Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) assessed the safety of Humulus Lupulus (Hops) Extract (reported functions include antimicrobial agent and hair conditioning agent) and Humulus Lupulus (Hops) Oil (reported function is fragrance). The Panel reviewed the relevant data related to these ingredients. Because final product formulations may contain multiple botanicals, each containing the same constituents of concern, formulators are advised to be aware of these constituents and to avoid reaching levels that may be hazardous to consumers. For these ingredients, the Panel was concerned about the presence of 8-prenylnaringenin, β-myrcene, and quercetin in cosmetics, which could result in estrogenic effects, dermal irritation, and genotoxicity, respectively. Industry should use current good manufacturing practices to limit impurities and constituents of concern. The Panel concluded that Humulus Lupulus (Hops) Extract and Humulus Lupulus (Hops) Oil are safe in cosmetics in the present practices of use and concentration when formulated to be non-sensitizing.
Introduction
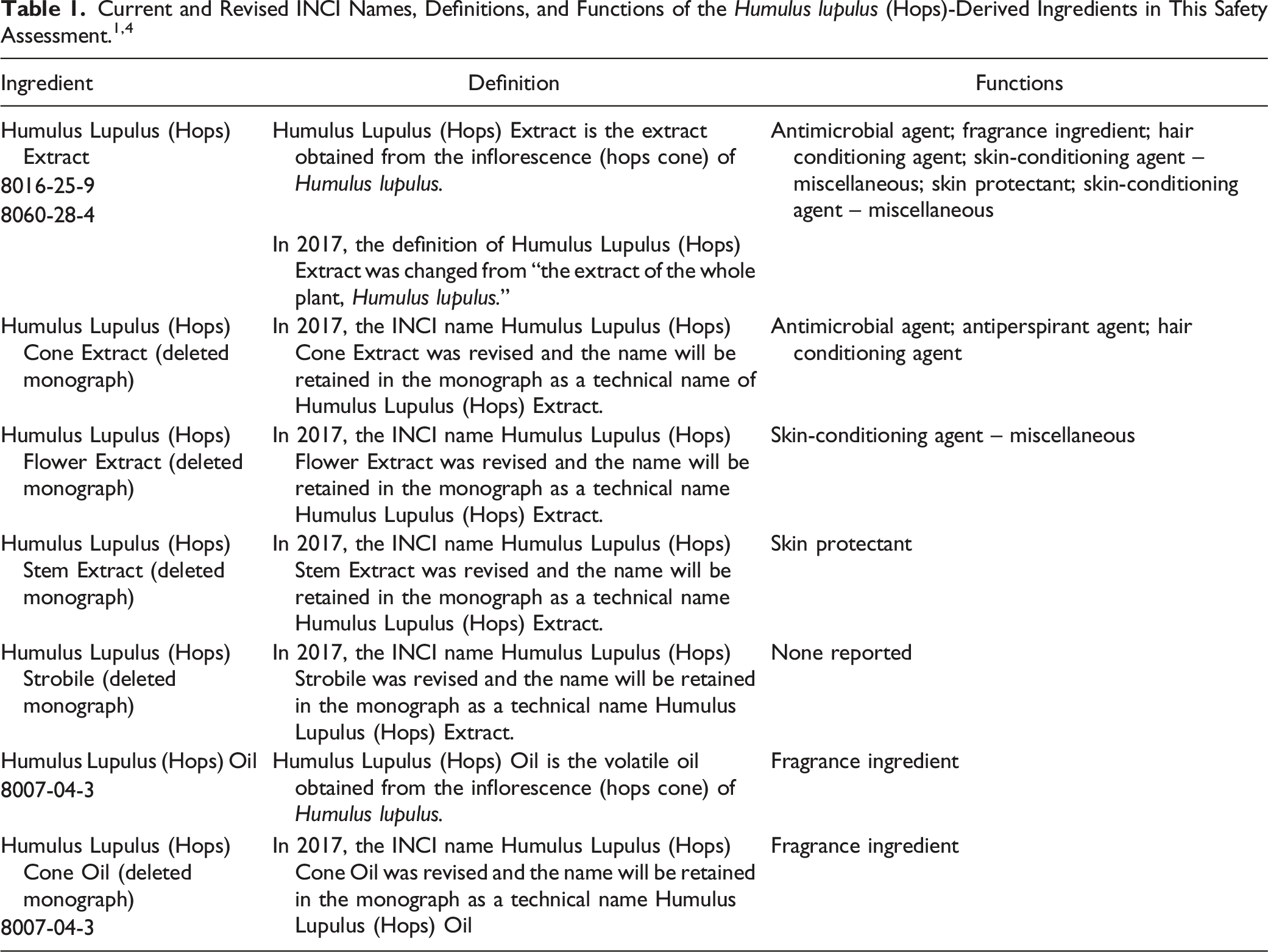
Previously, the Dictionary listed four other Humulus lupulus (hops)-derived ingredients: Humulus Lupulus (Hops) Cone Extract, Humulus Lupulus (Hops) Flower Extract, Humulus Lupulus (Hops) Stem Extract, and Humulus Lupulus (Hops) Strobile (Table 1). 4 It was determined by the International Nomenclature Committee (INC) that these ingredients were all extracts of the inflorescence (hops cone) of the Humulus lupulus (hops) plant. It was also determined that the previous definition of Humulus Lupulus (Hops) Extract, i.e., the extract of the whole plant, was erroneous and that this ingredient is also the extract of the inflorescence (hops cone). To correct these errors, these five ingredients were deemed synonymous and the single name Humulus Lupulus (Hops) Extract is now the official INCI name. Additionally, Humulus Lupulus (Hops) Cone Oil (the volatile oil obtained from the cones of Humulus lupulus), has been changed to Humulus Lupulus (Hops) Oil and the definition is now “the volatile oil obtained from the inflorescence (hops cone) of Humulus lupulus”. Data have been submitted under the revised names; with the exception of the cosmetic use data, these data are presented under the corrected names, Humulus Lupulus (Hops) Extract and Humulus Lupulus (Hops) Oil.
The terms “inflorescence,” “cone,” and “strobile” refer, synonymously, to the structures formed by the female Humulus lupulus (hops) plant after the flowers have bloomed, whether or not they have been fertilized. Thus, for example, “cone oil” is the same as “strobile oil. Both “cone” and “strobile” are used in the literature. Also, “hops” is used commonly for the plant name as well as for the harvested strobiles; in the literature, it is not always clear if authors are referring to the whole plant or just the strobiles. In this report, it is assumed that the authors are referring solely to the strobiles, unless otherwise indicated.
Most of the parts of Humulus lupulus (hops) (i.e., shoots, leaves, flowers, seeds, rhizomes, and essential oils) are edible; the strobile is the most commonly consumed in food (mostly in beer).5,6 The U.S. Food and Drug Administration (FDA) has determined that essential oils, oleoresins (solvent-free), and natural extractives (including distillates) of Humulus lupulus (hops) are generally recognized as safe (GRAS) for human consumption. [21CFR182.20] Consumption of these Humulus lupulus (hops)-derived foods would result in much larger systemic exposures than what is expected from use of ingredients in cosmetic products, even if there was 100% dermal absorption. Thus, the systemic toxicity potential of Humulus lupulus (hops)-derived ingredients is not the focus of this safety assessment (although such information may be included). The primary focus of this safety assessment is the review of safety based on local effects (e.g., topical exposures).
Botanical cosmetic ingredients, such as Humulus lupulus (hops)-derived ingredients, may contain hundreds of constituents, some of which have the potential to cause toxic effects. For example, sesquiterpene lactones, which are present in the Humulus lupulus (hops) plant, may cause Type IV allergic reactions (cell-mediated, delayed-type hypersensitivity) and other toxicity when present in sufficient amounts. Another example, β-myrcene, is reported to be a dermal irritant and a possible carcinogen (including in a National Toxicology Program (NTP) report).7-11 In this safety assessment, the Panel is reviewing information available to evaluate the potential toxicity of each of the Humulus lupulus (hops)-derived ingredients as whole, complex ingredients. Except for specific constituents of concern, the Panel is not reviewing information that may be available to assess the potential toxicity of the individual constituents derived from the Humulus lupulus (hops) plant.
The Panel has reported on related ingredients that can be used to support the safety of the Humulus lupulus (hops)-derived ingredients. The information on these related ingredients may be relevant for this safety assessment. The Panel reviewed the safety of phytosterols, which are plant-derived sterols that are likely constituents of most of the Humulus lupulus (hops)-derived ingredients 12 in 2013, and concluded that the phytosterols are safe as used. 13
The names of the ingredients in this report are written in accordance with the Dictionary, as shown above, capitalized without italics and without abbreviations. When referring to the plant from which these ingredients are derived, the standard taxonomic practice of using italics will be followed (e.g., Humulus lupulus).
Often in the published literature, the information provided is not sufficient to determine how well the tested substance represents the cosmetic ingredient; the taxonomic name is used, unless it is clear that the test substance is similar to the cosmetic ingredient. If it is similar to the cosmetic ingredients, then the INCI name is used.
This safety assessment includes relevant published and unpublished data that are available for each endpoint that is evaluated. Published data are identified by conducting an exhaustive search of the world’s literature. A listing of the search engines and websites that are used and the sources that are typically explored, as well as the endpoints that the Panel typically evaluates, is provided on the Cosmetic Ingredient Review (CIR) website (https://www.cir-safety.org/supplementaldoc/preliminary-search-engines-and-websites; https://www.cir-safety.org/supplementaldoc/cir-report-format-outline). Unpublished data are provided by the cosmetics industry, as well as by other interested parties.
Chemistry
Definition
The definitions of these Humulus lupulus (hops)-derived ingredients (with technical names, included for cross reference) are provided in Table 1.
The terms “cone” and “strobile” are synonymous and refer to the structures formed by the female Humulus lupulus (hops) plant after the flowers have bloomed, whether or not they have been fertilized.
