Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) assessed the safety of Triphenyl Phosphate, which is reported to function as a plasticizer in manicuring products. The Panel reviewed the available data to determine the safety of this ingredient. The Panel concluded that Triphenyl Phosphate is safe in cosmetics in the present practices of use and concentration described in this safety assessment.
Keywords
Introduction
Triphenyl Phosphate is reported to function as a plasticizer in cosmetics, as described by the web-based International Cosmetic Dictionary and Handbook (wINCI; Dictionary). 1 In cosmetic products, this ingredient is used exclusively in manicuring preparations, including nail polishes and enamels.
This safety assessment includes relevant published and unpublished data that are available for each endpoint that is evaluated. Published data are identified by conducting an exhaustive search of the world’s literature. A listing of the search engines and websites that are used and the sources that are typically explored, as well as the endpoints that the Panel typically evaluates, is provided on the Cosmetic Ingredient Review (CIR) website (respectively, https://www.cir-safety.org/supplementaldoc/preliminary-search-engines-and-websites; https://www.cir-safety.org/supplementaldoc/cir-report-format-outline). Unpublished data are provided by the cosmetics industry, as well as by other interested parties.
Some chemical and toxicological data on Triphenyl Phosphate included in this safety assessment were obtained from robust summaries of data submitted to the European Chemical Agency (ECHA) by companies as part of the REACH chemical registration process. Additionally, some data were obtained from an assessment by the Organisation for Economic Co-Operation and Development Screening Information Data Sets (OECD SIDS). These data summaries are available on the ECHA and OECD SIDS websites, respectively, and when appropriate, information from the summaries has been included in this report.
Chemistry
Definition
Triphenyl Phosphate is the organic compound that conforms to the structure in Figure 1.
1
It is reported to function as a plasticizer in cosmetic products. Triphenyl Phosphate.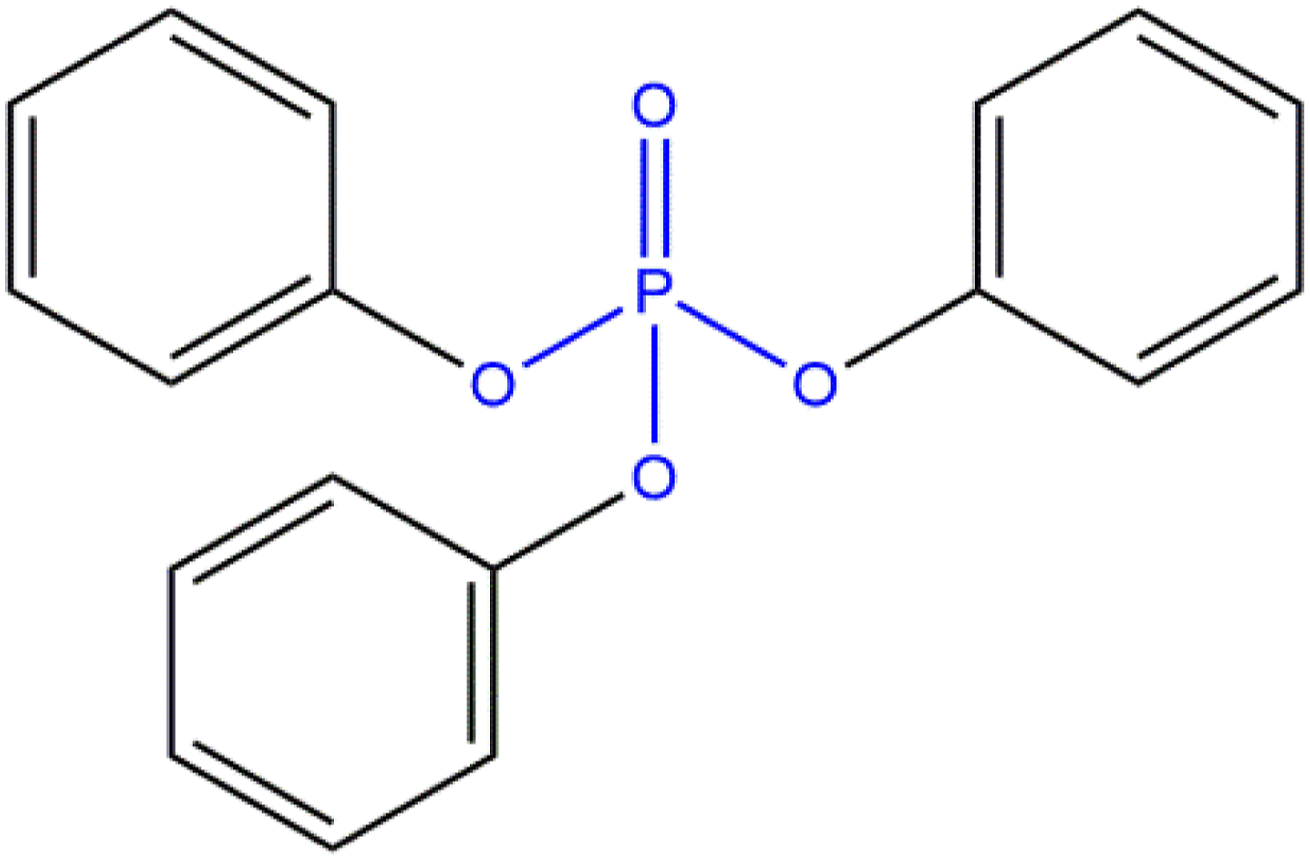
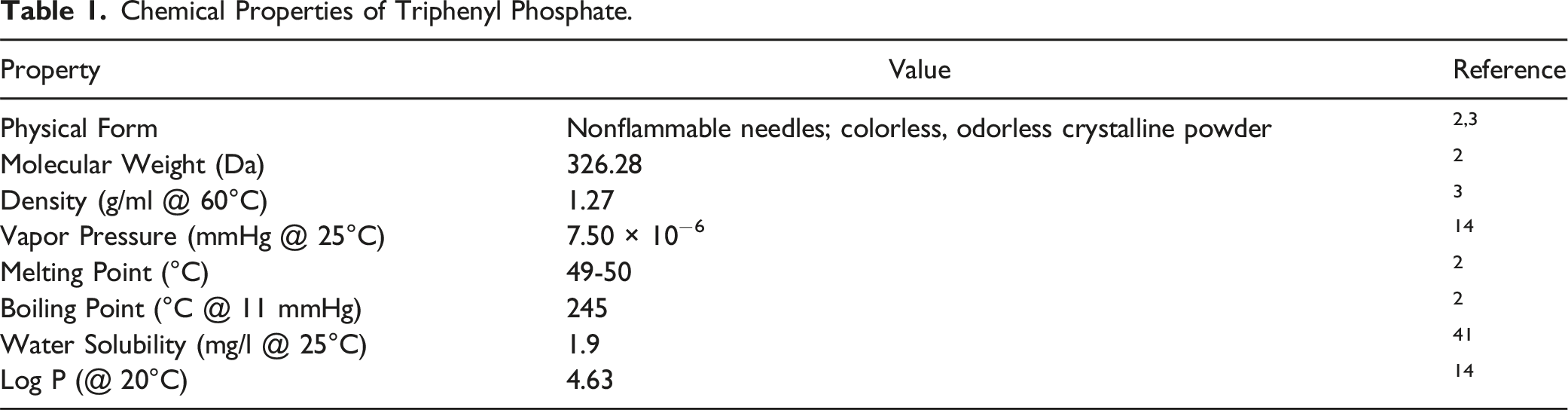
Chemical Properties
Method of Manufacturing
According to one source, Triphenyl Phosphate can be prepared by reacting metaphosphoric anhydride and phenol or by reacting triethyl phosphite with sodium p-toluenesulfonchloramide. 2 Triphenyl Phosphate can also be derived by reacting phenol and phosphorus oxychloride.3,4
Composition/Impurities
The purity of Triphenyl Phosphate is reported to be greater than or equal to 99.6% w/w. 4 Impurities may include water, phenol, and esters.
Ultraviolet (UV) Absorption
In spectral analysis of Triphenyl Phosphate, no maximum UV absorption peaks were observed in the UVA and UVB ranges. 5
Use
Cosmetic
