Abstract
Diethylpropion has been available in the market for treating obesity for over 50 years. Refined studies are lacking to fully elucidate its action spectrum. The aim of our study was to evaluate possible toxic effects of anorectic diethylpropion in Chinese hamster ovary (CHO) cells. Comet assay (detects breaks in the DNA strand), micronucleus test (detects clastogenic/aneugenic damage), and cell survival test (detects cytotoxic damage) were used to evaluate the toxic effects. In comet assay, we found that the damage scores with diethylpropion treatments at the concentrations of 20 and 40 μg/mL were more significant ( p < 0.05) than that of the negative control. When assessing the possible aneugenic and/or clastogenic damage caused by the drug in CHO cells, we found no difference ( p > 0.05) in the values of micronucleated cells when comparing different diethylpropion treatments and the negative control. Regarding the cell viability, for all the diethylpropion concentrations tested, higher values ( p < 0.05) of apoptosis were found compared with those of the negative control. In relation to the number of necrotic cells, no difference ( p > 0.05) was noted between the means of the three concentrations of diethylpropion evaluated and the negative control. In the experimental conditions, we conclude that diethylpropion has weak genotoxic and cytotoxic activities.
Introduction
Obesity is linked to the development of numerous diseases affecting humans. The etiology of obesity is multifactorial and may arise from genetic, environmental, or behavioral factors. In physiological terms, obesity occurs when there is an imbalance between caloric intake and energy expenditure. The excess energy is accumulated in the body as fat. The classification adopted for assessing whether an individual is obese considers the body mass index (BMI). The BMI is the defined as the ratio of the body weight in kilograms to the square of height. Individuals with a BMI > 30 kg/m2 are classified as obese. 1,2
The number of obese people has increased at an alarming rate worldwide. This medical condition is a cause of concern to public health services, particularly because it affects children or young people in the productive phase. The losses caused by the disease not only affect patients and their families but society as well because an increasing obese population also increases the prevalence of other related diseases such as diabetes, heart disease, and some cancers. Thus, there is a need for social mobilization to mitigate the damage caused by obesity and seek solutions to address this “epidemic” of modern times. 3,4
There are several strategies for the treatment of obesity, and the success of treatment usually results from the combination of therapeutic measures. Behavioral changes, physical activities, and nutritional education have been the initial prescriptions for treatment. When these measures fail, anorectics are prescribed as a complementary tool to therapy. After failure of all these attempts, the alternative of bariatric surgery is considered. 5 –7
Diethylpropion hydrochloride is a phenylethylamine derivative drug that is also known as amfepramone. Diethylpropion shows sympathomimetic action, stimulating the sympathetic nervous system by increasing the release of dopamine (DA) and norepinephrine (NE) and inhibiting their catchment. Despite the stimulating action of diethylpropion, its effect is weaker than that caused by using amphetamine. Diethylpropion has been available in the market for treating obesity for over 50 years, and refined studies are lacking to fully elucidate its action spectrum; additionally, its use as a low-cost medicine is associated with improper use, particularly in Brazil. 8 –10
Diethylpropion has anorectic effects similar to that of amphetamine, and it produces central nervous system (CNS) stimulation and elevation of blood pressure. Repeated use leads to the development of tolerance. The mechanism of action of diethylpropion is unknown; as an anorexigenic, it remains unknown if it only suppresses hunger or demonstrates other effects on metabolism and the CNS. The drug is rapidly absorbed and metabolized in the gastrointestinal tract, and some of its metabolites are active. The amfepramone and its metabolites can cross the blood–brain barrier. Elimination occurs through the kidneys after 48 h of ingestion in a form that is metabolized or unchanged. 11,12
