fed a diet without the test substance. All animals were killed at the end of the dosing period, and both gross and histopathologic examinations were performed. None of the animals died during the study and no treatment-related clinical signs were observed. Furthermore, the following parameters were unaffected by treatment: food consumption, ophthalmoscopic examinations, hearing tests, electrocardiograms, and blood pressure. There were no treatment-related body weight losses during the study. Hematology, clinical biochemistry, and urinalysis parameters were unaffected by feeding with the test substance; sporadic statistically significant intergroup differences were observed, but these findings were not dose-related.
There were no treatment-related or dose-related changes in organ weights or organ-to-body weight ratios. At gross examination, the type and incidence of findings were comparable between test and control groups. At microscopic examination, the incidence and severity of findings were comparable between test and control groups and were considered commonly observed changes in dogs of the age and strain used in this study. No inflammatory and/or degenerative changes (i.e., necrosis, granulomas, etc.) were associated with vacuolated histiocytes that were diagnosed in the sinusoids and trabeculae of some mesenteric lymph nodes. The NOAEL was determined to be the target dose of 2500 mg/kg/day (target dose for highest dose group). 36
The chronic oral toxicity of VP/VA Copolymer was evaluated using 3 groups of male and female Wistar rats of the Chbb:THOM (SPF) strain (50 males, 50 females/group). 36 The 3 groups were fed the test substance (in the diet) at the following doses for 24 months: group 1 (low dose: 686 mg/kg/day [males] and 691 mg/kg/day [females]), group 2 (mid dose: 1374 mg/kg/day [males] and 1378 mg/kg/day [females), and group 3 (high dose: 2625 mg/kg/day [males] and 2759 mg/kg/day [females]). A fourth group (control group: 50 males, 50 females) was fed a diet without the test substance for the same duration. The test substance (same doses) was also fed to 4 satellite groups (3 tests and 1 control) for 18 months. These 4 groups were included for hematological evaluation. For all groups in the study, the animals were killed after a 16-h to 20-h fasting period that began after the end of the dosing period. Numerous tissues were submitted for histopathological examination. The mortality rates ranged from 14% in the high-dose males to 36% in the control males, and 26% in the high-dose females to 30% in the control females. Data were comparable in the satellite groups. Food consumption was described as normal. Due to the absence of a dose-response relationship, and a higher mortality rate in control rats of both sexes, it was concluded that the test substance did not affect survival. No remarkable test substance-related clinical signs were observed in the study. Body weight and body weight change were statistically significantly reduced in high-dose males at most time points throughout most of the study. Marginal differences in hematological parameters (within historical control ranges) were observed, but there was no dose-response relationship and the differences were not considered treatment-related. The vast majority of the gross lesions in the main groups in the study were comparable to the incidence in controls, and there was no clear dose-response relationship. The NOAEL was determined to be the target dose of 2800 mg/kg/day (target dose for high dose group). Results relating to tumor formation are included in the Carcinogenicity section of this report.
A 2-year feeding study on VP/VA Copolymer (60% VP and 40% VA) was performed using 2 groups of Sprague-Dawley rats (51 males and 51 females/group, test and control groups). 15 Test animals were given feed containing 5% VP/VA Copolymer, and control animals were given feed containing 5% cellulose. Based on the consumption of the entire diet, test animals were fed ∼0.67 g VP/VA Copolymer (equivalent to ∼450 mg/kg/day) for the duration of the study. Hemoglobin content and leukocyte count were determined in 5 rats per sex (test and control groups) for up to 364 days of the study. Hematology, blood chemistry, and urinalysis parameters were evaluated after ∼500 days of the study. These 3 parameters were evaluated using 10 test and control rats of each sex, as well as in all test (20 rats) and control (11 rats) animals that remained alive after 675 days. Survival in both the control and test groups was described as poor (8% to 14%), due to inflammatory diseases of the respiratory tract. There were no signs of toxicity in test or control rats, and no treatment-related clinical chemistry changes. At histopathological examination of organs (liver, kidneys, and other organs [not stated]), an increased incidence of liver congestion and fatty degeneration in the test group, compared to the control group, was reported. No gross pathologically detectable lesions were observed. Results relating to carcinogenic potential are included in the Carcinogenicity section of this report.
Oral: VP Copolymers: PVP and Modified PVP Polymers: PVP
Neither toxic effects nor gross lesions attributable to PVP were found in rats maintained for 2 years on a diet containing up to 10% of a PVP trade name material. 8 A similar 2-year feeding study in dogs found swollen phagocytic cells in the lymph nodes.
Inhalation: VP Copolymers: VP/VA Copolymer
Thirty-six male and 36 female Syrian hamsters were exposed to the low concentration of 0.08 ± 0.08 mg/L VP/VA Copolymer in air, 4-32 minutes a day, once a week for up to 2 years. 2 The high-level group consisted of 36 male and 36 female hamsters exposed to 0.35 ± 0.09 mg/L, 9-35 minutes a day, once a week for up to 2 years. Necropsies were performed on all that were sacrificed or that died spontaneously. Survival time, body weight, and weight and appearance of lungs were similar in control and aerosol-exposed animals.
Developmental and Reproductive Toxicity Studies
In Vitro
PVP and Modified PVP Polymers
PVP
No teratogenic effects were observed when up to 500 µg of PVP (MW 11,500) was injected into the yolk sac of rabbit embryos. 8
Additional DART data were not discovered in the published literature, and unpublished data were not submitted.
Genotoxicity Studies
In Vitro
VP Acrylate Copolymers
Ammonium Acryloyldimethyltaurate/VP Copolymer
Ammonium Acryloyldimethyltaurate/VP Copolymer was not mutagenic (up to 5000 µg/plate) in bacterial reverse mutation assays. 6
PVP and Modified PVP Polymers
PVP
The genotoxicity of PVP was evaluated in invitro and in vivo mutagenicity assays. 8 Bacterial assay results were as follows: PVP-iodine, up to 20 µl (non-mutagenic in Ames test), PVP-iodine, in various amounts (mutagenic at > 2 µl in Ames test), PVP-iodine, amount not stated (mutagenic to Salmonella typhimurium strain TA1530 in Ames test). In the in vitro mouse lymphoma assay, PVP (up to 100 mg/ml) was not mutagenic with or without metabolic activation. In the same assay, PVP-iodine (up to 10 mg/ml) caused aberrant non-dose-related mutations with, but not without, activation. In the in vitro Balb/c 3T3 transformation assay, PVP (up to 100 mg/ml) was not mutagenic (non-dose-related transformations observed.