Plant Identification
The genus Humulus consists of dioecious, perennial, climbing vines and bines (i.e., a twining plant stem or flexible shoot). 14 This genus belongs to the Cannabaceae family of the Urticales order which, in 2003, was incorporated into the natural order of Rosales. 15 The plant, which is native to Europe and western Asia, is now cultivated in North and South America, Africa, Asia and Australia and is invasive in many areas. 3 Cultivation is predominately in the northwestern United States and Germany. 2 Within Humulus lupulus, there are five taxonomic subspecies based on morphological properties and geographical location: Humulus lupulus var. lupulus, Humulus lupulus var. cordifolius, Humulus lupulus var. neomexicanus, Humulus lupulus var. pubescens, and Humulus lupulus var. lupuloides. 16 Over 100 cultivars have been named. 17 It is not known whether a single or multiple varieties are used in cosmetics.
While the Humulus lupulus (hops) plant is typically dioecious (i.e., the male and female flowers usually develop on separate plants), occasional fertile monoecious individual plants have been reported. 18 When grown for beer, viable seeds are undesirable; therefore, only female plants are grown in hops fields to prevent pollination. Female plants are propagated vegetatively, and male plants are culled if plants are grown from seeds. Under natural conditions, the flowers are wind pollinated and the female inflorescence develops to form a strobile (or cone). Only the strobiles of the female plants are able to develop the lupulin glands that secrete a fine yellow resinous powder. 2 These glands secrete predominantly bitter acids and hop oil, the constituents of which include phytoestrogens such as 8-prenylnaringenin (8-PN) and other prenylflavonoids.19,20
Humulus lupulus (hops) is a climbing perennial bine, which means that it grows in a helix around a support and uses downward-facing bristles/hooked hairs for grip instead of tendrils or suckers as would a vine.3,6,21 Generally, the bines are trained to 25 ft (7.6 m) or higher on a trellis. Lateral arms develop at the nodes, producing flowers at their terminal buds. The green to yellowish-brown leaves have three or five lobes, depending on the variety, and are hairy on both sides; the margins of the cordate (heart-shaped) leaves are serrated and the petioles are slightly fleshy with stout hooked hairs. Humulus lupulus (hops) is a perennial plant that regrows each spring from the rhizomes of an underground rootstock in commercial hops production.
Physical and Chemical Properties
Chemical and Physical Properties of Humulus Lupulus (Hops) Extract.

aA CO2 extract of dried cones manufactured for use as a food ingredient.
Once harvested, Humulus lupulus (hops) strobiles deteriorate upon aging and exposure to the atmosphere. 3 The stability of stored strobiles and ethanol extracts, as measured by humulones, lupulones, and xanthohumol content, is optimal in 70% ethanol. 22
Green Humulus lupulus (hops) strobiles have a variety of odors including: citrus, tropical fruit, stone fruit, pine, cedar, floral, spicy, herbal, earthy, tobacco, onion/garlic and grassy. 23
Preparation/Extraction
Humulus lupulus (hops)
In general, when the strobiles are harvested from Humulus lupulus (hops) bines for beer production, the strobiles are immediately dried by forced hot air, and are often pressed into dense cylindrical pellets, 5 to 8 mm in diameter and up to 25 mm long. 17 The pelletization reduces the overall surface area and therefore reduces the rate of chemical oxidation/degradation, and provides a more compact product for shipping. Not all suppliers dry the strobiles; the strobiles may be harvested fresh as whole, wet cones at the farm, and shipped for immediate use within 36 h of harvest. 23
Sometimes, hops are treated with sulfur dioxide to improve the color and prevent change of active constituents. 3
Methylene chloride is the most common solvent used for the extraction of Humulus lupulus (hops) for beer brewing; hexane and methanol are also employed. 24 Typically, at least 95% of the available α-acids (source of the bitter flavorings) can be extracted from fresh Humulus lupulus (hops) strobiles. Methanol is the most efficient solvent for the extraction of α-acids (approximately 25%), followed by methylene chloride (approximately 20%) and hexane (approximately 18%).
