Abstract
Methyl ester sulphonate (MES) is an anionic surfactant that is suitable to be used as an active ingredient in household products. Four palm-based MES compounds with various carbon chains, namely C12, C14, C16 and C16/18 MES, were assayed by the in vitro bacterial reverse mutation (Ames) test in the Salmonella typhimurium strains TA98, TA100, TA1535, and TA1537 and the Escherichia coli strain WP2
Introduction
A compound is excellent and in high demand when it is safe to be used in product formulations besides its superior performance. Product safety is one of the most critical factors related to sustainability. In order to sustain a good product in the market, it is crucial to obtain the safety data of the product/compound. Therefore, a toxicity evaluation has to be performed. One of the toxicity evaluations is the genotoxic test.
Several methods can be used to evaluate the genotoxic potential of chemicals, namely the chromosome aberration test, the micronucleus test, the comet assay in human lymphocytes, and the bacterial reverse mutation assay. 1 The Ames test is the most widely used and accepted method for the detection of mutagenic activity of various chemicals and drugs, where it has a high predictive value for in vitro carcinogenicity. 2 It has been globally recognized by scientific communities and government agencies. It is a short-term biological assay used as one of a battery of tests to assess carcinogenic potential using mutagenicity in bacteria as an endpoint. The assay is conducted in the presence and absence of exogenous microsomal metabolic activation to understand the role of bioactivation to the mutagenic process. There are several methods used to perform the Ames test, namely the plate incorporation method,3–8 the preincubation method,9–13 the fluctuation method,14,15 and the suspension method. 16
Alpha-sulfonated methyl ester, more commonly known to the industry as methyl ester sulfonate (MES), can be derived from any vegetable oil-based material and offers interesting possibilities as a surfactant.17–19 Palm methyl ester is a potential feedstock as it is the most abundant raw material available in Malaysia. 20 The environmentally friendly properties of MES have been the driving force behind the industry’s interest in producing it commercially. In 2012, the global MES producers were from China, the USA, Japan, Indonesia and Malaysia. 21
The growing demand for bio-based raw materials is expected to boost the demand for MES, owing to its renewable nature and easy availability. In its report, Persistence Market Research provides decisive information on the global MES market and offers extensive information through structured market research. The global MES market was fixed at US$903.0 million by the end of 2018 and is expected to reach US$2,199.6 million by the end of 2026, with a significant compound annual growth rate (CAGR) of 11.8% over the forecast period. The global MES market is expected to present an incremental opportunity of US$1,296.6 million between 2018 and 2026. 22
MES is an anionic surfactant suitable to be used as an active ingredient in household products and has been promoted as an alternative to petroleum-based surfactants.23–25 MES can also be produced from other natural sources such as sesame oil, 26 soybean oil methyl ester, 27 castor oil, 28 jatropha oil,29–31 and other oleochemical feedstocks. Surfactants from the oleochemical origin are considered non-toxic and biodegradable, hence eco-friendly, besides being derived from renewable resources. 26 The utilization of oleochemical feedstock to produce surfactants is a technology that has been around for many years. The increasing consumption of renewable and sustainable bio-based chemicals in personal care, cosmetics, polymers, lubricants, pharmaceuticals, food and other industries has driven the global growth of oleochemical market. 20
Linear alkylbenzene sulphonate (LAS), a petroleum-based surfactant, is currently the most commonly used surfactant worldwide as the primary cleaning agent applied in many laundry detergents and cleaners at concentrations up to 30 percent. MES, which has good surface active properties and excellent detergency, 32 has a high potential to replace LAS in product formulations.
There are minimal toxicological data published on the palm-based MES. One of the published data was on the acute fish toxicity of palm-based MES. 33 A comparative ecotoxicity study of the MES to tilapia and Daphnia magna was also reported. 34 Results from the test indicated that Daphnia was more sensitive to toxic effects from MES than tilapia. The ecotoxicity increased as the hydrophobicity of the MES increased due to increase of alkyl chain length. In addition, the effects of palm-based MES on green algae using the algae growth inhibition test (OECD 201) were assessed. 35 In 2018, Yusof and co-workers investigated the eye irritation potential of palm-based MES using the bovine corneal opacity and permeability test method (BCOP), OECD Test Guideline 437, and reconstructed human corneal-like epitelium test method, OECD Test Guideline 492. 36
This study aimed to investigate the safety of palm-based MES in terms of its mutagenic potential. The Ames test was used to evaluate whether MES (Figure 1) can cause mutations in the DNA of the test organism. It uses amino acid-dependent bacterial strains of S. typhimurium and E. coli as test organisms to detect the point of mutation. The cells cannot grow to form colonies in the absence of an external histidine or tryptophan source. The production of histidine or tryptophan will be resumed if a reversion of the mutation occurs where colony growth is resumed. Mutagenic compounds cause an increase in the number of revertant colonies relative to the background level.