The safety of the cosmetic ingredient included in this assessment is evaluated based on data received from the US Food and Drug Administration (FDA) and the cosmetics industry on the expected use of this ingredient in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in the FDA Voluntary Cosmetic Registration Program (VCRP) database. Use concentration data are submitted by the cosmetics industry in response to surveys, conducted by the Personal Care Products Council (Council), of maximum reported use concentrations by product category.
According to 2018 VCRP data, Triphenyl Phosphate is used solely in 331 nail products, with the majority of the uses (286) being reported in nail polishes and enamels. 6 The results of the concentration of use survey conducted in 2017 by the Council indicate that Triphenyl Phosphate is used solely in leave-on manicuring preparations, at a maximum use concentration range of 1% to 14.5%, with the highest maximum concentration of use reported to be in polish strips. 7 Use concentrations were reported to be at up to 11.9% for nail enamels and at up to 1% in nail lotions.
Triphenyl Phosphate is not restricted from use in any way under the rules governing cosmetic products in the European Union. 8 OECD SIDS determined this chemical is low priority for further work regarding human health impact due to its low hazard potential. 4
Non-Cosmetic
Triphenyl Phosphate is a fire retarding agent and plasticizer for cellulose acetate and nitrocellulose. 3 Triphenyl Phosphate is a noncombustible substitute for camphor in celluloid; it is also used to render acetylcellulose, nitrocellulose, airplane “dope,” etc., stable and fireproof; impregnating roofing paper; plasticizer in lacquers and varnishes. 2 Triphenyl Phosphate is an approved indirect food additive in substances for use only as components of adhesives (21 CFR 175.105).
Toxicokinetics
Dermal Penetration
In Vitro
The dermal uptake and percutaneous penetration of Triphenyl Phosphate and other organophosphate esters was studied using human skin in Franz diffusion cells. 9 The exposed skin area in the mounted Franz diffusion cell was 2.64 cm2 and 16.6 ml was the average volume of the receptor chamber. The receptor fluid was an aqueous solution of 0.9% sodium chloride, 5% bovine serum albumin, 40 mg/l hexamycin, and disodium phosphate buffer (to pH 7.4). The skin was dosed with 1000 ng Triphenyl Phosphate in 500 μl ethanol:toluene (4:1) solution to cover the entire skin surface. The diffusion cells were studied at 24, 48, and 72 h after dosing and the donor cell wash, epidermis, dermis and receptor fluid were analyzed for the ester content. When compared to the other esters, Triphenyl Phosphate tended to build up in the skin tissues, primarily in the upper layers. Only “smaller amounts” of Triphenyl Phosphate permeated the skin and reached the receptor fluid within 72 h.
Absorption, Distribution, Metabolism, Distribution
In Vitro
In an in vitro metabolism study, Triphenyl Phosphate incubated with rat liver homogenate (without nicotinamide adenine dinucleotide phosphate (NADPH) and soluble fractions) was determined by gas chromatography to be metabolized to diphenyl phosphate via hydrolysis. 10 Triphenyl Phosphate was prepared in an ethanol solution at 0.0004 M.