Three formulations containing PVP-iodine were not genotoxic in a comet assay or a chromosome aberration test, with or without metabolic activation. 37 The solutions contained 3% or 10% PVP-iodine. In both tests, Chinese hamster ovary (CHO)-K1 cells were exposed for 4 h to the test solutions. Expected results were observed with positive and negative controls.
The genotoxicity of PVP was evaluated in the Ames test using the following Salmonella typhimurium strains: TA98, TA100, TA1535, and TA1537. 38 Each strain was tested with aqueous PVP (doses up to 10,000 µg/plate) with and without metabolic activation. The results were classified as negative in all bacterial strains, with and without metabolic activation.
Triacontanyl PVP
The genotoxicity of a Triacontanyl PVP trade name material was evaluated in the Ames test using the following S. typhimurium strains: TA98, TA100, TA1535, TA1537, and TA1538. 10 The test substance was evaluated (with and without metabolic activation) at doses up to 2500 µg/plate. 2-Aminoanthracene, 2-nitrofluorene, sodium azide, and ICR-191 served as positive controls. The test substance was not genotoxic in any of the S. typhimurium strains tested. Marked increases in the number of revertant colonies were observed in positive control cultures.
VP Crosspolymers
Sodium Acryloyldimethyltaurate/VP Crosspolymer
The genotoxicity of Sodium Acryloyldimethyltaurate/VP Crosspolymer was evaluated in the Ames test (bacterial strains and doses not stated) (Organization for Economic Co-operation and Development (OECD) Test Guideline (TG) 471). 12 The test substance was classified as non-genotoxic.
In Vivo
PVP and Modified PVP Polymers
PVP
PVP-iodine complex (11.2% available iodine) was evaluated in the following three in vivo genotoxicity assays: dominant lethal assay (male NMRI mice dosed intraperitoneally (i.p.) with 72 mg of PVP-iodine/kg), micronucleus test (male and female NMRI mice dosed i.p. with 36 mg of PVP-iodine/kg), and a bone marrow assay (male and female Chinese hamsters dosed i.p. with up to 82.5 mg PVP-iodine/kg). 8 In the dominant lethal assay, the conception rate decreased significantly during the first week, but the average number of implantations (and resultantly, the mutagenicity index) was not affected. In the remaining weeks, all parameters remained similar between control and treated groups. Micronucleus test results indicated a significant increase in the number of micronuclei when compared to controls, but the value was considered within normal range. In the bone marrow assay, there were no increases in the rates of aberrant metaphases.
Carcinogenicity Studies
Animal
Oral
VP Polymers: VP/VA Copolymer
The carcinogenicity of VP/VA Copolymer was evaluated using 3 groups of male and female Wistar rats of the Chbb:THOM (SPF) strain (50 males, 50 females/group). 36 The 3 groups were fed the test substance (in the diet) at the following doses for 24 months: group 1 (high dose: 2625 mg/kg/day [males] and 2759 mg/kg/day [females]), group 2 (mid dose: 1374 mg/kg/day [males] and 1378 mg/kg/day [females), and group 3 (low dose: 686 mg/kg/day [males] and 691 mg/kg/day [females]). A fourth group (control) was fed a diet without the test substance for the same duration. At histopathological examination, there was no treatment-related increase in the number of animals with the following: neoplasms (primary neoplasm or benign, malignant, systemic and metastasized neoplasms). There also was no treatment-related increase in the total number of primary neoplasms, or benign, malignant, systemic, or metastasized neoplasms. Additionally, there was no indication that the test substance caused any non-neoplastic alteration of organs or organ systems, when comparing the incidence and graded severity of microscopic findings of treated animals with the corresponding observations in control animals. All neoplastic and non-neoplastic microscopic findings were considered to have developed spontaneously and were not related to treatment. (Results relating to chronic toxicity were described earlier in this report.)
A 24-month feeding study on VP/VA Copolymer (60% VP and 40% VA) was performed using 2 groups of Sprague-Dawley rats (51 males and 51 females/group, test and control groups). 15 Test animals were given feed containing 5% VP/VA Copolymer, and control animals were given feed containing 5% cellulose. Based on the consumption of the entire diet, test animals were fed ∼0.67 g VP/VA Copolymer (equivalent to ∼450 mg/kg/day) for the duration of the study. No treatment-related tumors or other gross pathologically detectable lesions were induced. (Results relating to chronic toxicity were described earlier.)
Implantation
PVP and Modified PVP Polymers: PVP
The implantation of PVP sponges into mice and rats resulted in development of local sarcomas, but without metastases. 8
Human
PVP and Modified PVP Polymers
PVP
According to the International Agency for Research on Cancer, PVP is not classifiable as to its carcinogenicity to humans. 39
Anticarcinogenicity Study
PVP and Modified PVP Polymers
PVP
Orally administered PVP significantly decreased the rate of bladder tumors in mice exposed to bracken fern. 8
Other Relevant Studies
Cytotoxicity
PVP and Modified PVP Polymers
PVP
The effect of PVP on the ultrastructure of spermatozoa from 12 fertile patients was evaluated. 40 A sperm suspension (0.1 mL) from each patient was added to a 10% PVP solution (0.5 mL) and incubated for 30 minutes. An aliquot of the sperm suspension without PVP served as the control. The samples were analyzed by transmission electron microscopy. Results indicated that the untreated sperm fractions and the PVP-treated fractions were significantly different. The means of the percentages of spermatozoa devoid of defects in untreated sperm fractions versus PVP-treated fractions were 4.2808% and 0.5490%, respectively (P = 0.001). The sperm organelles that were deteriorated by PVP treatment were as follows: swollen, reacted or absent acrosomes, the granular and decondensed chromatin, and swollen and badly shaped mitochondria. The most affected organelle was the plasma membrane, which appeared broken in a high percentage of the cells. In cross sections of sperm tails after PVP treatment, the plasma membrane was broken, the mitochondria were swollen, and the axoneme was disassembled. Thus, the 10% PVP solution strongly affected the fine structure of spermatozoa. The authors concluded that the 10% PVP solution exerted a disintegrating effect on the various kinds of sperm membranes, and, as a secondary consequence of the eventual necrotic process, alteration of chromatin and cytoskeletal components.
