Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) assessed the safety of three methylxanthines, Caffeine, Theobromine, and Theophylline, as used in cosmetics. All of these ingredients are reported to function as skin-conditioning agents in cosmetic products. The Panel reviewed the data relevant to the safety of these ingredients and concluded that Caffeine, Theobromine, and Theophylline are safe in cosmetics in the present practices of use and concentration described in this safety assessment.
Keywords
Introduction
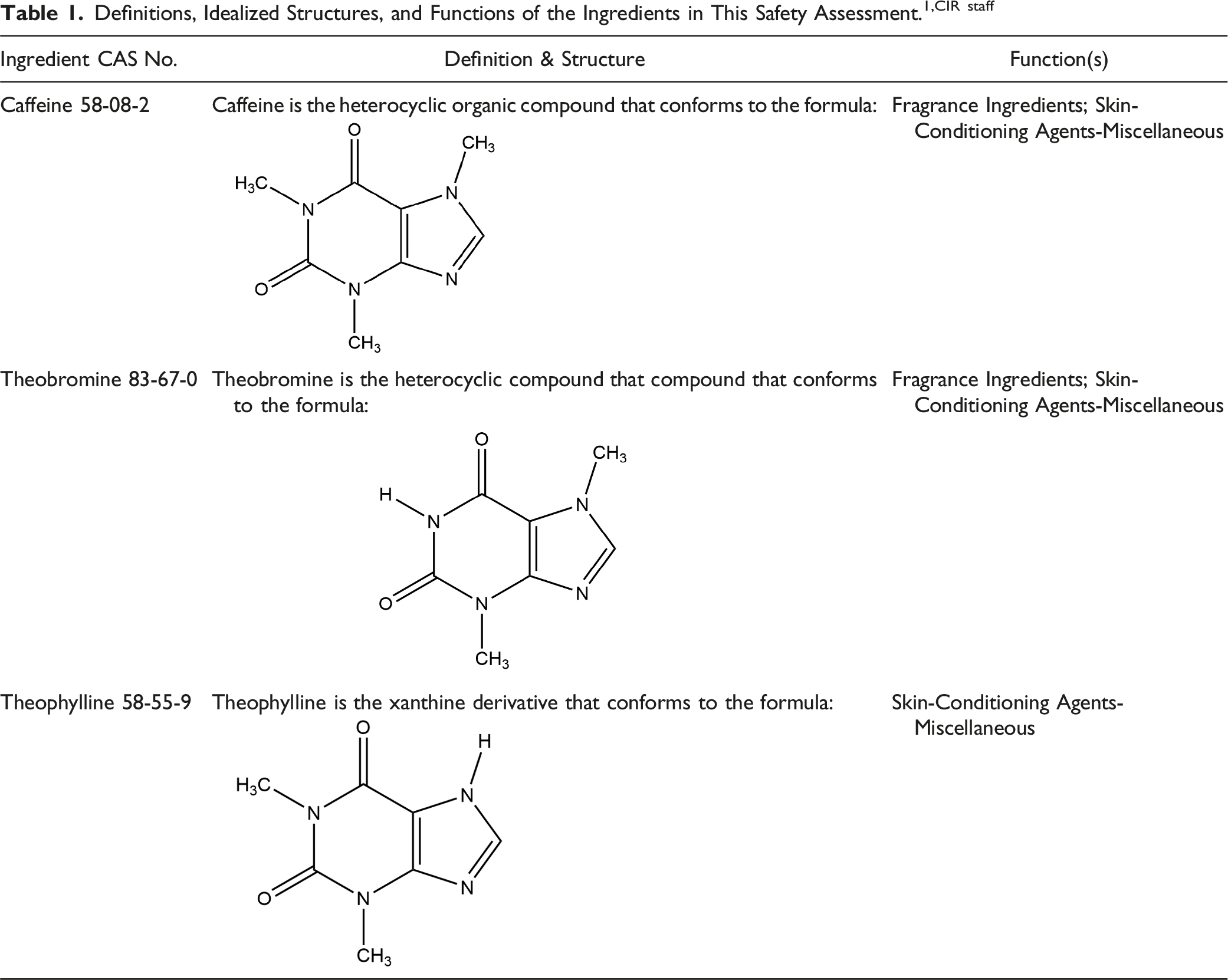
Definitions, Idealized Structures, and Functions of the Ingredients in This Safety Assessment.1,CIR staff
This safety assessment includes relevant published and unpublished data that are available for each endpoint that is evaluated. Published data are identified by conducting an exhaustive search of the world’s literature. A listing of the search engines and websites that are used and the sources that are typically explored, as well as the endpoints that the Expert Panel for Cosmetic Ingredient Safety (Panel) typically evaluates, is provided on the Cosmetic Ingredient Review (CIR) website (https://www.cir-safety.org/supplementaldoc/preliminary-search-engines-and-websites; https://www.cir-safety.org/supplementaldoc/cir-report-format-outline). Much of the information in this report was discovered in the European Chemicals Agency (ECHA) database2-4 or was available from the Organisation for Economic Cooperation and Development (OECD) Screening Information Dataset (SIDS) reports.5,6, Information from these sources is cited throughout the assessment. Please note that the ECHA website and OECD SIDS documents provides summaries of information generated by industry, and when cited herein, it is those summary data that are incorporated into this safety assessment.
Chemistry
Definition and Structure
Alkaloids are naturally-derived, nitrogen-containing compounds.
7
Methylxanthines are methyl-substituted alkaloid intermediates in the degradation (catabolic pathway) of adenosine monophosphate to uric acid. Caffeine, Theobromine, and Theophylline can be naturally or synthetically derived and are secondary metabolites derived from purine nucleotides.
8
The definitions and structures of the ingredients included in this report are presented in Table 1.
1
The ingredients in this group are all methylated xanthine derivatives and are in that way structurally similar. The placement of the N-methyl groups is the only structural difference between these three ingredients (Figure 1). Methylxanthines.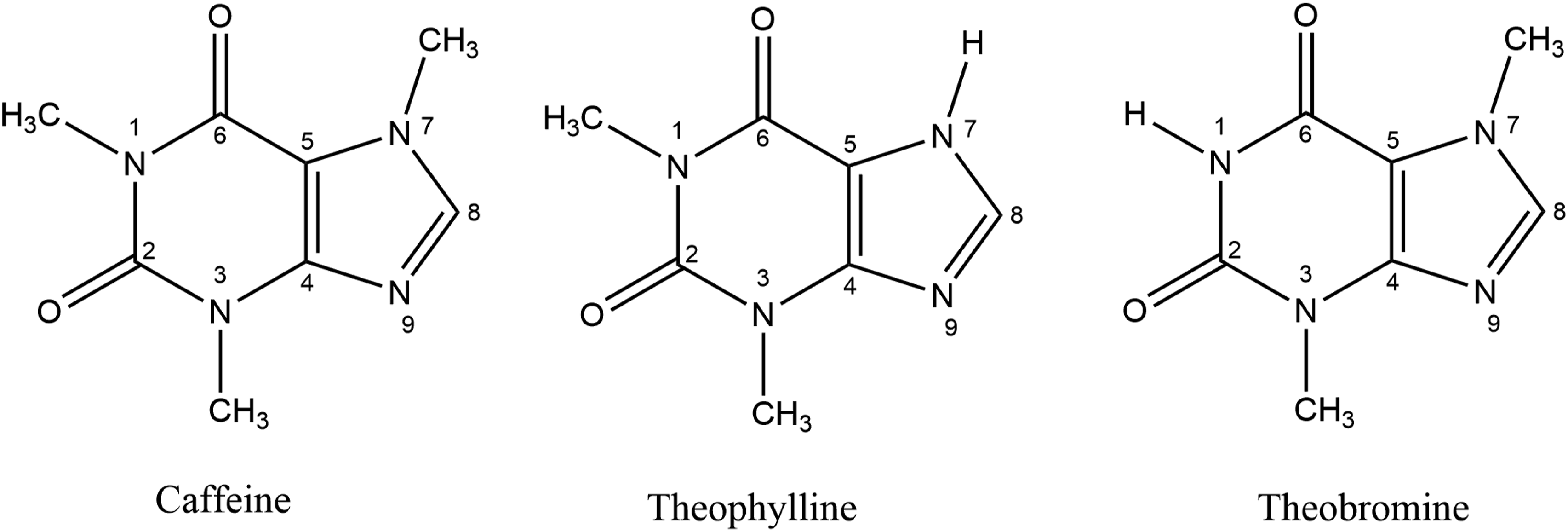
Physical and Chemical Properties
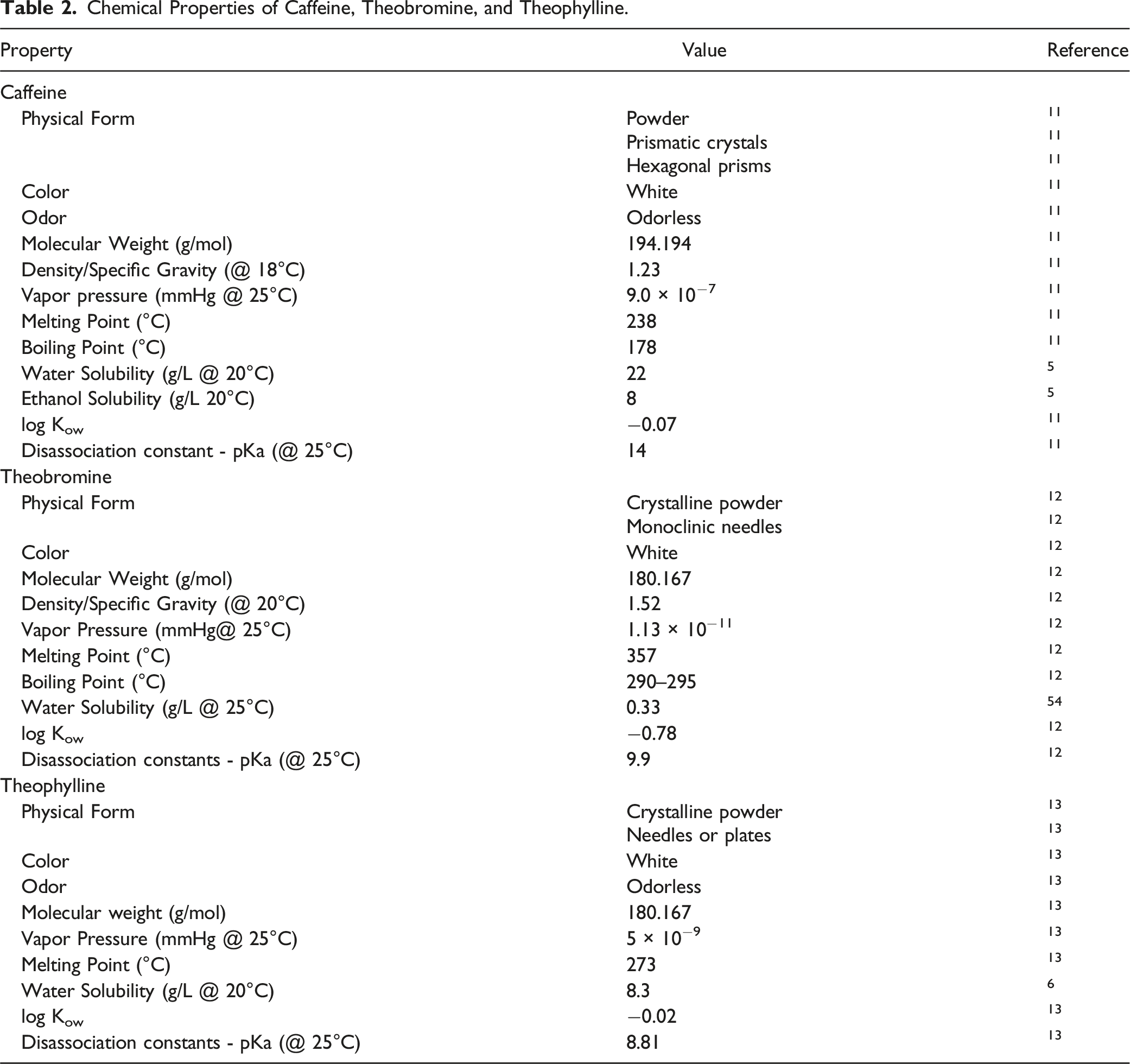
Chemical Properties of Caffeine, Theobromine, and Theophylline.