The main adverse events of diethylpropion include chest pain, arrhythmia, changes in the electroencephalogram profile, convulsive episodes (epileptics), psychotic episodes, anxiety, euphoria, depression, dysphoria, tremor, mydriasis, headache, changes in libido, gynecomastia, menstrual irregularities, hives, rash, agranulocytosis, leukopenia, dysuria, dyspnea, hair loss, psychic dependence, and schizophrenia. With abrupt discontinuation of consumption of the drug, the individual expresses extreme fatigue and mental depression. 11,12
The consumption of anorectics in Brazil has increased significantly in recent years. Between 1988 and 1992, 72,940 kg of anorectic drugs were consumed with the following annual consumption rates: 7780 kg in 1988, 10,040 kg in 1989, 13,366 kg in 1990, 18,113 kg in 1991, and 23,648 kg in 1992. In 1992, 17,002 kg of diethylpropion, 5516 kg of fenproporex, and 160 kg of mazindol were consumed. In 1988, the use of anorectics was set to 4.60 daily doses (defined daily doses (DDDs)/1000 inhabitants/day) and increased to 10.05 DDDs in 1992. In 1988, diethylpropion represented 45.2% of all anorectics commercialized in Brazil. From 1987 to 1991, the number of pharmacies using anorectics in their formulas increased by 536%. 13,14
The marketing of diethylpropion is controlled, but irregular marketing and self-medication are possible, with several lines of evidence of sales via the Internet. 13 The danger of indiscriminate use increases because, despite its availability in the market for decades, no conclusive study has evaluated its carcinogenic potential due to prolonged use and its mutagenic potential. 15
The present study aimed to contribute to the elucidation of possible genotoxic and/or cytotoxic damage caused by diethylpropion. Comet assay (detects breaks in the DNA strand), micronucleus test (detects clastogenic/aneugenic damage), and cell survival test (detects cytotoxic damage) were used to evaluate the effects of diethylpropion. These tests are widely used and approved by regulatory agencies worldwide. As an “in vitro” test system, cultured Chinese hamster ovary (CHO) cells were used.
Materials and methods
Chemicals
The chemical name for diethylpropion hydrochloride is 1-phenyl-2-diethylamino-1-propanone hydrochloride (CAS 134-80-5). The structure of diethylpropion hydrochloride is shown in Figure 1. The drug was obtained from Farmoderm (Ribeirão Preto, São Paulo (SP), Brazil).
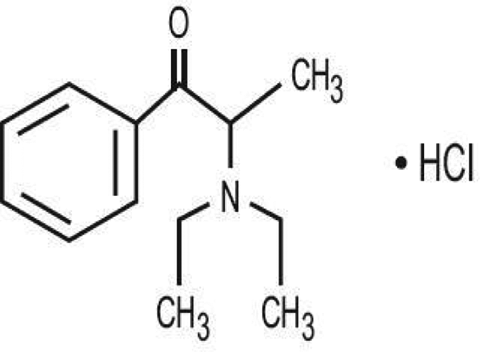
Chemical structure of diethylpropion hydrochloride. 15
Anthracycline antibiotic doxorubicin (Doxolem®—chlorohydrate of doxorubicin; CAS Registry 23214-92-8; Zodiac Produtos Farmacêuticos S/A, Pindamonhangaba, SP, Brazil; Figure 2), which is commonly used to treat various cancers, has been used as the positive control. The primary mechanism of doxorubicin action is likely to be through an interaction with topoisomerase II. The effects of doxorubicin involve the production of free radicals, an increase in DNA strand breakage, chromosomal aberrations, and increases in micronucleus formation in vitro and in vivo. 16

Chemical structure of anthracycline antibiotic doxorubicin.
Cell culture
The CHO cell line CHO-K1 was obtained from the Laboratory of Cytogenetics and Mutagenesis, Faculty of Medicine of Ribeirão Preto, Ribeirão Preto, SP, Brazil. CHO-K1 cells were grown in 25-cm2 culture flasks (Corning Glass, Corning, New York, USA) and allowed to grow until confluence using 10 mL of culture medium (Ham’s-F10 + Dulbecco’s modified Eagle’s medium (1:1); Invitrogen, Grand Island, New York, USA and Sigma-Aldrich, St Louis, Missouri, USA) supplemented with 10% fetal bovine serum (Cultilab, Campinas, SP, Brazil), 1.2 g/L sodium bicarbonate (Steve Regan, Salt Lake, Utah, USA), and antibiotics (0.06 g/L penicillin, 0.10 g/L streptomycin (Sigma-Aldrich), 0.05 g/mL kanamycin (Gibco, SP, Brazil), and 0.2% ciprofloxacin (Ciprofloxan®; Hipolabor, Sabará, Minas Gerais, Brazil) and 4-(2-hydroxyethyl)piperazine-1-ethanesulfonic acid (2.38 g/L; Serva, Heidelberg, Germany). The cells were then incubated at 37°C in an atmosphere with 5% carbon dioxide. Cells that reached confluence were transferred to a centrifuge tube and centrifuged at 1000 r/min (180 g) for 5 min. After centrifugation and discarding of the supernatant, the pelleted cells were resuspended and counted in a Neubauer chamber. A total of 5 × 105 cells were placed in each well of a six-well plate (Corning Glass).