The effect of a PVP trade name material on cultured HeLa cells (human cervical carcinoma cells) was evaluated. 41 HeLa cells were incubated with the trade name material (at concentrations of 5%, 10%, and 20%) for 24 h. Treatment with the test substance produced a dose- and time-dependent toxicity (i.e., inhibitory effect on cell proliferation) to HeLa cells. The hallmarks of apoptosis, such as chromatin condensation, DNA fragmentation, and formation of apoptotic bodies, were observed. Other results indicated that the apoptosis induced by the test substance may have been via cell cycle arrest at the G2/M phase.
Dermal Irritation and Sensitzation Studies
Irritation
In Vitro
VP Crosspolymers: Hydrolyzed Wheat Protein/PVP Crosspolymer
The skin irritation potential of Hydrolyzed Wheat Protein/PVP Crosspolymer (21% solids, i.e., concentration of Hydrolyzed Wheat Protein/PVP Crosspolymer) was evaluated using the EpiskinTM reconstituted human epidermis model. 42 The principle of this assay is based on the measurement of cytotoxicity in epidermal cultures, following topical exposure to the test substance, using the colorimetric MTT (3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyl-tertrazolium bromide) reduction assay. Cell viability is measured by enzymatic reduction of the yellow MTT tetrazolium salt to a blue formazan salt (within the mitochondria of viable cells) in test substance-treated cells relative to the negative controls. Dulbecco’s phosphate buffered saline (PBS) with calcium and magnesium served as the negative control, and 5% w/v aqueous sodium dodecyl sulfate served as the positive control. Triplicate tissues were treated with the test substance (as supplied, 10 µl) for 15 min, followed by a post-exposure incubation period of 42 h. After 42 h, each tissue was obtained for MTT-loading. After loading, a total biopsy of each epidermis was made and formazan crystals were extracted out of the MTT-loaded tissues. At the end of extraction, optical density was measured at 540 nm. Data were presented in the form of % viability (i.e., MTT reduction in test substance-treated tissues relative to negative control tissues). The relative mean viability of tissues treated with Hydrolyzed Wheat Protein/PVP Crosspolymer for 15 min was 86.6%, and the test substance was classified as a non-irritant.
Animal
VP Copolymers: VP/VA Copolymer
VP/VA Copolymer (50% in alcohol solution, 5 g dose) was mildly irritating to the skin in 24-h patch tests involving groups of 6 rabbits, whereas the undiluted ingredient was non-irritating to the skin of 6 rabbits. 2 In 24-h skin irritation tests, using groups of 3 to 9 rabbits, on product formulations containing various concentrations of VP/VA Copolymer, concentrations of 0.5%, 1.50% and 4% were non-irritating and a test concentration of 1.75% had the potential for minimal irritation.
Acute skin irritation studies of VP/VA Copolymer were conducted on the abraded and intact skin of rabbits. Formulations containing 0.25%-4.0% VP/VA Copolymer produced mild irritation. Solutions of 50% VP/VA in alcohol produced mild irritation, and one sample of the 100% powder moistened in water produced no irritation. 2
VP Acrylate Copolymers: Ammonium Acryloyldimethyltaurate/VP Copolymer
In a study involving rabbits (number and strain not stated), Ammonium Acryloyldimethyltaurate/VP Copolymer (assumed applied neat, not specified) was applied for 4 h to a 6 cm2 area of skin. The test substance was non-irritating to the skin of rabbits. 6
PVP and Modified PVP Polymers: PVP
A 10% PVP-iodine solution was applied, under an occlusive patch, for 96 h to hairless dorsal skin of 25 rabbits. 8 After a 2-week non-treatment period, another occlusive patch was similarly applied for 48 h. No dermal reactions were observed after either duration of patch application.
PVP and Modified PVP Polymers: Triacontanyl PVP
The skin irritation potential of a Triacontanyl PVP trade name material (moistened with saline) was evaluated using 6 new Zealand white rabbits. 10 The test substance (0.5 g) was applied, under an occlusive wrap, for 24 h to both an abraded and intact site on each animal. The area (cm2) of the application site was not stated. Very slight erythema was observed at 2 intact sites and 4 abraded sites (at 24 h) and at 1 intact site and 2 abraded sites (at 72 h). Slight edema was observed at 1 intact site and 2 abraded sites, only at 24 h. The test substance was classified as a slight skin irritant.
Human
VP Copolymers: VP/Hexadecene Copolymer
The skin irritation potential of a cosmetic base containing 14.95% VP/Hexadecene Copolymer (undiluted) was evaluated in an occlusive patch test involving 50 subjects, identified as follows: 27 subjects (normal, healthy), 6 (with eczema), 3 (with allergy), and 14 (with sensitive skin). 43 The product was applied to the back (dose per cm2 not stated) for 48 h using clear, square patch test chambers. Reactions were scored at 48 h (30 minutes after patch removal) and 72 h post-application. Sodium dodecyl sulfate (1% in water) and water served as positive and negative controls, respectively. There were no reactions to the product in any of the subjects tested. The positive control caused reactions in 21 subjects. Reactions to the negative control were not observed.
PVP and Modified PVP Polymers: PVP
In 48-h and 96-h Shelanski patch tests, both involving 200 subjects, undiluted PVP-iodine (10% PVP and 2% iodine) was not a skin irritant. 8 In 3 studies, groups of 20 subjects were patch tested with a foundation containing 2% PVP. One to 2 subjects in each group had minimal faint, uniform or spotty erythema.
