Abstract
This work investigated the safety of extracts obtained from plants growing in Colombia, which have previously shown UV-filter/antigenotoxic properties. The compounds in plant extracts obtained by the supercritical fluid (CO2) extraction method were identified using gas chromatography coupled to mass spectrometry (GC/MS) analysis. Cytotoxicity measured as cytotoxic concentration 50% (CC50) and genotoxicity of the plant extracts and some compounds were studied in human fibroblasts using the trypan blue exclusion assay and the Comet assay, respectively. The extracts from Pipper eriopodon and Salvia aratocensis species and the compound trans-β-caryophyllene were clearly cytotoxic to human fibroblasts. Conversely, Achyrocline satureioides, Chromolaena pellia, and Lippia origanoides extracts were relatively less cytotoxic with CC50 values of 173, 184, and 89 μg/mL, respectively. The C. pellia and L. origanoides extracts produced some degree of DNA breaks at cytotoxic concentrations. The cytotoxicity of the studied compounds was as follows, with lower CC50 values representing the most cytotoxic compounds: resveratrol (91 μM) > pinocembrin (144 μM) > quercetin (222 μM) > titanium dioxide (704 μM). Quercetin was unique among the compounds assayed in being genotoxic to human fibroblasts. Our work indicates that phytochemicals can be cytotoxic and genotoxic, demonstrating the need to establish safe concentrations of these extracts for their potential use in cosmetics.
Introduction
Phytochemicals are widely used as raw materials in the cosmetic industry. 1 The extensive use of plants as medicine has shown that they are not as safe as frequently claimed in the past.2,3 They can produce various toxic effects, including genotoxic damage (e.g., DNA adducts, DNA strand breaks, DNA-protein crosslinks, and chromosomal breakage). 4 Therefore, it is essential to study the adverse effects of plants before their use as a source of cosmetic ingredients.
Different guides or procedures using animal-free safety testing of cosmetic ingredients (e.g., colorants, preservatives, and UV filters) have been implemented in the European Union since the ban on animal use to assay cosmetic ingredients.5-9 There are different in vitro genotoxicity assays that provide information on relevant endpoints such as mutagenicity, clastogenicity, and aneugenicity. 10 According to our knowledge, no standard safety assay exists for testing phytochemicals used as cosmetic ingredients. However, it has been shown that the most commonly used genotoxicity assays for testing phytochemicals are those recommended by regulatory agencies for chemicals. 11 Among them, the Comet assay12,13 is a method widely used for genotoxicity evaluation of medicinal plants 11 and pharmaceuticals with inclusion into the ICH S2R1 guidance. 14 The use of a high-throughput platform (96-well microplate) has notably increased the overall capacity, reproducibility, and robustness of the Comet assay.15-18
We recently showed that different plant extracts and major compounds have UV-filter/antigenotoxic properties 19 ; therefore, they could potentially be used as cosmetic sunscreen ingredients. We hypothesize that these plant extracts and their constituents are neither cytotoxic nor genotoxic in human fibroblasts. To test this hypothesis, we evaluated the cytotoxicity and genotoxicity of these plants and constituents in human fibroblasts using the trypan blue exclusion and Comet assays, respectively. Here, we showed that some of the plant extracts under study were cytotoxic and genotoxic in human fibroblasts. We also provided evidence for the need for genotoxicity testing of plant-derived ingredients and for establishing safe phytochemical concentrations for use in cosmetics.
Materials and Methods
Plant Materials and Extracts
We studied extracts obtained from five plant species growing in Colombia, which have previously shown UV-filter/antigenotoxic properties. 19 The plants and their Colombian National Herbarium voucher numbers (in parenthesis) were as follows: Achyrocline satureioides (COL579420), Chromolaena pellia (COL559437), Lippia origanoides (COL560259), Piper eriopodon (COL578364 and COL578974), and Salvia aratocensis (COL560246). The plant species were collected in different areas of Colombia as follows: Los Santos – Santander (COL559437, COL560246, and COL560259); Zapatoca – Santander (COL579420 and COL578364); and Tame – Arauca (COL578974). Plant species collection was supported by collection permit and granted by the “Ministerio de Ambiente y Desarrollo Sostenible” of Colombia (Contract No. 270).
For each specimen, fresh leaves and flowers were used for supercritical fluid (CO2) extraction. 20 Extract stock solutions were prepared at 30 mg/mL, dissolved in methanol (1 mL), vortexed (3 min), exposed to ultrasound (10 min, 40°C), and centrifuged at 5000 r/min (10 min). Then, the supernatant (1 mL) was filtered and stored until use at 80°C. Before being used, the extracted stock solutions were defrosted and placed under refrigeration (5-8°C) for 24 h.
Gas Chromatography Coupled to Mass Spectrometry (GC/MS) Analysis
For the determination of major compounds in plant extracts, gas chromatography (GC) analyses were performed using a 6890 System Plus gas chromatograph (Agilent Technologies, Palo Alto, CA, USA), equipped with a mass selective detector MSD 5975 (Electron ionization, EI, 70 eV, AT, Palo Alto, CA, USA) and an automatic injector 7863 (Agilent Technologies, Palo Alto, CA, USA) that was operated in split mode (1:30), and its temperature was 250°C. A fused-silica capillary column DB-5MS (J&W Scientific, Folsom, CA, USA) of 60 m × .25 mm, i.d. x .25 μm, df, with stationary phase of 5%-phenyl poly (methysiloxane) was used for metabolite secondary separation. Chromatographic conditions were as follows: the initial column head pressure was 16.43 psi, and the operating mode was constant flow (1.0 mL/min) using helium as a carrier gas (99.995%, Messer, Bogotá, Colombia). The GC oven temperature was programmed from 45°C (5 min) to 150°C (2 min) at 4oC/min and then was increased to 250°C (5 min) at 5oC/min and finally to 300°C (5 min) at 10oC/min. The mass range used for mass spectra was m/z 45-450 with an acquisition speed of 3.58 scans/s using the MSD Chem Station G1701DA software (AT, Palo Alto, CA, USA).