37
The chemical structure of palm-based MES.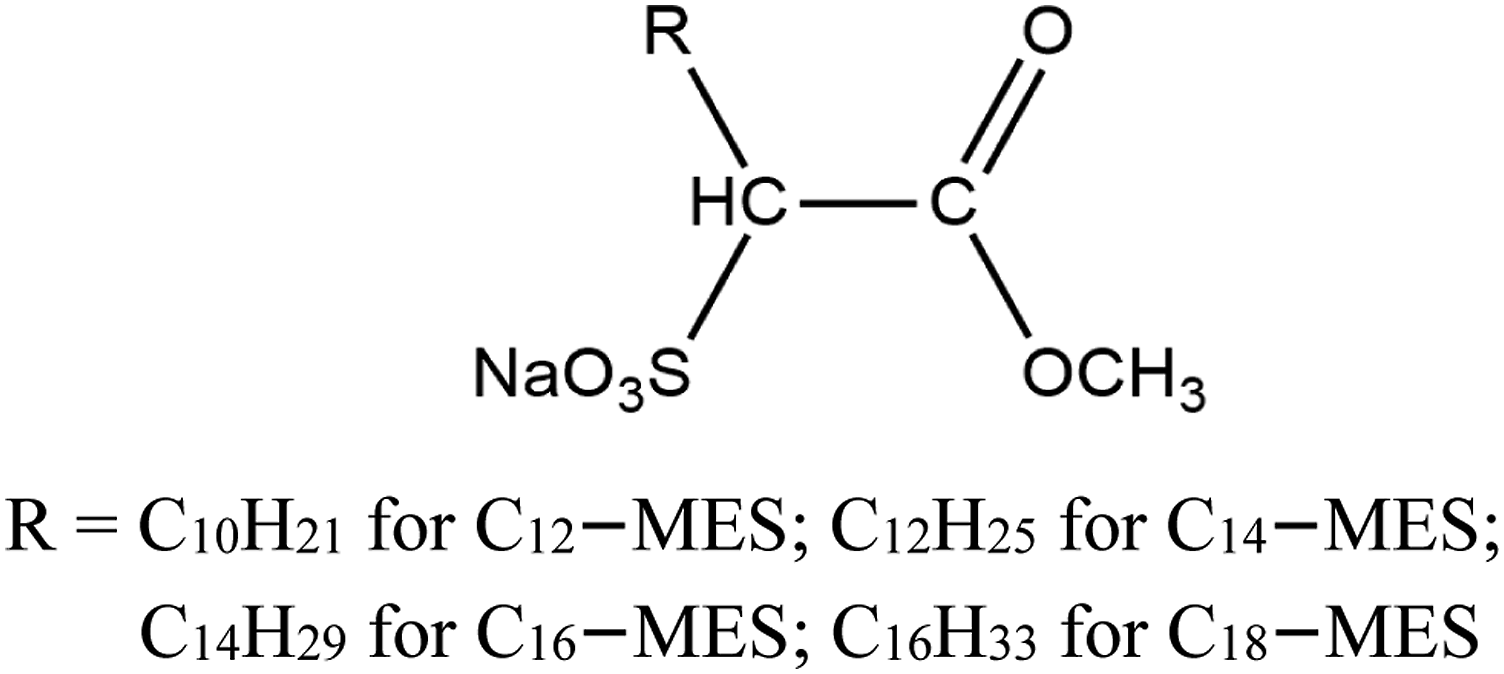
Materials and Methods
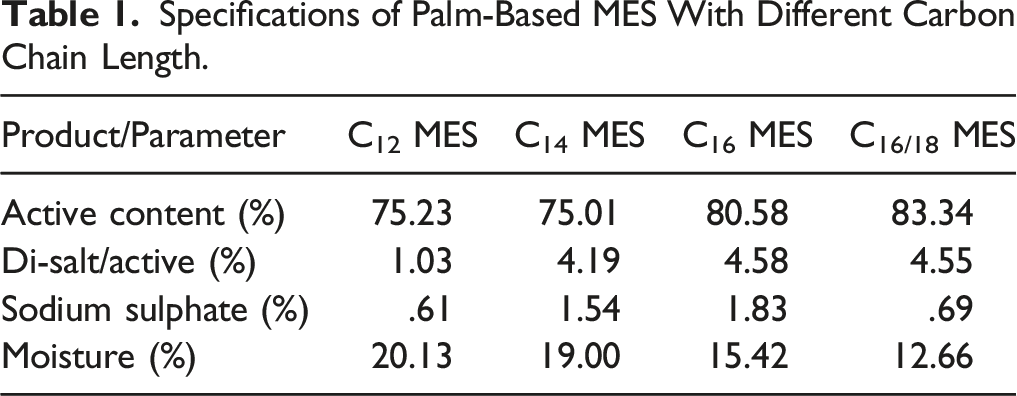
Specifications of Palm-Based MES With Different Carbon Chain Length.