The irritation and sensitization potential of different preparations that contain iodine, including PVP-iodine, was investigated in 24 fair-skinned, healthy subjects without a history of iodine allergy. 7 PVP-iodine was tested at concentrations of 1%, 5%, 7.5%, and 10% on the intrascapular area on the back or on the volar forearm (2-day application) with Finn Chambers on Scanpor tape. Only 1 subject reacted to PVP-iodine, at concentrations of 7.5% (vesiculation on day 4) and 10% (definite erythema on day 4).
Sensitization
Animal
VP Copolymers: VP/VA Copolymer
VP/VA Copolymer was not a sensitizer to guinea pigs after repeated intracutaneous injections. 2 The skin was inspected 24 h after each injection.
VP Acrylate Copolymers: Ammonium Acryloyldimethyltaurate/VP Copolymer
Ammonium Acryloyldimethyltaurate/VP Copolymer (neat) was not sensitizing to guinea pigs. 6
PVP and Modified PVP Polymers: PVP
A 10% PVP-iodine solution did not cause dermal sensitization in rabbits. 8
VP Crosspolymers: Sodium Acryloyldimethyltaurate/VP Crosspolymer
The skin sensitization potential of Sodium Acryloyldimethyltaurate/VP Crosspolymer (concentration not stated) was evaluated in the local lymph node assay (LLNA) (OECD TG 429). 12 There was no evidence of sensitization.
Human
VP Copolymers: VP/VA Copolymer
Repeated insult patch tests of a 5.0% formulation of VP/VA Copolymer caused no irritation or sensitization in 50 subjects. 2 Likewise, three solutions of 50% VP/VA Copolymer in alcohol caused no irritation in 150 subjects.
VP Acrylates Copolymer: VP/Acrylates/Lauryl Methacrylate Copolymer
In a human repeated insult patch test (HRIPT) involving 105 subjects, VP/Acrylates/Lauryl Methacrylate Copolymer (96%) induced minimal erythema in 6 and 2 subjects during the induction and challenge phases, respectively. 11 These results were not considered positive by the authors of this study. Details relating to the test protocol were not included.
PVP and Modified PVP Polymers: PVP
Undiluted PVP-iodine (10% PVP, 2% iodine) did not induce sensitization in an HRIPT involving 100 subjects. 8 In an exaggerated use study (Draize-Shelanski patch test technique) on a PVP trade name material (PVP concentration not stated) involving 150 subjects, results were negative for skin sensitization. Results were also negative in an HRIPT (27 subjects) on a PVP trade name material (10% aqueous solution). In a maximization test involving 25 subjects, 2% PVP did not induce contact allergy.
PVP and Modified PVP Polymers: Triacontanyl PVP
The skin sensitization potential of a Triacontanyl PVP trade name material was evaluated in an HRIPT involving 102 subjects (21 males, 81 females). 10 Nine 24-h induction patches (type not stated), each containing ∼200 mg of the test substance, were applied to the left upper back of each subject over a 3-week period. The area of application (cm2) was not stated. A 24-h challenge patch was applied 2 weeks after removal of the last induction patch. Reactions were scored at 48 h and 72 h post-application. A minimal reaction (not defined) was observed in 6 subjects during the induction phase. Reactions were not observed during the challenge phase. The test substance was a non-sensitizer.
Photosensitization/Phototoxicity
Animal
VP Codpolymers: VP/VA Copolymer
No photosensitization data on VP/VA Copolymer were available for review, but the UV absorption characteristics suggest that photosensitization is unlikely. 2
Human
PVP and Modified PVP Polymers: PVP
A PVP trade name mixture (10% aqueous solution) did not induce a phototoxic response in a study involving 10 human subjects. 8
PVP and Modified PVP Polymers: Triacontanyl PVP
The phototoxicity of a Triacontanyl PVP trade name material was evaluated using 10 subjects (1 male, 9 females). The test substance (∼200 mg) was applied, under an occlusive wrap, for 24 h to both forearms of each subject. 10 The area of application was not stated. After removal of the occlusive wrap, 1 forearm of each subject was irradiated with UVA light. Both arms of each subject were evaluated for reactions on days 2, 3, and 4, and reactions were not observed. The test substance did not induce a contact dermal phototoxic response.
During the induction phase of a photoallergenicity study, a Triacontanyl PVP trade name material (200 mg) was applied, under an occlusive wrap, for 24 h to both forearms of 28 subjects. 10 The area of application was not stated. The study involved a 3-week induction phase, 2-week non-treatment period, and then a challenge phase. After 24 h, the occlusive wraps were removed and 1 forearm of each subject was irradiated for 15 minutes with UVA light (3.3 J) and UVB light (108 to 144 mJ). Induction was repeated 6 times during a 3-week period. The challenge phase began 2 weeks after the last induction (followed by irradiation). The test substance was applied (under an occlusive wrap) for 24 h to a new site on the forearm. After removal of the occlusive wrap, 1 forearm was irradiated with UVA light. Test sites were evaluated immediately after irradiation and at 48 h and 72 h post-application. A challenge reaction (minimal reaction) was observed in 1 subject, only at the site that was irradiated after test substance application. The test substance did not induce contact photoallergy.
Ocular Irritation Studies
In Vitro
PVP and Modified PVP Polymers
PVP
PVP-iodine was severely toxic to corneal endothelium at concentrations of 5% and 10% in a rabbit eye model. 7 An in vitro study of cultured bovine corneal endothelial cells with PVP-iodine concentrations up to 0.1% found that concentrations of 0.05% or less did not induce endothelial cell damage. 7
VP Crosspolymers
Hydrolyzed Wheat Protein/PVP Crosspolymer
The ocular irritation potential of Hydrolyzed Wheat Protein/PVP Crosspolymer (21% solids, i.e., concentration of Hydrolyzed Wheat Protein/PVP Crosspolymer) was evaluated using the SkinEthicTM reconstituted human corneal epithelium model. 44 This test is based on the hypothesis that irritant chemicals are able to penetrate the corneal epithelial tissue and are sufficiently cytotoxic to cause cell death. The tissues were treated with 30 µl of the test substance for 10 min, and the study design consisted of a test for direct reduction of MTT. Triplicate tissues were also treated with 30 µl of a negative control solution (negative control) and 30 µl of 1% w/v sodium dodecyl sulfate (positive control). Following MTT loading, the reduced MTT was extracted from the tissues. After extraction, the absorbency of triplicate aliquots of the extracted MTT solution for each SkinEthicTM tissue was measured. The optical density was measured at 540 nm, and data were presented in the form of % viability (i.e., MTT conversion relative to negative controls). The % relative mean tissue viability of Hydrolyzed Wheat Protein/PVP Crosspolymer was ≥ 60%, classifying the test substance as a non-irritant.