In a qualitative in vitro metabolism study on phosphate flame retardants and plasticizers in human liver S9 fraction and microsomes, Triphenyl Phosphate was mainly transformed to a diester metabolite and to a hydroxylated metabolite. 11
In a related study of phosphate flame retardants, the metabolite formation from Triphenyl Phosphate was characterized using primary human hepatocytes. 12 Cryopreserved human hepatocytes were thawed and suspended in media with 20 μM Triphenyl Phosphate for up to 2 h. Extracts of these materials were then analyzed by liquid chromatography-quadrupole-time-of-flight mass spectrometry. This analysis found that diphenyl phosphate corresponded to less than half of the depletion of Triphenyl Phosphate following the 2-hour exposure. Other metabolites, mainly sulfate and glucuronide conjugates, were produced at lower rates.
Human
The potential for Triphenyl Phosphate to be absorbed during cosmetic application was assessed in human volunteers. 13 Two cohorts (26 volunteers total) were recruited to assess the exposure of Triphenyl Phosphate by fingernail painting. The volunteers provided urine samples before and after applying a polish containing 0.97% Triphenyl Phosphate by weight. The metabolite, diphenyl phosphate, was then measured in urine samples (n = 411). Prior to application, the geometric mean of diphenyl phosphate for the control samples was 0.96 ng/ml. The concentration of diphenyl phosphate was found to increase nearly seven-fold approximately 10–14 hours after fingernail painting (13.02 ng/ml; P < 0.001). To determine relative contributions of inhalation and dermal exposure, 10 volunteers total also painted their own nails and synthetic nails adhered to gloves on two separate occasions. Urine was then collected for 24 hours following applications for metabolite analysis. Urinary diphenyl phosphate was significantly diminished (near background concentration; geometric mean not reported) when the volunteers wore gloves, allowing the researchers to suggest that the primary route of exposure is dermal.
Toxicological Studies
Acute Toxicity Studies
Acute Toxicity Studies of Triphenyl Phosphate.

Short-Term and Subchronic Toxicity Studies
Short-Term and Subchronic Toxicity Studies for Triphenyl Phosphate.

Developmental and Reproductive Toxicity (DART) Studies
The effects of 300 to 7500 ppm Triphenyl Phosphate on the reproductive organs were also investigated in the 90-day dietary study (see Short-Term and Subchronic Toxicity Studies, summarized in Table 3). 14 No adverse effects were observed during microscopic examination or weight measurements of the gonads (males: testes and epididymes, seminal vesicles including coagulating glands; females: ovaries, uterus including cervix, vagina) at dietary doses up to 7500 ppm.
The effects of Triphenyl Phosphate on prenatal development were studied in pregnant New Zealand rabbits in accordance with the OECD Test Guideline (TG) 414. 14 The dams received Triphenyl Phosphate in 1% aqueous carboxymethyl cellulose once daily via gavage from days 6 to 28 post-coitum at doses of 0, 32, 80 and 200 mg/kg bw/day. The dams were checked daily for clinical signs of toxicity, and feed consumption and body weights were measured periodically. Dams that survived to day 29 post-coitum were killed and underwent external, thoracic, and abdominal macroscopic examinations. The uteri, placentas, and ovaries were examined, and the numbers of fetuses, early and late resorptions, total implantations, and corpora lutea were recorded. Gravid uterine weights were recorded, and net body weights and net body weight changes were calculated. The fetuses were weighed, sexed, and examined for external, visceral, and skeletal malformations and developmental variations.
No adverse effects were observed in any of the maternal parameters investigated in this study, including mortality, clinical signs, body weights, food consumption, and macroscopic examination. No adverse effects were noted in any of the developmental parameters investigated in this study, including litter size, sex ratio, fetal body weights, external, visceral and skeletal developmental malformations or variations. The authors of this study concluded that the maternal and developmental NOAELs for Triphenyl Phosphate are at least 200 mg/kg bw/day each, based on the absence of adverse effects. 14
The teratogenic potential of Triphenyl Phosphate was investigated in Sprague-Dawley rats. 19 Groups of 40 male and 40 female rats received 0%, 0.25%, 0.50%, 0.75%, or 1.0% Triphenyl Phosphate in their feed from 4 weeks post-weaning through mating and gestation (91 days). Daily intake of Triphenyl Phosphate during pregnancy was determined to be 0, 166, 341, 516, and 690 mg/kg bw, respectively (no further details on the males were provided). Body weights of the pregnant rats and feed consumption were measured on days 7 and 14 of gestation and before laparotomies were performed on day 20. The dams were observed daily for clinical signs of toxicity. The major organs were examined, and the ovaries were removed and examined for numbers of corpora lutea. The gravid uterus was removed and weighed. Litter size and resorptions were recorded. The fetuses were examined for gross abnormalities, sexed, weighed, measured, and underwent skeletal or visceral examinations.