Supercritical fluid extraction (SFE) with carbon dioxide is also used to collect the extract for beer brewing.25-28 Another modern method is pressurized fluid extraction (PFE), which is employed for extracting different polyphenols.29-33 PFE has been developed to extract bitter acids.
FDA Regulations on the Method of Manufacture and Residual Solvents in “Modified Hop Extract,” as a Food Additive Used or Intended for Use as a Flavoring Agent in the Brewing of Beer [21CFR172.560].

In general, oils are extracted from Humulus lupulus (hops) by steam distillation at 100°C. 34
Humulus Lupulus (Hops) Extract
One manufacturer reported that the method of manufacture of Humulus Lupulus (Hops) Extract for use in cosmetics begins with extraction with water and propylene glycol. 35 The extract solution is then pressed, clarified, and decontaminated. Further details were not provided.
Another manufacturer reports that Humulus Lupulus (Hops) Extract may be extracted with either a 50% volume ethanol solution or a 50% volume butylene glycol solution. 36 After the dried raw material (the flower head) is extracted with the solution, the extract is filtered and concentrated. After sedimentation, the filtrate is “adjusted” and packaged.
To manufacture a product mixture containing Humulus Lupulus (Hops) Extract, dried whole strobiles of Humulus lupulus (hops) are dispersed and solubilized with stirring in caprylic/capric triglyceride. 37 The solution is then filtered to obtain the desired mixture.
Composition
Humulus lupulus (hops)
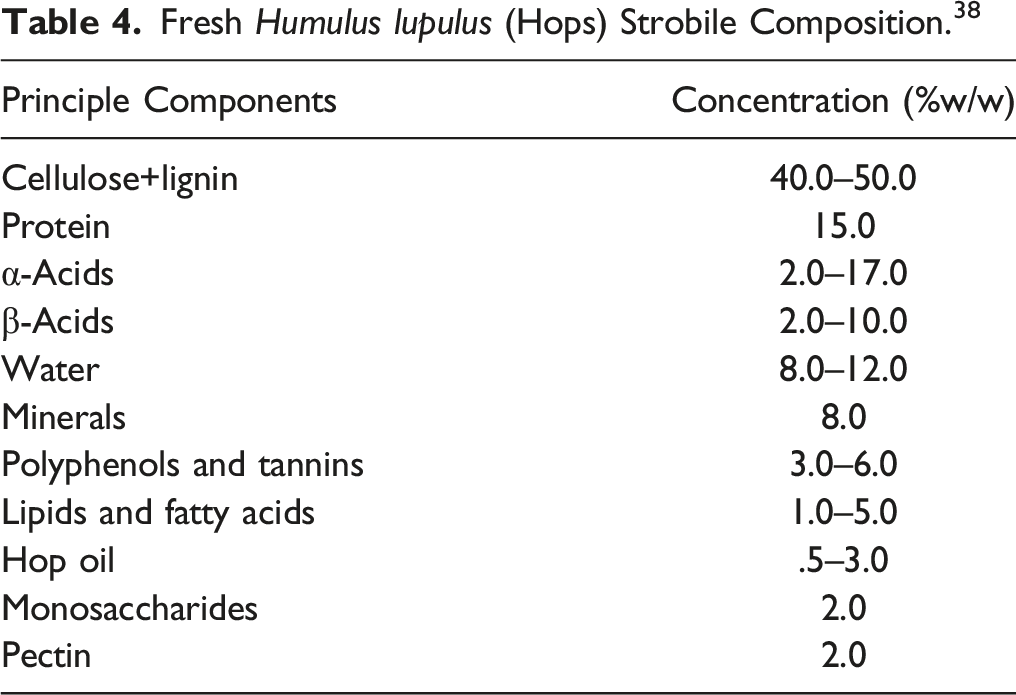
Fresh Humulus lupulus (Hops) Strobile Composition. 38
Dried Humulus lupulus (Hops) Strobile Composition. 2

Composition of Polyphenols and Their Concentrations in Humulus lupulus (Hops). 2
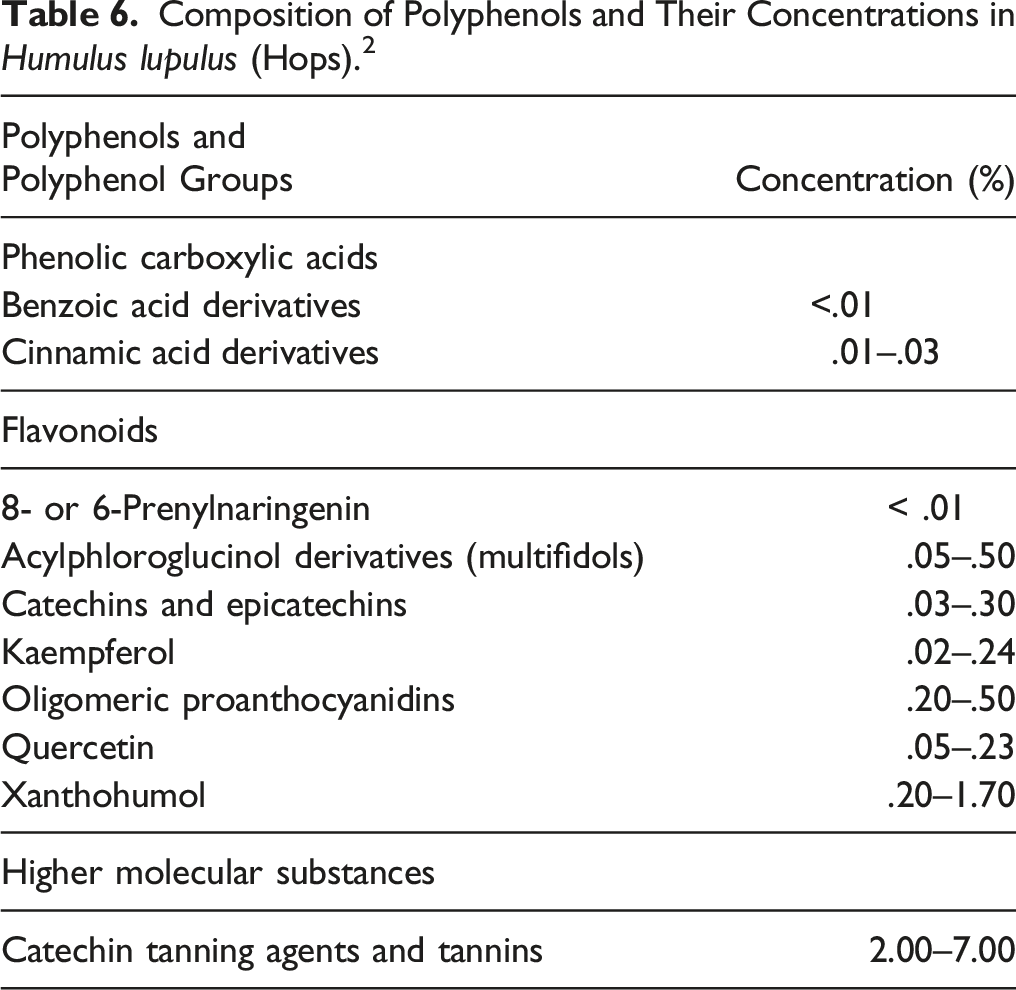
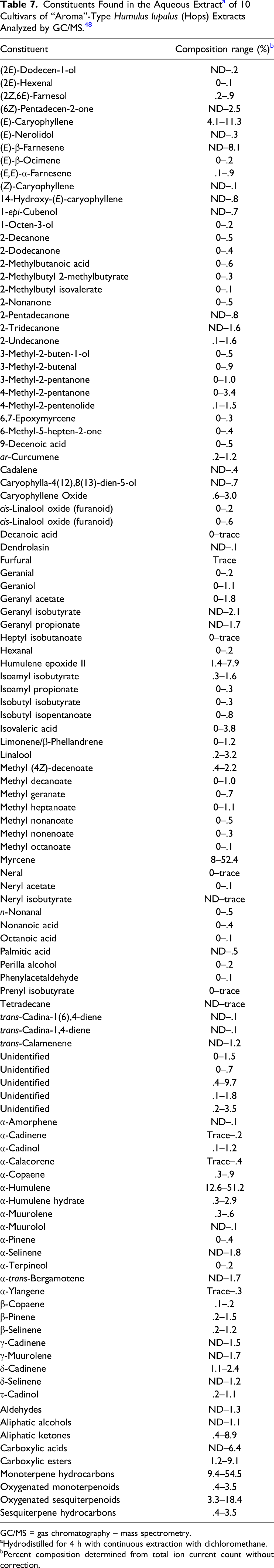
Flavonoids are composed of different chemical classes such as flavones, isoflavones, flavonols, flavanols, flavanones, and chalcones. These compounds differ in the level of oxidation of the flavane nucleus and in the number and position of hydroxyl, methyl, and methoxyl substituents. 41 Flavonoids, which make up .5% to 1.5% of the dried strobile, include quercetin and kaempferol glycosides.42,43 Thirty prenylated, geranylated, oxidized and/or cyclized chalcones have been isolated from the secretions of the lupulin glands. 17 The prenylated, geranylated flavonoids constitute up to 1% of the dried strobile and 80% to 90% of total flavonoids.19,20,44-46 The chalcone xanthohumol is the most abundant prenylated flavonoid in fresh and properly preserved strobiles (approximately .01 to .5%); desmethylxanthohumol, dehydrocycloxanthohumol, and the flavanones isoxanthohumol, 8-PN (25 to 60 mg/kg) and 6-prenylnaringenin are also found in the strobiles.19,20,44-47 A majority of the known flavonoids from Humulus lupulus (hops) strobiles can be considered to be derivatives of the compound 2’,4,4’,6’-tetrahydroxy-3’-prenylchalcone (chalcone numbering), commonly known as desmethylxanthohumol.
GC/MS = gas chromatography – mass spectrometry.
aHydrodistilled for 4 h with continuous extraction with dichloromethane.
bPercent composition determined from total ion current count without correction.
Humulus lupulus (hops) oil
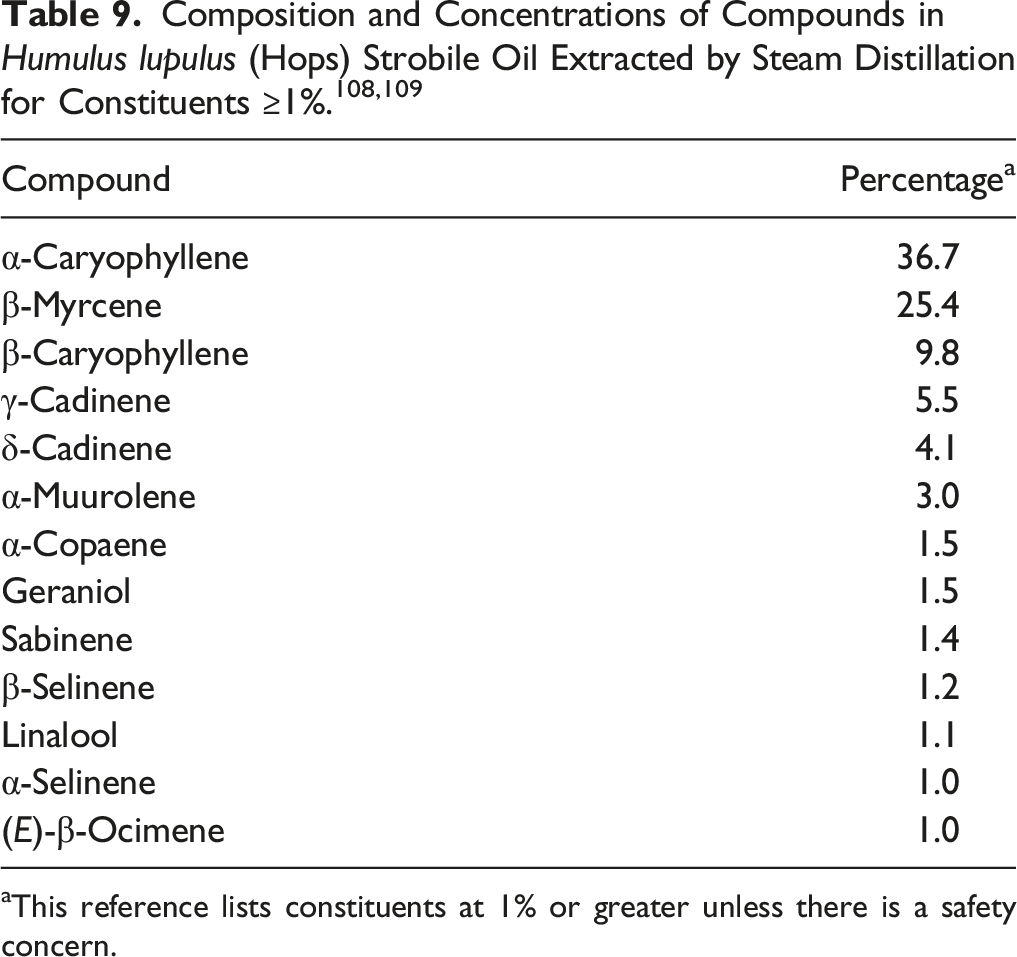
Compounds in Humulus Lupulus (Hops) Strobile Oil. 52

aThis reference lists constituents at 1% or greater unless there is a safety concern.
Humulus lupulus (hops) extract
The ethanol extract of Humulus Lupulus (Hops) Extract is reported to contain flavonoid and tannin. 36 The butylene glycol extract of Humulus Lupulus (Hops) Extract is reported to contain tannin and amino acid.
Constituents of Concern
Humulus Lupulus (hops)
Humulus lupulus (hops) plants are reported to contain linalool, quercetin, β-myrcene, 8-PN (or hopein), and other prenylated flavonoids (Figure 1).42,43,52,54-56 Humulus lupulus (hops) oil is reported to contain sesquiterpene lactones.17,50-52 The potential adverse effects of exposures to these constituents are summarized in Table 10. Constituents of concern of Humulus lupulus (hops) plants. Constituents of Concern Found in Humulus lupulus (Hops).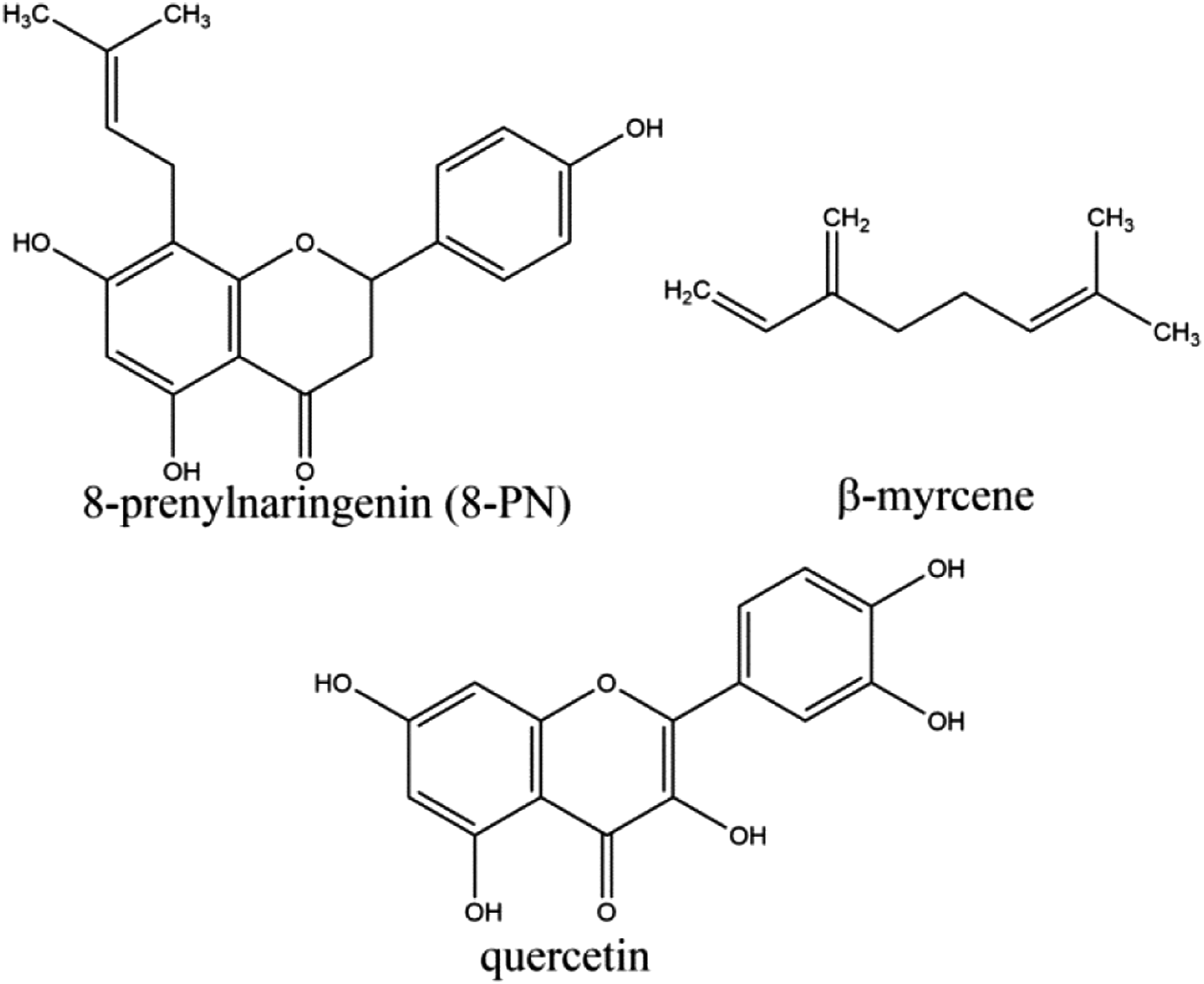

Constituents of Humulus lupulus (Hops) that Have IFRA Standards. 118

IFRA – International Fragrance Association.