Method of Manufacture
Caffeine
The methods below are general to the processing of Caffeine for food or food ingredients, and it is unknown if they apply to cosmetic ingredient manufacture.
Caffeine can be extracted from plants or developed synthetically. Caffeine is most commonly extracted from green coffee beans, tea, or cola nuts. 10 Dichloromethane, ethyl acetate, edible fats and oils, supercritical carbon dioxide, and acid-activated carbon can each be used in the Caffeine extraction process.
Theophylline
According to one method, the synthetic manufacturing of Theophylline comprises the reaction of dimethylurea and ethyl cyanoacetate. 10 However, Theophylline is also naturally occurring and can be found in green coffee beans (Coffea arabica or Coffea canephora), black tea (Camellia sinensis), cocoa (Theobroma cacao) cotyledon, and dried mate (Ilex paraguariensis).
Theobromine
Theobromine can be produced synthetically from 3-methyluric acid, but is not typically derived this way. 10 More commonly, Theobromine is obtained from botanical sources, mainly found in the cocoa bean (Theobroma cacao). The extraction of this ingredient typically occurs from the husks of the cocoa beans.
Impurities
Caffeine
The Food Chemicals Codex states that Caffeine must contain at least 98.5% pure Caffeine. 14 In addition, Caffeine must also not exceed a 0.5% or 8.5% weight loss upon drying the anhydrous form or hydrous from, respectively, and the residue on ignition must not be more than 0.1%. 14 According to the British Pharmacopoeia, Caffeine must not contain less than 98.5%, and not more than the equivalent of 101.5% of 1,3,7-trimethyl-3,7-dihydro-1-H-purine-2,6-dione (Caffeine), calculated with reference to the dry substance. 16
Theobromine
According to the British Pharmacopoeia, Theobromine should be at least 99.0% and not more than 101% of 3,7-dimethyl-3,7-dihydro-1-H-purine-2,6-dione, calculated with reference to the dried substance. 16
Theophylline
Specifications for Theophylline indicate that it be at least 97.0% of the active ingredient according to the United States Pharmacopeia (USP), and should not contain less than 99.0% active ingredient according to the British Pharmacopoeia.15,16 Theophylline also must not exceed a 0.5% weight loss upon drying for the anhydrous form, or 7.5%–9.5% weight loss for the monohydrate form. In addition, the residue on ignition must not be more than 0.15%.
Natural Occurrence
Caffeine
Caffeine can be found naturally in many plants. 10 The most common sources include coffee (Coffea canephora and Coffea arabica), cocoa beans, tea leaves, and guarana (Paullinia cupana). Coffee beans contain, on average, 1.1% Caffeine in green arabica coffee beans (Coffea arabica), and 2.2% in green robusta (Coffea canephora) beans. Tea plants can contain up to 5% Caffeine, but levels are dependent on seasonal variation, origin, and fertilizers. 10
Theophylline
Theophylline is commonly found in black tea (Camellia sinensis), green coffee beans (Coffea arabica), dried mate (Ilex paraguariensis), and cacao (Theobroma cacao). 10
Theobromine
Theobromine is also found in the same sources as identified above, but according to one textual authority, is primarily sourced commercially from the cocoa plant (Theobroma cacao). 10
Use
Cosmetic
The safety of the cosmetic ingredients addressed in this assessment is evaluated based on data received from the US Food and Drug Administration (FDA) and the cosmetics industry on the expected use of these ingredients in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in the FDA Voluntary Cosmetic Registration Program (VCRP) database. Use concentration data are submitted by the cosmetic industry in response to a survey, conducted by the Personal Care Products Council (Council), of maximum reported use concentrations by product category.
Frequency and Concentration of Use.
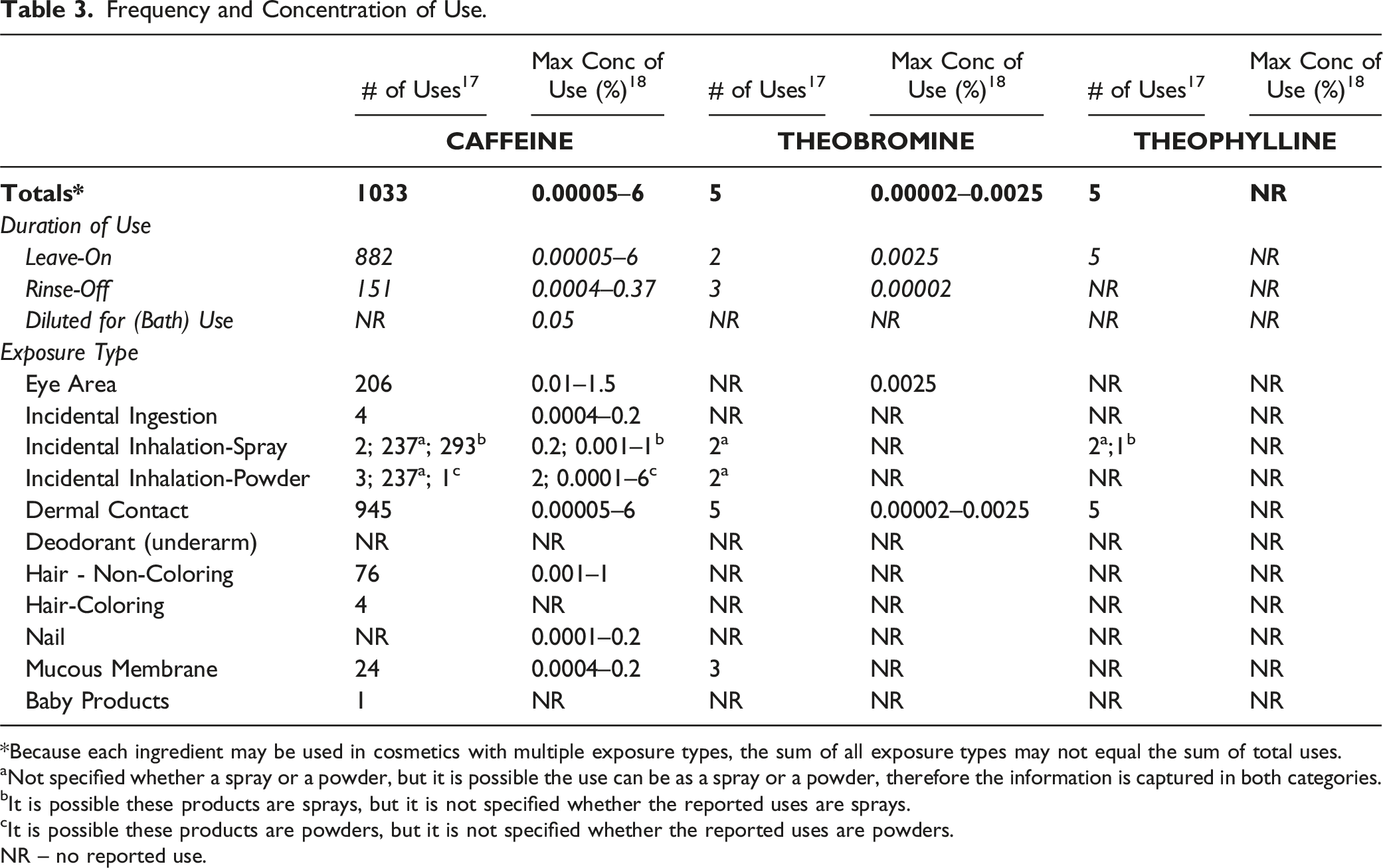
*Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
aNot specified whether a spray or a powder, but it is possible the use can be as a spray or a powder, therefore the information is captured in both categories.
bIt is possible these products are sprays, but it is not specified whether the reported uses are sprays.
cIt is possible these products are powders, but it is not specified whether the reported uses are powders.
NR – no reported use.
Cosmetic products containing Caffeine and Theobromine are applied near the eyes (e.g., at maximum concentrations of 1.5% and 0.0025%, respectively, in eye lotions), and Caffeine is used in products that can result in incidental ingestion (e.g., at 0.2% in lipstick). Caffeine is also used in sprays (e.g., up to 0.2% in face and neck sprays) and powders (e.g., up to 2% in face powders), and these products can result in incidental inhalation. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters > 10 μm, with propellant sprays yielding a greater fraction of droplets/particles < 10 μm compared with pump sprays.19,20 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and thoracic regions of the respiratory tract and would not be respirable (i.e., they would not enter the lungs) to any appreciable amount.21,22 Conservative estimates of inhalation exposures to respirable particles during the use of loose powder cosmetic products are 400-fold to 1000-fold less than protective regulatory and guidance limits for inert airborne respirable particles in the air.23-25
The methylxanthines named in the report are not restricted from use in any way under the rules governing cosmetic products in the European Union. 26
Non-Cosmetic
Caffeine
Caffeine is most commonly present in or added to beverages, such as coffee, tea, and soft drinks. It can also be used as a flavoring agent in baked goods, desserts, and candy. 27 The US FDA has categorized Caffeine as “generally recognized as safe” (GRAS) when included in cola-type beverages at concentrations that are equal to or less than 0.02% [21CFR182.1180]. Dietary Caffeine intake was studied in 37,602 subjects in the US. 28 The 90th percentile intake was 380 mg/d.
A smaller percentage of Caffeine is used in over-the-counter (OTC), FDA-approved, drug products, prescription drug products, and dietary supplements. 27 Caffeine is frequently used in analgesic products, as it may thereby augment the relief of headaches and pain associated with migraines or menstruation. Caffeine is also used in some medications to treat bronchopulmonary dysplasia and apnea in preterm infants. The US FDA has determined that Caffeine is safe as an active ingredient in OTC weight control drug products and when used as an active ingredient in stimulant drug products at dosage limits of 100–200 mg every 3–4 hours for adults and children ages 12 and up. [21CFR182.1180]
Theobromine
Theobromine is mainly used in the production of Caffeine. 10 The majority of consumed Theobromine is through chocolate/cocoa products. Theobromine is also used as a bronchodilator, diuretic, and vasodilator.
Theophylline
Theophylline is a bronchodilator and can be used to treat lung diseases such as bronchitis, asthma, and emphysema. 10 It can also be used for the relief of biliary colic, and in diuretics.