The plate incorporation method, with and without S-9 metabolic activation, was conducted according to the Organization for Economic Cooperation and Development (OECD) Test Guideline 471 37 and Mortelmans and Zeiger. 38 The S-9 used in this study is a supernatant fraction of a rat liver homogenate (S-9 microsomal fraction). Bacterial strains were freshly prepared from working stock by pre-culturing for 12 hours at 37°C with agitation in nutrient broth. Then, the test substance was dissolved in dimethyl sulfoxide (DMSO). A mixture of test solution (.1 mL), .1 mL of fresh bacterial culture (1-2 × 109 CFU/mL), buffer (.5 mL) and overlay agar (2.0 mL) was prepared for the system without metabolic activation. To evaluate genotoxicity of MES metabolites, a mixture of test solution (.1 mL), .1 mL of fresh bacterial culture (1-2 × 109 CFU/mL), .5 mL of a 10% S-9 mixture (S-9 fraction and co-factors) and overlay agar (2.0 mL) was prepared for the system with metabolic activation. The mixture was then poured over the surface of a minimal agar plate. Concurrent strain-specific negative and positive controls were included in the assay. Negative control consists of DMSO alone, without the test substance, but treated the same way as the treatment groups. Triplicate platings were used for an adequate estimate of variation. The plates were then incubated between 48 to 72 hours at 37°C. The number of revertant colonies per plate was counted after the incubation period.
The reference substance for positive controls employed for TA98, TA100/TA1535, TA1537 and E. coli was 2-nitrofluorene (2.5 μg/plate), sodium azide (5 μg/plate), 9-aminoacridine (50 μg/plate) and nitroquinoline oxide (.75 μg/plate), respectively. The MES samples were dissolved in DMSO according to their concentrations. When the test was carried out in the presence of S-9 metabolic activation, the reference substance for positive control was 2-aminoanthracene for all five strains. Colony numbers were determined by manual counting. The student’s t-test was performed to statistically analyse the data obtained. A P-value < .05 was considered significant.
Results
The Number of Revertant Colonies Obtained From the Ames Test Without and With S9 on C12 Palm-Based MES at Five Analysable Concentrations Against all the Five Strains.
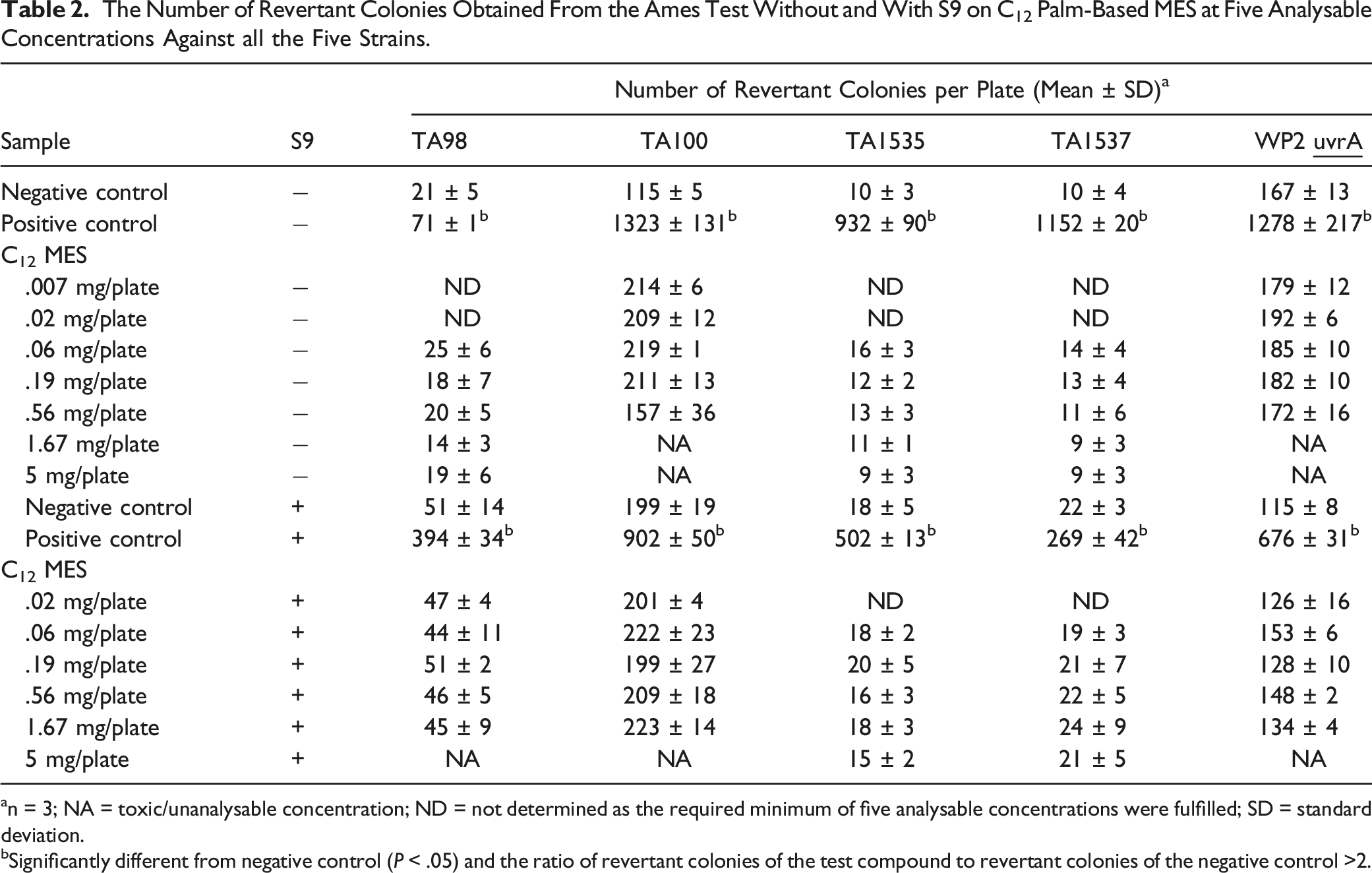
