Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) reviewed the safety of 12 polyfluorinated polymers in cosmetic products; most of these ingredients have the reported function of film former in common. However, PTFE, the only ingredient that is reported as currently used in cosmetics, functions as a bulking agent and slip modifier, but not as a film former. The Panel reviewed data relevant to the safety of these ingredients under the intended conditions of use in cosmetic formulations, and concluded that PTFE and Hexafluoropropylene/Tetrafluoroethylene Copolymer are safe in cosmetics in the present practices of use and concentration described in the safety assessment; the data are insufficient to determine the safety of the 4 fluorinated-side-chain polymers and 6 fluorinated polyethers.
Keywords
Introduction
The Panel assessed the safety of the following 12 polyfluorinated polymers as used in cosmetics:
Fluoropolymers
PTFE
Hexafluoropropylene/Tetrafluoroethylene Copolymer
Fluorinated-Side-Chain Polymers
Acrylates/Perfluorohexylethyl Methacrylate Copolymer,
Behenyl Methacrylate/Perfluorooctylethyl Methacrylate Copolymer,
C6-14 Perfluoroalkylethyl Acrylate/HEMA Copolymer,
Stearyl Methacrylate/Perfluorooctylethyl Methacrylate Copolymer
Fluorinated Polyethers
Acrylates/Methoxy PEG-23 Methacrylate/Perfluorooctyl Ethyl Acrylate Copolymer,
PEG-10 Acrylate/Perfluorohexylethyl Acrylate Copolymer
Polyperfluoroethoxymethoxy Difluoroethyl PEG Diisostearate
Polyperfluoroethoxymethoxy Difluoroethyl PEG Ether,
Polyperfluoroethoxymethoxy Difluorohydroxyethyl Ether,
Polyperfluoroethoxymethoxy Difluoromethyl Ether
Definitions, Idealized Structures, and Functions of the Ingredients in This Safety assessment.(1; CIR Staff)
Monomer Components of Polyfluorinated Polymers.