The identification of major compounds from plant extracts was based on chromatographic retention lineal indices (LRI) and interpretation and comparison of mass spectra with databases. The LRI index was calculated using the retention times of a linear hydrocarbon standard (C8 to C40) mixture as indicated by the following equation:
Chemicals, Buffer, Enzymes, and Culture Media
Five plant compounds (trans-β-caryophyllene, squalene, pinocembrin, quercetin, and resveratrol), the filter titanium dioxide, the mutagens 4-nitroquinoline 1-oxide (4NQO) and bleomycin (BLM), trypan blue solution (.4%), lyophilized proteinase K, and high-resolution agarose, were obtained from Sigma-Aldrich Co. Inc. (Milwaukee, WI, USA). Quercetin and resveratrol have been chosen for comparative purposes as plant compounds that are frequently used in cosmetic industry for their antioxidant properties. YOYO solution was purchased from Thermo Scientific (MA, USA). Dulbecco’s modified Eagle’s medium (DMEM), fetal bovine serum (FBS), phosphate buffered saline (PBS), ethylenediaminetetraacetic acid (EDTA)-trypsin solution, and antibiotic mixture (penicillin/streptomycin) were purchased from Gibco (Grand Island, New York, USA). The remaining reagents were obtained from J.T. Baker (Phillipsburg, New Jersey, USA) or Merck (Kenilworth, New Jersey, USA). Other reagents and solvents were obtained from the commercial houses J.T. Baker (Phillipsburg, NJ, USA) or Merck (Kenilworth, NJ, USA). Stock compound solutions were prepared in ethanol or DMSO, depending on their solubility. From these solutions, working solutions for the tests were prepared. The standard mutagen stock solutions (4NQO and BLM) were prepared in acetone. Titanium dioxide stock solution was prepared in methanol.
Extract and Compound Cytotoxicity in Human Fibroblast (MRC5) Cells
Plant extracts and compounds were evaluated using the trypan blue exclusion assay. 21 Lung embryonic fibroblast (MRC5) cells, 22 graciously provided by Dr. Carlos Frederico Martins Menck from Universidade de Sao Paulo (Sao Paulo, Brazil), were grown in DMEM/F-12 medium (5 mL) supplemented with 10% heat-inactivated fetal bovine serum (FBS) and 1% GIBCO penicillin-streptomycin at 37°C under CO2 (5%) conditions in a Midi 40 incubator (Thermo Scientific, Marietta, OH, USA). Every 3 days, cells were grown in fresh medium to reach a confluence of 80%. Cell cultures were mixed with each extract, compound, or mutagen at different concentrations and kept at 37°C (24 h) under CO2 (5%) atmosphere conditions. After 24 h, trypsin-EDTA-treated cells were centrifuged (2000 r/min, 6 min), dissolved in PBS buffer (100 μL), and mixed (10 μL) with the same volume of trypan blue (.4%) to assess cell viability. Live and dead cells were counted using a Neubauer chamber and an Eclipse E200 optical microscope (Nikon Instruments Inc., NY, USA). At least three independent experiments were carried out for each treatment. The results were expressed as percentages of cell viability (% CV) as follows: % CV = (living cells)/(total cells) x 100. The cytotoxic concentrations 50% (CC50) and 30% (CC30) were calculated using the graphic method. 23 Extracts were considered cytotoxic at values ≥ CC50 and noncytotoxic at values ≤ CC30.
Extract and Compound Genotoxicities in Human Fibroblast (MRC5) Cells
The genotoxicity of plant extracts and major compounds was evaluated in MRC-5 cells using the Comet assay. 12 For this purpose, a high-throughput Trevigen CometChip® platform (Gaithersburg, Maryland, USA) was used as indicated by Sykora et al, 18 with minor modifications. First, a CometChip® slide, cleaned previously with ethanol, was covered with an agarose solution prepared in PBS at 1.3% and tempered at 45°C; then, the agarose was solidified for 24 h at 4°C. Trypsin-treated cells (3 mL) were collected using centrifugation (2000 r/min, 6 min), washed two times using NaCl solution (.75%), centrifuged and suspended in fresh NaCl solution (3 mL), and then quantified using a Neubauer counting chamber. A cell suspension (3 mL) prepared at 4.4 × 104 cells/mL was mixed with an equal volume of low-melting-point agarose prepared in PBS at 1%, and the mix was poured on a CometChip® slide. The agarose was solidified for 15 min at 4°C. Finally, the CometChip® chamber was assembled by hermetically sealing to prevent mixing between the wells. For cell treatments, 100 μL of extract, compound, or standard mutagen prepared as indicated above, were loaded into wells of the CometChip® chamber. A sample of DMEM (100 μL) was considered a negative control. The chamber with treatments was incubated for 30 min at 37°C under CO2 (5%) atmosphere conditions. The solutions of each treatment were removed from the wells, and 30 μL of Bioultra Proteinase K (.19 mg/mL) were loaded in each well for cell enzymatic lysis (1 h, 37°C).
After lysis, the CometChip® slide was removed from the CometChip® chamber and submerged for 15 min at 4°C in a comet electrophoresis tank (Cleaver Scientific Ltd, Rugby, Warwickshire,UK), which contained alkaline buffer (.3 N NaOH, 1 mM EDTA, and pH 13). Electrophoresis was carried out for 30 min at 300 mA and 25 V. The CometChip® slide was submerged for 15 min in a tray containing neutralizing solution (.4 M TRIS and pH 7.5), then washed with distilled water, and dried at 37°C in a Midi 40 incubator. Finally, cell nuclei contained in each microgel were stained with 7 μL of YOYO solution (1 mM prepared in 5% DMSO) and were immediately scored using a Zeiss Axio Observer 7 fluorescence microscope (GmbH, Oberkochen, Germany), as shown in Figure 1. From left (non-damaged nuclei) to right (highly damaged nuclei), the figure shows increases in the number of DNA breaks. The intensity of the comet’s tail with respect to the head reflects the number of DNA breaks. Comets were classified into five categories based on this intensity of DNA damage, as was proposed by Collins et al.