aUse categories are based on types of skin contact (e.g., skin, lips), length of contact (e.g., leave-on, rinse-off), or type of use (e.g., mouthwash).
Humulus Lupulus (Hops) Extract
According to one supplier, the butylene glycol extract of Humulus Lupulus (Hops) Extract does not contain β-myrcene (detection limit .01 mg/100 g). 36
A product mixture that contains approximately .18% Humulus Lupulus (Hops) Extract (extracted with caprylic/capric triglyceride) reports a theoretical content of β-myrcene of 22 ppm based on the content of the starting materials. 37 However, it is noted that the method of manufacture does not favor β-myrcene retention. The HPLC profile of this mixture shows a peak at xanthohumol and no other prenylflavonoids.
Humulus Lupulus (Hops) Oil
Humulus Lupulus (Hops) Oil is reported to contain geraniol (.2%), limonene (1%), and linalool (.6%). 57
Impurities
Humulus lupulus (hops)
Multiple fungi and bacteria may be found on Humulus lupulus (hops) plants. 3 Analysis of the dust in air samples collected during harvest showed that total concentrations of microorganisms ranged between 2.08 and 129.58 × 103 cfu/m3; the concentrations of endotoxin ranged between 26 and 6250 ng/m3. 58 In samples of the settled dust after harvest, the concentrations of total microorganisms ranged from .25 × 106 to 2.87 × 108 cfu/g; the concentrations of endotoxin ranged between 312.5 and 6250 μg/g (median 6250 μg/g).
FDA regulations restrict the amounts of residuals from solvents in the manufacture of “modified hop extract” as a food additive (Table 3). [21CFR172.560] These restrictions include: boron, 310 ppm; benzene, 1.0 ppm; light petroleum spirits, 1.0 ppm; methyl alcohol, 250 ppm; hexane, 125 ppm; ethylene dichloride, 150 ppm; methylene chloride, 250 ppm; trichloroethylene, 250 ppm; and isopropyl alcohol, 250 ppm.
Analysis of dried food grade Humulus lupulus (hops) strobiles (possible source material for Humulus Lupulus (Hops) Extract) produced for beer production had the following results: lead <1.0 ppm, arsenic <.5 ppm, cadmium <.03 ppm, and total heavy metals <10 ppm. 23 Heavy metals, pesticides, herbicides, fungicides, nitrates, and radioactivity are reported to be below tolerance levels. Another analyses of leaves and strobiles of Humulus lupulus (hops) plants had the following results: copper, 102.3 and 81.1 ppm; vanadium, .07 and .05 ppm; molybdenum, .07 and .12 ppm; iron, 49.3 and 54.2 ppm; tin, 1.4 and 1.2 ppm; lead, 3.1 and 2.3 ppm, and nickel, 7.9 and 5.5 ppm, respectively. 59
The levels of residual solvent present in commercial hop extracts used for brewing beer are reported to be <100 ppm. 60
Humulus Lupulus (Hops) Extract
Authors of an analysis of a product mixture that contained Humulus Lupulus (Hops) Extract (.6 to 1.2%) reported that heavy metals were certified to be present at ≤5 ppm, microbes at <100 cfu/mL, yeasts and molds at < 100 cfu/mL, and enterobacteria absent. 61
Use
Cosmetic
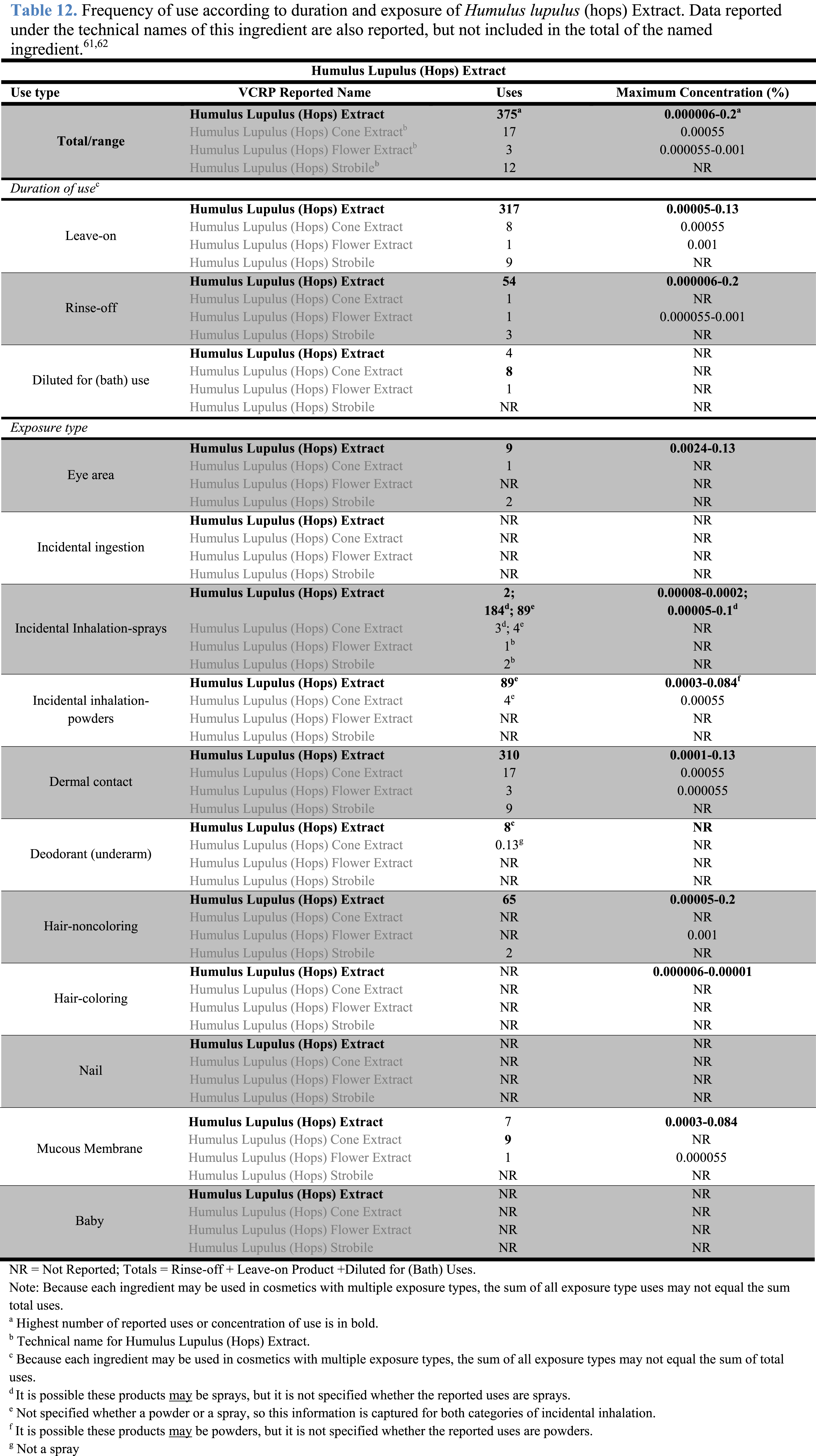
The safety of the cosmetic ingredients included in this assessment is evaluated based on data received from the US FDA and the cosmetic industry on the expected use of these ingredients in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in FDA’s Voluntary Cosmetic Registration Program (VCRP) database. Use concentration data are submitted by the cosmetic industry in response to surveys, conducted by the Personal Care Products Council (Council), of maximum reported use concentration by product category.