Toxicokinetic Studies
Dermal Penetration
In Vitro
Caffeine
The follicular penetration of Caffeine was studied using a combination of the Franz diffusion cell technique, and the follicle closing technique (FCT). 29 Caffeine, 12.5 mg (study design 1) or 2500 mg (study design 2), was dissolved in 100 mL Dulbecco’s phosphate-buffered saline (DPBS). Three types of skin samples were used as test barriers: a human reconstituted model, a human heat-separated model, and full-thickness human skin. Study design 1 involved the study of the permeability of both the reconstituted human epidermis model and the heat-separated human epidermis. The skin samples had a high, low, or no follicular density. These samples were exposed to the test substance for 24 hours. DPBS was used as the receptor medium. Samples taken from the receptor chamber were examined using high-performance liquid chromatography. After 24 hours, the skin sample that had a normal follicular density had the lowest absorption of the three types of samples (36.1 ± 9.85%). The skin sample without follicles had absorption of 43.4 ± 9.73%, and the high follicular density sample had the highest percentage of absorption (47.1 ± 9.10%).
In study design 2, the full-thickness skin membranes were mounted in the diffusion cell and subsequently exposed to the FCT. Shunts in the closed follicular pathway samples were blocked with a varnish-wax mixture. The varnish-wax mixture was also applied to the open follicular pathway samples, but only near the follicles preventing shunt blockage. Breast skin was more penetrable than abdominal skin in both types of skin samples (blocked or open follicular orifices). For example, in skin samples with artificially blocked follicular orifices, 3.6 ± 0.59% of the test substance penetrated in the epidermis of the breast skin, while 2.5 ± 0.94% penetrated into the epidermis of the abdominal skin. In both abdominal and breast skin samples, test substance penetration was higher in samples with open follicular orifices. In breast skin with open follicular orifices, 7.9 ± 0.56% of the test substance penetrated into the epidermis, while 3.6 ± 0.59% penetrated into the epidermis of blocked follicular orifice samples.
A solution containing 4 mg/mL Caffeine was applied at a dose of 50 μL/cm2 to mounted human skin. 30 Six hours after application, skin samples were cleaned with soap and isotonic water. Permeation was measured for 42 hours. Caffeine permeation was reported to be 24% in cells that were not washed, and 8% in cells that were washed.
Studies have also been done to examine the effect of skin thickness and occlusion on the absorption of Caffeine.31,32 Human abdominal skin samples were mounted on Dianorm Teflon macro 1 cells or Franz diffusion cells. Occlusion did not have an effect on the dermal absorption of Caffeine. When Caffeine in saline was applied to skin samples varying in thickness, it was observed that the maximum flux of Caffeine was increased with decreasing thickness, but these increases were considered to be non-significant. The amount of Caffeine in the skin membrane was not affected by skin thickness.
Theophylline
Human skin was used to examine the metabolism and absorption of 98% Theophylline. 6 Absorption varied among skin samples. The lowest reported absorption was 3.6%, while the highest was 33.4%. Diffusion ranged from 2.2%–7.7%. Approximately 0.2%–4.6% of the applied substance was metabolized, and more than 60% of the metabolites diffused through skin samples. Reported metabolites were 1,3,7-trimethyluric acid, 1,3-dimethyluric acid, and 3-methylxanthine. The amount of metabolites varied per skin sample. 6
A flow-through in vitro diffusion system was used to study percutaneous absorption of 8-[14C]-Theophylline (radiolabeled) through 5 different samples of excised human skin. 33 Donors of the skin samples differed by age, sex, and ethnicity. Eagle’s medium containing gentamicin sulfate and 10% fetal bovine serum was continuously perfused along the well. Receptor fluid was aerated with 95% O2; 5% CO2 and pumped underneath the skin’s surface at a rate of 3 mL/h. The diffusion area was 1.0 cm2. Skin cells were exposed to a solution of Theophylline (6.8 and 306.8 μg/cm2) and receptor fluid was collected hourly for 20 hours. The percentage of the applied dose that diffused through the skin was similar between all skin samples, with a range of 2.8%–7.8%. The percentage of the applied dose that was absorbed varied greatly between skin samples (3.6%–33.4%). The metabolites were estimated by thin-layer chromatography. Between 0.2 ± 0.1%–4.6 ± 0.2% of the applied doses were metabolized, and over 60% of the total formed metabolites penetrated through the skin. The metabolites that were observed were 1,3,7-trimethyluric acid, 1,4-dimethyluric acid, and 3-methylxanthine.
Absorption, Distribution, Metabolism, and Excretion (ADME)
Caffeine
Caffeine is readily absorbed through body membranes. The typical half-life of Caffeine in humans, after oral consumption, is 2.5–4.5 hours, but the time is increased during the third trimester of pregnancy and in women taking oral contraceptives. 5 Caffeine is widely distributed throughout body tissues, and is metabolized by hepatic enzyme systems only. The majority of Caffeine, 70%–80%, is metabolized through 3-N-demethylation to paraxanthine, carried out by the liver enzyme CYP1A2. 34 Approximately 7%–8% of Caffeine is metabolized by 1-N-demethylation to Theobromine, while 7-N-demethylation to Theophylline also accounts for about 7%–8% of Caffeine metabolism products. 34 The remaining Caffeine is metabolized through C-8 hydroxylation, resulting in the formation of 1,3,7-trimethyluric acid. However, outside of the liver, no significant metabolism of Caffeine occurs in other organs. The majority of Caffeine is excreted via urine (> 95% in humans). 5
Theobromine
Theobromine can be seen in the body as a result of Caffeine metabolism. In humans, Theobromine is metabolized in the liver into xanthine (i.e. fully demethylated), and further metabolized into methyluric acid, facilitated by CYP1A2 and CYP2E1. 35 Some mammals, such as cats and dogs, metabolize Theobromine at a slower pace. The lethal dose for Theobromine in dogs is reported to be 100–500 mg/kg bw. In humans, Theobromine is metabolized at a faster rate. The approximate half-life of Theobromine is 7.1 ± 0.7 hours in humans, and approximately 18 hours in dogs. 36 An estimated 10% of Theobromine is excreted via urine unchanged, while the rest is excreted as metabolites.
Theophylline
Theophylline is extensively metabolized in the liver (up to 90% in adults). This ingredient undergoes N-demethylation and 8-hydroxylation via cytochrome P450 1A2 into 1-methlxanthine, 3-methylxanthine, and 1,3-dimethyluric acid. 37 Metabolites are excreted into bile and eliminated via urine. 6 Only 7%–12% of orally ingested Theophylline is excreted unchanged in urine. After review of the kinetics and metabolism of Theophylline in rats, the International Agency for Research on Cancer (IARC) concluded that Theophylline is quickly and completely absorbed from the digestive tract, readily crosses placental boundaries, can be distributed in breast milk, and is dispersed throughout all organs, with the exception of adipose tissue. 10 In plasma, about 50% of the ingested Theophylline is bound to plasma proteins. The elimination half-time of Theophylline is approximately 3–11 hours in adults, which can be shortened or prolonged by certain medications and behaviors, such as smoking and oral contraceptives.
Animal
Oral
Theobromine
Theobromine was given to rabbits orally in doses of 1 and 5 mg/kg bw. 10 Rabbits given these doses showed complete gastrointestinal absorption. Rabbits given high doses (10–100 mg/kg bw) displayed a reduction in absorption rate. Despite the reduced absorption rate, absolute bioavailability of the given Theobromine reached 100%.
A single dose of 15–50 mg/kg bw of Theobromine was given to dogs orally. 10 Peak plasma concentrations were observed within 3 hours of dosing. Dogs were then given a high dose (150 mg/kg bw), and displayed peak plasma concentrations 14–16 hours after dosing, implying slowed intestinal absorption.
Human
Dermal
Caffeine
A non-occlusive 2.5 cm2 patch of 4 μg Caffeine in acetone was applied to the forearms of people from the age of 22–40 or 65–86. 30 After 24 hours, the site was washed and urine was collected. For the 22–40 age group, the dose recovered in urine was 32.1 ± 4.2%. For the 65–86 age group, the dose recovered was 61.8 ± 5.4%.
The role of hair follicles in the dermal absorption of Caffeine was studied. 38 A solution that contained Caffeine (2.5%), ethanol, and propylene glycol was applied to the skin of six male subjects who had not ingested Caffeine for at least 48 hours before testing. The average hair follicle density was 20–32 follicles/cm2. Hair was clipped down to 0.5 mm in the 25 cm2 application area of the chests of volunteers. For the first test, a microdrop of varnix-wax mixture was applied beside each hair follicle orifice, and 2 mg/cm2 of the Caffeine solution were applied to the test area of each subject for 8 h (formulation allowed to evaporate); subjects were not allowed to shower or bathe for 72 hours. A blood sample was taken at 5, 10, 20, and 30 minutes, and 1, 2, 5, 8, 24, and 72 hours after application. The same procedure was repeated in the same test areas after 3 more days of a Caffeine-free diet, but with each hair follicle orifice blocked with a microdrop of the varnish-wax mixture. Caffeine was extracted from the serum samples with dichloromethane. An aliquot of each serum sample was measured by a surface ionization mass spectrometry technique.
When hair follicles were left open, the average Caffeine levels in the blood 5 minutes after application were 3.75 ng/mL, and when hair follicle orifices were blocked, Caffeine was not detected in the blood until 20 minutes after application. After 20 minutes, the average amount of Caffeine detected in the blood in the blocked follicle group was 2.45 ng/mL, compared to an average blood concentration of 7.57 ng/mL in the open follicle group. The highest concentration in the open follicle group was approximately 11.75 ng/mL an hour after application, while the highest concentration in the blocked follicle group was 6.65 ng/mL at 2 hours after application. After 72 hours, no Caffeine was detected in the blood serum in either test group.
Oral
Caffeine
An absorption study was performed using four male subjects given 0, 1, 5, and 10 mg/kg in water. Plasma peak was reached at 47 ± 5 minutes. The mean plasma concentration was 8.3 ± 0.1 μg/mL, with an apportion rate constant of 6.3 ± 1.9 per hour and elimination rate constant of 0.11 ± 0.02 per hour. Ninety-nine percent of the given dose was absorbed. The metabolites 3,7-dimethylxanthine, 1,3-dimethylxanthine, and 1,7-dimethylxanthine were measured in the plasma. Approximately 85% of the administered dose was recovered in the urine within 48 hours. The main metabolites excreted were 1-methyluric acid, 7-methylxanthine, and 1,7-dimethylxanthine. 5
Nine pregnant and 4 post-partum women were exposed to Caffeine. 5 No other study details were provided. Results showed a significant prolongation of Caffeine elimination in the pregnant women. Post-partum women showed normal rates of Caffeine elimination.
Thirteen males, 9 females not on oral contraceptive steroids (OCS), and 9 females taking OCS were examined for the disposition and elimination of Caffeine. 5 The elimination half time of Caffeine was significantly longer in women taking OCS. The average elimination half time for women taking OCS was 10.7 hours, versus 6.2 hours in women not taking OCS. Women on OCS had a total plasma clearance of 0.79 mml/min/kg, and free clearance of 1.12 mL/min/kg. Women not taking OCS had a total plasma clearance of 1.3 mml/min/kg and free clearance of 1.97 mL/min/kg. Kinetic parameters were similar in men and women, with the exception of volume of distribution, which was significantly higher in women.