Diethylpropion treatments
The doses of diethylpropion used in the present study were determined through pilot testing. The treatment scheme follows the following stages:
At the zero time point, 5 × 105 cells were seeded per well of a culture plate; After 20 h, the culture medium was exchanged and treatment with diethylpropion was initiated; After 40 h, the rates for performing the comet assay and cell viability was halted and cytochalasin B treatment was applied to accomplish in the micronucleus test; After 54 h, the rates for performing the micronucleus test was halted.
Later, the following treatments were carried out:
negative control (culture medium); positive control (doxorubicin, 0.15 µg/mL); diethylpropion, 10 µg/mL; diethylpropion, 20 µg/mL; and diethylpropion, 40 µg/mL.
We performed three independent experiments, and both the drug and positive control were dissolved in the culture medium. To measure cytotoxicity in pilot test, trypan blue assay was used.
Micronucleus test in CHO cell culture
The technique used to examine micronuclei in binucleated cells is the same as that described by Fenech and Morley. 17 After exposure at different time period to the treatments, the cells were washed with 6 mL of Hanks’ solution, trypsinized (0.24% trypsin-0.02% Versene; Instituto Adolfo Lutz, Brazil), and centrifuged for 5 min at 900 r/min. The cells were resuspended in 5 mL of hypotonic solution (1% sodium citrate) at 4°C. Three drops of formaldehyde were added, and the mixture was homogenized gently using a Pasteur pipette. This cell suspension was centrifuged again at 900 r/min for 5 min and resuspended in 5 mL of fixative solution (methanol/acetic acid 3:1 v/v). This operation is repeated twice, and the supernatant was discarded. The cell suspension was applied to a microscopy slide with a film of distilled water at 4°C. The slides were stained with Giemsa stain diluted in 3% phosphate-buffered solution (PBS; 0.06 M disodium hydrogen phosphate and 0.06 M monopotassium phosphate, pH 6.8) for 5–10 min, washed with distilled water, and allowed to dry at room temperature for microscopic analysis. Quantification of micronuclei was determined in 1000 binucleated cells with preserved cytoplasm. The criteria for identifying the micronucleus are the same as those recommended by Fenech 18 and Titenko-Holland et al. 19
Nuclear division index
The nuclear division index (NDI) was determined by analyzing 1000 cells in each slide. Only cells with a preserved cytoplasm containing from 1 to 4 nuclei were counted. Counting was aided by a digital cell counter and an optical microscope. NDI was calculated according to the method described by Eastmond and Tucker
20
using the following formula:
where M1, M2, M3, and M4 represent the number of cells with 1, 2, 3, or 4 nuclei, respectively and N represents the total number of cells analyzed.
Determination of the apoptotic index
First, 100 µL of dye solution consisting of 25 µL of propidium iodide dissolved at a concentration of 5 µg/mL in distilled water, 50 µL of fluorescein diacetate at a concentration of 15 µg/mL in dimethyl sulfoxide (DMSO), and 10 μL Hoechst 33342 at a concentration of 2 µg/mL in water and 15 µg/mL of PBS (pH = 8) was prepared.
Next, both the culture medium and the adherent cells (after trypsinization) from each flask were placed into a centrifuge tube, and the mixture was centrifuged at 1000 r/min for 5 min. The supernatant was then discarded, leaving a small amount to resuspend the cells.