The Humulus lupulus (hops)-derived ingredients were reported to the 2017 VCRP and surveyed by the Council in 2015 and 2016 under the former INCI names, and that is how they are reported here.62,63
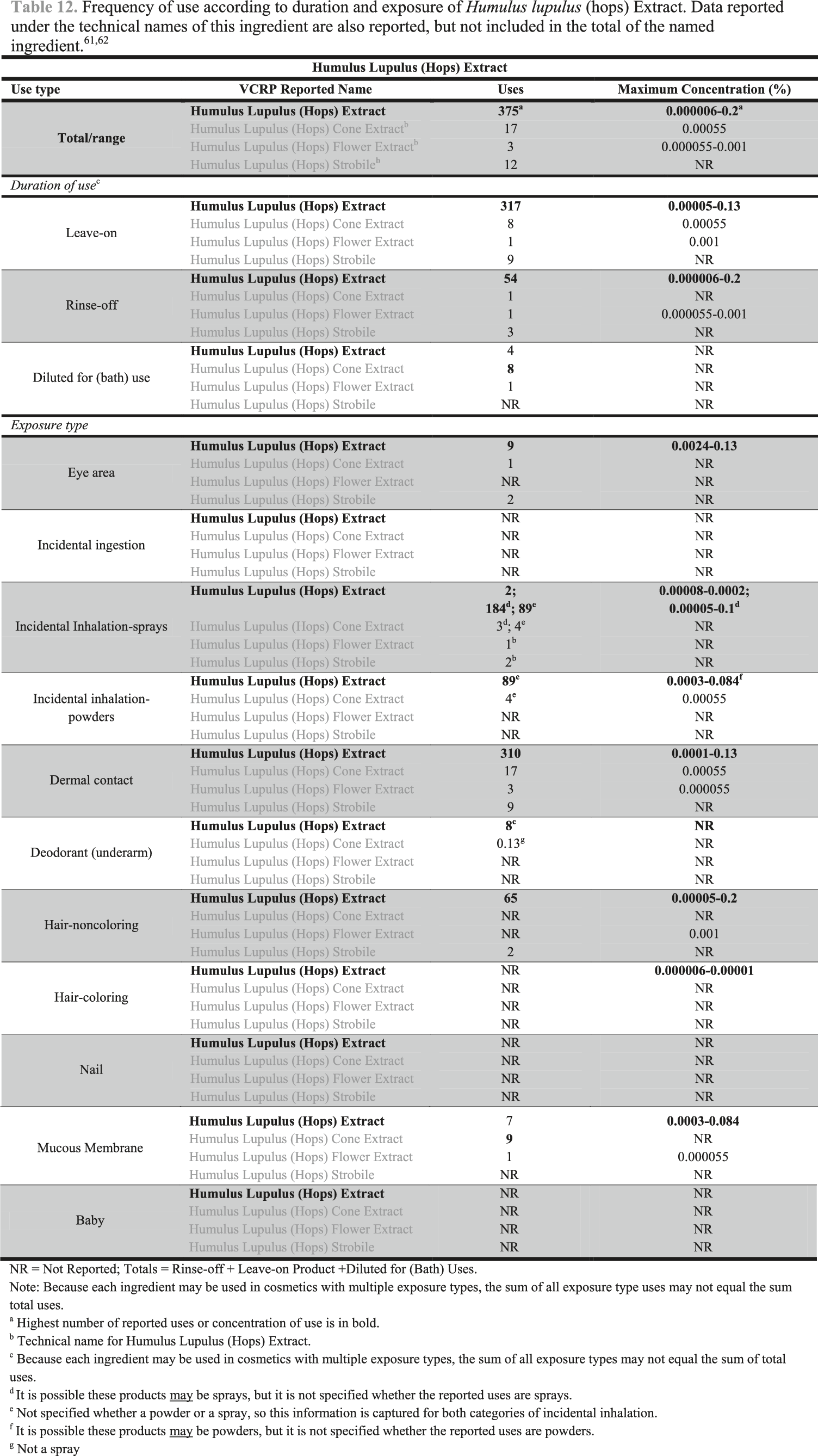
The results of the concentration of use survey conducted by the Council in 2015 (and updated in 2016) indicate that Humulus Lupulus (Hops) Extract is used at up to .2% in hair conditioners.63,64 The highest reported maximum concentration of use with dermal contact was reported to be .13% in eye lotion and in the category of other skin care preparations.
Humulus Lupulus (Hops) Cone Oil is not in use according to the VCRP and the industry survey results.
Humulus Lupulus (Hops) Extract is reported to be used in formulations that are used around the eyes at up to .13% and in formulations that come in contact with mucus membranes at up to .084% (e.g., bath soaps and detergents, bubble baths).
Humulus Lupulus (Hops) Extract is used in cosmetic sprays and could possibly be inhaled; for example, this ingredient is reported to be used at up to .0002% in hair sprays. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles <10 µm compared with pump sprays.65,66 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and thoracic regions of the respiratory tract and would not be respirable (i.e., they would not enter the lungs) to any appreciable amount.67,68 There is a reported use in face powders at up to .00055%. Conservative estimates of inhalation exposures to respirable particles during the use of loose-powder cosmetic products are 400-fold to 1000-fold less than protective regulatory and guidance limits for inert airborne respirable particles in the workplace.69-71
Neither of the Humulus lupulus (hops)-derived ingredients named in the report (under the new or revised names) are restricted from use in any way under the rules governing cosmetic products in the European Union. 72
Non-Cosmetic
Humulus lupulus (hops) strobiles are predominantly used to make beer.2,3 They were originally added to beer for their antimicrobial properties. Brewers then began using strobiles (and their extracts) to add bitterness, flavor, and aroma. 24
FDA determined that essential oils, oleoresins (solvent-free), and natural extractives (including distillates) of Humulus lupulus L. (hops) are GRAS for human consumption. [21CFR182.20] Modified Humulus lupulus (hops) extract may be safely used in beer in accordance with the following prescribed conditions: (a) the food additive is used or intended for use as a flavoring agent in the brewing of beer, and (b) the food additive is manufactured by one of the prescribed processes (Table 3). [21CFR172.560]
Most parts of the Humulus lupulus (hops) plant (shoots, leaves, flowers, seeds, rhizomes, and essential oils) are edible.5,6 The shoots are consumed as a delicacy and resemble asparagus.
In Europe, Humulus lupulus (hops) is administered as an herbal supplement in the form of powders, liquid extracts (ethanol extract drug ratio/dry extract ratio [DER] 1:1; sweet wine extract DER 1:10), tinctures (ethanol extract DER 1:5), and dry extracts (50% methanol extract DER 4 to 5:1) of the inflorescence of the plant.49,73 It is also administered as a tea. Humulus lupulus (hops) strobiles are used in European, Indian-Ayurvedic, and Native American traditional medicines for the relief of insomnia, excitability, and specifically for restlessness associated with nervous tension, headache and/or indigestion.
It has been shown that Humulus lupulus (hops) byproducts, after harvesting of the strobiles, can be used to absorb lead from contaminated waters. 74
Toxicokinetic Studies
Obtaining data on the toxicokinetics of Humulus lupulus (hops)-derived ingredients would not be practical because these ingredients are complex mixtures. Exposure to the components of these ingredients in cosmetics is expected to be lower than that from dietary exposure because these ingredients are incorporated into cosmetic products only at very low concentrations.
Toxicological Studies
Acute Toxicological Studies
Acute toxicity data on Humulus lupulus (hops)-derived ingredients were not found in the published literature and no unpublished data were submitted.
Short-Term Toxicity Studies
Oral
Wistar rats (n = 7/group) were fed a low-fat diet, a high-fat diet, or high-fat diet supplemented with 1% xanthohumol-rich Humulus lupulus (hops) extract for 41 days. 75 There were no mortalities or other adverse effects observed in any of these groups. The addition of the extract reduced the effects of the high-fat diet on weight gain from days 21 to 41 of the study. The weights of livers of rats fed the supplemented high-fat diets were similar to the controls, as were the plasma glucose levels, at the end of the test period.
Subchronic Toxicity Studies
Oral
In a study on the effects of Humulus lupulus (hops) extract on high-fat diets, male C57BL/6J mice (n = 10/group) were fed a normal diet, a high-fat diet, or a high-fat diet supplemented with 2 or 5% of various Humulus lupulus (hops) extracts for 20 weeks. 76 The high-fat diet was supplemented with one of the following: aqueous Humulus lupulus (hops) extract, ethyl acetate-soluble fraction of the aqueous Humulus lupulus (hops) extract, ethyl acetate-insoluble fraction of the aqueous Humulus lupulus (hops) extract, methanol-soluble fraction of the ethyl acetate-insoluble fraction of the aqueous Humulus lupulus (hops) extract, or methanol-insoluble fraction of the ethyl acetate-insoluble fraction of the aqueous Humulus lupulus (hops) extract. There were no mortalities or adverse effects reported for any group. The addition of any Humulus lupulus (hops) extract reduced the effects of the high-fat diet on weight gain. The weights of livers and mesenteric and epididymal adipose tissues of mice fed the supplemented high-fat diets were similar to that of the controls, as were plasma glucose levels, at the end of the test period; the extract had no additional effect on the effects of the high-fat diets.
Developmental and Reproductive Toxicity (Dart) Studies
Developmental and reproductive toxicity data on Humulus lupulus (hops)-derived ingredients were not found in the published literature and no unpublished data were submitted.
Genotoxicity Studies
In Vitro
Humulus Lupulus (Hops) Extract
An aqueous Humulus Lupulus (Hops) Extract (10 to 400 mg/µL in ethanol) was weakly mutagenic (a 2- to 4-fold increase in induced revertants compared with controls) in Salmonella typhimurium (strains TA98 and TA100), with or without metabolic activation. 77 No further details were provided.
A Humulus Lupulus (Hops) Extract (0, 1000, 2500, 5000, 7500, and 10,000 µg/plate; extract solvent not specified; water control) was not mutagenic in S. typhimurium (strains TA98 and TA100) or Escherichia coli (strain pKM101), with or without metabolic activation. 78 The positive and negative controls yielded the expected results.
An Ames test was performed on a product mixture containing 5% Humulus Lupulus (Hops) Extract (extracted in water/glycerin 50/50) at 10% in deionized water (effective concentration of .5% hops) with and without metabolic activation using S. typhimurium (strains TA97a, TA98, TA100, TA201, and TA1535). 79 The test substance was not mutagenic in this assay with or without metabolic activation.
Carcinogenicity Studies
Carcinogenicity data on Humulus lupulus (hops)-derived ingredients were not found in the published literature and no unpublished data were submitted.