Animal
VP Copolymers
VP/VA Copolymer
The acute ocular irritation potential of VP/VA Copolymer, as supplied and in formulation, was evaluated using albino rabbits. 2 For the solutions that were tested, moderate to severe ocular irritation was observed at a concentration of 50% and mild irritation was observed at concentrations of 25% and 37.5% VP/VA Copolymer. For the product formulations that were tested, ocular irritation (degree not stated) was observed at concentrations of 2.4% and 24% VP/VA Copolymer, minimal to moderate ocular irritation was observed at a concentration of 4%, and concentrations of 0.25%, 0.5%, and 1.75% were non-irritating.
VP Acrylate Copolymers
Ammonium Acryloyldimethyltaurate/VP Copolymer
In an ocular irritation assay, Ammonium Acryloyldimethyltaurate/VP Copolymer (undiluted, 0.1 mL) was non-irritating to the eyes of rabbits. 45
VP/Acrylates/Lauryl Methacrylate Copolymer
Undiluted VP/Acrylates/Lauryl Methacrylate Copolymer was slightly irritating to the eyes of rabbits. 11 The number of animals tested and details relating to the test protocol and study results are not included.
PVP and Modified PVP Polymers
PVP
In ocular irritation studies using rabbits, a 10% PVP-iodine solution (without detergent) was minimally irritating, whereas repeated instillations of 0.5% PVP-iodine did not cause ocular irritation. 8
An in vivo study on rabbits with PVP-iodine up to 1% found concentrations of 0.1% or less did not damage the corneal endothelium. 46
Triacontanyl PVP
In a study involving 6 New Zealand white rabbits, a Triacontanyl PVP trade name material (unknown concentration; 50 mg was instilled into the conjunctival sac of 1 eye of each animal. 10 Untreated eyes served as controls. The eyes of 3 rabbits were rinsed after instillation. Reactions were scored for up to 7 days post-instillation according to the Draize scale. In all treated eyes, slight erythema, edema, and discharge were observed at 1 h post-instillation. Conjunctival irritation persisted for 4 days in 1 eye (unrinsed) and, for 1 day, in 1 rinsed eye. The test substance was classified as a slight ocular irritant.
VP Crosspolymers
Sodium Acryloyldimethyltaurate/VP Crosspolymer
The ocular irritation potential of Sodium Acryloyldimethyltaurate/VP Crosspolymer (test concentration unknown) was evaluated using rabbits in accordance with OECD TG 405. 12 Slight conjunctival effects were observed and had resolved by 24 h. The test substance was classified as slightly irritating.
Human
VP Copolymers
VP/Hexadecene Copolymer
A controlled use test of an eye shadow containing 12.22% VP/Hexadecene Copolymer was performed using 10 healthy female subjects, 5 of whom wore contact lenses. 47 The product was applied to the face (eye region) daily for 2 weeks, and the subjects were examined by an ophthalmologist. No subject had subjective or objective eye irritation in the form of tears or pain, and there was no evidence of eyelid irritation or incompatibility (redness, itching). Furthermore, examination of the eye with a slit-lamp microscope did not reveal any evidence of irritant contact conjunctivitis with chemosis. The authors concluded that the product should be classified as harmless regarding the possibility of eye or eyelid irritation.
Clinical Studies
Case Reports
VP Copolymers
VP/Eicosene Copolymer
An atopic male with a history of xerosis and pruritus of the hands, lower arms, and legs applied a prescribed emollient cream containing VP/Eicosene Copolymer (concentration not stated) daily. 48 Within a month, the patient developed an itchy, vesicular dermatitis of the limbs. Patch testing of the cream was performed, and reactions were scored on days 2 and 3. A mild erythemato-edematous (+) reaction to the cream was observed on both days. In a ROAT in which the cream was applied to the antecubital fossa, a positive reaction was observed within 3 days. At 6 months after resolution of the dermatitis, the patient was patch tested with the cream and its ingredients. Reactions were scored on days 2, 3, and 4. A delayed, but clearly positive, erythemato-edematous reaction (+reaction) to 10% VP/Eicosene Copolymer in petrolatum was observed on day 4. A positive reaction to the cream (+/+) was observed on days 3 and 4. The patch test reaction to VP/Eicosene Copolymer was considered allergic and clinically relevant. Positive reactions were not observed in the 15 control subjects patch tested with VP/Eicosene Copolymer.
Acute facial eczema was observed in a female patient after application of a sunscreen containing VP/Eicosene Copolymer (concentration not stated) and 23 other ingredients. 49 Product application was followed by moderate sun exposure. The patient had a childhood history of eczema. One month later, patch testing (Finn chambers, applied to back) of the ingredient and product was performed. Reactions were scored after days 2 and 3, and a positive reaction (+/+) to the sunscreen was observed. In a second patch test on the sunscreen, the test site was irradiated with UVA (10 J/cm2) on day 2. A positive reaction was observed on days 2 and 3 (+/+). Patch testing of the individual ingredients was also performed, and test results indicated that VP/Eicosene Copolymer was the only ingredient that caused a positive reaction. A positive reaction to this ingredient (1% in petrolatum) was observed on days 2 and 3 (+/+).