Into a centrifuge tube was placed 100 µL of the cell suspension and 2 µL of the dye solution. The contents were mixed and incubated in a water bath at 37°C for 5 min. Next, 15 µL of this mixture was placed on a slide and covered with a coverslip. The cells were then immediately counted using a fluorescence microscope with a triple filter. In normal cells, the cytoplasm appears green and has an intact nucleus; apoptotic cells show green cytoplasm with a fragmented nucleus (apoptotic bodies stained blue), and necrotic cells in the nucleus and cytoplasm appear homogeneously stained in red. Three independent experiments were performed, and 500 cells per treatment were evaluated in each experiment.
Comet assay
The single-cell electrophoresis assay, or comet assay, was performed under alkaline conditions. Briefly, 15 μL of each culture cell sample was mixed with 160 μL of 0.5% low-melting-point agarose (Invitrogen, Carlsbad, California, USA) at 37°C and layered onto a conventional microscope slide precoated with 1.5% normal melting point agarose (Invitrogen). The slides were placed overnight in cold, freshly prepared lysing solution (2.5 mol/L sodium chloride, 100 mmol/L ethylenediaminetetraacetic acid (EDTA) disodium salt, 10 mmol/L, pH 10, tris(hydroxymethyl)aminomethane (Tris) buffer, 10% DMSO, and 1% Triton X-100) and then into a horizontal electrophoresis buffer (1 mmol/L EDTA disodium salt and 0.3 mol/L sodium hydroxide) at 4°C for 20 min. Using the same buffer, electrophoresis was performed at 25 V and 300 mA for 20 min. After electrophoresis, the slides were rinsed three times in neutralizing buffer (0.4 mol/L, pH 7.5, Tris-hydrochloric acid), fixed for 5 min in absolute alcohol, air-dried, and stored at room temperature. Immediately before analysis, DNA was stained with silver and examined with a 40× objective using a light microscope. Coded slides were scored in a blind test, and 100 culture cells were randomly analyzed for each treatment (50 cells per slide from two slides per treatments). The cells were scored visually into five classes according to tail size from undamaged (0) to maximally damaged, 4 and a value (damage index) was assigned to each comet according to its class. The damage index thus ranged from 0 (completely undamaged: 100 cells × 0) to 400 (with maximum damage: 100 cells × 4). The comets with a “cloudy” appearance or a very small head and a tail like a balloon (necrotic/apoptotic cells) were excluded from the evaluation under the assumption that they represented dead cells. 21 –23
Statistical analysis
We used the statistical software Sigma Stat for Windows version 1.0. We applied analysis of variance and Student’s t tests with α = 0.05.
Results
In comet assay, we found that the DNA damage scores in cells treated with diethylpropion at concentrations of 10, 20, and 40 µg/mL in culture medium had higher values ( p < 0.05) than those in the negative control cells. No difference ( p > 0.05) in the DNA damage score was noted between cells that were treated with diethylpropion at the concentrations of 10 and 40 μg/mL (Figure 3). In the latter experiment, observations made after 2 h of treatment showed increased damage scores directly related to the increased concentration of diethylpropion. Additionally, after 6 h of treatment, the DNA damage scores in cells treated with diethylpropion concentrations of 20 and 40 µg/mL were equivalent and superior to those in cells treated with 10 µg/mL diethylpropion (data not shown).

DNA damage scores (average values divided by 1000 and standard deviation) in CHO cells 20 h after treatment. C: negative control, DEM-F10; DOX: doxorubicin, 0.15 µg/mL; 10, 20, and 40: diethylpropion, 10, 20 and 40 μg/mL in culture medium, respectively. CHO: Chinese hamster ovary.
We evaluated the possible aneugenic and/or clastogenic damage (Figure 4) caused by diethylpropion in CHO cells and verified that no difference ( p > 0.05) was found in the values of the frequencies of micronucleated cells when we compared the cells with different diethylpropion treatments and negative control cells. However, with 20 µg/mL of diethylpropion, a slight increase in numerical value could be observed, but the difference was not statistically significant ( p > 0.05) to be attributed to interference by the drug.

Average frequency of micronuclei in CHO cells (1000 cells) after treatment for 20 h. C: negative control, DEM culture medium-F10), DOX: doxorubicin, 0.15 μg/mL; 10, 20, and 40: diethylpropion, 10, 20, and 40 μg/mL in culture medium, respectively. CHO: Chinese hamster ovary.