Other Relevant Studies
Estrogenic Activity
Historically, there is circumstantial evidence of potential estrogenic activity connected to Humulus lupulus (hops) exposure, including menstrual disturbances reported to be common among female Humulus lupulus (hops) harvesters.80,81 In an investigation of the reported observation that women who normally live “a distance” from hop gardens regularly begin to menstruate two days after arriving to pick hops, it was reported that hops contain “the equivalent of 20 to 300 μg estradiol/g”. 49 Humulus lupulus (hops) extracts have been reported to reduce hot flashes in menopausal women and, in Germany, hops baths containing approximately 30% Humulus lupulus (hops) extracts (which have been discontinued) were used to treat gynecological disorders.49,82 However, early studies to confirm this activity experimentally were inconclusive or contradictory because of inadequate sensitivity of the methods used.80,83
More recently, 8-PN has been shown to be the source of the estrogenic activity of Humulus lupulus (hops). 8-PN mimics the action of 17β-estradiol, albeit with less (10- to 20,000-fold) potency.84-88 It is a potent ligand for the α-estrogen receptor (ER) with an IC50 value in the nanomolar range; it stimulates the production of alkaline phosphatase in Ishikawa cells, and stimulates the growth of estrogen-dependent MCF7 breast cancer cells.44,89 It was reported that 8-PN has a greater affinity for the ERα (where it is 70-fold less potent than estradiol) than for ERβ (reported to be 20,000-fold less potent than estradiol). 90
In a screening for drugs derived from plants for estrogenic activity, an ethanolic Humulus lupulus (hops) extract (50%; .2 g/mL) exhibited binding to ERs in intact, estrogen-dependent [ER(+)], human breast cancer MCF-7 cells with a potency equivalent to .5 μg of estradiol per 2 g of dried Humulus lupulus (hops) strobile (for comparison, the potencies of 2 g of thyme or red clover were equivalent to .5 or 3 μg of estradiol, respectively). 91 Humulus lupulus (hops) extract also showed significant ability to stimulate cell proliferation in ER (+) T47D, but not in ER(−) MDA 468, breast cancer cells. 91 In contrast, in a different series of experiments, a similarly prepared Humulus lupulus (hops) extract at concentrations of .01–1.0% v/v was found to inhibit serum-stimulated growth of ER(+)T47D breast cancer cells. 92 Ovarian cells isolated from immature female rats, which 48 h previously had been injected (primed) with pregnant mare’s serum gonadotropin, were incubated with follicle-stimulating hormone to induce estradiol secretion. The addition of purified water-soluble fractions from defatted Humulus lupulus (hops) extract to the culture medium reduced the estrogen E2 released from the ovarian cells with a probably related decrease in cyclic adenosine monophosphate (cAMP) release. 49
A Humulus lupulus (hops) extract activated the estrogen response element (ERE) in Ishikawa cells and induced ERE-luciferase expression in MCF-7 cells. In the MCF-7 cell line, progesterone receptor (PR) mRNA was significantly upregulated by Humulus lupulus (hops) extract with an EC50 of 1.1 μg/mL. 93 Humulus lupulus (hops) consisted of a chloroform partition of a methanolic extract from a previously SFE-CO2-extracted Nugget Humulus lupulus (hops) cultivar; the individual constituents included prenylated flavanones and isoflavonoids. The estrogenic activity proved to be considerably greater than that of established phytoestrogens such as coumestrol (present in red clover) and genistein and daidzein (present in soy).
Dermal Irritation and Sensitization Studies
Irritation
Human
Humulus Lupulus (Hops) Extract
In a 2-week cumulative irritation test (n = 26) of a formulation containing Humulus Lupulus (Hops) Extract (.125%), the test formulation did not demonstrate a significant irritation potential in human subjects. 94
A formulation containing Humulus Lupulus (Hops) Extract (.6 to 1.2%; 20 µL) was patch tested (n = 12) at 10% (final concentration .06 to .12%) using 8-mm aluminum cups covering 50 mm2 skin.95,96 The patches were administered to the upper back for 24 h. Controls were water and sodium lauryl sulfate (1%). The test sites were examined 30 min to 1 h and 24 h after patch removal. One subject had a reaction at the site of the negative control and was not included in the final analysis of the results. No reactions to the test material were observed in 9 subjects; two subjects showed very slight erythema. The controls had the expected results in the rest of the subjects. The irritation index was .04 (out of 5) and the test article was rated a non-irritant.
A patch test (n = 12) was conducted on a product mixture that contained Humulus Lupulus (Hops) Extract (approximately .18% in caprylic/capric triglyceride). 37 The test substance was applied to the skin on the back under a patch for 48 h. No adverse reactions were observed. It was concluded that the test substance had good cutaneous compatibility.
Sensitization
Humulus lupulus (hops) extract
In a human maximization test (n = 26) of a product containing Humulus Lupulus (Hops) Extract (.125%), the test product did not demonstrate contact sensitization potential. 94 No further information was provided.
A human repeated insult patch test (HRIPT; n = 52) of Humulus Lupulus (Hops) Extract (10%; extracted with butylene glycol) had negative results. 36 No further information was provided.
An HRIPT (n = 102) was conducted of a product mixture that contained Humulus Lupulus (Hops) Extract (approximately .18% in caprylic/capric triglyceride). 37 The test substance was applied to the same site three times per week for 9 applications. The challenge was applied two weeks after the last induction application. All patches were in place for 48 h. Patch sites were examined for reactions when patches were removed and at 72 and 96 h. No reactions were observed.
An HRIPT (n = 102) was conducted of a product mixture that contained Humulus Lupulus (Hops) Extract (approximately 5% in glycerin/water). 97 The induction and challenge phases were conducted at 10% (.5% Humulus Lupulus (Hops) Extract). Induction and challenge patches were in place for 24 h. The test sites were observed before the application of the next patch and at 24 and 72 h after the application of the challenge patch. There were no signs of irritation or sensitization at any time during the test period; there was no indication of a potential to cause dermal irritation or contact sensitization.
Ocular Irritation Studies
In Vitro
In vitro Ocular Assays of Humulus Lupulus (Hops) Extract.
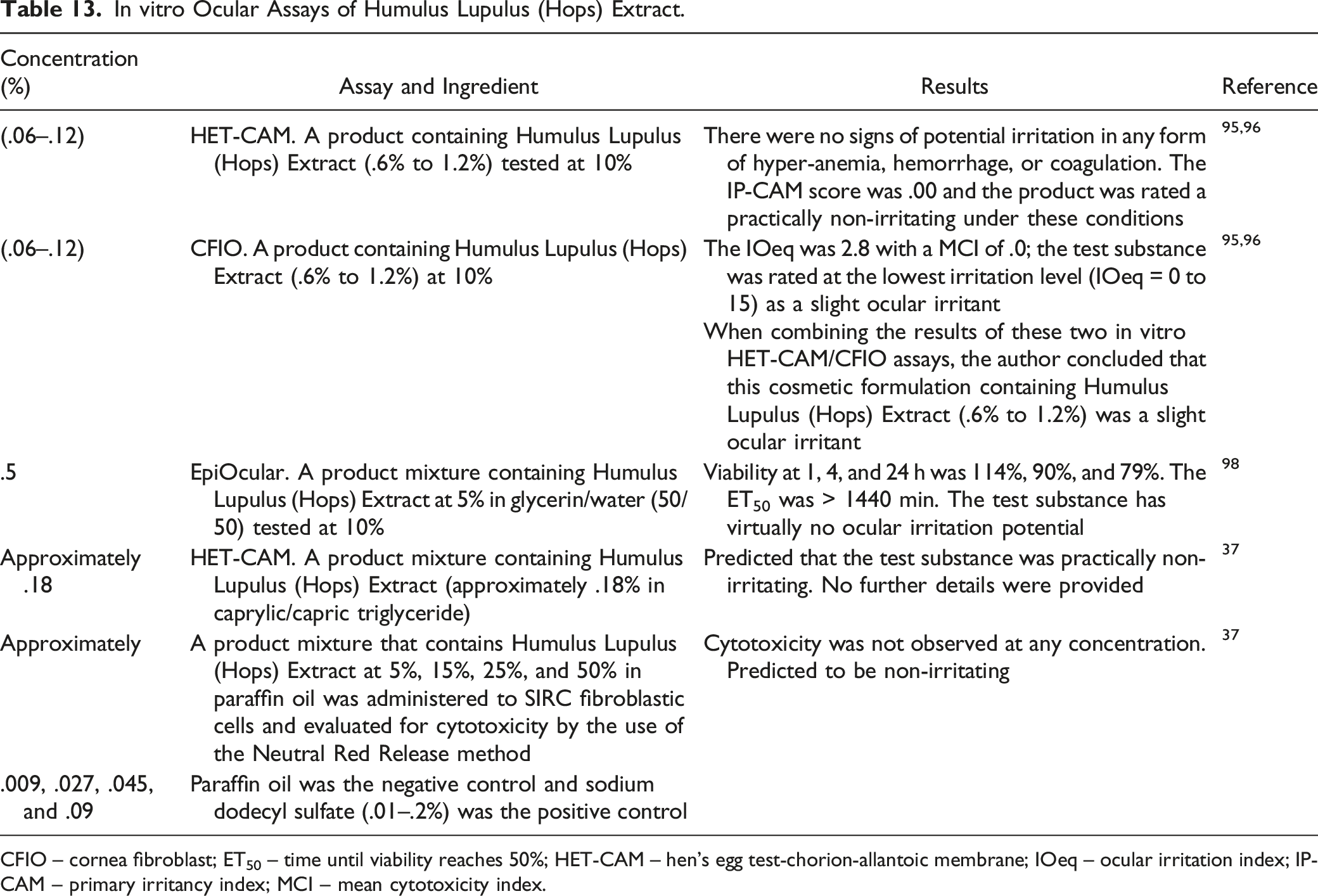
CFIO – cornea fibroblast; ET50 – time until viability reaches 50%; HET-CAM – hen’s egg test-chorion-allantoic membrane; IOeq – ocular irritation index; IP-CAM – primary irritancy index; MCI – mean cytotoxicity index.