In general, feed consumption was slightly greater in the treated animals than in the controls, except during days 0–7 of gestation. Maternal body weights of the treated animals on gestation day 0 were similar to the controls, except for the high dose group, which were significantly lower. Body weight gains during pregnancy and adjusted body weight gain excluding the gravid uterus had dose-dependent decreases, but were not significant. No toxic effects to reproduction or development were observed in the dams or the offspring at any dose level. Slight increases in the number of soft tissue variations were observed, but these were not dose-related. Number and type of developmental anomalies in the treated groups were comparable to those in the controls. The authors of the study concluded that Triphenyl Phosphate was not teratogenic in this rat study. 19
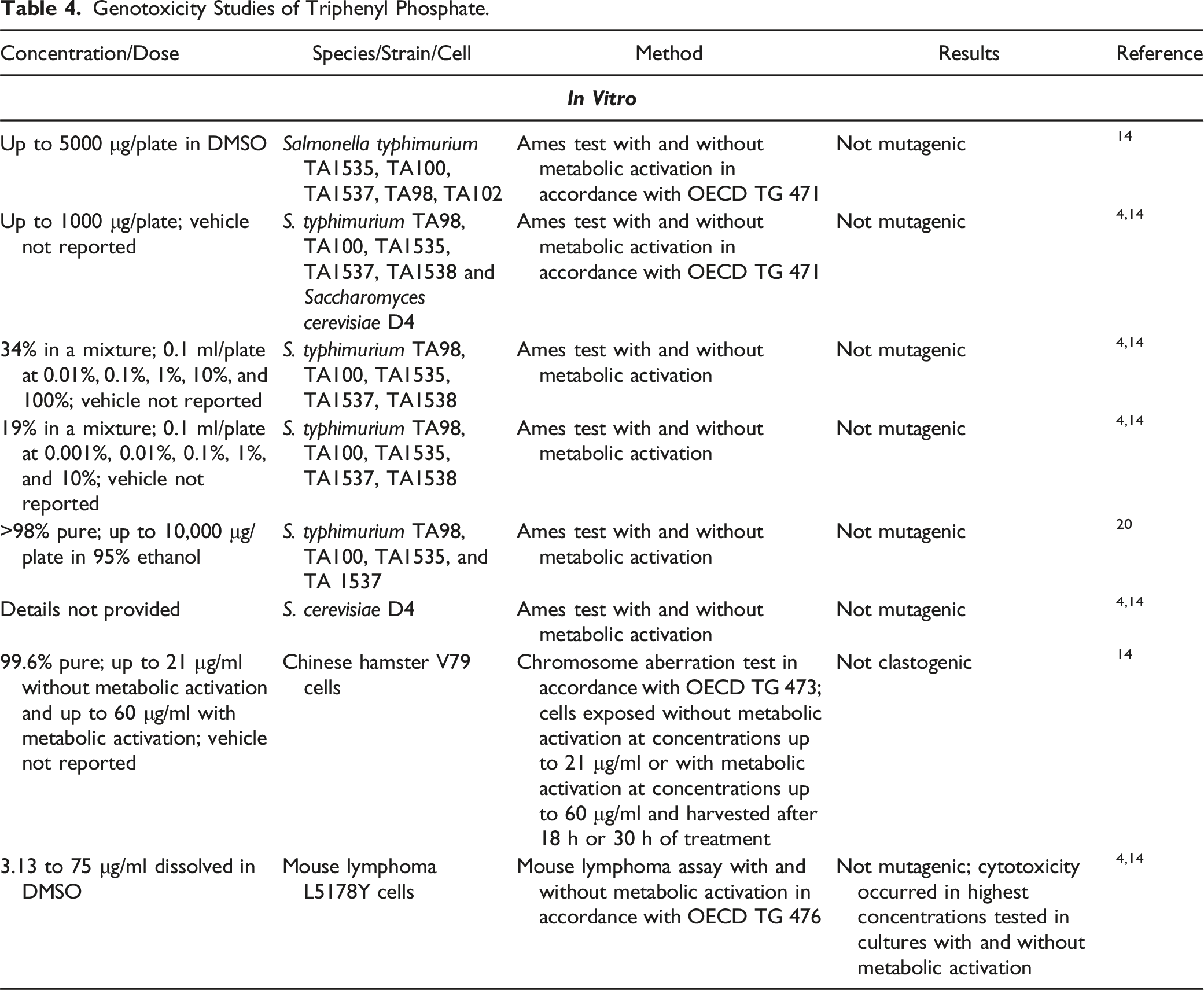
Genotoxicity Studies
Genotoxicity Studies of Triphenyl Phosphate.
Carcinogenicity Studies
No relevant published carcinogenicity studies on Triphenyl Phosphate were identified in a literature search for this ingredient, and no unpublished data were submitted.
Other Relevant Studies
Endocrine Activity
In Vitro
The effects of Triphenyl Phosphate (>99% pure, dissolved in 0.1% dimethyl sulfoxide [DMSO]) on induction of oxidative stress and gene expression were investigated in the murine Leydig cell line, TM3. 21 The TM3 cells were cultured in 0, 20, or 60 μg/ml Triphenyl Phosphate for up to 24 h. After 24 h exposure, cell growth declined and morphology changed in the high dose groups. Significant increases were observed in superoxide dismutase, catalase, glutathione peroxidase, and glutathione S-transferase activities and their respective gene expressions in a dose-dependent and/or time-dependent manner in Triphenyl Phosphate treated groups. Triphenyl Phosphate significantly reduced the expression of main genes related to testosterone (T) synthesis, especially in the high dose group at 24 h. Triphenyl Phosphate treatments for 24 h caused significant decreases in T levels in the medium. Co-treatments of human chorionic gonadotropin (hCG) with Triphenyl Phosphate could inhibit hCG-induced changes in the expression of testosterone and testosterone synthesizing genes. The authors of the study concluded that Triphenyl Phosphate could induce oxidative stress and endocrine activation in TM3 Leydig cells.