24
.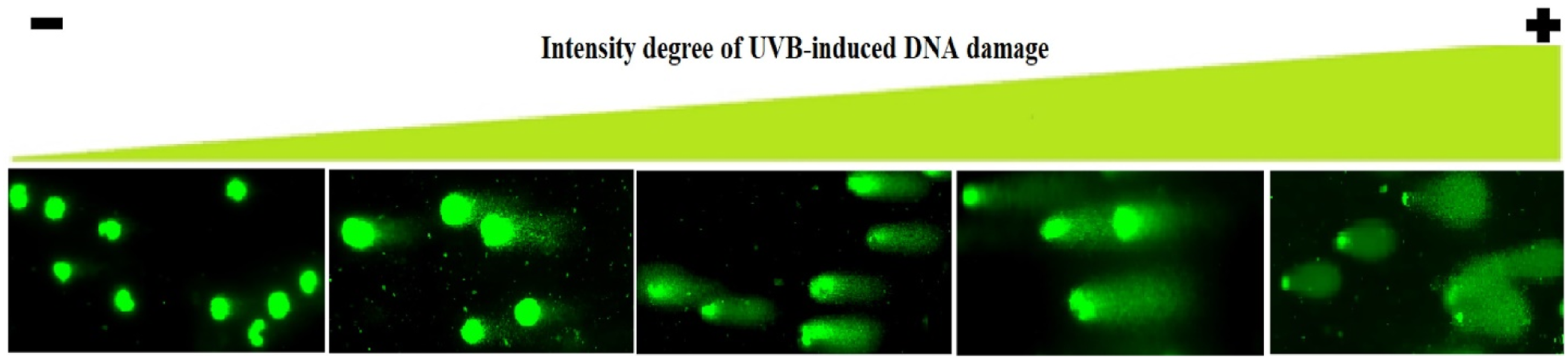
DNA damage was expressed in arbitrary units based on the classification of Comets into five categories (0-4), as proposed by Collins et al. 24 A genetic damage index (GDI) was calculated for each treatment as follows: GDI = (N0x0 + N1x1 + N2x2 + N3x3 + N4x4)/n, where N is the number of nuclei scored in each category and n is the number of scored cells per slide. 25 Two hundred cells per slide and two slides per treatment were analyzed. The results from at least three independent experiments were averaged to obtain the GDI values for each treatment.
Statistical Analysis
The survival (%) and GDI values and their corresponding standard errors were calculated. In all cases, the data passed the Kolmogorov–Smirnov and F-maximum tests for normality and variance homogeneity, respectively; therefore, parametric tests were used in subsequent data analyses. When a significant F-value was obtained in one-way analysis of variance (ANOVA), the groups were subsequently compared with Tukey’s test. A Pearson correlation was performed to measure the relationship between GDI values and extract concentrations. For all statistical analyses, a value of P < .05 indicated significance. The R program 26 was used for all analyses.
Results