A similar study was done using 9 females who had been taking a low-dose estrogen-containing oral pill for at least 3 months, compared to 9 females who did not take OCS. 5 Each subject was given an oral dose of 162 mg of Caffeine. In subjects who took the low-dose estrogen-containing oral pill, the elimination half-life of Caffeine was 7.88 hours, the plasma clearance was 1.05 mL/min/kg, and peak plasma concentration was 3.99 μg/ml. In subjects who did not take OCS, the elimination half-life was 5.37 hours, plasma clearance was 1.75 mL/min/kg, and the peak plasma concentration was 4.09 μg/ml.
Theophylline
The distribution of Theophylline into breast milk was studied in five women. On average, less than 10% of the mother’s Theophylline intake was distributed into breast milk. 6
Toxicological Studies
Acute Toxicity Studies
Acute Toxicity Studies.
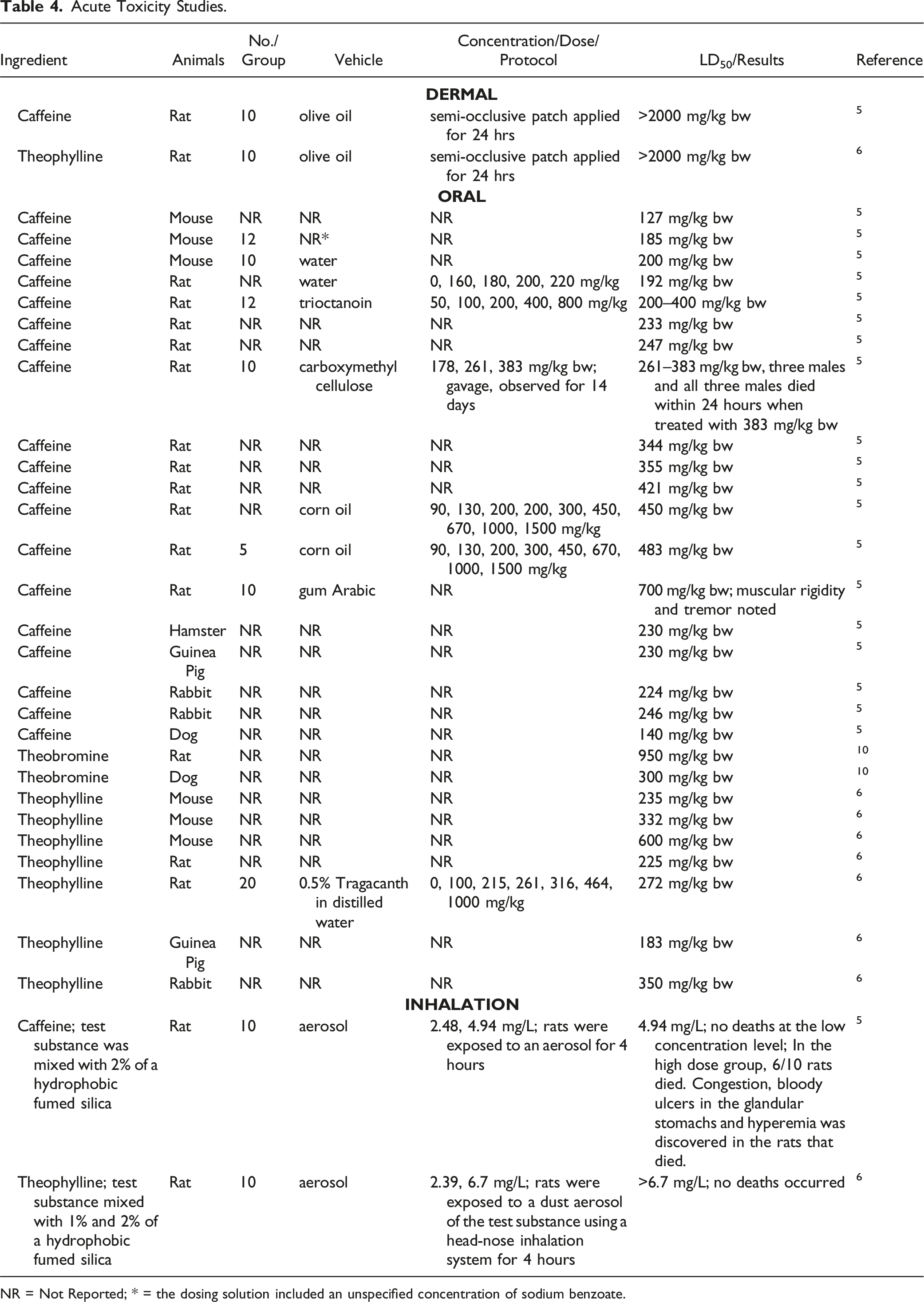
NR = Not Reported; * = the dosing solution included an unspecified concentration of sodium benzoate.
Acute dermal toxicity studies were performed in which both Caffeine and Theophylline, in olive oil, were applied under a semi-occlusive patch for 24 h to rats. The LD50 was > 2000 mg/kg bw for both test substances.5,6
Numerous acute oral toxicity studies were performed with Caffeine. The lowest reported LD50s in mice and rats are 127 mg/kg bw (vehicle not specified) and 192 mg/kg bw aqueous (aq.), respectively. 5 In other species, LD50s of Caffeine were 230 mg/kg bw (guinea pigs and hamsters), 224 mg/kg bw (rabbits), and 240 mg/kg bw (dogs). For Theobromine, the reported LD50s in rats and dogs (vehicle not specified) were 950 and 300 mg/kg bw, respectively. 10 For Theophylline, the lowest LD50s reported for mice, rats, guinea pigs, and rabbits are 235, 225, 183, and 350 mg/kg bw, respectively. 6
Acute inhalation studies were performed in rats on Caffeine and Theophylline mixed with a hydrophobic fumed silica.5,6 The LC50 following aerosol exposure or Caffeine mixed with 2% of a hydrophobic fumed silica was 4.94 mg/L. Following a dust aerosol exposure to Theophylline mixed with 1% and 2% silica, an LC50 of > 6.7 mg/L was established.
Short-Term, Subchronic, and Chronic Toxicity Studies
Repeated Dose Oral Toxicity Studies.
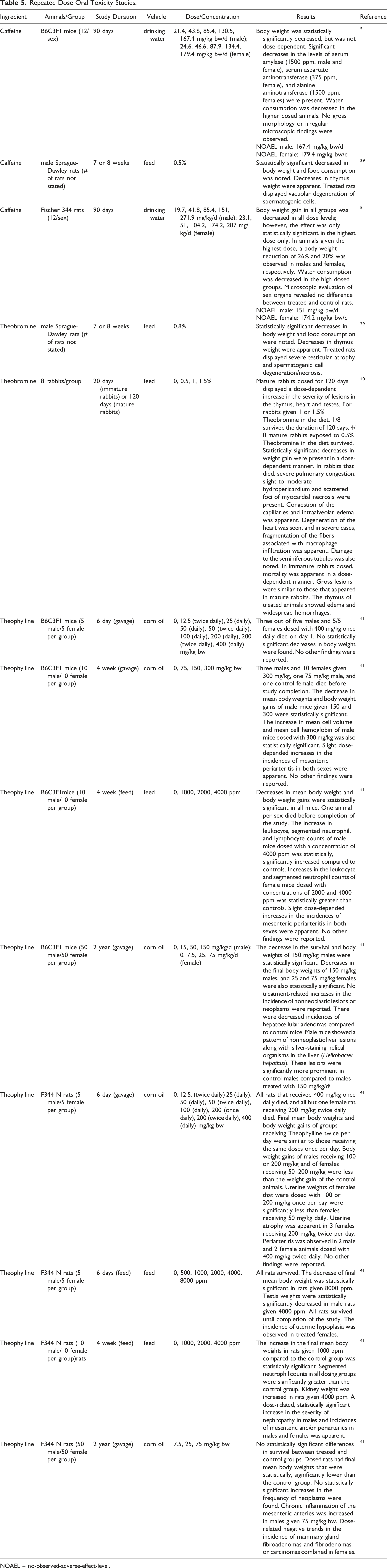
NOAEL = no-observed-adverse-effect-level.
Repeated dose oral toxicity studies were performed with the methylxanthines. In a study in which rats were given diets containing 0.5% Caffeine or 0.8% Theobromine for 7 or 8 weeks, treated rats of both groups displayed statistically significant decreases in thymus weights and vacuolar degeneration of spermatogenic cells; the effects were more severe with Theobromine. 39 In 90-day studies in which mice and rats were dosed with Caffeine in drinking water, the no-observable-adverse effect-levels (NOAELs) were 167 and 179 mg/kg bw/day in male and female mice, respectively, and 151 and 174 mg/kg bw/day in male and female rats, respectively; the highest doses administered in these studies were 167 and 179 mg/kg bw/day to male and female mice, respectively, and ∼272 and 287 mg/kg bw/day in male and female rats, respectively. 10
In a study in which immature and mature rabbits were fed a diet containing ≤ 1.5% Theobromine for 20 or 120 days, respectively, only 25% of the immature rabbits survived until study termination in each group, including controls. 40 Rabbits placed in groups 0, 1, 2, and 3 were given doses of 0, 0.5, 1, and 1.5%, respectively. In mature rabbits, on day 30 of treatment, group 1 gained 6% of their original body weight, while groups 2 and 3 lost an average of 5.9 and 18.1%, respectively. By day 30, 0/8 animals died in group 1, 5/8 animals died in group 2, and 4/8 animals died in group 3. By day 120, 4/8, 7/8, and 7/8 animals died in groups 1, 2 and 3, respectively. In immature rabbits, mortality was clearly dose-dependent. Lesions were apparent in the thymus in both immature and mature rabbits. Mature rabbits displayed severe pulmonary congestion and slight to moderate hydropericardium. Slight ascites were also present in the liver, as well as kidney congestion and redness of the gastro-intestinal mucosa. In immature rabbits, lesions were similar to mature rabbits. In addition, edema of the thymus and extensive hemorrhaging was present.
A 16-day gavage study was performed using mice given Theophylline in corn oil at doses as high as 400 mg/kg bw once daily. 41 All females (5/5) and 3/5 males dosed with 400 mg/kg bw died on day 1. A similar study was performed using rats. Animals were given up to 400 mg/kg Theophylline in corn oil via gavage once per day for 16 days; all male and females died after exposure of 400 mg/kg Theophylline given once daily and 9/10 animals died after exposure to 200 mg/kg Theophylline given twice a day. A 16-day feed study was also performed using groups of 5 rats/sex. Rats were given Theophylline at concentrations of up to 8000 ppm. All rats survived; the final mean body weight was statistically decreased in rats given 8000 ppm. In a study in which Theophylline was given to 10 mice/group/sex via diet at a maximum concentration of 4000 ppm for 14 weeks, statistically significant decreases in mean body weights and increases in leukocyte, segmented neutrophil, and lymphocyte counts were recorded at the 2000 and 4000 ppm levels. In a study in which 10 mice/group/sex were given Theophylline in corn oil for 14 weeks via gavage at a maximum dose of 300 mg/kg bw, a statistically significant decrease in mean body weights of male mice given 150 or 300 mg/kg bw was apparent.