In vitro reporter gene assays indicated Triphenyl Phosphate had potential endocrine-disrupting properties. As an agonist, Triphenyl Phosphate could stimulate estrogen receptor α/β (ER α/ERβ), and pregnane X receptor (PXR) activity. As an antagonist, Triphenyl Phosphate could inhibit androgen receptor (AR) and glucocorticoid receptor (GR) activity. In comparison, Triphenyl Phosphate showed no effects on the activity of thyroid hormone receptor α (TRα), TRβ, retinoic acid receptor α (RARα), retinoid X receptor α (RXRα), peroxisome proliferator-activated receptor (PPARα), or PPARγ. 22 In rat pituitary cells (GH3), significant up-regulation of thyroid hormone receptor α/β (trα/trβ) genes was observed following exposure to 10 or 100 μg/l Triphenyl Phosphate, and the expression of thyroid-stimulating hormone β (tshβ) gene was significantly up-regulated by exposure to 100 μg/l Triphenyl Phosphate. 23 In rat thyroid follicular cells (FRTL-5), the expression of the sodium/iodide symporter (nis) and thyroid peroxidase (tpo) genes was significantly up-regulated after exposure to after exposure to 3 or 10 mg/l Triphenyl Phosphate. The authors of the study pointed out these results suggested Triphenyl Phosphate could stimulate thyroid hormone synthesis in the thyroid gland.
Triphenyl Phosphate showed statistically significant estrogenic activity, with an EC20 value of 88 μM, measured by flow-cytometric proliferation assay, in MCF-7 human breast adenocarcinoma cells. 24 In MVLN cells, a bioluminescent MCF-7-derived cell line, Triphenyl Phosphate acted as estrogen receptor antagonists by inhibiting binding of 17β-estradiol (E2) to estrogen receptor. 25
In Vivo
The same researchers evaluated the effects of Triphenyl Phosphate (>99% pure) on the induction of oxidative stress and endocrine activation in groups of 7 ICR male mice. 26 The mice received 0, 100, or 300 mg/kg/bw Triphenyl Phosphate in feed daily for 35 days. At the end of the exposure period, the mice were killed, and livers and testes were removed and weighed. The livers were then homogenized and underwent enzyme analysis, while the testes underwent histopathological examination. Gene expression analysis was performed on the total RNA from the livers and testes.
Compared to the control group, statistically significant decreases in body and testes weights were observed in the 300 mg/kg Triphenyl Phosphate-treated mice. Hepatic malondialdehyde content increased significantly in a dose-dependent manner, while the contents of glutathione decreased significantly in the 300 mg/kg dose group. Triphenyl Phosphate exposure affected hepatic activities of antioxidant enzymes including glutathione peroxidase (increased activity), catalase (increased activity), and glutathione S-transferase (decreased activity) as well as related gene expression. In the testes, exposure to 300 mg/kg Triphenyl Phosphate resulted in histopathological damage and a decrease of testicular testosterone levels, whereas no morphologic changes, except a slight reduction of Sertoli cells, were observed in the 100 mg/kg dose group. The expression of the main genes related to testosterone synthesis, including steroidogenic acute regulatory protein, low-density lipoprotein receptor, cytochrome P450 cholesterol side-chain cleavage enzyme, and cytochrome P450 17α-hydroxysteroid dehydrogenase in the testes also was decreased after the exposure to 300 mg/kg Triphenyl Phosphate. The authors of the study concluded that Triphenyl Phosphate induced oxidative stress and endocrine activation in mice. 26
In zebrafish (Danio rerio) larvae at 7 days post-fertilization, exposure to Triphenyl Phosphate from 40 to 500 μg/l led to significant increases in both triiodothyronine (T3) and thyroxine (T4) concentrations and influenced the expression of several genes associated with the thyroid system, including corticotrophin-releasing hormone (crh), tshβ, trα, trβ, nis and tg genes. 23 Exposure to Triphenyl Phosphate also significantly upregulated the expression of the genes related to the metabolism (dio1), transport (ttr), and elimination (ugt1ab) of thyroid hormones.
After 14-day of zebrafish exposure, Triphenyl Phosphate significantly increased plasma E2 concentration and decreased testosterone (T) and 11-ketotestosterone (11-KT) among male fish. 25 Among females, both plasma T and E2 concentrations increased but there was no change of 11-KT. Transcription of cytochrome P450 (CYP)17 and CYP19a genes in gonad was significantly up-regulated in both sexes, while the vitellogenin (VTG) 1 gene in the liver was down- and up-regulated in female and male fish, respectively. In another study, after 21-day of zebrafish exposure to Triphenyl Phosphate, there was a significant decrease in fecundity along with significant increases of plasma E2 concentrations, VTG levels, and E2/T and E2/11-KT ratios were observed. 27 Several genes of the hypothalamus–pituitary–gonad (HPG) axis changed after the exposure in a sex-dependent manner. Overall, the zebrafish study authors concluded Triphenyl Phosphate could alter sex hormone balance through several mechanisms including alterations of steroidogenesis or estrogen metabolism.
Neurotoxicity
The effects of dietary exposure of Triphenyl Phosphate on neuromotor function were studied in a 4 month study in rats. 17 Groups of 10 male Sprague-Dawley rats received 0, 0.25%, 0.50%, 0.75%, or 1.0% Triphenyl Phosphate in their feed ad libitum. Daily doses were determined to be 0, 161, 345, 517, and 711 mg/kg/day, respectively. Behavioral tests including measures for motility, exploratory behavior, balance and general motor coordination, and muscular strength were performed on a monthly basis. No treatment-related effects were noted in the behavioral assessments at any of the monthly test sessions. The study authors concluded that Triphenyl Phosphate at up to 1.0% in a 4-month dietary study in rats did not cause neurotoxicity.