Additionally, no difference was observed ( p > 0.05) in the NDI among the treatments. Both the controls and different concentrations of diethylpropion showed equal NDI values (Figure 5).

Average NDI values in CHO cells 20 h after treatment. C: negative control, DEM-F10; DOX: doxorubicin, 0.15 μg/mL; 10, 20, and 40: diethylpropion, 10, 20 and 40 μg/mL of culture medium, respectively; 1000 cells were analyzed per treatment. NDI: nuclear division index; CHO: Chinese hamster ovary.
We evaluated the cell viability and found that, for all the concentrations tested, diethylpropion treatment showed higher values ( p < 0.05) of apoptosis compared with the negative control. Additionally, no difference ( p > 0.05) was found in the rates of apoptosis among the diethylpropion treatments at concentrations of 10 and 40 μg/mL. Treatment with an intermediate diethylpropion concentration (20 μg/mL) produced an apoptosis rate slightly lower ( p < 0.05) than that for the other two concentrations (Figure 6).
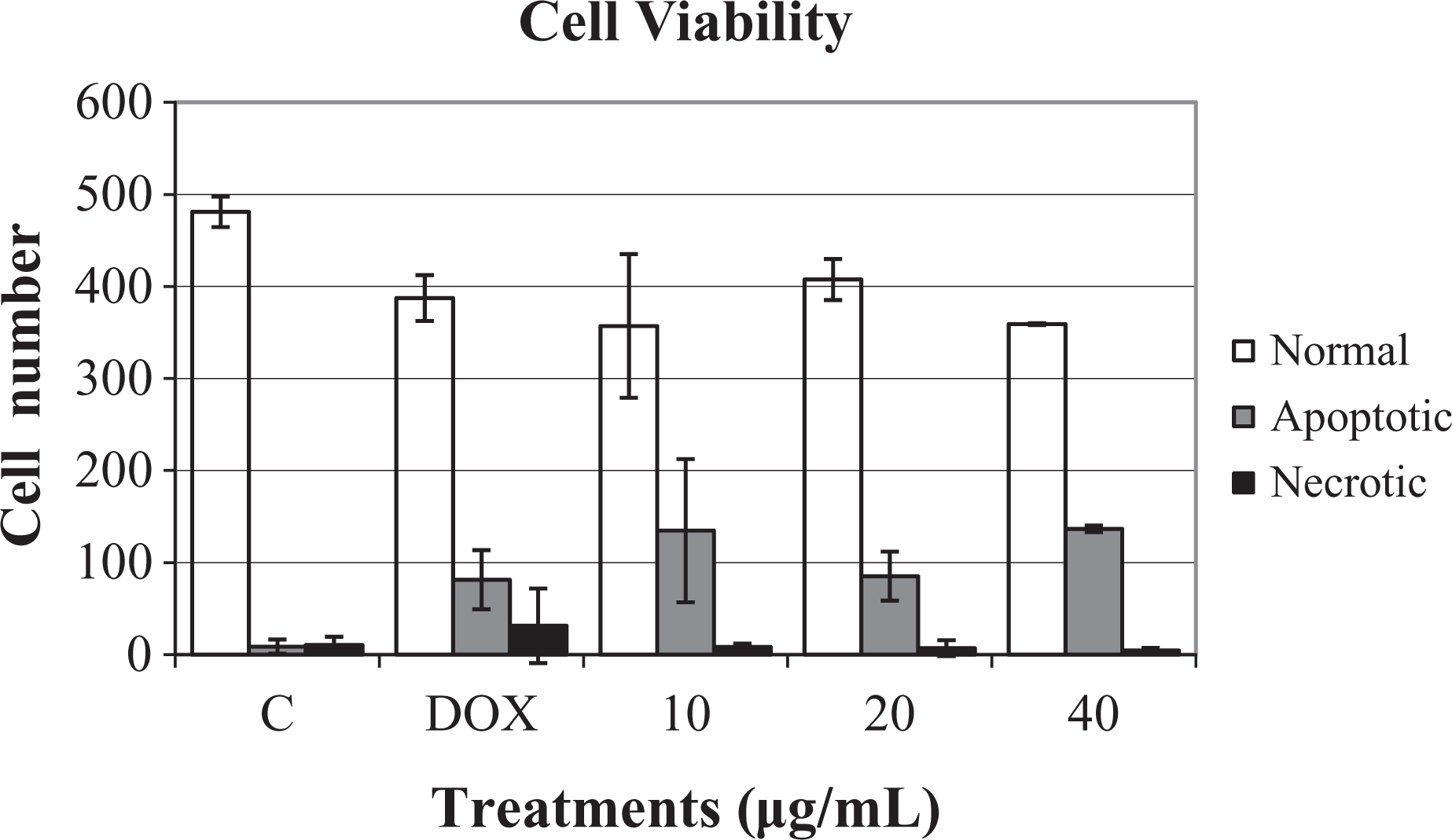
Cell viability in CHO cells 20 h after treatment. C: negative control, DEM-F10; DOX: doxorubicin, 0.15 μg/mL; 10, 20, and 40: diethylpropion, 10, 20 and 40 μg/mL of culture medium, respectively; 500 cells were analyzed per treatment.