Two-year studies were also performed. 41 Theophylline in corn oil, at up to 150 mg/kg bw, was given to 50 mice/group/sex for 2 years via gavage. Administration resulted in decreases in survival, and decreases of body weights of male mice dosed with 150 mg/kg and female mice dosed with 25 mg/kg. Final body weights of female mice dosed with 75 mg/kg were significantly less than the control groups. In a 2-year gavage study using 50 rats/sex/group, animals were dosed with up to 75 mg/kg bw Theophylline in corn oil. No statistically significant differences in the survival between treated and control groups were seen. Dosed rats had a statistically significant decrease in final mean body weights compared to the control group. In addition, chronic inflammation of the mesenteric arteries was increased in male rats given 75 mg/kg bw.
Developmental and Reproductive Toxicity Studies
Developmental and Reproductive Toxicity Studies.
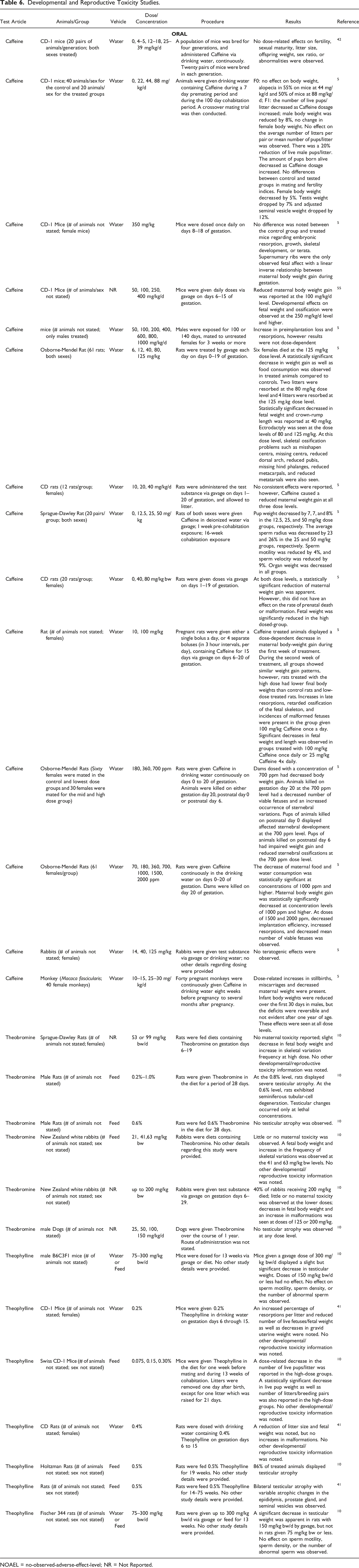
NOAEL = no-observed-adverse-effect-level; NR = Not Reported.
Studies were performed evaluating the developmental toxicity of Caffeine in mice. Mice dosed with up to 39 mg/kg/d of Caffeine in drinking water did not display any consistent dose-related effects on fertility, litter size, offspring weight, sex ratio, or fetal abnormalities. 42 Details of this study were not provided. When mice were given up to 350 mg/kg Caffeine on days 8–18 of gestation via gavage, animals did not display differences in embryonic resorption, growth, skeletal development, or terata, compared to control groups. No other details of this study were provided. 5 In different studies, mice displayed reduced maternal body weight gain (100–400 mg/kg/d Caffeine; gestation day (GD) 6–15) a reduction of live male pups/litter, female body weight, and adjusted seminal vesicle weight (up to 88 mg/kg/d; 21–35 weeks). When male mice were dosed with up to 1000 mg/kg/d Caffeine for 100 or 140 days (before mating) via drinking water and mated with untreated females, an increase of preimplantation loss and resorptions was seen.
Multiple reproductive toxicity studies were performed using rats. Dose-dependent maternal weight gain decreases were reported when rats were given Caffeine in doses of 10–40 mg/kg/d (GD 1–20; 12 females/group; oral administration), 40–80 mg/kg/d (GD 1–19; 20 females/group; oral administration), 10–100 mg/kg/d (GD 6–20; administered as single bolus or 4 divided doses), 100 mg/kg/d (GD 7–19, 7–16, 16–19, or day 19; oral administration), and 70–2000 mg/kg (GD 0–20; 61 females/group; drinking water). 5 In a study in which 61 Osborne-Mendel rats were treated with up to 125 mg/kg Caffeine via gavage on GD 0–19, 6/61 females died at the highest dose level. At the 80 mg/kg dose level, 2 litters were resorbed, and at the 125 mg/kg dose level, 4 litters were resorbed. No other details regarding this study were given. Resorption was also noted at concentrations of 1500 and 2000 ppm in a different study involving 61 female rats given Caffeine in drinking water on days 0 -20 of gestation. At these doses, decreased implantation efficiency, and a decreased number of viable fetuses was also noted.
A 23% average sperm radius decrease, as well as a decrease in sperm motility and velocity, was observed in 8 rats given 25 mg/kg Caffeine via gavage for 17 weeks. 5 No teratogenic effects were observed when rabbits were given up to 125 mg/kg of Caffeine via gavage on gestation days 6 -16. When 40 pregnant monkeys (Macaca fascicularis) were given Caffeine (10–15, 25–30, 25–30 mg/kg/d) in drinking water eight weeks before pregnancy to several months after pregnancy, a dose-related increase in stillbirths, decreased maternal weight, and miscarriages were present.
Female Sprague-Dawley rats were fed diets containing up to 99 mg/kg bw/d Theobromine during GD 6–19. 10 No maternal toxicity was reported, but a slight decrease in fetal body weight, as well as an increase in skeletal variation frequency, was apparent. Male rats were fed Theobromine in the diet for 28 days at concentrations of 0.2%–1.0%. 10 At the 0.8% level, rats displayed severe testicular atrophy. At the 0.6% level, rats exhibited seminiferous tubular-cell degeneration. Testicular changes occurred only at lethal concentrations. A similar study was performed using a concentration of 0.6% Theobromine in the diet for 28 days. No testicular atrophy was reported.
Rabbits given up to 63 mg/kg bw Theobromine via feed displayed little to no maternal toxicity. Details regarding dosing procedures were not provided. In a different study, female rabbits were given up to 200 mg/kg bw Theobromine via gavage on GD 6–29. At the 200 mg/kg dose level, 40% of the dams died, but little to no maternal toxicity was reported in rabbits given 25–125 mg/kg.
Theobromine was fed to male dogs at doses of 25, 50, 100, or 150 mg/kg/d for one year. 10 No testicular atrophy was seen at any dose level.
Up to 300 mg/kg bw Theophylline was given to male B6C3F1 mice via gavage for 13 weeks. 10 Mice that were dosed with 300 mg/kg bw/d displayed a slight but statistically significant decrease in testicular weight. When dosed with 150 mg/kg bw/d or less, no effects were observed.
CD1 mice given 0.2% Theophylline on GD 6–15 via drinking water displayed an increased percentage of resorptions/litter and a reduced number of live fetuses. 41 Dose-related decreases in the number of live pups/litter was also reported in a different study in which mice (sex not stated) were given 0.3% Theophylline in feed for one week before mating and during 13 weeks of cohabitation. 10 A statistically significant decrease in testicular weight was observed at the 300 mg/kg bw dose level in male mice dosed for 13 weeks via gavage or diet. No effects were reported at the 150 mg/kg bw dose level. The group treated with 0.4% Theophylline via drinking water on GD 6–15 displayed decreases in litter size and fetal weight. 41
Male Holtzman rats were given Theophylline at a concentration of 0.5% for 19 weeks (method of administration not provided). 10 Eighty-six percent of the treated rats displayed testicular atrophy. A similar study was performed in male rats given 0.5% Theophylline for 14 to 75 weeks (method of administration not provided). 41 Bilateral testicular atrophy and atrophic changes in the epididymis, prostate gland, and seminal vesicles was noted.
Male Fischer 344 rats were given up to 300 mg/kg/d Theophylline for 13 weeks. 10 A statistically significant decrease in testicular weight was reported after dosing by gavage with 150 mg/kg bw/d, but no effects were reported when animals were dosed with 75 mg/kg bw/d or less.
Genotoxicity
Genotoxicity Studies.
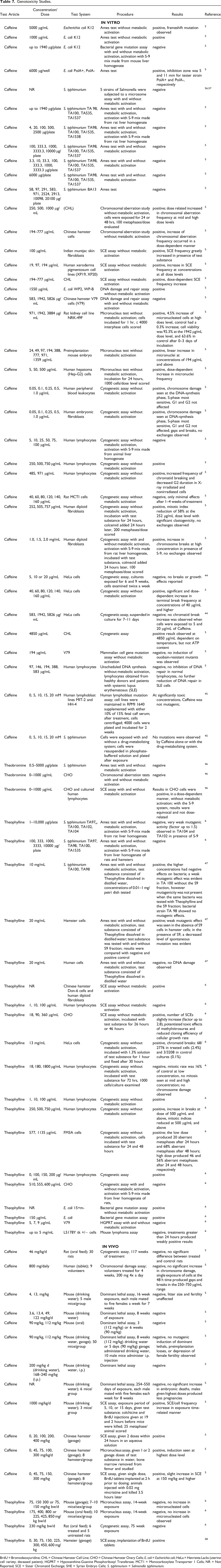
BrdU = Bromodeoxyuridine; CHL = Chinese Hamster Cell Line; CHO = Chinese Hamster Ovary Cells; E. coli = Escherichia coli; HeLa = Henrietta Lacks (uterine cell variety; deceased patient); HGPRT = Hypoxanthine-Guanine Phosphoribosyl Transferase; MCT1 = Monocarboxylate Transporter 1 Cells; NR = Not Reported; SCE = Sister Chromatid Exchange; SHE = Syrian Embryo Cells; S. typhimurium = Salmonella typhimurium.
In Vitro
Caffeine
Multiple tests were available regarding the genotoxicity of Caffeine. Ames tests performed without metabolic activation, at concentrations as low as 1000 μg/mL, yielded positive results. 5 In Ames tests performed in Salmonella typhimurium with metabolic activation at doses up to 6000 μg/plate, only negative results were reported. However in a different study, positive results were observed in Escherichia coli at concentrations as low as 6000 μg/well. In another study, S. typhimurium cells were exposed to Caffeine with and without metabolic activation at concentrations as high as 20 mM. 45 No mutations were observed. Bacterial gene mutation assays performed on Caffeine without metabolic activation yielded negative results in concentrations as high as 20,100 μg/plate (S. typhimurium). All chromosomal aberration studies performed without metabolic activation yielded positive results at concentrations as low as 194 μg/mL (Chinese hamster cell line (CHL)). Sister chromatid exchange (SCE) assays performed on Caffeine with metabolic activation yielded both positive and negative results. Positive results were apparent with concentrations as low as 19 μg/mL (human xeroderma pigmentosum cell lines) and negative results were seen with doses as high as 400 mg/kg bw (Chinese hamster cells). 43 DNA damage and repair assays performed without metabolic activation yielded negative results in Chinese hamster lung fibroblast V79 (V79) cells at up to 5826 μg/mL and positive results in E. coli at concentrations as low as 1550 μg/mL. 5 A DNA damage and repair assay performed with metabolic activation on E. coli resulted in bacterial growth and a minimal inhibitory concentration range of 187–1125 μg/plate. All micronucleus tests performed on Caffeine without metabolic activation, concentrations as low as 5 μg/mL in human hepatoma cells, resulted in positive results.