an = 3; NA = toxic/unanalysable concentration; ND = not determined as the required minimum of five analysable concentrations were fulfilled; SD = standard deviation.
bSignificantly different from negative control (P < .05) and the ratio of revertant colonies of the test compound to revertant colonies of the negative control >2.
The Number of Revertant Colonies Obtained From the Ames Test Without and With S9 on C14 Palm-Based MES at Five Analysable Concentrations Against all the Five Strains.
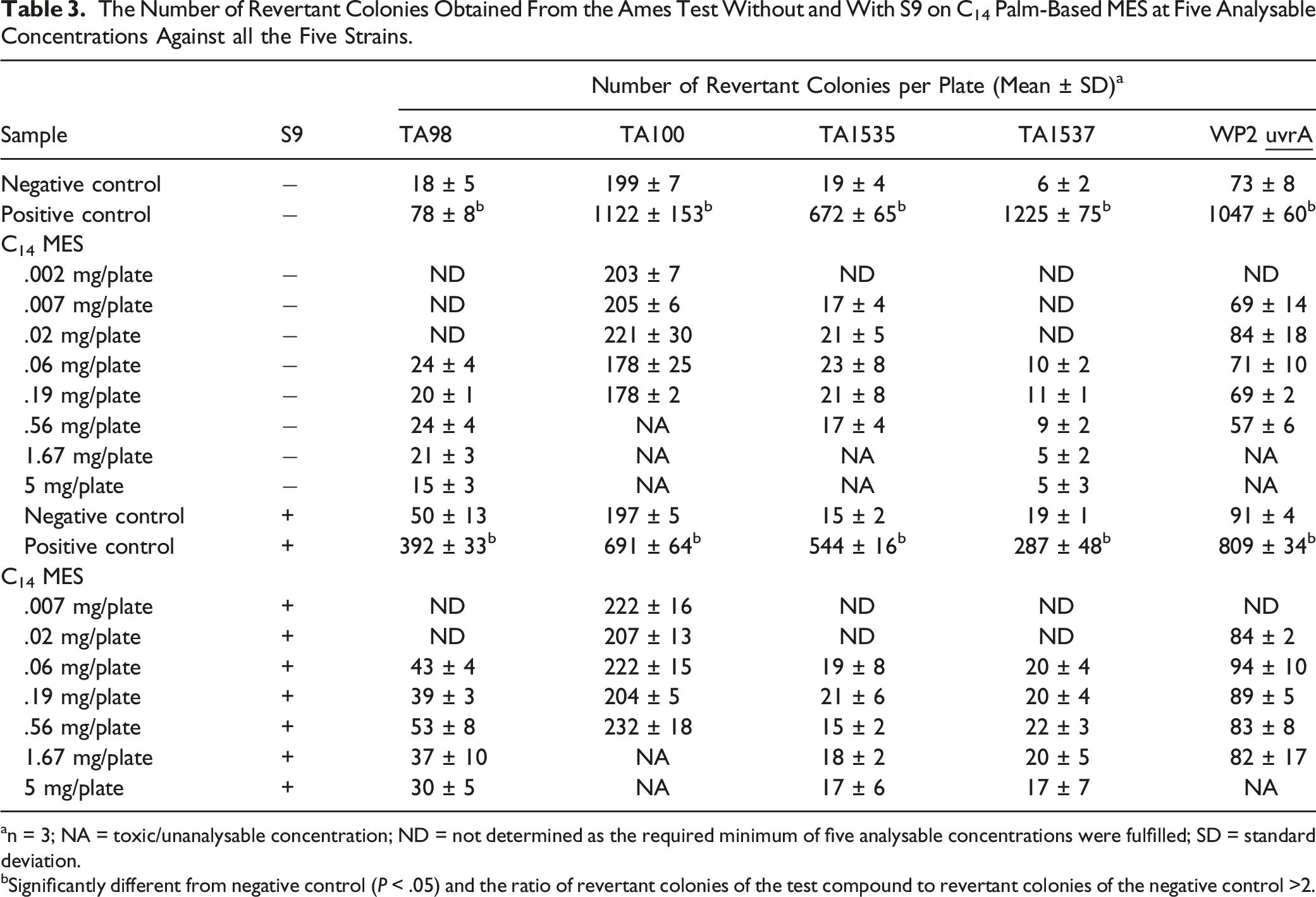
an = 3; NA = toxic/unanalysable concentration; ND = not determined as the required minimum of five analysable concentrations were fulfilled; SD = standard deviation.
bSignificantly different from negative control (P < .05) and the ratio of revertant colonies of the test compound to revertant colonies of the negative control >2.
The Number of Revertant Colonies Obtained From the Ames Test Without and With S9 on C16 Palm-Based MES at Five Analysable Concentrations Against all the Five Strains.
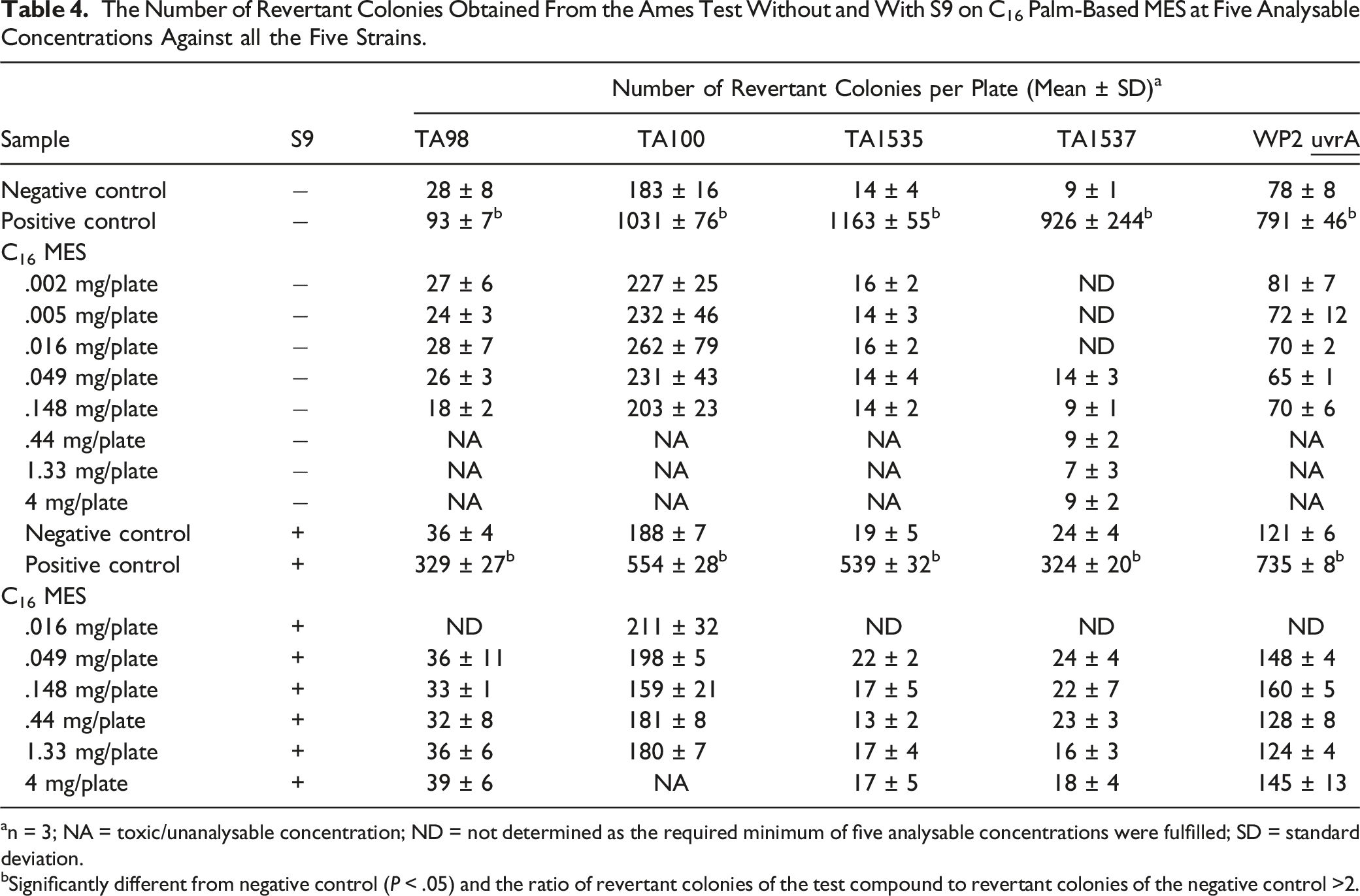
an = 3; NA = toxic/unanalysable concentration; ND = not determined as the required minimum of five analysable concentrations were fulfilled; SD = standard deviation.