Human
In a 4-week use study (n = 48) of an eye cream that contained Humulus Lupulus (Hops) Extract (.125%), the test material did not demonstrate a potential to cause eye irritation. 94
Clinical Studies
Occupational Exposure
Dermal
The causative agents of Humulus lupulus (hops) plant-induced contact skin reactions have not been established.99,100 Both irritant and allergic effects have been described. In Humulus lupulus (hops) harvesters, dermatitis has been attributed to mechanical abrasion by the rough hairs on the climbing stem. It has also been suggested that lupulin, the yellow powdery secretion of the glandular hairs on the scales of the strobiles, may be responsible for the irritation.
Farmers (n = 73) who cultivated Humulus lupulus (hops) plants and other crops from 18 randomly selected farms filled out a questionnaire on their skin diseases and were administered skin prick tests (SPT) for allergens of Humulus lupulus (hops) as well as grain dust, straw dust, hay dust, storage mites, and antigens of microorganisms typical of farm environments. 101 Only the results of the Humulus lupulus (hops) are reported here. Fresh strobiles and leaves were cut into small pieces, and extracted with glycerol and saline at 1:2 (w/w) for 48 h at 4°C. The extracts were centrifuged and clear supernatants were used in the testing. The subjects consisted of 42 males and 31 females, aged 16 to 84 (median 46) years, with duration of employment resulting in exposure ranging from 2 to 73 (median 31) years.
The questionnaire showed that Humulus lupulus (hops) was reported to cause the greatest number of skin problems; 14 farmers (19.2%) reported work-related skin symptoms, 2 (11%) of which were caused by Humulus lupulus (hops). There were no reported skin problems associated with working with Humulus lupulus (hops) by 65 subjects. The reported skin symptoms of the subjects with skin problems were mostly mild: four reported rashes on uncovered skin (the description of which was sufficient to diagnose airborne dermatitis), two subjects reported hand dermatitis, and two reported pruritus without visible skin changes. One case of airborne dermatitis to Humulus lupulus (hops) was severe enough to be classified as debilitating.
Positive skin reactions to four Humulus lupulus (hops) allergen preparations, i.e., cone extract in glycerol, cone extract in saline, leaf extract in glycerol, and leaf extract in saline, were found in one, two, three, and four of 65 subjects, respectively. In all, six subjects (8.2%) reacted to at least one extract. Among the subjects reporting skin problems related to Humulus lupulus (hops), SPTs gave positive results in two subjects, and the tests were negative in six subjects. The tests were also positive in four persons who did not report any Humulus lupulus (hops)-related skin problems. The predictive values for SPT of the extracts (skin reaction to at least one of the preparations) were: positive predictive value (PPV) = .33 and negative predictive value (NPV) = .91. 101
Inhalation
Washington State Workers’ Compensation claims filed by Humulus lupulus (hops) workers for respiratory disease between 1995 and 2011 were systematically identified and reviewed in a study of occupational respiratory disease in Humulus lupulus (hops) workers. 102 Incidences of respiratory disease in Humulus lupulus (hops) workers were compared with rates in field vegetable crop farm workers. A total of 57 cases of respiratory disease associated with Humulus lupulus (hops) dust inhalation were reported. The attending health care practitioner diagnosed 61% of these cases as having work-related asthma. Chronic obstructive pulmonary disease was diagnosed in 7% of these cases; the remaining cases were diagnosed as allergic respiratory disorders (e.g., allergic rhinitis [18%] or asthma-associated symptoms [e.g., dyspnea; 14%]). All cases were associated with Humulus lupulus (hops) harvesting, secondary hops processing, or indirect exposure. The incidence of respiratory disease in Humulus lupulus (hops) workers was 15 cases per 10,000 full-time workers, which was 30 times greater than the incidence for field vegetable crop workers. A strong temporal association between Humulus lupulus (hops) dust exposure and respiratory symptoms and a clear association between an increase in Humulus lupulus (hops) dust concentrations and the clinical onset of symptoms were apparent in 3 cases. The authors concluded that occupational exposure to Humulus lupulus (hops) dust is associated with respiratory disease; respiratory disease rates were higher in Humulus lupulus (hops) workers than in a comparison group of agricultural workers.
In a study of occupational exposure of brewery workers to organic dusts such as Humulus lupulus (hops), barley, and brewery yeast, the potential to affect respiratory function and immunological status was examined. 103 Male subjects (n = 97) employed in a brewery plant had a mean age of 40 years, and the mean duration of employment was 16 years. The control group consisted of unexposed workers (n = 76). Respiratory symptoms were recorded. Lung function was measured by recording maximum expiratory flow-volume (MEFV) curves. Immunological testing was performed on all brewery workers and 37 of the control volunteers using SPTs with Humulus lupulus (hops), barley, and yeast antigens as well as other non-occupational allergens, and by determining total serum IgE levels. There was a higher prevalence of most of the chronic respiratory symptoms in brewery workers compared to controls. Occupational asthma was recorded in only 2 (2.1%) of the brewery workers; smoking was reported to be the major factor (that was examined in this study) responsible for the high prevalence of chronic respiratory symptoms in workers, not exposure to dust of the brewing ingredients, including Humulus lupulus (hops). A large number of brewery workers complained of acute symptoms that developed during the work shift. Lung function test scores were decreased compared to predicted levels. Multivariate analysis of these respiratory function parameters suggested the importance of workplace exposure in explaining lung function abnormalities. There was a greater instance of positive SPTs in brewery workers for Humulus lupulus (hops) than in controls (15% vs 3%). There were increased serum levels of total IgE in 34 out of 97 (45.1%) brewery workers compared to controls, 1 out of 76 (2.7%). However, workers with positive SPTs had a prevalence of chronic respiratory symptoms and lung function changes similar to those of workers with negative SPTs. The authors concluded that the data suggest that both smoking and dust exposure in the brewery industry may be responsible for the development of respiratory impairment and immunological reactions.
Case Reports
Summary
This is a safety assessment of Humulus Lupulus (Hops) Extract and Humulus Lupulus (Hops) Oil as used in cosmetics. Both of these ingredients are derived from the strobile of the Humulus lupulus plant, commonly called hops. The reported functions of Humulus Lupulus (Hops) Extract in cosmetics include antimicrobial agent, hair conditioning agent, and skin-conditioning agent – miscellaneous; Humulus Lupulus (Hops) Oil is reported to function as a fragrance ingredient.
Previously, the wINCI listed four other names for Humulus Lupulus (Hops) Extract. Data submitted under those deleted names have been assigned to Humulus Lupulus (Hops) Extract and the deleted names are now technical names for Humulus Lupulus (Hops) Extract.
Humulus lupulus (hops) strobile is an ingredient in food (most commonly in beer) and most of the other parts of this plant (shoots, leaves, flowers, seeds, rhizomes, and essential oils) are edible. The FDA determined that essential oils, oleoresins (solvent-free), and natural extractives (including distillates) of Humulus lupulus L. (hops) are GRAS for human consumption.
Humulus lupulus (hops) plants are reported to contain several constituents of concern, including 8-PN, β-myrcene, and quercetin; these constituents could result in estrogenic activity, dermal irritation, and genotoxicity, respectively, if concentrations were high enough. Geraniol, limonene, linalool, and sesquiterpene lactones are potential dermal sensitizers. Humulus Lupulus (Hops) Oil is reported to contain sesquiterpene lactones.
The butylene glycol extract of Humulus Lupulus (Hops) Extract is reported to contain no detectable β-myrcene. A product mixture that contains approximately .18% Humulus Lupulus (Hops) Extract is reported to have a theoretical content of β-myrcene of .0022% based on the content of the starting materials; it is noted that the method of manufacture does not favor β-myrcene retention.
The Humulus lupulus (hops)-derived ingredients were reported to the VCRP database and surveyed by the Council in 2015(and updated in 2016) under the revised INCI names, and that is how they are reported herein.
According to VCRP data received in 2017, Humulus Lupulus (Hops) Extract was reported to be used in 375 formulations, including 317 leave-on formulations and 54 rinse-off formulations. The results of the concentration of use survey conducted by the Council in 2015 (and updated in 2016) indicate that the highest reported maximum concentration of use of Humulus Lupulus (Hops) Extract is up to .2% in hair conditioners.
Humulus Lupulus (Hops) Oil is not in use according to the VCRP and the industry survey.
Rats fed a low-fat diet, a high-fat diet, or high-fat diet supplemented with 1% xanthohumol-rich extract of Humulus lupulus (hops) extracts for 41 days and male mice fed a normal diet, a high-fat diet, or high-fat diets supplemented with 2% or 5% of various Humulus lupulus (hops) extracts for 20 weeks had no mortalities or adverse effects reported for any group. The additions of any of the Humulus lupulus (hops) extracts reduced the effects of the high-fat diet on weight gain. The weights of livers and mesenteric and epididymal adipose tissues of mice fed the supplemented high-fat diets were similar to those of the controls, as were the plasma glucose levels at the end of the test period.
An aqueous Humulus Lupulus (Hops) Extract (10 to 400 mg/µL in ethanol) was weakly mutagenic (an increase in induced revertants 2 to 4 times the controls) to S. typhimurium with and without metabolic activation. In another assay, a Humulus Lupulus (Hops) Extract was not mutagenic to S. typhimurium and E. coli at up to 10,000 µg/plate, with or without metabolic activation. A product mixture containing 5% Humulus Lupulus (Hops) Extract at 10% (.05% hops in deionized water) was not mutagenic to S. typhimurium with or without metabolic activation.
Historically, there is circumstantial evidence of potential estrogenic activity connected to Humulus lupulus (hops) exposure, including menstrual disturbances reported to be common among female Humulus lupulus (hops) harvesters. 8-PN has been shown to be the source of the estrogenic activity of Humulus lupulus (hops) plants. It mimics the action of 17β-estradiol, albeit with a lesser (10- to 20,000-fold) potency.
In a 2-week cumulative irritation test of a product containing Humulus Lupulus (Hops) Extract (.125%), the test product did not demonstrate a significant irritation potential in human skin. In a human patch test of a product containing Humulus Lupulus (Hops) Extract at .06 to .12%, the irritation index was .04 (out of 5) and the test article was rated a non-irritant. No adverse reactions were observed in a patch test of a product mixture that contains Humulus Lupulus (Hops) Extract (approximately .18%).