The majority of cytogenetic tests performed using Caffeine yielded positive results at concentrations as low as 0.05 μg/mL in both human peripheral blood leukocytes and human embryonic fibroblasts. 5 However, negative results were seen in studies at concentrations as high as 160 μg/mL (rat MCT1cells). A cytogenetic test performed with metabolic activation resulted in an increase in chromosome breaks at a concentration of 2.0 mg/mL in the presence of S-9 (human diploid fibroblasts). In cytogenetic studies in which the use of metabolic activation was not noted, positive and negative results were seen (HeLa cells). In one study, Caffeine yielded positive results at a concentration of 4850 μg/mL (CHL). Caffeine did not promote breaks or growth effects in two studies using immortalized human cervical cancer cells (HeLa) cells at doses as high as 5826 μg/mL. 44 Multiple mammalian cell gene mutation assays performed without metabolic activation yielded negative results in concentrations as high as 194 μg/mL in V79 cells. An unscheduled DNA synthesis assay performed without metabolic activation on human lymphocytes obtained from both healthy donors and patients with systemic lupus erythematosus (SLE), yielded negative results at concentrations as high as 583 μg/mL. No inhibition of DNA repair in normal lymphocytes or reduction of DNA repair in SLE cells were reported. In a human lymphoblast mutation assay, Caffeine was considered to be non-mutagenic when dosed with concentrations as high as 20 mM. 45
Theobromine
S. typhimurium (strains not specified) cells were treated with Theobromine at concentrations of 0.5–5000 μg/plate in an Ames test performed with and without metabolic activation. 46 Results were negative. Negative results were also obtained in a chromosomal aberration assay (Chinese hamster ovary (CHO) cells; 0–1000 μg/mL Theobromine) performed with and without metabolic activation. When CHO cells and cultured lymphocyte cells were used in an SCE assay performed without metabolic activation (up to 1000 μg/mL Theobromine), results were positive. When tested with metabolic activation, results were equivocal and not dose-related.
Theophylline
Negative and positive results were also seen in genotoxicity studies involving Theophylline. In one study involving hamster V79 cells and human cells, positive results were only obtained when testing was done without metabolic activation.
47
No DNA damage was observed when human cells were dosed with up to 20 mg/mL Theophylline. When V79 cells were exposed to up to 20 mg/mL Theophylline without metabolic activation, weak mutagenic effects were present. However, in the presence of metabolic activation, negative results were yielded. An Ames test using E. coli in concentrations as low as 150 μg/mL produced negative results.
6
Another bacterial gene mutation assay using E. coli also yielded negative results, however information on dosing was not provided. Cytogenetic assays performed without metabolic activation also had conflicting results, with positive results at concentrations as low as 500 μg/mL in human lymphocytes and negative results at concentrations as high as 1800 μg/mL in human lymphocytes. A hypoxanthine-guanine phosphoribosyltransferase (HGPRT) assay performed with and without metabolic activation produced negative results (V79 cells; up to 9 μg/mL). A mouse lymphoma assay also yielded negative results when L5178Y tk +/− cells were dosed with up to 5 mg/mL. SCE assays in Chinese hamster Don-6, human diploid fibroblast (dose not stated) and CHO cells (18–360 μg
In Vivo
Caffeine
Assays testing the cytogenetic potential of Caffeine were performed using rats as well as human volunteers. When 30 rats were dosed with 46 mg/kg/d via feed for 117 weeks, there were no statistically significant differences compared to control rats. 5 In a different cytogenetic assay involving humans, 9 volunteers were given 800 mg of Caffeine in tablet form each day. No significant increase in chromosome damage was seen.
Multiple dominant lethal assays performed on mice via drinking water or gavage yielded negative results at doses as high as 200 mg/kg/d Caffeine. 5 However, positive results were seen when a micronucleus assay was performed on Chinese hamsters. 5 Induction was apparent at the 300 mg/kg/d dose level.
An SCE assay was performed in Chinese hamsters given a single dose of up to 300 mg/kg Caffeine via gavage. 5 Bromodeoxyuridine (BrdU) tablets were implanted two hours before Caffeine dosing. A slight increase in SCEs was apparent at the 150 mg/kg dose level and higher. A similar SCE assay was performed using mice given up to 1000 mg/kg Caffeine each day for 5, 10, or 15 days. The frequency of SCEs increased in a time-dependent manner.
Theophylline
The majority of in vivo genotoxicity studies involving Theophylline yielded negative results. 6 However, when hamsters were given up to 600 mg/kg in an SCE assay, positive results were noted. In a micronucleus assay in which mice were given up to 150 mg/kg Theophylline for 14 weeks via gavage, no increase in micronucleated cells were seen. Similar results were observed in a micronucleus assay in which mice were given up to 850 mg/kg bw/d Theophylline in the diet for 14 weeks. Negative results were also seen in a cytogenetic assay in which rats were given up to 230 mg/kg bw/d Theophylline via oral feed for 75 weeks.
Carcinogenicity Studies
IARC concluded there is inadequate evidence for the carcinogenicity of Caffeine in experimental animals and in humans; IARC had an overall evaluation that Caffeine is not classifiable as to its carcinogenicity to humans. 10
Carcinogenicity Studies.

Sprague-Dawley rats (50 rats/sex) were given Caffeine (up to 2000 ppm) for 104 weeks via drinking water. 5 No statistically significant difference between the incidences of tumors in control and treated rats were apparent except for mammary fibroadenomas. The incidence of mammary fibroadenomas showed a significant inverse dose-response relationship. Fifty percent of the control animals displayed mammary fibroadenomas, while 26% of rats dosed with the highest concentration showed mammary fibroadenomas.
Forty female Wistar rats were given a 0.2% (2000 mg/L) Caffeine solution as their drinking fluid for 12 months. 48 Twenty-two of the 40 treated rats had pituitary adenomas, while 9 of the 30 untreated rats had pituitary adenomas. Pituitary hyperplasia was seen in 5/40 treated rats, and in 1/30 untreated rats.
Three groups each of 50 male and 50 female Wistar rats were maintained on a basal diet and given either tap-water (controls), a 0.1% solution of synthetic Caffeine (purity 100%), or a 0.2% Caffeine solution as the drinking fluid for 78 weeks. 10 Rats that survived were then given tap-water only for 26 more weeks. A total of 65/96 untreated rats had developed tumors. In the 0.1% solution group, 75/88 rats were tumor-bearing, and in the 0.2% group, 55/94 rats were tumor-bearing.
National Toxicology Program (NTP) studies regarding the carcinogenic potential of Theophylline were found. Theophylline was not carcinogenic in rats and mice when administered at up to 150 mg/kg bw/d in male B6C3F1 mice and up to 75 mg/kg in male and female Fischer 344 rats. 41 Authors of an NTP study concluded there was no evidence of carcinogenic activity based on 2 year gavage studies performed on F344/N rats and B6C3F1 mice.
No information regarding the carcinogenicity of Theobromine was found in the published literature.
Co-carcinogenicity
Osborne-Mendel rats were given a diet consisting of 0.5% Caffeine, Theobromine, or Theophylline alone or with sodium nitrite. 49 In the group fed both nitrite and the methylxanthines, the mortality rate was significantly increased. Food intake was decreased in Caffeine-treated rats, and a further reduction of food intake was noted in rats treated with both Caffeine and sodium nitrite. Food intake was not affected in rats treated with Theobromine alone, but rats treated with both Theobromine and sodium nitrite displayed significant decreases in intake. There was no effect on feed consumption in rats treated with sodium nitrite only. Terminal mean body weights were decreased in all treated rats. The addition of sodium nitrite created a slight increase in the reduction of body weight. No neoplastic or pre-neoplastic lesions were observed.
In a different study, groups of 50 rats were given 100 mg/kg N-butyl-N-(4-hydroxybutyl)-nitrosamine (BBN). 5 After administration of BBN, rats were given 110 mg/kg/d Caffeine for 21 months via drinking water. Groups received either Caffeine-treated water only, Caffeine-treated water and phenacetin incorporated into their diet, or phenacetin alone. No carcinogenic potential was seen in rats given only Caffeine treated water, but an increase in tumor incidences was seen in rats given Caffeine and phenacetin combined. The increase in tumor incidences was greater in rats treated with both Caffeine and phenacetin versus rats treated with phenacetin alone.
Groups of 12–20 Fischer 344 rats were given one 200 mg/kg dose of diethylnitrosamine (DEN) intraperitoneally. 5 After a 2-wk period, the animals were then given 90 mg/kg/d Caffeine via drinking water for 6 weeks. Control rats were given a DEN injection only. Carcinogenetic potential was evaluated by comparing the number and area of glutathione-S-transferase placental form positive (GST-P+) foci in the liver of treated rats with control rats. There was no increase in the number and area of GST-P + -foci in treated animals.
Tumor Promotion
Groups of mice were given an injection of 7,12-dimethylbenz[a]anthracene (DMBA) once a week for 6 weeks. 50 The test substance (250 and 500 mg Caffeine/L of drinking water) was then given to groups of 54–55 BD2F1 or 37–42 C3H mice, one week after the end of DMBA injections. In BD2F1 mice, the low dose of the test substance revealed a 20% increase of mammary carcinoma multiplicity, while the high dose revealed a 40% increase. C3H mice had an increase of mammary carcinomas of 13% at 250 mg/L Caffeine, and an increase of 117% at 500 mg/L Caffeine.
Dermal Irritation and Sensitization
Irritation
Animal
Caffeine
A study was performed in which 500 mg of a test substance containing 50% Caffeine was applied to the skin of three White Vienna rabbits under a semi-occlusive patch for 4 hours. 5 The irritation index was 0.
Theophylline
Doses of 2 g/kg bw Theophylline in olive oil were applied to Wistar rats via a semi-occlusive patch for 24 hours. 6 After patch removal, sites were washed. No substance-related irritation was observed.
Semi-occlusive patches containing 0.5 g of 50% aqueous Theophylline were applied to two White Vienna rabbits. 6 Patches remained on the skin for 4 hours. After removal of patches, sites were washed and scored. Four hours after patch removal, very slight reddening of the skin was present in both rabbits. No other signs of irritation were reported. The reported irritation index was 0.0.
One male and two female White Vienna rabbits were given a 0.5 g dose of a test substance consisting of a 50% aqueous solution of Theophylline under a semi-occlusive patch. 6 Patches were kept on for four hours. After patch removal, the exposed area was washed. Sites were scored 4, 24, 48, and 72 hours after removal. The female rabbits had slight redness four hours after patch removal. No other signs of irritation were reported.
Sensitization
Animal
A local lymph node assay was performed (LLNA) using concentrations of 0, 0.5, 2, and 5% Caffeine in an ethanol:water (70:30) vehicle. 51 The assay was performed according to OECD guideline 429. Four female mice (CBA) per dose level were used. No other study details were provided. No adverse effects were observed.
Human
A human repeated insult patch test (HRIPT) was performed on 105 test subjects. 52 The test substance (a body product containing 6% Caffeine) was applied (20 μL) to the backs of the subjects under an occlusive patch. Applications occurred 9 times over a period of 3 weeks during the induction phase. After a 2 week rest period, the test substance was applied to the original test site and to a previously untreated test site, under an occlusive patch. No allergic reactions were noted during the induction or challenge phases.