PVP and Modified PVP Polymers
PVP
A woman with pollinosis developed anaphylaxis after vaginal application of a PVP-iodine solution for disinfection during a medical examination. 50 Wheal and flare responses (3+) to the PVP-iodine solution (10% aq.), PVP-iodine (0.1% aq.) and PVP (0.001% aq.) were observed following prick tests. In another case study, a man had an anaphylactic reaction minutes after oral ingestion of acetaminophen-containing tablets. 51 A positive test reaction to PVP (5% in water), one of the components of the drug, was reported.
A case of a boy with a history of anaphylactic reactions following treatment for impetigo contagiosum was reported. 52 Skin prick tests with PVP-iodine solution (0.1-100 mg/dl in water) and PVP (K30; 0.1-10 mg/ml in water) were negative. However, in a histamine release test (using peripheral blood basophils), histamine release was observed in a dose-dependent manner after stimulation with PVP in the presence of autologous serum. A rare case of iododerma was reported in a man with a history of diabetes, hypertension, asthma, and gout. 53 Treatment with a 10% topical solution of PVP-iodine resulted in multiple pinpoint pustules (consistent with iododerma) on both lower extremities.
Four days following surgery to treat carpal tunnel syndrome, a woman presented with an acute vesicular dermatitis on her left hand, palm and dorsal surface, and interdigital spaces. 54 These reactions were observed after application of a 10% PVP-iodine solution to the surgical site. Patch testing with PVP-iodine solution (1% diluted in water) caused a positive (4+) reaction. A positive reaction (++) was also observed in the repeated open application test (ROAT).
Severe irritant contact dermatitis resulting in necrosis of the skin occurred in a woman following surgical preparation of her chest and upper abdomen with 10% PVP-iodine solution. 55 A woman with no significant medical history developed transient hypotension, anuric renal failure, hemolysis, coagulopathy, and uterine infarction following intra-uterine injection of 2% PVP-iodine solution as a dye in a hydrotubation procedure. 56 In another report, PVP-iodine-induced irritant contact dermatitis was diagnosed in a woman following antiseptic preparation of a spinal anesthesia site for an emergency Caesarean section. 57
A case of a girl with an anaphylactic reaction to eye drops containing PVP (excipient) has been reported. The patient prick-by-prick test results for a PVP-iodine 7.5% antiseptic solution were positive. 58
Other Clinical Reports
PVP and Modified PVP Polymers
PVP
In the patch testing of 500 consecutive patients with 10% PVP-iodine solution (diluted 10 times in water), 14 patients (2.8%) had a positive reaction to the test material. 59 These patients then underwent ROATs with a PVP-iodine solution and only 2 of the 14 patients tested positive.
Patch testing was performed on 10 patients with a history of contact dermatitis following application of PVP-iodine preparations and positive patch test reactions to the preparations. 60 On days 3 and 5, “+” reactions or stronger were observed in 10/10 patients with 10% PVP-iodine, in 9/9 patients with 5% PVP-iodine, and in 5/9 patients with 2% PVP-iodine. All patients (10/10) had positive reactions to the PVP-iodine preparation tested neat. In the control group, “+” reactions were observed in 3/10 to 5% and 10% PVP-iodine and to the PVP-iodine preparation. No reactions were observed to lower test concentrations or to any of the other components tested. The strong reactions were classified as allergic sensitization.
In a survey of physicians in Japan for occupational allergy, 17 out of 307 reported contact allergy to PVP-iodine. 61 Nineteen patients (12 men and 7 women) developed extensive patchy or linear erythema, sometimes accompanied by bullae and erosion, on both sides of the buttocks, the back and posterior areas of the thighs a few days after operations or cardioangiography. 62 The patients were patch tested with 10% PVP-iodine solution and had strongly positive results (irritant contact dermatitis).
Summary
The safety of 30 vinylpyrrolidone polymers as used in cosmetics is reviewed in this safety assessment; 9 of these ingredients have been reviewed previously. All of these ingredients share in common a vinylpyrrolidone monomer. Most of these ingredients have the reported function of film former in cosmetics in common. Viscosity increasing agent and binder are 2 other functions that are reported for many of these ingredients.
VP/VA Copolymer is produced by free radical copolymerization of NVP and VA in an isopropanol solution, in the presence of initiators. The process is continuous and temperature-controlled. Hydrazine is formed from amines present in this reaction mixture; but specifications limit the concentration to a maximum of 0.1%. Some of the proposed maxima for VP/VA Copolymer, as a food ingredient, in a petitioner’s submission to the EFSA are: vinylpyrrolidone (5 mg/kg), vinyl acetate (5 mg/kg), and hydrazine (1 mg/kg).
Due to the method of production (radical polymerization) provided by one supplier, residual monomers may be present in VP/VA Copolymer at a maximum of 50 ppm vinylpyrrolidone and a maximum of 100 ppm vinyl acetate. Other suppliers reported that radical polymerization is used to make PVP. Due to production via this method, residual monomers may be present at a maximum of 100 ppm vinylpyrrolidone and a maximum of 100 ppm vinyl acetate. Impurities that may be present are acetaldehyde at a maximization concentration of 100 ppm and heavy metals in sum (as lead) at a maximum of 10 ppm.
One supplier reported that Maltodextrin
According to 2018 VCRP data, the greatest use frequency is reported for PVP, which is being used in 900 cosmetic products (798 leave-on products + 101 rinse-off products + 1 product diluted for bath use). The second highest use frequency is being reported for Ammonium Acryloyldimethyltaurate/VP Copolymer (597 cosmetic products: 525 leave-on products + 62 rinse-off products). In general, the differences in current use frequencies of vinylpyrrolidone polymers in cosmetics versus those reported in previous years are unremarkable.
The results of a concentration of use survey conducted in 2017 indicate that VP/VA Copolymer is being used at concentrations up to 44% in rinse-off products (paste masks and mud packs), which is the highest maximum ingredient use concentration that is being reported for vinylpyrrolidone polymers. Notably, in 2003, the highest maximum use concentration of VP/VA Copolymer in rinse-off products was 10%, which is 4-fold lower than the current highest maximum use concentration in rinse-off products. The highest maximum ingredient use concentration of vinylpyrrolidone polymers in leave-on products is being reported for PVP, which is used at concentrations up to 35% in leg and body paints. Notably, in 2013, the highest maximum use concentration of PVP in leave-on products was lower, 12%. Thus, the highest maximum use concentration of PVP in leave-on products is approximately 3-fold greater than the highest maximum use concentration of this ingredient in leave-on products that was reported in 2013. It should also be noted that 35% was the maximum cosmetic use concentration that was reported in the original final report on PVP that was published in 1998.