During cytotoxicity assays, we observed three well-differentiated responses of the fibroblasts to extract treatments: cells with intact membranes that were not stained, cells with disturbed membranes that were stained but kept their shape, and stained cell fragments that resulted from cell destruction (data not shown). None of the cells survived treatments with extracts from S. aratocensis and P. eriopodon species at the concentrations studied. Using graphical interpolation, the concentrations of the other plant extracts that produced 50% (CC50) and 30% (CC30) of cell cytotoxicity in human fibroblasts were calculated. The cytotoxicity (CC50) of these extracts was as follows: L. origanoides (89 μg/mL; most cytotoxic) > A. satureioides (173 μg/mL) > C. pellia (184 μg/mL). These extracts were innocuous to fibroblast cells at concentrations less than or equal to (≤) the CC30 values as follows (Figure 2): L. origanoides (74 μg/mL), A. satureioides (140 μg/mL), and C. pellia (149 μg/mL). Viability of untreated (controls) MRC5 human fibroblasts and viability of cells treated with different non-cytotoxic extract concentrations. The cell viability (%) mean values and their corresponding standard errors (SEM), calculated from at least three independent experiments (n = 3), are presented.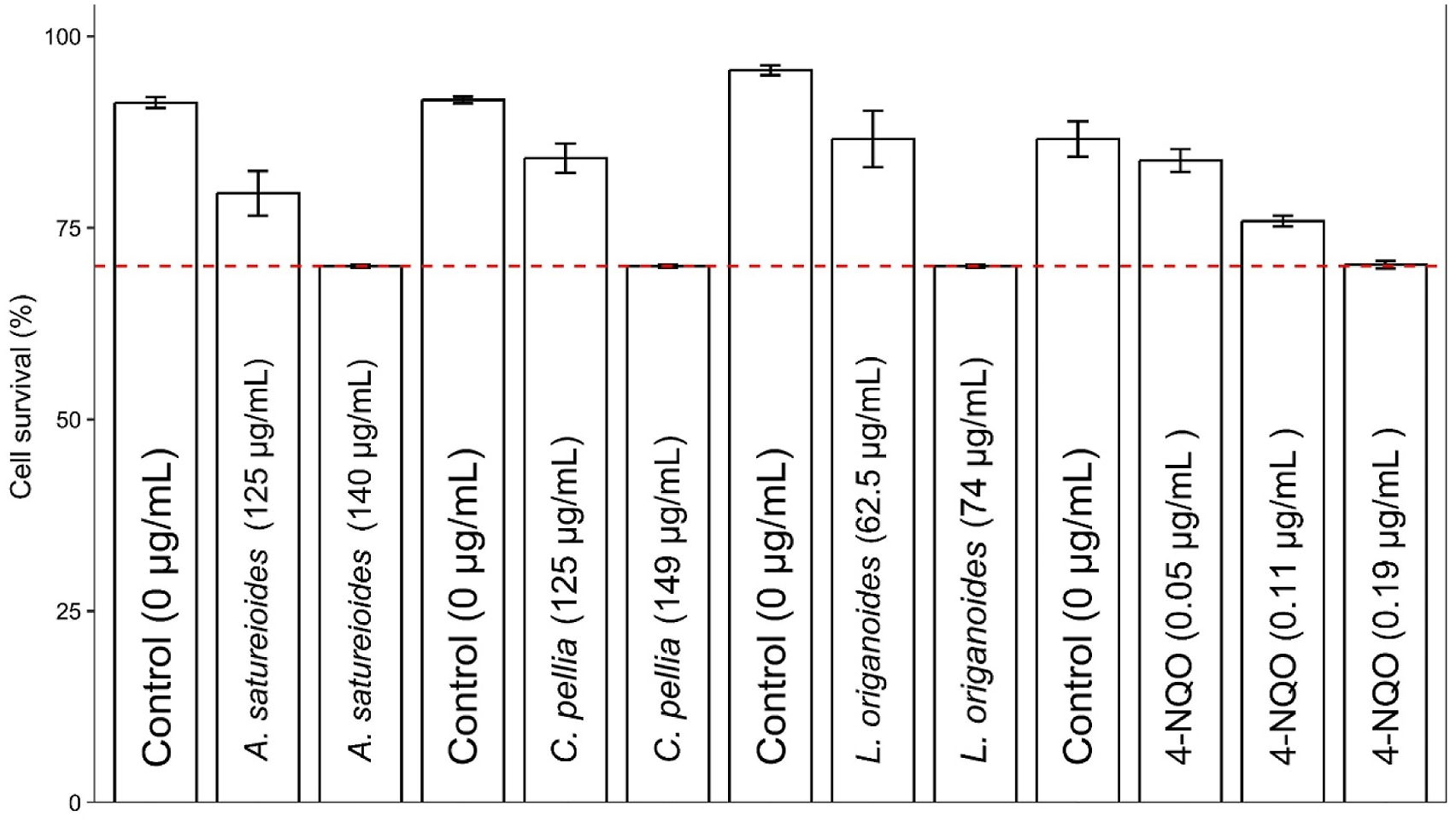
Major Compounds in Plant Extracts Obtained by Supercritical Fluid (CO2) Extraction From:
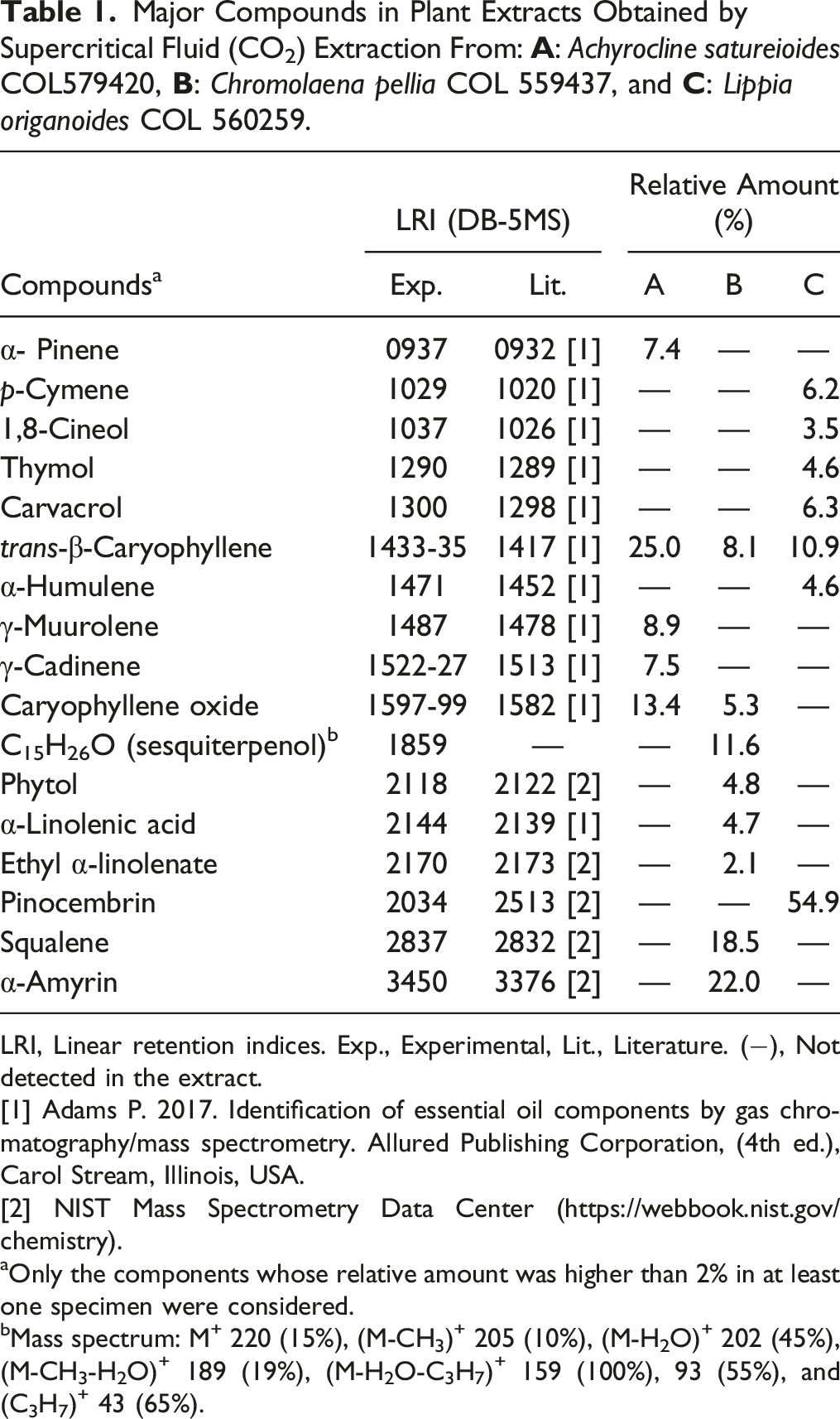
LRI, Linear retention indices. Exp., Experimental, Lit., Literature. (−), Not detected in the extract.