bSignificantly different from negative control (P < .05) and the ratio of revertant colonies of the test compound to revertant colonies of the negative control >2.
The Number of Revertant Colonies Obtained From the Ames Test Without and With S9 on C16/18 Palm-Based MES at Five Analysable Concentrations Against all the Five Strains.
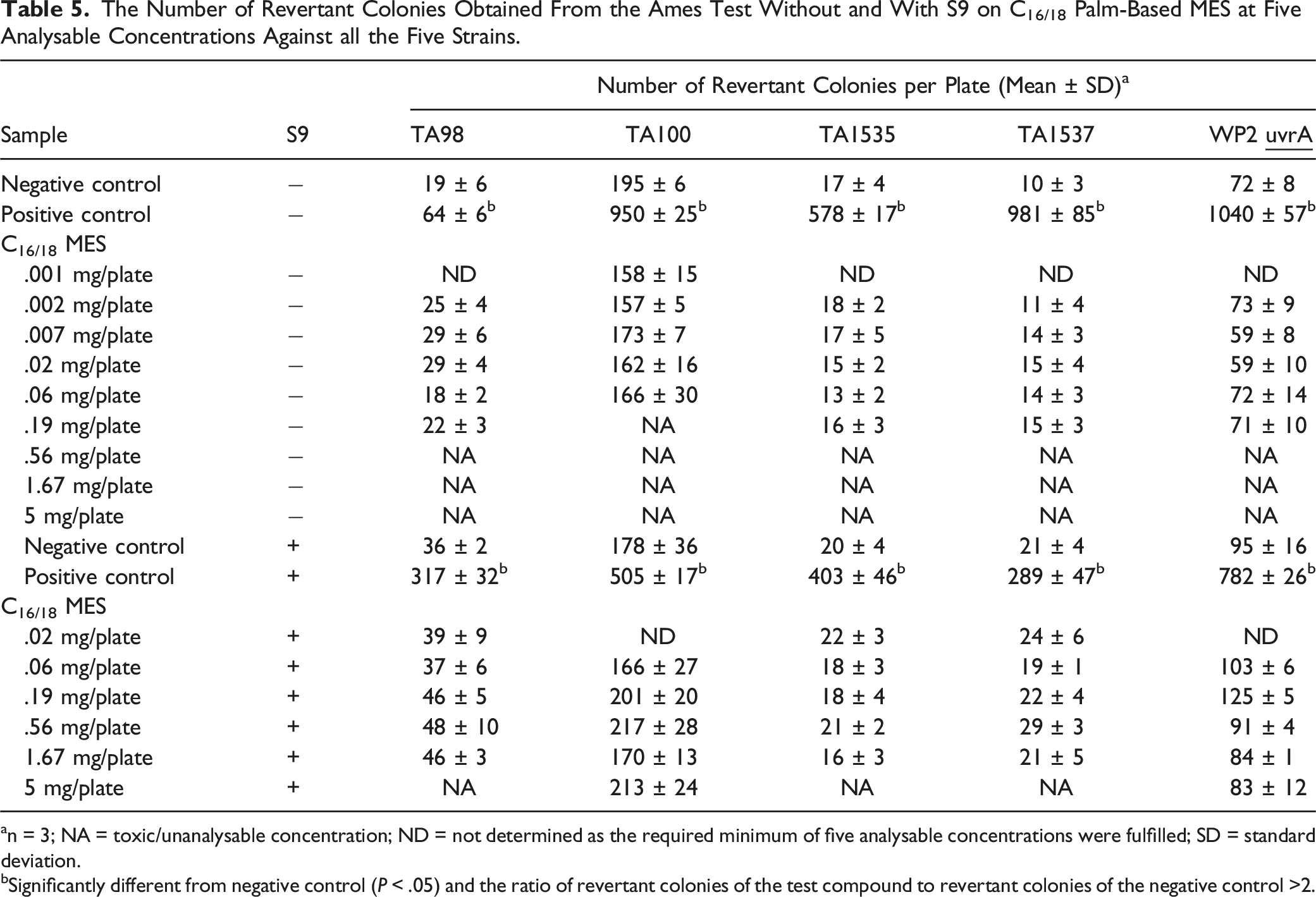
an = 3; NA = toxic/unanalysable concentration; ND = not determined as the required minimum of five analysable concentrations were fulfilled; SD = standard deviation.