Immunotoxicity
The potential immunotoxic effects of Triphenyl Phosphate were examined in a dietary study in rats.14,18 Groups of 10 male and 10 female Spartan Sprague-Dawley rats received feed containing 0, 0.25%, 0.5%, 0.75%, and 1% Triphenyl Phosphate for 120 days. Total protein analysis and electrophoretic analyses of serum proteins were performed. Immunotoxicity was assessed by measurements of the weights of lymphoid organs, immunohistochemical evaluation of spleen, thymus, and lymph nodes using immunoperoxidase staining (rabbit serum anti-rat Ig and sheep serum anti-rabbit IgG adsorbed with rat Ig), and the humoral response to antigens in sheep red blood cells.
A trend towards an increase in thymus weights was observed in male rats in the 0.75% dose group, but little to no differences were observed in the 1% dose group. No significant changes in spleen weights were observed. No significant changes were found in these organs and lymph nodes during histopathologic examinations. No significant alterations of serum protein were detected. Electrophoresis revealed increased levels of alpha- and beta-globulin in male and female rats but effects were similar at all dose levels, relative to the control group. There were no significant differences between animals immunized with sheep red blood cells and non-immunized animals. Only non-dose-dependent variation was found in the humoral immune response to sheep red blood cells in female rats. The authors of this dietary rat study concluded that the NOEL for immunotoxicity was 1% Triphenyl Phosphate.14,18
Cytotoxicity
The cytotoxic potential of Triphenyl Phosphate was studied is several different cultured cell lines. 28 The test material was dissolved in DMSO (0.5%) and diluted in minimum essential medium and cultured with human (KB and HEL-R66), monkey (Vero) or dog (MDCK) cells for 72 h. After the incubation period, the number of viable cells was determined and compared to the DMSO control. Inhibition of growth by Triphenyl Phosphate was observed in a dose dependent manner in all cell lines. The dose that inhibited cell multiplication by 50% (ID50) was 0.6 mM and 0.5 mM for the KB and HEL-R66 cell lines, respectively, 0.4 mM for the Vero cell line, and 0.5 mM for the MDCK cell line. The authors concluded Triphenyl Phosphate is toxic to the human, monkey and dog cell lines described in this study.
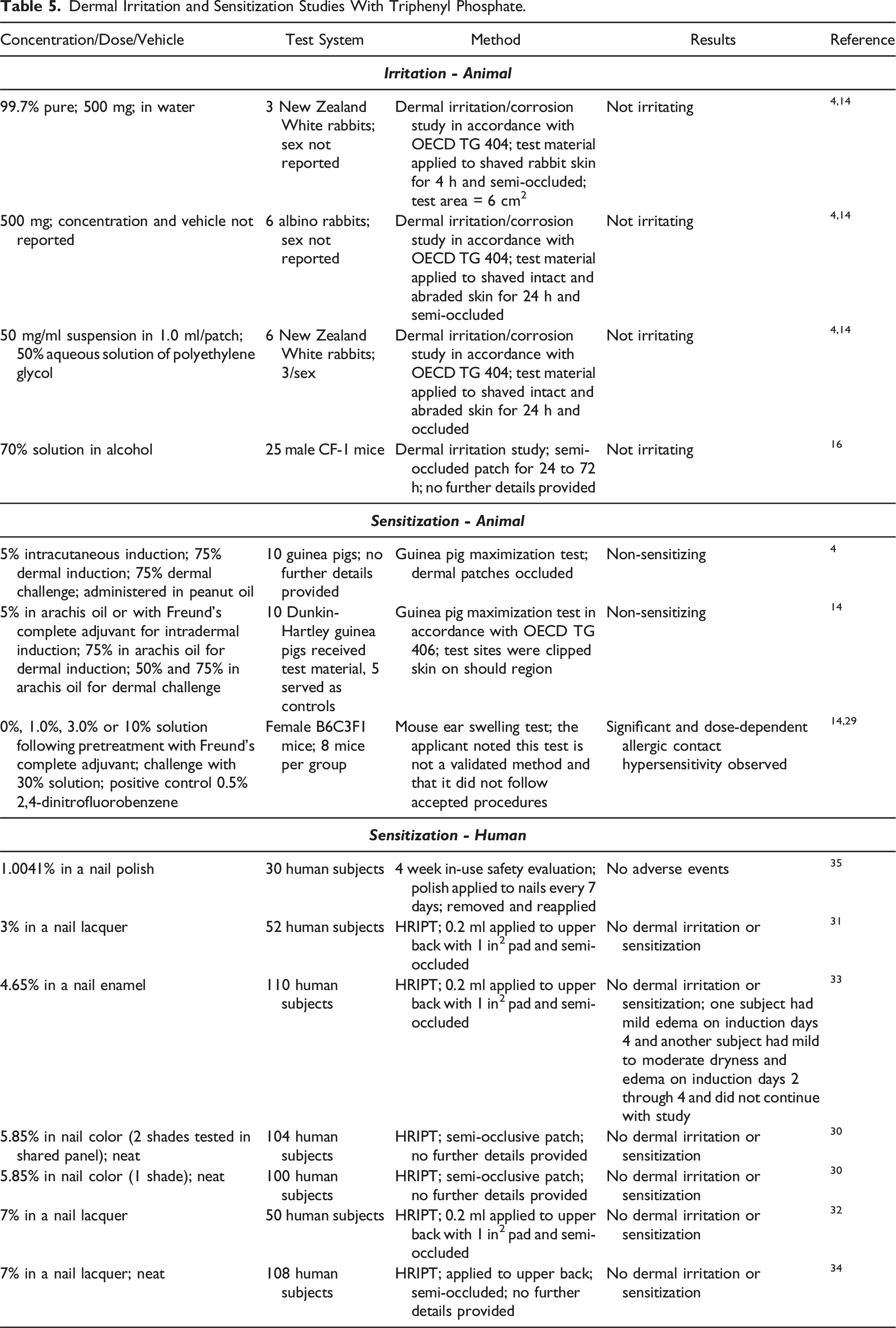
Dermal Irritation and Sensitization Studies
Dermal Irritation and Sensitization Studies With Triphenyl Phosphate.