Regarding the number of necrotic cells, no difference ( p > 0.05) was found between the means of the three concentrations of diethylpropion evaluated and the negative control (Figure 6).
Discussion and conclusion
Diethylpropion induces the release and inhibits the reuptake of DA and NE by neuronal terminals. Diethylpropion, similar to fenproporex, is structurally similar to phenylethylamine found in amphetamine. 14
Diethylpropion is rapidly absorbed from the digestive tract and is extensively metabolized to 2-ethylamino-1-phenyl-propan-1-one and N,N-diethylnorephedrine, both of which are excreted almost exclusively by the kidneys. In the study by Yu et al., 23 to verify the physiological activity of diethylpropion they found that the drug was inactive (prodrug) for the release and uptake of biogenic amines. That study also indicated that the metabolite 2-ethylamino-1-phenyl-propan-1-one is responsible for its biological effects. Another interesting aspect of that study was that the metabolite 2-ethylamino-1-phenyl-propan-1-one is a substrate for the carrier NE, which inhibits the reuptake of DA and its transporter. This finding suggests that diethylpropion in therapeutic doses is related to increased NE in the brain and not to increased DA. 24 Because we used an in vitro system in the present study, we could not ensure that the drug was metabolized by the cellular system. Therefore, we interpret our data with caution and suggest that a future “in vivo” study is warranted for better understanding of the metabolism of diethylpropion.
When evaluating possible DNA damage using the comet assay, we observed the that the DNA damage scores in CHO cells that received the intermediate concentration of diethylpropion (20 µg/mL) was higher than those in treatments with the lower dose (10 µg/mL) and higher dose (40 µg/mL) of diethylpropion. One possible explanation for this behavior is that, at 20 µg/mL diethylpropion, a balance is observed between drug internalization into the cell and cell death. At this concentration, the cell can internalize the drug and remain viable, enabling the drug (or its metabolites) to reach the nucleus and interfere with DNA activity. At the lower diethylpropion concentration (10 µg/mL), the drug may exert a direct weak genotoxic action and does not reach the effective dose to significantly increase the DNA damage score. The higher (40 µg/mL) diethylpropion concentration may be destroying the cells or saturating membrane transporters, preventing the internalization of the drug. Similar results were reported by da Silva et al. in their in vivo study with fenproporex. 25
The weak genotoxicity observed in our study may be attributable to culture conditions not producing the neuroamines that accelerate cellular metabolism to form excess free radicals and consequently intensify the damage to biological macromolecules. Galvan-Arzate and Santamariá 26 studied the effect of diethylpropion administered at a concentration of 5 mg/kg in rats for 15 consecutive days and found the occurrence of excitotoxic/cytotoxic effects, release of free radicals and cause of oxidative stress. They also found that, in certain areas of the CNS (hippocampus), lipid peroxidation increased by 75% compared with the control. The results are in compliance with other derivatives of amphetamine and methamphetamine that the production of free radicals is responsible for the toxic mechanism. 26 Thus, the damage observed, in the present study, is possibly due to the direct action of the drug under culture conditions. Regarding the biological action of diethylpropion in vivo occurring through the increase in NE, and the study by Mao et al, too revealed that the neurotransmitter NE, at high concentrations, causes damage to various cellular components via generation of free radicals. They claimed that NE causes apoptosis in many cell types, including neurons and heart cells. Oxidative stress also causes changes in the endoplasmic reticulum that may result in cell death. 27