[1] Adams P. 2017. Identification of essential oil components by gas chromatography/mass spectrometry. Allured Publishing Corporation, (4th ed.), Carol Stream, Illinois, USA.
[2] NIST Mass Spectrometry Data Center (https://webbook.nist.gov/chemistry).
aOnly the components whose relative amount was higher than 2% in at least one specimen were considered.
bMass spectrum: M+ 220 (15%), (M-CH3)+ 205 (10%), (M-H2O)+ 202 (45%), (M-CH3-H2O)+ 189 (19%), (M-H2O-C3H7)+ 159 (100%), 93 (55%), and (C3H7)+ 43 (65%).
Cell Viability of Human Fibroblast Treated With Different Concentrations of the Compounds are presented.
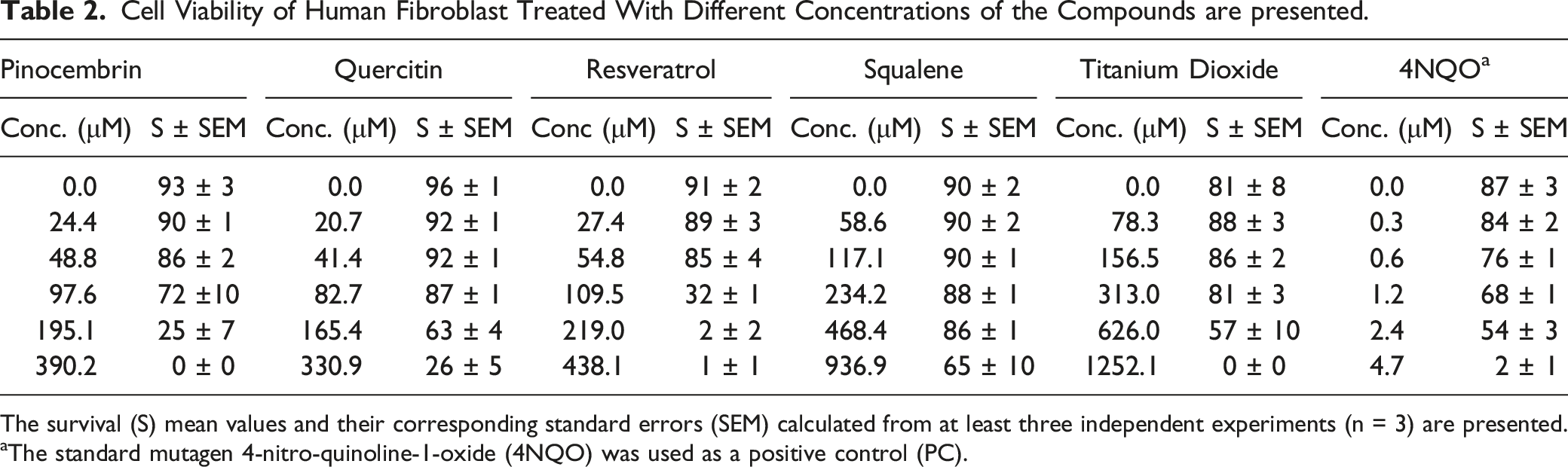
The survival (S) mean values and their corresponding standard errors (SEM) calculated from at least three independent experiments (n = 3) are presented.
aThe standard mutagen 4-nitro-quinoline-1-oxide (4NQO) was used as a positive control (PC).
Genotoxicity of the Flower Extracts in MRC5 Human Fibroblasts Cells.
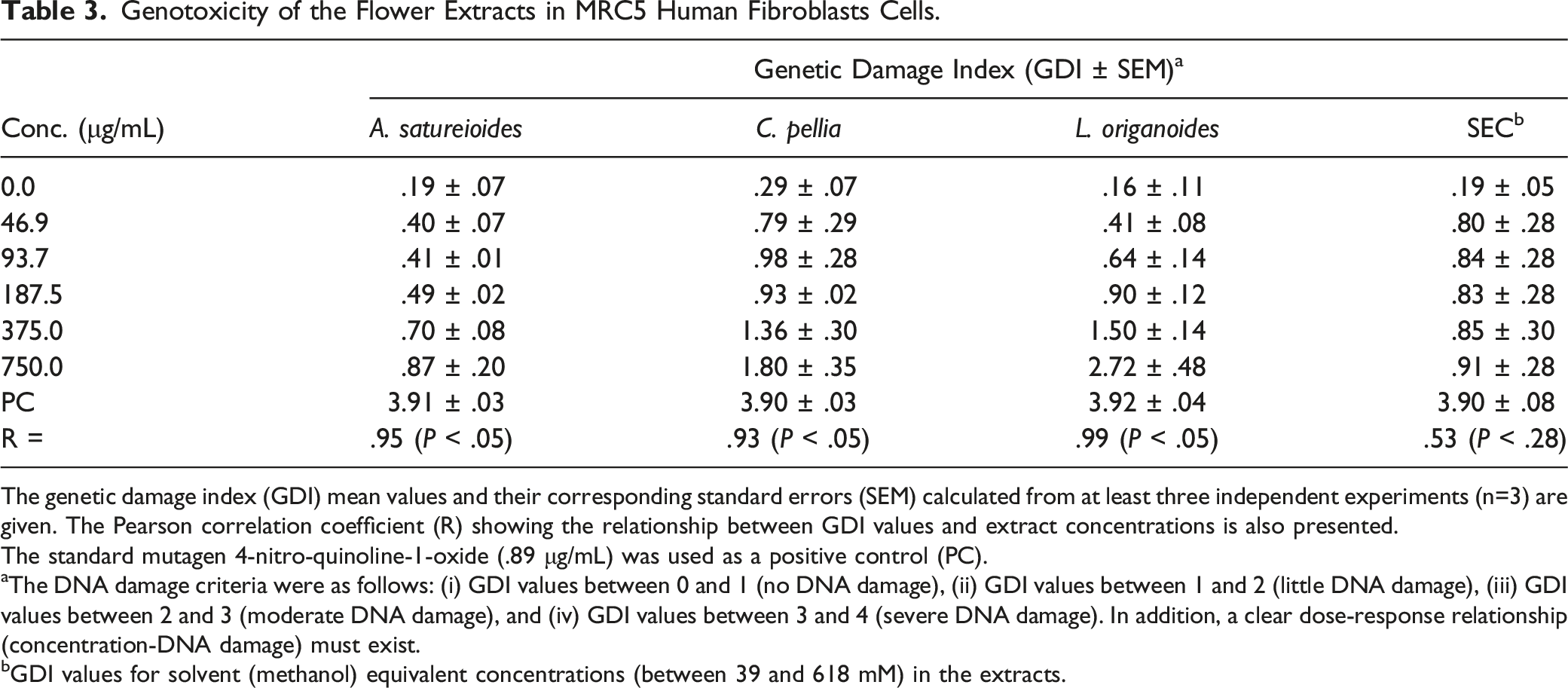
The genetic damage index (GDI) mean values and their corresponding standard errors (SEM) calculated from at least three independent experiments (n=3) are given. The Pearson correlation coefficient (R) showing the relationship between GDI values and extract concentrations is also presented.