A single dose of a Triacontanyl PVP (unknown concentration) trade name material was applied, under an occlusive wrap, for 24 h to the backs of 10 New Zealand white rabbits; the acute dermal LD50 was > 2 g/kg. An oral LD50 of > 5 g/kg (rats) has been reported for undiluted VP/Acrylates/Lauryl Methacrylate Copolymer.
A single 5 g/kg oral dose of a Triacontanyl PVP trade name material (ground into a powder and mixed with peanut butter and honey) was fed to 10 Sprague-Dawley rats. None of the animals died and no gross organ changes were observed at necropsy. The LD50 was > 5 g/kg.
VP/VA Copolymer was administered in the diet of 3 groups of male and female Sprague-Dawley rats (5 animals/sex/group) for 28 days at doses up to 1000 mg/kg/day. There were no clinical signs of toxicity or test substance-related macroscopic or microscopic tissue changes. The short-term (28 days) oral toxicity of a PVP trade name material (5% w/v in water) was evaluated using 2 groups of 6 HanWistar rats (RccHan:WIST strain). There were no toxicologically relevant effects on body weight gain, food consumption, or water consumption, and there were no treatment-related microscopic changes.
In a 90-day study, VP/VA Copolymer was also administered in the diet of 3 groups of male and female Sprague-Dawley rats (10 animals/sex/group) at doses up to 1000 mg/kg/day. There were no clinical signs of toxicity or test substance-related macroscopic or microscopic tissue changes.
The chronic oral toxicity of VP/VA Copolymer was evaluated using groups of 50 male and 50 female Wistar rats of the Chbb:THOM (SPF) strain. The groups were fed the test substance (in the diet) for 2 years, and 2759 mg/kg/day was the highest dose that was administered. There were no effects on survival and no remarkable test substance-related clinical signs in any of the dose groups. The vast majority of the gross lesions were comparable to the incidence in controls, and there was no clear dose-response relationship. A 2-year feeding study on VP/VA Copolymer (5% in diet) was also performed using groups of 102 Sprague-Dawley rats. Survival in both the control and test groups was described as poor (8% to 14%), due to inflammatory diseases of the respiratory tract. There were no signs of toxicity in test or control rats, and no treatment-related clinical chemistry changes. No gross pathologically detectable lesions were observed. However, at microscopic examination, an increased incidence of liver congestion and fatty degeneration was observed.
In a chronic (52 weeks) feeding study, groups of 8 to 12 Beagle dogs were fed VP/VA Copolymer in the diet, and 2522 mg/kg/day was the highest dose that was administered. None of the animals died and no treatment-related clinical signs were observed. At gross and microscopic examinations, the type and incidence of findings were comparable between test and control groups.
The genotoxicity of aqueous PVP (doses up to 10,000 µg/plate) was evaluated in the Ames test (with and without metabolic activation) using the S. typhimurium TA98, TA100, TA1535, and TA1537. Results were negative. Sodium Acryloyldimethyltaurate/VP Crosspolymer was also non-genotoxic (doses not stated) in the Ames test, and the same was true for a Triacontanyl PVP trade name material (doses up to 2500 µg/plate, with and without metabolic activation).
The carcinogenicity of VP/VA Copolymer was evaluated using groups of 100 male and female Wistar rats of the Chbb:THOM (SPF) strain. The groups were fed the test substance (in the diet) for 2 years, and 2759 mg/kg/day was the highest dose that was administered. All neoplastic and non-neoplastic microscopic findings were considered to have developed spontaneously and were not related to treatment. A 2-year feeding study on VP/VA Copolymer (5% in diet) was also performed using groups of 102 Sprague-Dawley rats. No treatment-related tumors or other gross pathologically detectable lesions were induced.
The skin irritation potential of Hydrolyzed Wheat Protein/PVP Crosspolymer (21% solids, i.e., concentration of Hydrolyzed Wheat Protein/PVP Crosspolymer) was evaluated using the EpiskinTM reconstituted human epidermis model. The relative mean viability of tissues treated with Hydrolyzed Wheat Protein/PVP Crosspolymer for 15 min was 86.6%, and the test substance was classified as a non-irritant.
A Triacontanyl PVP trade name material (0.5 g, moistened with saline) was slightly irritating to the skin of 6 New Zealand white rabbits. The skin irritation potential of a cosmetic base containing 14.95% VP/Hexadecene Copolymer (undiluted) was evaluated in an occlusive patch test involving 50 subjects (27 with normal skin; remainder with eczema, allergy, or sensitive skin). Skin irritation was not observed.
Results for an LLNA of Sodium Acryloyldimethyltaurate/VP Crosspolymer (test concentration not stated) were negative. The skin sensitization potential of a Triacontanyl PVP (unknown concentration) trade name material (∼200 mg) was evaluated in an HRIPT involving 102 subjects, and results were negative. In an HRIPT involving 105 subjects, VP/Acrylates/Lauryl Methacrylate Copolymer (96%) induced minimal erythema in 6 and 2 subjects during the induction and challenge phases, respectively. These reactions were not considered positive.
In a study involving 10 subjects, a Triacontanyl PVP (unknown concentration) trade name material (∼200 mg) did not induce a contact dermal phototoxic response in the presence of UVA light. During the induction phase of a photoallergenicity study, a Triacontanyl PVP (unknown concentration) trade name material (200 mg) was applied to the forearms of 28 subjects. A challenge reaction (minimal reaction) was observed in 1 subject, only at the site that was irradiated after test substance application. It was concluded that the trade name material did not induce contact photoallergy.