bSignificantly different from negative control (P < .05) and the ratio of revertant colonies of the test compound to revertant colonies of the negative control >2.
The Number of Revertant Colonies Obtained From the Ames Test Without and With S9 on LAS at Five Analysable Concentrations Against all the Five Strains.
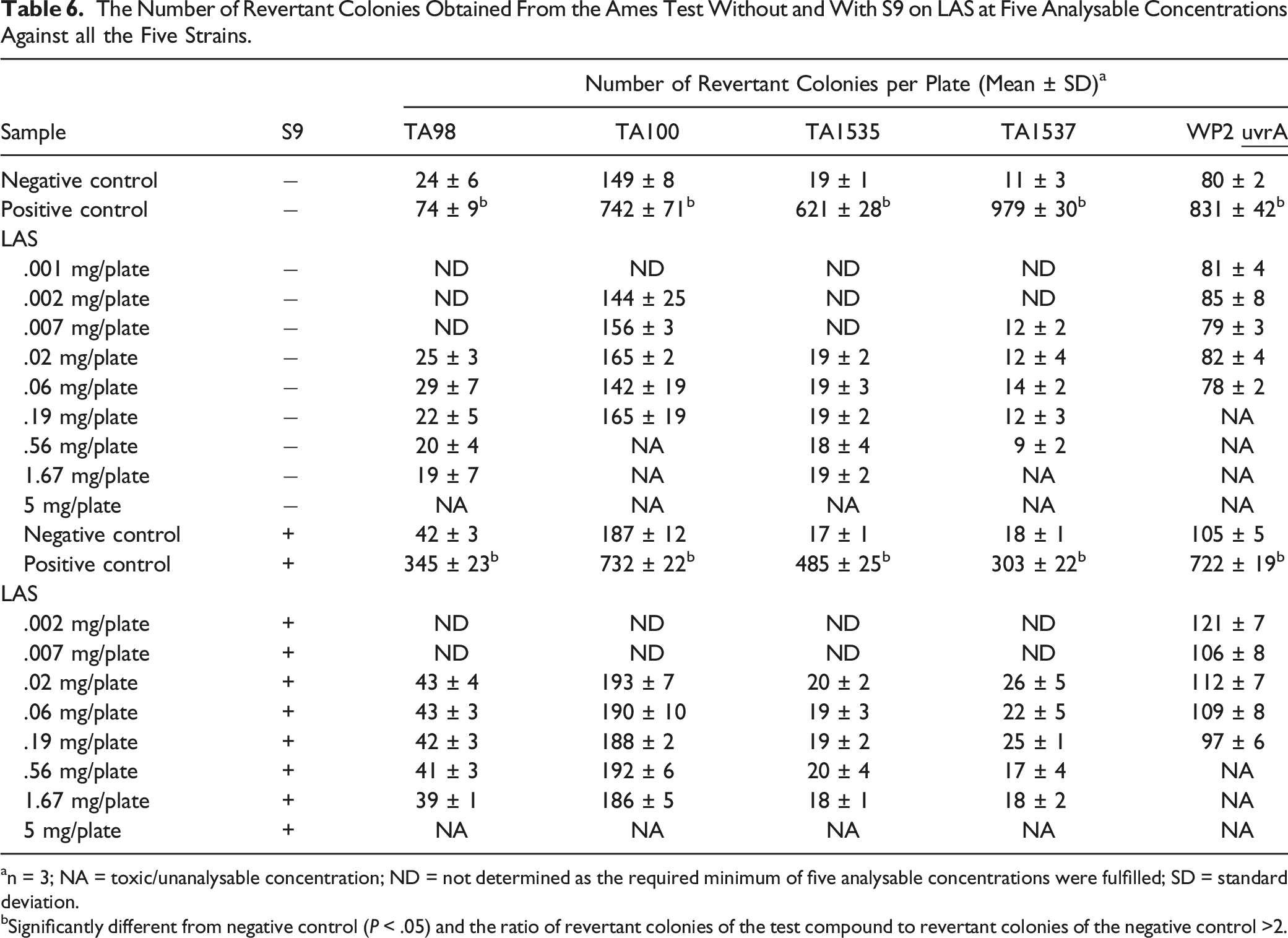
an = 3; NA = toxic/unanalysable concentration; ND = not determined as the required minimum of five analysable concentrations were fulfilled; SD = standard deviation.