The causative agents of Humulus lupulus (hops) plant-induced contact skin reactions have not been established. In Humulus lupulus (hops) harvesters, dermatitis has been attributed to mechanical abrasion by the rough hairs on the climbing stem. It has also been suggested that lupulin, the yellow powdery secretion of the glandular hairs on the scales of the strobiles, may be responsible for the irritation.
In a human maximization test of a product containing Humulus Lupulus (Hops) Extract (.125%), the test product did not demonstrate contact sensitization potential. An HRIPT of Humulus Lupulus (Hops) Extract (10%; extracted with butylene glycol) gave negative results. No reactions were observed in an HRIPT that was conducted of a product mixture that contains Humulus Lupulus (Hops) Extract (approximately .18%). There were no signs of irritation or sensitization in an HRIPT of a product mixture that contained Humulus Lupulus (Hops) Extract (approximately 5%).
In vitro assays showed that products and product mixtures containing Humulus Lupulus (Hops) Extract up to .5% were predicted to be either non-irritating or slight ocular irritants. Humulus Lupulus (Hops) Extract was predicted to be a slight ocular irritant in HET-CAM and cornea fibroblast assays and a non-irritant in an EpiOcular assay. Another Humulus Lupulus (Hops) Extract was predicted to be non-irritating in an HET-CAM assay and to SIRC fibroblastic cells.
In a 4-week use study of an eye cream that contained Humulus Lupulus (Hops) Extract (.125%), the test material did not demonstrate potential for eliciting ophthalmic irritation.
In a survey of farmers, exposure to Humulus lupulus (hops) was reported to cause the greatest number of skin problems; 14 of 73 (19.2%) of the farmers reported work-related skin symptoms, 11% of which were caused by Humulus lupulus (hops). There were one, two, three, and four positive SPTs to the following Humulus lupulus (hops) allergen preparations, respectively: cone extract in glycerol, cone extract in saline, leaf extract in glycerol, and leaf extract in saline.
In a study of occupational respiratory disease in Humulus lupulus (hops) workers, using Workers’ Compensation claims filed by Humulus lupulus (hops) workers for respiratory disease, the incidence rate of respiratory disease in Humulus lupulus (hops) workers was 15 cases per 10,000 full-time workers, which was 30 times greater than the incidence rate for field vegetable crop workers. The authors concluded that occupational exposure to Humulus lupulus (hops) dust is associated with respiratory disease; respiratory disease rates were higher in hop workers than in a comparison group of agricultural workers.
In a study of occupational exposure of brewery workers to organic dusts, including Humulus lupulus (hops), the potential to affect respiratory function and immunological status was examined. A large number of brewery workers complained of acute symptoms that developed during the work shift. Lung function tests were decreased compared to predicted levels. There was a greater instance of positive SPTs in brewery workers for Humulus lupulus (hops) than in controls (15% vs 3%). There were increased serum levels of total IgE in 34 of 97 (45.1%) brewery workers compared to controls, 1 of 76 (2.7%). However, workers with positive SPTs had a prevalence of chronic respiratory symptoms and lung function changes similar to those of workers with negative SPTs. The authors concluded that the data suggests that both smoking and dust exposure in the brewery industry may be responsible for the development of respiratory impairment and immunological reactions.
There were case studies of subjects becoming sensitized to Humulus lupulus (hops) plants or the constituent β-myrcene while working with the plant on farms, laboratories, and in breweries.
Discussion
The Panel examined the oral toxicity, genotoxicity, dermal and ocular irritation, and sensitization studies of Humulus lupulus (hops)-derived ingredients, as well as studies on occupational exposure. Essential oils, oleoresins, and natural extracts of Humulus lupulus (hops) are GRAS for human consumption.
The Panel noted the presence of β-myrcene at up to 25.4% in Humulus lupulus (hops) oil. This constituent is a potential irritant, and there is an NTP study showing increased incidences of kidney tumors in male rats and liver tumors in male mice after oral administration of 1.0 g/kg/day β-myrcene for 2 years. The increased incidence of kidney tumors in this study is likely attributable to a mechanism that is not relevant to humans, and the increased incidence of liver tumors is attributable to the high background incidence and susceptibility to the development of liver tumors that is characteristic of the mouse strain used in the study, and is also not predictive of carcinogenicity in humans. Further, the daily dose of β-myrcene administered orally to the rats and mice in the study were much greater than the highest possible exposure to β-myrcene that could occur from Humulus lupulus (hops)-derived ingredients in cosmetics. However, concerns about β-myrcene, and possibly other constituents, cannot be addressed fully by the Panel, because the available information is not sufficient to characterize adequately the compositions of Humulus lupulus (hops)-derived cosmetic ingredients. The Panel emphasized the importance, generally, of adequately characterizing the compositions of cosmetic ingredients derived from plants, as manufactured and supplied to formulators of cosmetic products.
There were possible estrogenic effects in persons who worked with Humulus lupulus (hops) in the field. The studies showed that the purported estrogenic effects were weak and the degree of the exposure to the workers is far greater than any exposure that could occur from the use of cosmetics that contain these ingredients at the reported concentrations of use.
Because final product formulations may contain multiple botanicals, each possibly containing similar constituents of concern, formulators are advised to be aware of these constituents and to avoid reaching levels that may be hazardous to consumers. For example, in Humulus lupulus (hops)-derived ingredients, the Panel’s concerns included the presence of 8-PN, β-myrcene, and quercetin in cosmetics, which could result in estrogenic effects, dermal irritation, and genotoxicity, respectively, as well as other constituents of concern. The Panel noted that IFRA standards to avoid adverse effect have been published for several Humulus lupulus (hops) constituents (Table 11). At the reported concentrations of use of these ingredients, the constituents that may cause these effects will be present at levels far below levels of concern, including for sensitization. However, when formulating products with multiple botanically-derived ingredients, manufacturers should avoid reaching levels of plant constituents that may cause sensitization or other adverse health effects.
There were instances of sensitization of persons who worked with Humulus lupulus (hops) in the field and in laboratories. The studies showed that the degree of the exposure of these persons is far greater than any exposure that could occur from the use of cosmetics that contain these ingredients at the reported concentrations of use. Additionally, the plant has hairs and bristles that could abrade the skin, thus increasing the chance of dermal penetration of constituents that could cause sensitization. Such abrasion would not occur with exposure to these cosmetic ingredients.
The Panel is reassured by the HRIPT of 10% Humulus Lupulus (Hops) Extract that sensitization should not be a problem under the reported conditions and concentrations of use in cosmetics because the highest reported concentration of use was .2% Humulus Lupulus (Hops) Extract in hair products and .13% in products that come in contact with the skin. Overall, sensitization and constituent profile data show that there should not be any issue with sensitization at the low concentrations of use of these ingredients. The Panel cautions that manufactures use cGMP to formulate products that are non-sensitizing.
There is a substantial data profile on the constituents of the Humulus Lupulus (Hops) Oil; many of these constituents, including potential sensitizers, are similar to those in the Humulus Lupulus (Hops) Extract. Therefore, the safety data on one ingredient informs the other. Concern, however, was expressed about alternative approaches to extraction that might not produce material with the same safety profile described in this safety assessment. There are multiple methods of extraction with multiple solvents (e.g., water/propylene glycol, water/ethanol, water/butylene glycol, and caprylic/capric triglyceride) presented for the ingredient Humulus Lupulus (Hops) Extract and steam distillation for Humulus Lupulus (Hops) Oil. The Panel’s safety conclusion is applicable only for Humulus Lupulus (Hops) Extract and Humulus Lupulus (Hops) Oil that are prepared in a manner that produces a similar chemical profile as that described in this report, especially for the constituents of concern. When prepared in a manner resulting in this chemical profile, the Panel’s conclusion is that these ingredients do not have significant estrogenic activity, genotoxicity, irritation, or sensitization potential. Ingredients not prepared in a manner that produces a similar chemical profile would be considered safe only if a similar safety test profile was demonstrated.
The Panel expressed concern about pesticide residues, heavy metals, and substances from plants of other species (weeds) that may be present in botanical ingredients. Also, multiple fungi and bacteria have been detected co-localized with Humulus lupulus (hops) plants. To address these concerns, the cosmetics industry should continue to use cGMPs to limit impurities.
There were no constituents of concern associated with phototoxicity and there were no indications of phototoxicity in the farmer workers exposed to high amounts of Humulus lupus (hops). Accordingly, phototoxicity assays were not deemed to be required.
Also, the Panel noted the limited scope of the in vitro genotoxicity assay in which an aqueous Humulus Lupulus (Hops) Extract increased revertants 2-4 times that of controls. The Panel concluded that these test results were not statistically significant and that there was no significant risk of genotoxicity.
The pulmonary disease associated with working with Humulus lupus (hops) plants was reported to be caused by constant inhalation exposure to the plant dust over extended periods (e.g., years). This exposure is far greater than any exposure would be associated with the use of cosmetic products. In this context, the Panel discussed the issue of incidental inhalation exposure from aerosol and pump hair sprays. The limited data on occupational inhalation suggests some potential for respiratory effects at large doses over extended periods of time. Otherwise, there were no inhalation toxicity data available. These ingredients are reportedly used at concentrations up to .0002% in cosmetic products that may be aerosolized and up to .00055% in loose powder products that may become airborne. The Panel noted that most droplets/particles would not be respirable to any appreciable amount. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. The Panel considered other data available to characterize the potential for Humulus lupulus (hops)-derived ingredients to cause toxicity, genotoxicity, irritation, and sensitization. They noted the lack of systemic toxicity, genotoxicity, irritation, and sensitization at relevant doses by Humulus lupulus (Hops)-derived ingredients. They also noted that parts of the plants are edible and extractives are GRAS as food additives. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at https://www.cir-safety.org/cir-findings.
Conclusion
The Expert Panel for Cosmetic Ingredient Safety concluded Humulus Lupulus (Hops) Extract and Humulus Lupulus (Hops) Oil are safe in cosmetics in the present practices of use and concentration described in this safety assessment when formulated to be non-sensitizing.
Footnotes
Author Contributions
The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Author's Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.