The standard mutagen 4-nitro-quinoline-1-oxide (.89 μg/mL) was used as a positive control (PC).
aThe DNA damage criteria were as follows: (i) GDI values between 0 and 1 (no DNA damage), (ii) GDI values between 1 and 2 (little DNA damage), (iii) GDI values between 2 and 3 (moderate DNA damage), and (iv) GDI values between 3 and 4 (severe DNA damage). In addition, a clear dose-response relationship (concentration-DNA damage) must exist.
bGDI values for solvent (methanol) equivalent concentrations (between 39 and 618 mM) in the extracts.
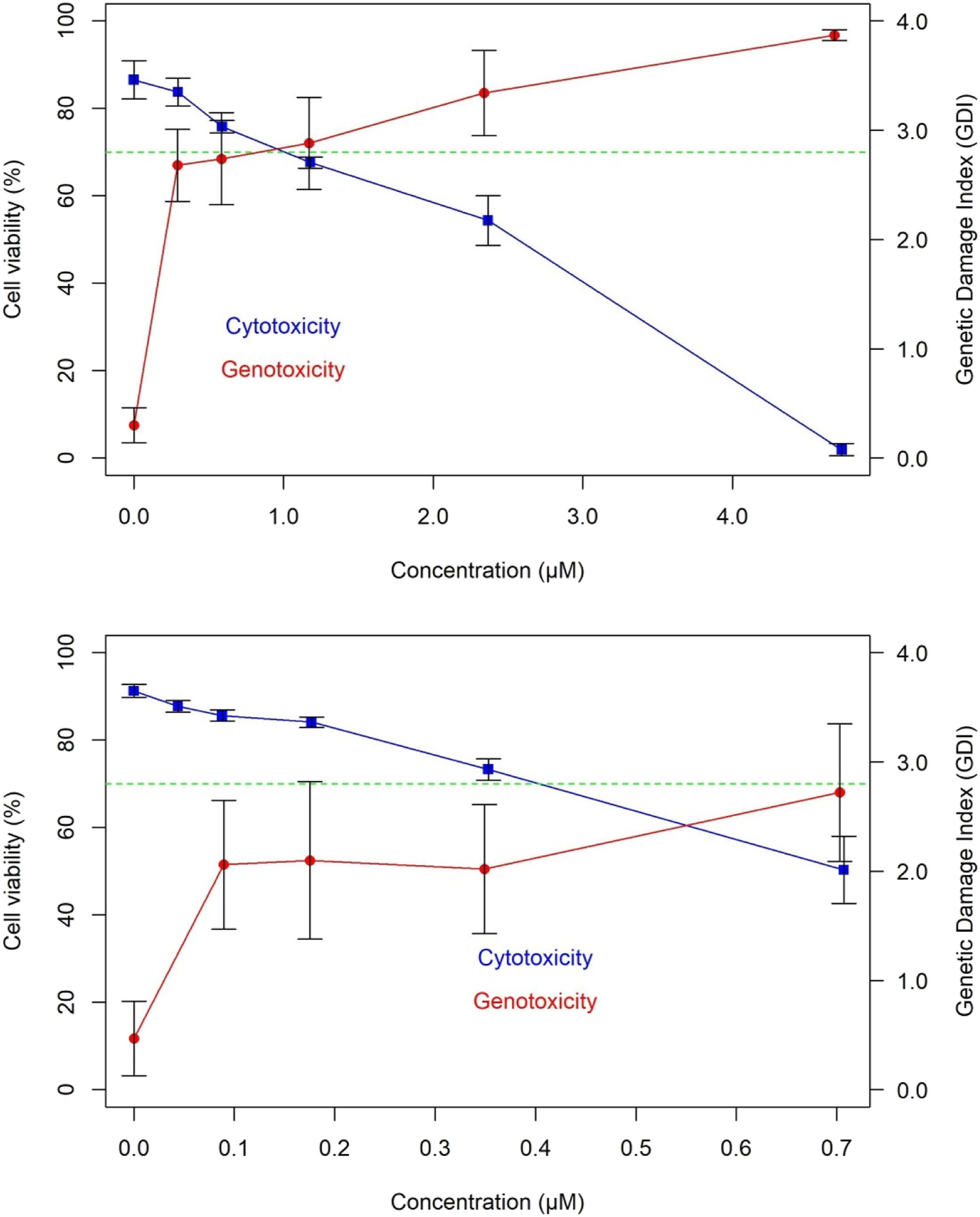
Cytotoxicity versus genotoxicity curves in human fibroblasts (MRC5) of the standard mutagens 4-nitro-quinoline-N-oxide (above) and bleomycin (below). The mean values of cell viability (%), genetic damage index (GDI), and their corresponding standard errors (SEM), calculated from at least three independent experiments (n = 3), are given.