Ocular Irritation Studies
Caffeine
Undiluted Caffeine (0.1 mL) was instilled into the eyes of three rabbits. 5 Average irritation indices were 0.9 (corneal opacity), 0 (iritis), 1.6 (conjunctival erythema) and 0.6 (conjunctival edema). Irritation was observed in all three animals within the first 24 hours, and only 1 animal showed minimal corneal and conjunctival irritation by day 8. The test substance was considered to be non-irritating.
Theophylline
Fifty-one mg of undiluted Theophylline (0.1 mL) was instilled into one eye of 3 male White Vienna rabbits. 6 Eyes were not rinsed. Tested eyes were examined 1, 24, 48, and 72 hours and 8 days after application. Minimal corneal opacity was reported for 1–2 rabbits each day, for up to 8 days. Minimal to well-defined redness and swelling was observed in the conjunctiva in all tested rabbits for up to 3 days. By day 8, only one animal showed conjunctival redness (grade 2), corneal opacity (grade 1) and keratitis. The irises of test animals were unaffected. Mean irritation indices for corneal opacity, iritis, conjunctival redness, and conjunctival swelling were 0.6, 0.0, 1.8, and 0.6 respectively. The test substance was considered to be non-irritating.
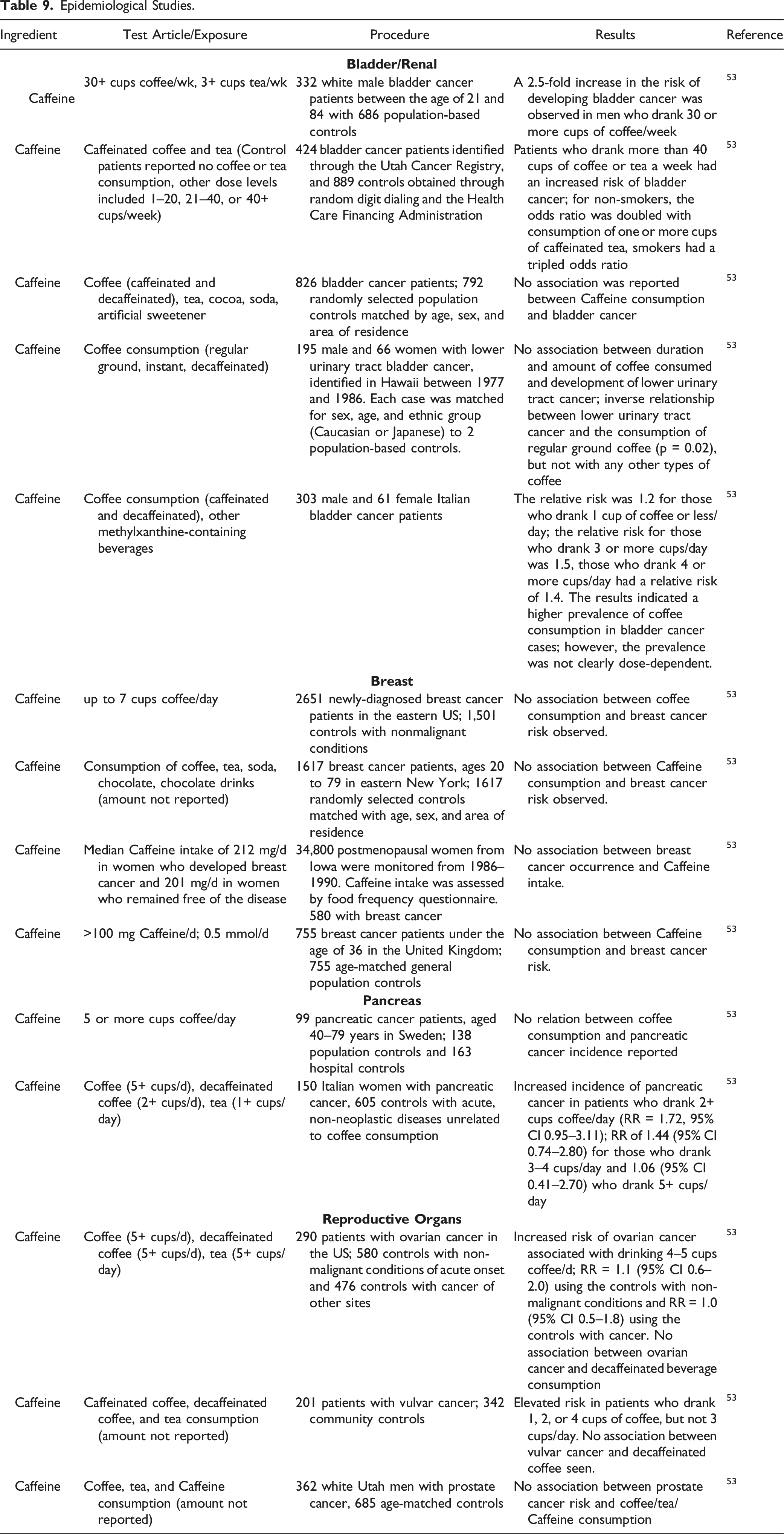
Epidemiological Studies
Epidemiological Studies.
Summary
The safety of three methylxanthines as used in cosmetics is reviewed in this safety assessment. According to the Dictionary, Caffeine, Theobromine, and Theophylline are reported to function as skin-conditioning agents, and Caffeine and Theobromine also are reported to function as a fragrance ingredient.
According to 2018 VCRP survey data, Caffeine is reported to be used in 1033 formulations, 882 of which are leave-on products and 151 are rinse-off. Theobromine and Theophylline are reported to have a much smaller frequency of use of 5 formulations each. The results of the concentration of use survey conducted by the Council indicate Caffeine also has the highest concentration of use in a leave-on formulation; it is used at up to 6% in non-spray body and hand products.
Follicular penetration of Caffeine was studied using a combination of the Franz diffusion cell and follicle closing techniques; the skin sample without follicles had absorption of 43.4 ± 9.73%, and the high follicular density sample had the highest percentage of absorption (47.1 ± 9.10%). When different areas of the body were tested for Caffeine penetration, breast skin was more penetrable than abdominal skin. The effect of washing skin on dermal Caffeine penetration was tested. A 24% permeation rate was reported for cells that were not washed, and a 8% permeation rate was reported for cells that were washed. In another study, it was observed that skin thickness did not have a significant effect on Caffeine penetration.
Human skin subjected to 98% Theophylline displayed a diffusion range of 2.2%–7.7%. Approximately 0.2%–4.6% of applied substance was metabolized, and more than 60% of the metabolites diffused through skin samples. Reported metabolites were 1,3,7-trimethyluric acid, 1,3-dimethyluric acid, and 3-methylxanthine. The amount of metabolites varied per skin sample. Theophylline absorption was tested among various skin samples. Theophylline absorption ranged from 3.6%–33.4%, while diffusion ranged from 2.2%–7.7%. In a different study, 8-[14C]-Theophylline (radiolabeled) was diffused through excised human skin. Between 0.2 ± 0.1%–4.6 ± 0.2% of the applied doses were metabolized, and over 60% of the total formed metabolites penetrated through the skin.
Caffeine is readily absorbed through bodily membranes and is distributed throughout the body. Theobromine and Theophylline are both metabolites of Caffeine, and make up 14% of Caffeine’s metabolism, combined. Factors such as pregnancy, oral contraceptives, and age affect the metabolism of Caffeine. All three methylxanthines are metabolized by the hepatic enzymes.
Theobromine can be present in the body as a result of Caffeine metabolism. Theobromine is metabolized in the liver into xanthine (i.e., fully demethylated), and further metabolized into methyluric acid, facilitated by CYP1A2 and CYP2E1. Theophylline is metabolized by ring oxidation and N-demethylation facilitated by microsomal enzymes in the liver (cytochrome P-450). After a review of the kinetics and metabolism of Theophylline in rats, IARC concluded that Theophylline is quickly and completely absorbed from the digestive tract, readily crosses placental boundaries, can be distributed in breast milk, and is dispersed throughout all organs, with the exception of adipose tissue.
Theobromine was given to rabbits, and the absolute bioavailability of the given Theobromine reached 100%. Peak plasma concentrations were reached within 3 hours when dogs were given a single dose of 15–50 mg/kg bw Theobromine.
Different age groups were dermally dosed with 4 μg Caffeine in acetone. The 65–86 age group displayed the highest dose recovery (61.8 ± 5.4%). The role of hair follicles in the dermal absorption of Caffeine (2.5%) was studied. The highest concentration in the open follicle group was approximately 11.75 ng/mL an hour after application, while the highest concentration in the blocked follicle group was 6.65 ng/mL at 2 hours after application.
Four men were treated with up to 10 mg/kg Caffeine orally. Ninety-nine percent of the dose was absorbed, with 85% of the given dose excreted in the urine. Significant prolongation of Caffeine elimination was observed in pregnant women compared to post-partum women. The elimination half time of Caffeine between women taking OCS and women not taking OCS was examined. Women on OCS had an average elimination half time of 10.7 hours, while women not taking OCS had an average elimination half time of 6.2 hours. In a different study, the elimination half times of women taking OCS and not taking OCS were 7.88 and 5.37 hours, respectively. The distribution of Theophylline in breast milk was studied in 5 women. It was observed that less than 10% of the mother’s Theophylline intake was distributed into the breast milk.
Studies involving acute dermal, oral and inhalation toxicity of the relevant ingredients reported low toxicity. The reported dermal LD50 for Caffeine and Theophylline were > 2000 mg/kg bw when test substances were applied via a semi-occlusive patch.
In oral studies, the lowest reported LD50s in mice and rats are 127 mg/kg bw (vehicle not specified) and 192 mg/kg bw (aq.), respectively. In other species, LD50s of Caffeine were 230 mg/kg bw (guinea pigs and hamsters), 224 mg/kg bw (rabbits), and 240 mg/kg bw (dogs). For Theobromine, the reported LD50s in rats and dogs (vehicle not specified) were 950 and 300 mg/kg bw, respectively. For Theophylline, the lowest LD50s reported for mice, rats, guinea pigs, and rabbits are 235, 225, 183, and 350 mg/kg bw, respectively.
The LC50 following aerosol exposure or Caffeine mixed with 2% of a hydrophobic fumed silica was 4.94 mg/L. Following a dust aerosol exposure to Theophylline mixed with 1% and 2% silica, an LC50 of > 6.7 mg/L was established.
Caffeine (0.5%) was given to rats in the diet for 7 or 8 weeks. A statistically significant decrease in thymus weight and vacuolar degeneration was apparent. Similar results were seen when 0.8% Theobromine was given to rats for the same duration. In 90-day studies in which mice and rats were dosed with Caffeine in drinking water, the NOAELs were 167 and 179 mg/kg bw/day in male and female mice, respectively, and 151 and 174 mg/kg bw/day in male and female rats, respectively; the highest doses administered in these studies were 167 and 180 mg/kg bw/day to male and female mice, respectively, and ∼272 and 287 mg/kg bw/day in male and female rats, respectively.
Immature and mature rabbits were fed a diet containing ≥ 1.5% Theobromine. In immature rabbits, mortality was present, and dose-dependent. Mature and immature rabbits displayed pulmonary congestion, ascites in the liver, kidney congestion, and redness of the gastro-intestinal mucosa.