The possible clastogenic/aneugenic effects of diethylpropion allow us to suggest that a weak direct action exists. Similar to what happened with the damage index, the average frequency of micronucleated cells was higher at the intermediate diethylpropion concentration (20 µg/mL), likely because, at this dose, the drug reaches the optimal peak cell absorption to interfere with the more effective mechanisms of duplication and segregation of chromosomes. The clastogenic/aneugenic effect may be attributed to N-dialkyl present in the molecule. Da Silva Nunes et al., contrary to what was observed in our study, found increase of micronucleus in buccal exfoliated cells of female due to intake of amfepramone at 120 mg/day, confirming that the metabolites are responsible for the strong genotoxic effect. 28 However, increases in fixing DNA damage at this dose have no dose–response correlation, and no difference is found between the treatments with the drug and the negative control. Because the above are important factors in interpreting positive genotoxic effects by the researcher, we interpret our results with caution and suggest a weak genotoxic effect of the drug at the µg/mL concentration in vitro, which is positive in vitro, is not always a risk in vivo.
The verification of damage by the comet assay and the nonconfirmation by the micronucleus test can be interpreted that most DNA damage injuries are transitory and can be repaired. 29 The in vitro tests used alone serve as indicators for characterizing risk. When combined with other toxicological tests, the in vitro tests are an important tool for application in the field of cytotoxicity and genotoxicity. The tests are low cost and can be performed quickly, allowing to the selection of various compounds for detailed studies. The limitation of in vitro tests include the following: they do not evaluate cells or tissues of all the organs; it is not possible to assess systemic kinetics (absorption, penetration, distribution, and excretion); they do not allow confirming an integral action of the various systems on the effects the drug; and the physicochemical properties of the compound may hinder testing. Despite these difficulties, the in vitro tests are universally accepted and used to compose a battery of genotoxic/cytotoxic tests. 30
Programmed cell death or apoptosis occurs in several pathological situations during development. Apoptosis can be triggered by various stimuli, including DNA damage, starvation, ultraviolet radiation, anticancer drug, and viral infection. Dysfunction of apoptotic mechanisms can lead to degenerative diseases, immunodeficiency, cancer, autoimmune diseases, and viral infections. 31 Necrosis occurs when cells are exposed to an extreme variation of the physiological conditions occurring in the membrane damage leading to cell death.
In cell viability tests, diethylpropion was verified to interfere with the metabolism of CHO cells, triggering programmed cell death. Confirming what was stated above in relation to DNA damage in the cells that received the drug at a concentration of 20 μg/mL, the apoptosis rate was smaller with 10 μg/mL and 40 μg/mL diethylpropion. The latter finding suggests the same explanation given above that 10 μg/mL diethylpropion allowed internalization of the drug into the cell and interference with the mechanism of apoptosis. Additionally, at 20 μg/mL, diethylpropion reaches equilibrium, allowing the cell to be viable. Furthermore, at 40 μg/mL diethylpropion, saturation occurs, activating the mechanisms of apoptosis. In necrosis test was not verified significant differences.
The lack of evidence of teratogenic and mutagenic effects of diethylpropion 15 should be seen as being due to lack of in vivo studies and not that the drug was harmless. This is because diethylpropion is expected to produce similar toxic behavior to amphetamine, which has demonstrated teratogenic and mutagenic effects. 32 In the experimental condition, we conclude that diethylpropion has weak genotoxic and cytotoxic activity.
Footnotes
Acknowledgments
The authors are grateful to Álvaro Bisol, Salvador de Carvalho, and Marinez SR Goyano for their scientific contributions and insights. We also thank Dr Aline Poerch and Dr Patricia Carminati for their technical and scientific support.
Conflict of interest
The authors declared no conflicts of interest.
Funding
This work was sponsored by CNPq, CAPES, FAEPA, HCFMRP, SEEGO e FMRP.