Genotoxicity of the Polyphenol Compounds in MRC5 Human Fibroblasts Cells.
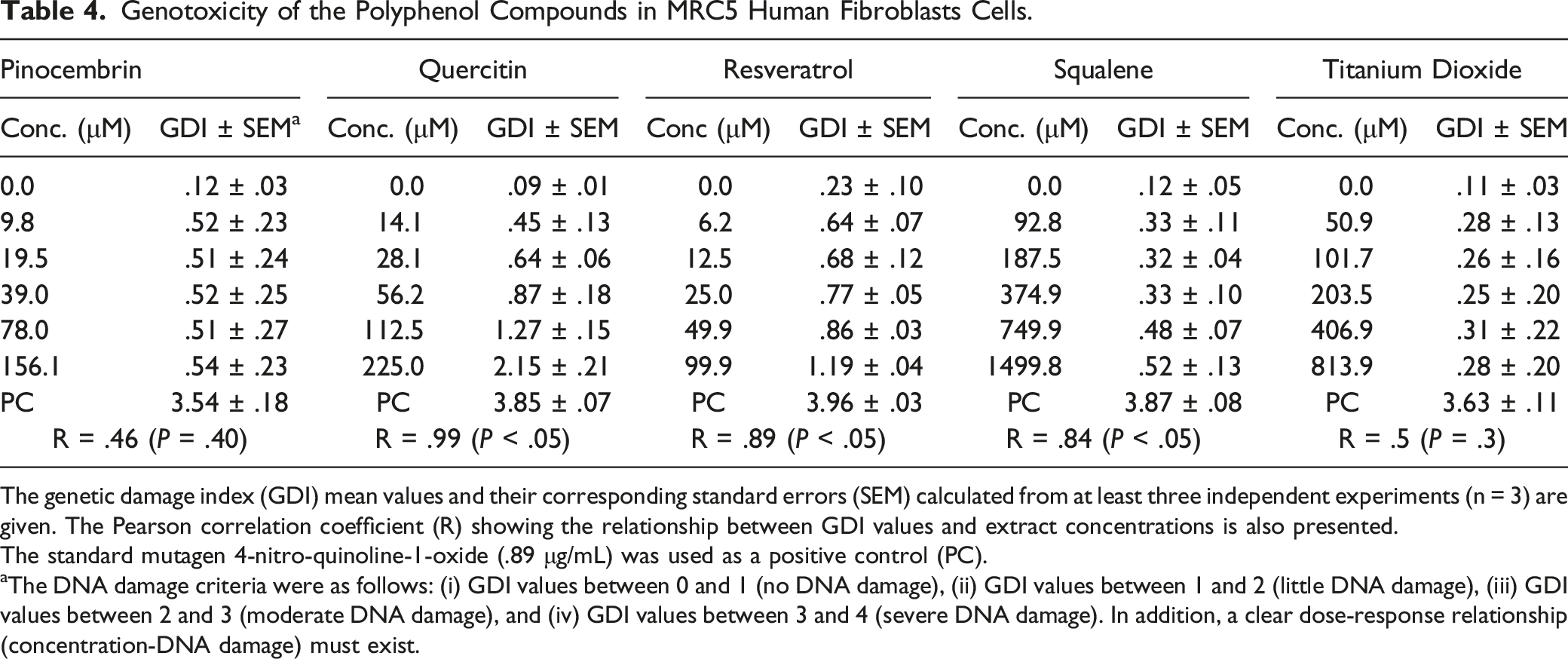
The genetic damage index (GDI) mean values and their corresponding standard errors (SEM) calculated from at least three independent experiments (n = 3) are given. The Pearson correlation coefficient (R) showing the relationship between GDI values and extract concentrations is also presented.
The standard mutagen 4-nitro-quinoline-1-oxide (.89 μg/mL) was used as a positive control (PC).
aThe DNA damage criteria were as follows: (i) GDI values between 0 and 1 (no DNA damage), (ii) GDI values between 1 and 2 (little DNA damage), (iii) GDI values between 2 and 3 (moderate DNA damage), and (iv) GDI values between 3 and 4 (severe DNA damage). In addition, a clear dose-response relationship (concentration-DNA damage) must exist.
Discussion
Relevant Information Available in the Literature on Activities of the Studied Plants.

There are some reports on cytotoxicity of these plant species, or closely related, on human non-skin cells. For example, the essential oils obtained from P. eriopodon and Salvia officinalis, a closely related species to S. aratocensis, showed high cytotoxicity in human breast and cervical cancer cell lines. 28 Although there are no reports in humans, the S. aratocensis essential oil has demonstrated cytotoxicity to mycobacteria. 29 The essential oil from C. odorata, a related species to C. pellia, resulted toxic in human breast and cervical cancer and embryonic kidney cell lines, 30 whereas the flavonoids and steroids from C. hirsute and C. squalida species show antimicrobial activity. 31 The synonymous genus Eupatorium has extensively demonstrated antimicrobial properties. 32 Finally, L. origanoides essential oil bioactivity has been widely studied, 33 including its toxic effects in different models such as protozoa, 34 insects,35–37 and crustaceans. 38
Conversely, we do not know positive cytotoxicity or toxicity reports of the A. satureioides species. This plant is used in Brazilian folk medicine as a digestive, eupeptic, emmenagogue, antispasmodic, anti-inflammatory, expectorant, and antidiarrheal agent. 39 The immunomodulatory and neuroprotective properties of this plant have been confirmed in pharmacological studies.40,41 This plant also shows anticancer activity42,43 and is non-cytotoxic and non-irritating to human skin, 44 supporting its use in topical administration. All these findings support the potential use of A. satureioides in cosmetic photoprotection.