In 16-day gavage studies involving Theophylline, the majority of rats died after being dosed with 200 mg/kg, and all rats died when dosed with 400 mg/kg. A 16-day feed study was also performed using rats given Theophylline at concentrations of up to 8000 ppm. All rats survived; the final mean body weight was statistically decreased in rats given 8000 ppm. Administration of Theophylline in corn oil to mice for 2 years via gavage resulted in decreases in the survival, and body weights of male mice dosed with 150 mg/kg were statistically significant. Mice given 4000 ppm Theophylline for 14 weeks displayed statistically significant decrease in mean body weights and increases in leukocyte, segmented neutrophil, and lymphocyte counts. Statistically significant decreases in the mean body weights and body weight gains of male mice was apparent after administration of 150 mg/kg bw via gavage for 14 weeks. In a 2 year study, mice were administered Theophylline at up to 150 mg/kg bw via gavage. Decreases in survival and body weights in treated mice were reported. In a different 2 year gavage study with Theophylline in corn oil using rats, no statistically significant differences in the survival between treated and control groups were seen.
Studies were performed evaluating the developmental toxicity of Caffeine in mice. Mice dosed with up to 350 mg/kg/d of Caffeine in drinking water did not display any consistent dose-related effects. In different studies, mice displayed reduced maternal body weight gain (100–400 mg/kg/d Caffeine; GD 6–15; gavage) a reduction of live male pups/litter, female body weight, and adjusted seminal vesicle weight (up to 88 mg/kg/d; 21–35 weeks; drinking water). Mice were dosed with up to 1000 mg/kg/d for 100 or 140 days via drinking water. An increase of preimplantation loss and resorptions were seen. Dose-dependent maternal weight gain decreases were reported when rats were dosed with 10–40, 40–80, 10–100, 100 and 70–2000 mg/kg via drinking water. In a study where 61 Osborne-Mendel rats were treated with up to 125 mg/kg via gavage on GD 0–19, 6/61 females died at the highest dose level. At the 80 mg/kg dose level, 2 litters were resorbed, and at the 125 mg/kg dose level, 4 litters were resorbed. Resorptions was also noted at concentrations of 1500 and 2000 ppm in a different study involving 61 female rats given Caffeine in drinking water on days 0 -20 of gestation. A 23% average sperm radius decrease, as well as a decrease in sperm motility and velocity, was observed in 8 rats given 25 mg/kg Caffeine via gavage. No teratogenic effects were observed when rabbits were given up to 125 mg/kg of Caffeine via gavage on gestation days 6 -16. Forty pregnant monkeys were given Caffeine in drinking water eight weeks before pregnancy to several months after pregnancy. A dose-related increase in stillbirths, decreased maternal weight, and miscarriages were present.
In a study in which rats were fed diets containing up to 99 mg/kg bw/d Theobromine, no maternal toxicity was reported, but a slight decrease in fetal body weight as well as an increase in skeletal variation frequency was apparent. Rats given Theobromine in the diet for 28 days displayed testicular atrophy, and rats fed 0.6% exhibited seminiferous tubular-cell degeneration. However, in a similar study where rats were fed 0.6% Theobromine, no testicular atrophy was noted. Rabbits given up to 63 mg/kg Theobromine bw via feed displayed little to no maternal toxicity. In a different study, rabbits were given up to 200 mg/kg bw Theobromine via gavage on GD 6–29. At the 200 mg/kg dose level, 40% of the dams died, but little to no maternal toxicity was reported in rabbits given 25–125 mg/kg. No testicular atrophy was noted when dogs were given up to 150 mg/kg/d Theobromine for one year. The route of administration for this study was not stated.
A statistically significant decrease in testicular weight was observed at the 300 mg/kg bw dose level in mice dosed for 13 weeks via gavage or diet. Mice given 0.2% Theophylline via drinking water displayed an increased percentage of resorptions/litter and a reduced number of live fetuses.41,43 Dose-related decreases in the number of live pups/litter was also reported in a different study in which mice were given 0.3% Theophylline in feed. Decreases in litter size and fetal weight were noted in rats dosed with 0.4% Theophylline via drinking water on GD 6–15. Rats given 0.5% Theophylline for 19 or 14–75 weeks displayed testicular atrophy. Statistically significant decreases in testicular weight were reported after rats were administered 150 mg/kg bw/d via gavage.
Multiple tests were available regarding the genotoxicity of Caffeine. The majority of bacterial in vitro tests yielded positive results, however the majority of mammalian cell in vitro genotoxicity assays yielded negative results. Bacterial studies were mostly positive without metabolic activation, and mostly negative with metabolic activation. Results were negative in an Ames test testing up to 5000 μg/plate Theobromine with and without metabolic activation. Negative results were also obtained in a chromosomal aberration assay performed with and without metabolic activation. CHO cells and cultured human lymphocytes were dosed with 1000 μg/mL. Positive results were obtained when metabolic activation was not present. Negative and positive results were also seen in genotoxicity studies involving Theophylline. However, in vivo genotoxicity assays using Caffeine produced predominately negative results.
IARC concluded there is inadequate evidence for the carcinogenicity of Caffeine in experimental animals and in humans; IARC had an overall evaluation that Caffeine is not classifiable as to its carcinogenicity to humans.
No statistically significant differences between the incidences of tumors in control and treated rats were present when Sprague-Dawley rats were given up to 2000 ppm Caffeine. Rats given 0.2% Caffeine in their drinking fluid for 12 months displayed pituitary adenomas and pituitary hyperplasia. Wistar rats were given either 0.1 or 0.2% Caffeine in drinking fluid. Tumor incidences was higher in untreated rats. Theophylline was not carcinogenic in rats and mice when administered up to 150 mg/kg bw/d in male B6C3F1 mice and up to 75 mg/kg in male and female Fischer 344 rats. Authors of an NTP study stated there was no evidence of carcinogenic activity based on 2 year gavage studies performed on F344/N rats and B6C3F1 mice.
Osborne-Mendel rats were given a diet consisting of 0.5% Caffeine, Theobromine, or Theophylline alone or with sodium nitrite. In the group fed both nitrite and the methylxanthines, the mortality rate was significantly increased. Food intake was decreased in Caffeine-treated rats, and a further reduction of food intake was noted in rats treated with both Caffeine and sodium nitrite. Food intake was not affected in rats treated with Theobromine alone, but rats treated with both Theobromine and sodium nitrite displayed significant decreases in intake. No neoplastic or pre-neoplastic lesions were observed. In a different study, rats were first given BBN followed by 110 mg/kg/d Caffeine or Caffeine and phenacetin for 21 months via drinking water. No carcinogenic potential was seen in rats given only Caffeine treated water, but an increase in tumor incidences was seen in rats given Caffeine and phenacetin combined. The increase in tumor incidences was greater in rats treated with both Caffeine and phenacetin versus rats treated with phenacetin alone. DEN injections were given to Fischer 344 rat followed by 90 mg/kg/d Caffeine via drinking water for 6 weeks. Carcinogenetic potential was evaluated by comparing the number and area of glutathione-S-transferase placental form positive (GST-P+) foci in the liver of treated rats with control rats. There was no increase in the number and area of GST-P + -foci in treated animals.
Mice were given an injection of DMBA once a week for 6 weeks. The test substance (250 and 500 mg Caffeine/L of drinking water) was given to groups of 54–55 BD2F1 or 37–43 C3H mice, one week after the end of DMBA injections. In BD2F1 mice, the low dose of the test substance revealed a 20% increase of mammary carcinoma multiplicity, while the high dose revealed a 40% increase. C3H mice had an increase of mammary carcinomas of 13% at 250 mg/L Caffeine, and an increase of 117% at 500 mg/L Caffeine.
The irritation index was 0 when White Vienna rabbits were subjected to 50% Caffeine under a semi-occlusive patch. Slight reddening was reported when White Vienna rabbits had semi-occlusive patches containing 0.5 g of 50% aqueous Theophylline applied to the skin. The reported irritation index was 0.0. In an LLNA using four female mice at concentrations of up to 5%, and an HRIPT (105 subjects; 6%), Caffeine was not considered a sensitizer.
Average irritation indices of 0.9 (corneal opacity), 0 (iritis), 1.6 (conjunctival erythema) and 0.6 (conjunctival edema) were recorded when undiluted Caffeine was instilled into the eyes of 3 rabbits. In a similar study, 51 mg of undiluted Theophylline was instilled into the eyes of 3 White Vienna rabbits. Mean irritation indices for corneal opacity, iritis, conjunctival redness, and conjunctival swelling were 0.6, 0.0, 1.8, and 0.6 respectively.
Case-control studies regarding the carcinogenicity through the intake of coffee (≥ 7 cups/day) provided no evidence of a potential breast cancer risk. Multiple studies confirmed this result. Different studies showed no or irregular association between Caffeine intake and cancer in the reproductive organs or pancreas. Cohort studies showed no correlation between risks for bladder cancer and Caffeine consumption, while a number of case-control studies showed a weak positive association between bladder cancer and coffee intake.
Discussion
The 3 ingredients in this report are methylxanthines, each of which is commonly ingested in food products, and can be naturally or synthetically derived. The Panel found that the data in this report were sufficient to support the safety of Caffeine, Theobromine, and Theophylline. In addition, the Panel noted that Caffeine, the methylxanthine with the highest frequency and concentration of use, is considered a GRAS foods substance in the US, with widespread frequent dietary exposure. Since the ingestion of this ingredient is safe, and exposure resulting from ingestion of food would be far greater than exposure due to cosmetic use, the concern for systemic toxicity was mitigated.
The Panel recognized several positive genotoxicity studies, but considered those to be potentially misleading. Indeed, positive results were only observed for in vitro studies without metabolic activation (those in vitro studies with metabolic activation were negative); the positive results of studies performed with mammalian cell cultures were also in sharp contrast to the in vivo mammalian studies which yielded negative results (suggesting that those positive results were not of concern). Furthermore, the Panel noted the negative results of the carcinogenicity studies performed by the NTP, further mitigating any concern with respect to the positive genotoxicity studies. Positive results for development and reproductive studies were also noted, but were considered negligible considering that these effects were only seen at lethal concentrations far exceeding reported cosmetic use concentrations.
The Panel discussed the issue of incidental inhalation exposure from formulations that may be aerosolized (e.g., face/neck products and face powders at up to 6%). The acute inhalation data suggest little potential for respiratory effects at relevant doses. Also, the Panel noted that in aerosol products, most droplets/particles would not be respirable to any appreciable amount. Furthermore, droplets/particles deposited in the nasopharyngeal or bronchial regions of the respiratory tract present no toxicological concerns based on the chemical and biological properties of these ingredients. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. The Panel considered other data available to characterize the potential for Caffeine, Theobromine, and Theophylline to cause systemic toxicity, irritation, sensitization, reproductive and developmental toxicity, and genotoxicity. They noted the lack of systemic toxicity at high doses in acute and chronic oral exposure studies, minimal or no irritation or sensitization in tests of dermal exposure at relevant concentrations, and the absence of relevant genotoxicity in multiple assays. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at https://www.cir-safety.org/cir-findings.
Conclusion
The Expert Panel for Cosmetic Ingredient Safety concluded that Caffeine, Theobromine, and Theophylline are safe in cosmetics in the present practices of use and concentration described in this safety assessment.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 555 13th St., NW, Suite 300W, Washington, DC 20004, USA.
Author Contributions
The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Declaration of Conflicting Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