The ocular irritation potential of Hydrolyzed Wheat Protein/PVP Crosspolymer (21% solids, i.e., concentration of Hydrolyzed Wheat Protein/PVP Crosspolymer) was evaluated using the SkinEthicTM reconstituted human corneal epithelium model. The % relative mean tissue viability of Hydrolyzed Wheat Protein/PVP Crosspolymer was ≥ 60%, classifying the test substance as a non-irritant.
An unknown concentration of Sodium Acryloyldimethyltaurate/VP Crosspolymer was classified as slightly irritating to the eyes of rabbits. Undiluted VP/Acrylates/Lauryl Methacrylate Copolymer was also slightly irritating to the eyes of rabbits. In a study involving 6 New Zealand white rabbits, a Triacontanyl PVP (unknown concentration) trade name material (50 mg) was classified as slightly irritating. A controlled use test of an eye shadow containing 12.22% VP/Hexadecene Copolymer was performed using 10 healthy female subjects. The product was applied to the face (eye region) daily for 2 weeks, and neither ocular or eyelid irritation was observed in any of the subjects.
An allergic (erythemato-edematous) reaction was observed in an atopic patient patch-tested with 10% VP/Eicosene Copolymer in petrolatum, but not in 15 control subjects. Acute facial eczema was observed in a female patient after application of a sunscreen containing VP/Eicosene Copolymer (concentration not stated). When the patient was patch-tested with the ingredient (1% in petrolatum), a positive reaction was observed.
Discussion
The Panel determined that the data were sufficient to conclude on the safety of 27 vinylpyrrolidone polymers, but additional data are needed for completion of the safety assessment of the remaining 3 vinylpyrrolidone polymers. The 27 vinylpyrrolidone polymers for which safety could be assessed comprise the following 4 subgroups combined: VP Copolymers, VP Acrylate Copolymers, Polyvinylpyrrolidone (PVP) and Modified PVP Polymers, and PVP Crosspolymers. For these 27 ingredients deemed safe, the Panel noted that concern over the lack of dermal absorption data was mitigated by large ingredient molecular weights, low residual monomer content, and related compositional characteristics and physical properties, despite differences in monomer identities. Regarding 2 of the 27 ingredients, Methacrylic Acid/Styrene/VP Copolymer and VP Acrylates/Lauryl Methacrylate Copolymer, the Panel noted that were these ingredients to be used in products that are applied to the nail they would be safe as used in nail products when skin contact is avoided. Furthermore, the Panel noted that products containing these 2 ingredients should be accompanied with directions to avoid skin contact, because of the sensitizing potential of methacrylates.
The Panel also noted that the production process for VP/VA Copolymer can yield hydrazine from amines that are present in the reaction mixture, and also considered that the Food Chemicals Codex acceptance criteria specifies that food use of PVP cannot contain more than 1 ppm hydrazine. The cosmetics industry should continue to use current good manufacturing practices (cGMPs) to limit impurities of prospective toxicological concern, including hydrazine and acrylates.
The issue of incidental inhalation exposure from powders and hair sprays was discussed by the Panel. The Council’s survey results indicate that VP/VA Copolymer is being used in both pump hair sprays (maximum use concentrations up to 9%) and aerosol hair sprays (maximum use concentrations up to 10%). VP/Eicosene Copolymer is being used in face powders at concentrations up to 0.5%. The Panel noted that in aerosol products, most droplets/particles would not be respirable to any appreciable amount. Furthermore, droplets/particles deposited in the nasopharyngeal or bronchial regions of the respiratory tract present no toxicological concerns based on the chemical and biological properties of these ingredients. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at https://www.cir-safety.org/cir-findings.
The subgroup of 3 vinylpyrrolidone polymers for which data are insufficient to determine safety comprise, in part, the residues of urethane monomers; and the data needed for this subgroup are: • Residual monomer concentration for at least one representative ingredient from this subgroup
In addition to the above request, the Panel agreed that data on the molecular weight range for the 3 urethane monomers should be provided.
Conclusion
The Expert Panel for Cosmetic Ingredient Safety determined that the following 27 vinylpyrrolidone polymers are safe in cosmetics in the present practices of use and concentration described in the safety assessment:
Acrylic Acid/VP Crosspolymer
Maltodextrin/VP Copolymer
PVP/Decene Copolymer*
PVP/VA/Itaconic Acid Copolymer*
PVP/VA/Vinyl Propionate Copolymer*
Styrene/VP Copolymer
Triacontene/VP Copolymer*
VP/Eicosene Copolymer
VP/Hexadecene Copolymer
VP/VA Copolymer
VP/Vinyl Alcohol Copolymer*
Acrylates/Stearyl Methacrylate/VP Copolymer*
Acrylates/VP Copolymer
Ammonium Acryloyldimethyltaurate/VP Copolymer
Ethylhexyl Acrylate/VP/Dimethicone Methacrylate Copolymer*
Ethylhexyl Methacrylate/Methyl Methacrylate/VP Copolymer*
Methacrylic Acid/Styrene/VP Copolymer*
Vinyl Caprolactam/VP/Dimethylaminoethyl Methacrylate Copolymer
VP/Acrylates/Lauryl Methacrylate Copolymer
VP/Dimethylaminoethylmethacrylate Copolymer
VP/DMAPA Acrylates Copolymer
VP/Vinyl Caprolactam/DMAPA Acrylates Copolymer
Butylated PVP
PVP
Triacontanyl PVP
Hydrolyzed Wheat Protein/PVP Crosspolymer
Sodium Acryloyldimethyltaurate/VP Crosspolymer
*Not reported to be in current use. Were the ingredients in this group not in current use to be used in the future, the expectation is that it would be used in product categories and at concentrations comparable to others in this group.
The Panel also concluded that the available data are insufficient to make a determination that the vinylpyrrolidone polymers listed below are safe under the intended conditions of use in cosmetic formulations:
VP/Dimethiconylacrylate/Polycarbamyl/Polyglycol Ester
VP/Dimethylaminoethylmethacrylate/Polycarbamyl Polyglycol Ester
VP/Polycarbamyl Polyglycol Ester
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 555 13th St., NW, Suite 300W, Washington, DC 20004, USA.
Author Contributions
The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Declaration of Conflicting Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