Ocular Irritation Studies
Clinical Studies
Provocative Studies
In occlusive patch testing with 5% Triphenyl Phosphate in petrolatum (pet) in accordance with the International Contact Dermatitis Research Group (ICDRG) recommendations with a variety of plastic and glue allergens, no sensitization was observed in 174 patients with suspected occupational dermatoses. 36 One patient was observed with an irritation response. No further details were provided.
Case Reports
A 71-year-old female hospital patient with no prior history of allergies to plastics was treated with oxygen with an EN46001 System 22 clear facemask. 37 Erythema developed around her nose and mouth on the second day of admission that corresponded with the areas where the facemask had been in contact with her skin. By day 5, she had an acute facial eczema, which was diagnosed as allergic contact dermatitis. The patient was treated with mometasone cream, and the reaction cleared within 2 weeks. Patch tests were performed using the British Contact Dermatitis Society standard series, the plastics/glue series, the rubber chemicals series, a piece of the EN46001 System 22 oxygen facemask, a piece of the elastane strap, Triphenyl Phosphate, and tricresyl phosphate. Positive patch test results were observed to Triphenyl Phosphate (5% pet., + on day 2 and ++ on day 4), the facemask (as-is; ++ on day 2 and ++ on day 4), wool alcohols (30% pet.; ?+ on day 4 – likely an irritant reaction), and Amerchol L101 (100%; ?+ on day 4 – likely an irritant reaction). Prick tests to latex were negative. The facemask manufacturer reported that the facemask did not contain Triphenyl Phosphate, but it did contain triphenyl phosphite, which may have produced a cross-reaction.
A 29-year-old man with no previous allergic or atopic history reported a 6-month history of itchy fissured psoriasiform dermatitis on both palms. 38 The patient has a hobby that involves working with plastic glues. Positive patch test results of a standard series, balsams, plastics, and lacquers were observed for paraben-mix (15% pet.; ++), cobalt chloride (1% pet.; +), potassium dichromate (0.5% pet.; ++), formaldehyde (1% aq.; +), and Triphenyl Phosphate (5% pet.; ++).
In another case report, a 67-year-old woman reported an itchy eczematous eruption on the bridge of her nose and temples that were believed to be caused by her eyeglasses. 39 Patch tests were performed with the ICDRG standard series on Finn chambers. Additional tests were performed with the patient’s facial products and acetone-moistened scrapings from her eyeglass frames. Patch test results were negative for the standard series (including benzocaine), but were positive for benzocaine liniment with phenyl salicylate and the scrapings from the frames. Further patch tests results were negative for tris (2,3-dibromopropyl)-phosphate (5% pet.), dibutylphthalate (5% pet.), methyl salicylate (2% pet.), and positive (++) for phenyl salicylate (1% pet.) and tricresyl phosphate (5% pet.). Tests with pure triphenyl phosphate (>98%) and tri-m-cresyl- and tri-p-cresyl phosphate at 0.05%, 0.5%, and 5% pet. were positive to triphenyl phosphate down to 0.05% (++ to +) and tri-m-cresyl phosphate down to 0.5% (++ to +), but no reactions were observed to tri-p-cresyl phosphate.
Occupational Exposure
The National Institute for Occupational Safety and Health (NIOSH) recommended exposure limit (REL) and the Occupational Safety Health Administration (OSHA) permissible exposure limit (PEL) are both 3 mg/m3 time weighted average (TWA). 40 NIOSH established the immediately dangerous to life or health concentration (IDLH) as 1000 mg/m3.
Summary
Triphenyl Phosphate is an organic compound reported to function as a plasticizer in cosmetics. According to 2018 VCRP data, Triphenyl Phosphate is used solely in 331 nail products, with the majority of the uses being reported in nail polishes and enamels. The results of the concentration of use survey conducted in 2017 by the Council indicate that Triphenyl Phosphate is used solely in leave-on manicuring preparations at 1% to 14.5%, with the highest maximum concentration of use reported to be in polish strips.
Triphenyl Phosphate is a fire retarding agent and plasticizer for cellulose acetate and nitrocellulose. It is a noncombustible substitute for camphor in celluloid; it is also used to render acetylcellulose, nitrocellulose, airplane “dope,” etc., stable and fireproof; impregnating roofing paper; plasticizer in lacquers and varnishes. Triphenyl Phosphate is an approved indirect food additive in substances for use only as components of adhesives.
In a dermal penetration study of organophosphate esters performed in vitro, Triphenyl Phosphate tended to build up in the skin tissues, primarily in the upper layers. Only “smaller amounts” of Triphenyl Phosphate permeated the skin and reached the receptor fluid within 72 h.
Triphenyl Phosphate has been reported to metabolize to diphenyl phosphate and sulfate and glucuronide conjugates in metabolism studies performed in vitro. An absorption study of 0.97% Triphenyl Phosphate in nail polishes in 26 human volunteers found that the primary route of exposure was dermal exposure.