bSignificantly different from negative control (P < .05) and the ratio of revertant colonies of the test compound to revertant colonies of the negative control >2.
Discussion and Conclusion
The biggest potential for human exposure to MES is the use of laundry and cleaning products. Laundry and cleaning products may be used as it is or diluted prior to or during use. Exposure to humans could be caused by direct or indirect skin or eye contact, aerosol inhalation from cleaning sprays and many more. Human exposure will be further reduced by the fact that residues from cleaning products on the skin are usually rinsed or washed off.
This work is based on the need to study the mutagenic potential of MES surfactants that are used in detergent formulations. To the best of our knowledge, the assessment of mutagenicity of MES has never been reported. Even though there are several methods to assess the mutagenicity of a substance, the Ames test was selected since it has been widely used for screening purposes with precise results.
Four samples, namely C12, C14, C16 and C16/18 palm-based MES, were assessed for their mutagenicity properties using the Ames test. The cytotoxicity and solubility of the test substances are the criteria to be considered when determining the highest amount of test substances to be used for the Ames test. Cytotoxicity may be detected by a reduction in the number of revertant colonies, a clearing or diminution of the background lawn, or the degree of survival of the treated culture. According to OECD Test Guideline 471, 5 mg/plate is the maximum recommended test concentration for soluble, non-cytotoxic substances. A minimum of five different analysable concentrations of the test substance should be used. Therefore, the maximum test concentration for soluble non-cytotoxic C12, C14, C16 and C16/18 palm-based MES on each strain was determined, and five different analysable concentrations were used in the Ames test. In addition, the validation of the Ames test in the absence and presence of S-9 metabolic activation was also done. Obtaining the data on positive and negative controls for all five strains is essential for validating the test. Each tester strain has a characteristic of revertant colonies spontaneous mutant frequency. The spontaneous mutant frequency refers to the negative control. The number of spontaneously induced revertant colonies per plate is relatively constant, and it will usually increase, that is, dose-dependent, when a mutagen is added to the plate. 38
Based on OECD Test Guideline 471, 5 mg/plate of the sample was used as the maximum concentration for the Ames test as all samples were completely soluble in the solvent, except for C16 palm-based MES. The maximum concentration used for C16 palm-based MES was 4 mg/plate.
A compound is categorized as a potentially mutagenic agent when the ratio of revertant colonies of the test compound to revertant colonies of the negative control is 2 or higher. 13 In this study, the ratio of revertant colonies of each C12, C14, C16 and C16/18 palm-based MES to negative control was less than 2-fold, indicating no evidence of changes in the base-pair substitution or frame-shift mutants of the bacteria. Results were negative in all studies, while the expected results were obtained from the negative and positive controls. Meanwhile, the Ames test was also performed on LAS. Similarly, LAS showed no evidence of mutagenicity, either with or without S-9 metabolic activation, against all five bacteria. This is in agreement with what was reported earlier, based on four well-designed in vitro bacterial (Salmonella) mutagenicity studies on LAS. 39 Even though both MES and LAS have no potential mutagenic properties, the use of MES in detergent and cleaning products is preferable to LAS because MES is derived from renewable sources and is more biodegradable than LAS.18,33
In conclusion, under the Ames test conditions described in this study, C12, C14, C16 and C16/18 palm-based MES were deemed non-mutagenic up to the maximum recommended soluble non-cytotoxic concentration in the S. typhimurium strains TA98, TA100, TA1535, and TA1537 and the E. coli strain WP2
Footnotes
Acknowledgements
The authors would like to thank the Director-General of the Malaysian Palm Oil Board (MPOB). Special thanks are also extended to Nur’ Ain Ali and Nurul Ain Mohd Arzani from Safety and Efficacy Assessment Group, Oleo Product Development Unit, Advanced Oleochemical Technology Division, MPOB, for their technical assistance.
Author Contributions
Y.Y. contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Z.H. contributed to conception and design, and critically revised manuscript; Z.M. contributed to conception, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work, ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors declare that they have no competing interests.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors disclose that financial support for the research described herein was provided by the Malaysian Palm Oil Board, Malaysia.