On the other hand, the C. pellia and L. origanoides extracts also produced low-to-moderate DNA damage at cytotoxic concentrations. This genotoxic pattern contrasted with the results observed with known mutagens (Figure 3) that were genotoxic at cell viabilities ≥70%. This indicated that genotoxicity in C. pellia and L. origanoides extracts was related to cytotoxicity. 45 Previous studies46-48 have recommended testing concentrations presenting viabilities ≥70-75% to avoid false genotoxicity results with the Comet assay. In this sense, trypan blue exclusion and comet assays, which measure cytotoxicity (membrane disruption) and genotoxicity (DNA breaks), respectively, were complementary for this purpose. As in previous studies,15-18 our work demonstrated the usefulness of the high-throughput (96-well microplate) Comet platform, in this case, for safety testing of phytochemicals. Here, we replaced alkaline per enzymatic (proteinase K) lysis of the cells since step alkaline lysis produced high levels of DNA breaks in untreated fibroblast cells (data not shown), as previously observed by García-Forero et al. 49 Proteinase K also digests proteins associated with nuclei,50,51 an action that facilitates DNA unwinding similar to alkaline treatments. Our data support the idea that enzymatic lysis and alkaline incubation prior to electrophoresis are sufficient to unwind DNA, as previously shown in human buccal 52 and keratinocytes 53 cells.
We determined chemical composition of the studied plant extracts using gas chromatography coupled to mass spectrometry (GC/MS) analysis (Table 1). In accordance with previous studies, 19 we have confirmed their chemical composition. Some of the major compounds present in these plants were evaluated for their cytotoxicity and genotoxicity. Two relevant findings resulted from this analysis. First, trans-β-caryophyllene was the only compound that was cytotoxic over the entire range of concentrations evaluated. That is, the compound destroyed the membranes of human fibroblasts at all concentrations as was previously demonstrated54-56 These studies have explained cytotoxic activity of plant terpenoid compounds by their membrane disrupter capability that increase its permeability. Like previous studies,57,58 pinocembrin and resveratrol also showed some degree of cytotoxicity depending on concentration. Second, pinocembrin, resveratrol, squalene, and titanium dioxide were non-genotoxic and basically innocuous to human fibroblasts. Overall, the genotoxicity was related to cytotoxicity, except for quercetin, whose genotoxicity has been well documented.59,60 Our findings suggest that cytotoxic/genotoxic activities of the plant extracts could be related to a synergistic effect between its major compounds, as previously proposed, 61 but this hypothesis requires confirmation in further studies. In addition, the results emphasize the necessity of establishing safe plant extract and compound concentrations for their potential use in cosmetics.
Conclusion
We showed that the P. eriopodon and S. aratocensis extracts were clearly cytotoxic in human fibroblast cells. The A. satureioides, C. pellia, and L. origanoides extracts showed different degrees of cytotoxicity (CC50) as follows: L. origanoides (89 μg/mL) > A. satureioides (173 μg/mL) > C. pellia (184 μg/mL). L. origanoides and C. pellia extracts showed genotoxicity in human fibroblasts related to their cytotoxicity. A. satureioides, C. pellia, and L. origanoides extracts were neither cytotoxic nor genotoxic at relevant concentrations for photoprotection. Pinocembrin, squalene, and titanium dioxide were basically innocuous to human fibroblasts. Our findings unequivocally indicated the necessity of establishing safe plant extract concentrations for their potential use in cosmetics. That is, the plant extracts should be used at noncytotoxic (≤CC30 values) and nongenotoxic (GDI values ≤1) concentrations. Our data were based on fibroblast cell assays; therefore, at least a second evaluation in different skin cell models (e.g., keratinocytes and melanocytes) is recommended before its use for skin photoprotection.
Footnotes
Acknowledgements
The authors thank funding from the Ministry of Science, Technology, and Innovation, the Ministry of Education, the Ministry of Industry, Commerce, and Tourism, and ICETEX, Programme Ecosistema Científico-Colombia Científica, from the Francisco José de Caldas Fund, Grant RC-FP44842-212-2018. The Ministry of Environment and Sustainable Development of Colombia supported the Universidad Industrial de Santander through access permits to genetic resources and derivatives for bioprospecting (contract no. 270). The authors thank Carlos Frederico Martins Menck from Universidade de Sao Paulo (Sao Paulo, Brazil) for supplying the human fibroblast (MRC5) cell line and thank Dr. Robert Tulio González from Universidad del Pacífico (Buenaventura, Colombia) for supplying the Achyrocline satureioides photo.
Author Contributions
Flórez González, S. J. contributed to acquisition, analysis, and interpretation; Stashenko, E. E. contributed to analysis and interpretation; Ocazionez, R. E. contributed to analysis and interpretation; Vinardell, M. P. contributed to analysis and interpretation; Fuentes, J. L. contributed to conception and design, contributed to analysis and interpretation, and drafted the manuscript. All authors critically revised the manuscript, gave final approval, and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Science, Technology, and Innovation, the Ministry of Education, the Ministry of Industry, Commerce, and Tourism, and ICETEX, Programme Ecosistema Científico-Colombia Científica, from the Francisco José de Caldas Fund, Grant RC-FP44842-212-2018.
Live Subject Statement
The project RC-FP44842-212-2018 was approved by the Scientific Research Ethical Committee (Record No. 15 – 2017 and File No. 4110) from Universidad Industrial de Santander. The experiments and chemical management were performed according to the national law (Resolution No. 008430- 1993) from the Ministry of Health of Colombia and the Institutional Manual of Integrated Management and Processes (PGIR–PGGA.05).