In rabbits, the dermal LD50 for Triphenyl Phosphate (concentration not reported) was greater than 10,000 mg/kg. The oral LD50 values for Triphenyl Phosphate in guinea pigs, rats, and mice were greater than 4000 mg/kg (concentration not reported), greater than 20,000 mg/kg (25% aqueous solution), and greater than 5000 mg/kg (20% emulsion in gum Arabic), respectively. Additional oral studies in mice at up to 500 mg/kg Triphenyl Phosphate found choline esterase activity was partially inhibited in the whole blood in a dose-dependent manner (87%–88% activity in 10–50 mg/kg to 30.4% in 500 mg/kg). The LC50 for inhalation exposure to Triphenyl Phosphate in rats was greater than 200 mg/l/hr (concentration not reported). Inhalation studies in mice at up to 757 mg/m3 for up to 4 h observed mean cholinesterase activity lower in treated groups than in controls; however, significance was only observed in the 2 h exposure in the 757 mg/m3 dose group.
The NOAEL for 50% (w/v) Triphenyl Phosphate in a 3-week dermal repeated dose study in rabbits was 1000 mg/kg/day, the maximum dose tested. In oral studies in cats at doses up to 50 mg/kg/day 2% Triphenyl Phosphate, mortalities, dyspnea, weakness, and decrease body weight were observed. Cholinesterase activity was 64% to 71% of normal values. In rat dietary studies up to 90 days in duration, the NOAEL was 1500 ppm based on liver weight increases. No adverse effects were observed during microscopic examination or weight measurements of the gonads (males: testes and epididymes, seminal vesicles including coagulating glands; females ovaries, uterus including cervix, vagina) at dietary doses up to 7500 ppm in this 90 day study. In a 4 month rat dietary study of the effects of Triphenyl Phosphate at up to 1.0% on neuromotor function, body weight gains were significantly reduced starting at 0.5%. The NOEL for non-immunotoxic effects in a 120 day rat dietary study on immunotoxic effects was 0.75% Triphenyl Phosphate due to reduction of body weight gains.
The maternal and developmental NOAELs in female rabbits was 200 mg/kg/day Triphenyl Phosphate (maximum dose tested) due to the lack of observed adverse effects. Triphenyl Phosphate was not teratogenic in a rat study at doses up to 1.0% (690 mg/kg).
Triphenyl Phosphate was not mutagenic in Ames tests at up to 10 mg/plate nor was it mutagenic in a mouse lymphoma test at up to 75 μg/ml. Triphenyl Phosphate (99.6% pure) was not clastogenic in a Chinese hamster assay at up to 60 μg/ml.
In studies researching effects on the endocrine system, Triphenyl Phosphate (>99% pure) was found to induce oxidative stress and reduce the expression of main genes related to testosterone synthesis in TM3 Leydig cells and in male mice at 300 mg/kg/day, but not at 100 mg/kg/day. In studies using animal and human cell lines and a zebrafish model, investigators found that Triphenyl Phosphate produced effects on sex hormone balance through various mechanisms including alterations of steroidogenesis and/or estrogen metabolism.
No neurotoxicity was observed in a subchronic dietary rat study of Triphenyl Phosphate at up to 1.0%. In another dietary rat study of Triphenyl Phosphate, the NOEL for immunotoxicity was 1% (maximum dose tested). Triphenyl Phosphate was toxic to human, monkey, and dog cell lines at 0.5 mM or 0.6 mM, 0.4 mM, and 0.5 mM, respectively.
Triphenyl Phosphate was not a dermal irritant in rabbits at up to 50% or mice at 70% in alcohol. No dermal sensitization was observed to Triphenyl Phosphate in guinea pig maximization tests up to 75%; however, significant and dose-dependent allergic responses were observed in a non-validated mouse ear swelling test at 3.0% or 10%. In HRIPTs with nail products, concentrations of up to 7% Triphenyl Phosphate did not induce irritation or sensitization in human subjects. No adverse events were reported in an in-use safety evaluation of a nail polish containing 1.0041% Triphenyl Phosphate.
Minimal ocular irritation effects were observed in rabbits tested with Triphenyl Phosphate, neat.
Sensitization was not observed in patch testing of dermatitic patients with 5% Triphenyl Phosphate in petrolatum. Case reports of allergic contact dermatitis were reported in patients that had been exposed to various plastic products containing Triphenyl Phosphate or triphenyl phosphite.
No relevant published carcinogenicity studies on Triphenyl Phosphate were identified in a literature search for this ingredient, and no unpublished data were submitted.
Discussion
The Panel found that the systemic toxicity data, including developmental and reproductive toxicity and short-term toxicity studies, and dermal irritation and sensitization studies, in this report were sufficient. The Panel noted that Triphenyl Phosphate can be absorbed at a very low rate through the skin, but the safety profile and use solely in nail products suggests that no adverse effects are likely to occur. The Panel also noted the lack of carcinogenicity data, but this gap was mitigated by multiple genotoxicity studies that were negative.
The Panel discussed the endocrine disruption potential of Triphenyl Phosphate in available in vitro and in vivo studies, and determined that the results were not sufficient to characterize this ingredient as an endocrine disrupting chemical. For further explanation of what qualifies as endocrine activity or disruption, please refer to the CIR resource document: https://www.cir-safety.org/supplementaldoc/cir-precedents-endocrine-activity.
Conclusion
The Expert Panel for Cosmetic Ingredient Safety concluded that Triphenyl Phosphate is safe in cosmetics in the present practices of use and concentration described in this safety assessment.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 555 13th Street, NW, Suite 300W, Washington, DC 20004, USA.
Author Contributions
The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.