This safety assessment includes relevant published and unpublished data for each endpoint that is evaluated. Published data are identified by conducting an exhaustive search of the world’s literature. A list of the typical search engines and websites used, sources explored, and endpoints that the Panel evaluates, is available on the Cosmetic Ingredient Review (CIR) website (https://www.cir-safety.org/supplementaldoc/preliminary-search-engines-and-websites; https://www.cir-safety.org/supplementaldoc/cir-report-format-outline). Unpublished data are provided by the cosmetics industry, as well as by other interested parties.
Chemistry
Definition and General Characterization
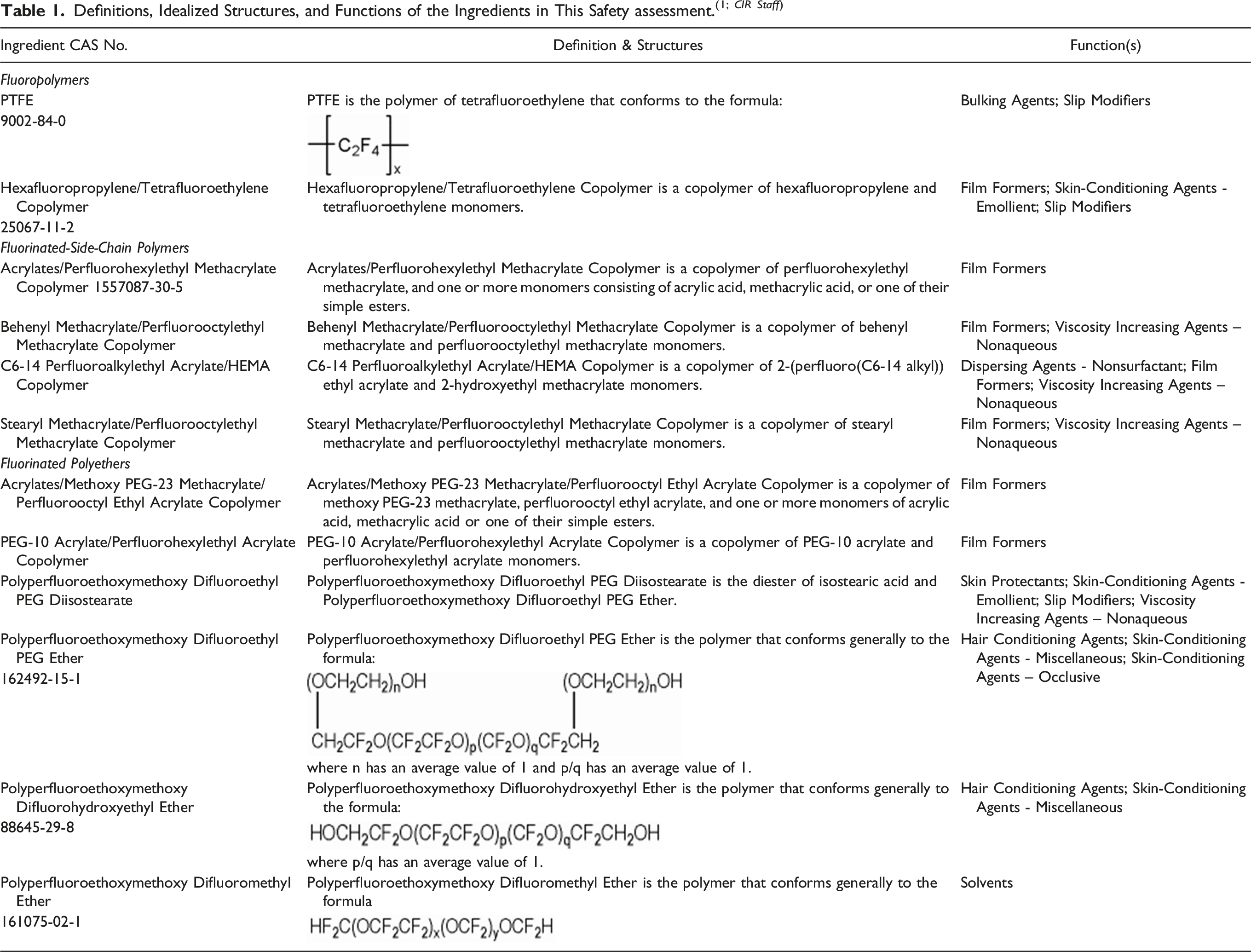
The definitions, structures, and reported functions in cosmetics of these ingredients are presented in Table 1.
1
These ingredients share in common a fluorinated organic polymer backbone, wherein at least some of the carbons in that backbone are perfluorinated. PTFE is a perfluorinated homopolymer, comprising only carbon and fluorine (Figure 1). Together with Hexafluoropropylene/Tetrafluoroethylene Copolymer, these two ingredients comprise the fluoropolymers sub-group. PTFE (Polytetrafluoroethylene).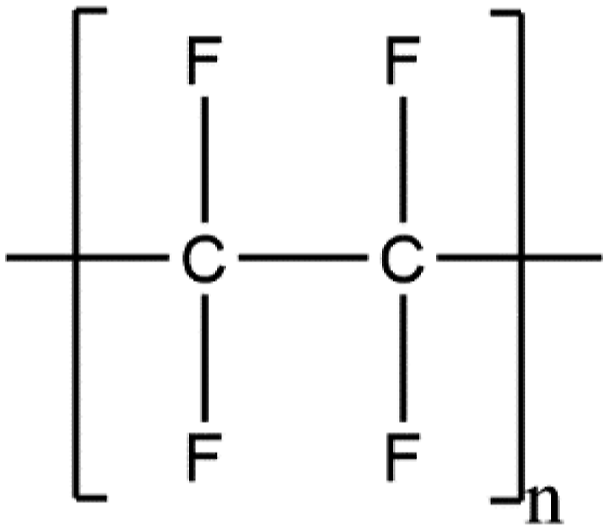
Certain other polyfluorinated polymer ingredients can be classified as fluorinated-side-chain polymers. These ingredients comprise polyacrylates with polyfluorinated side-chains. For example, Acrylates/Perfluorohexylethyl Methacrylate Copolymer is a copolymer of acrylates and methacrylate, wherein the methacrylic acid residues are esterified with perfluorinated, branched chains (Figure 2). Acrylates/Perfluorohexylethyl Methacrylate Copolymer – wherein R is methyl, ethyl, propyl, or butyl.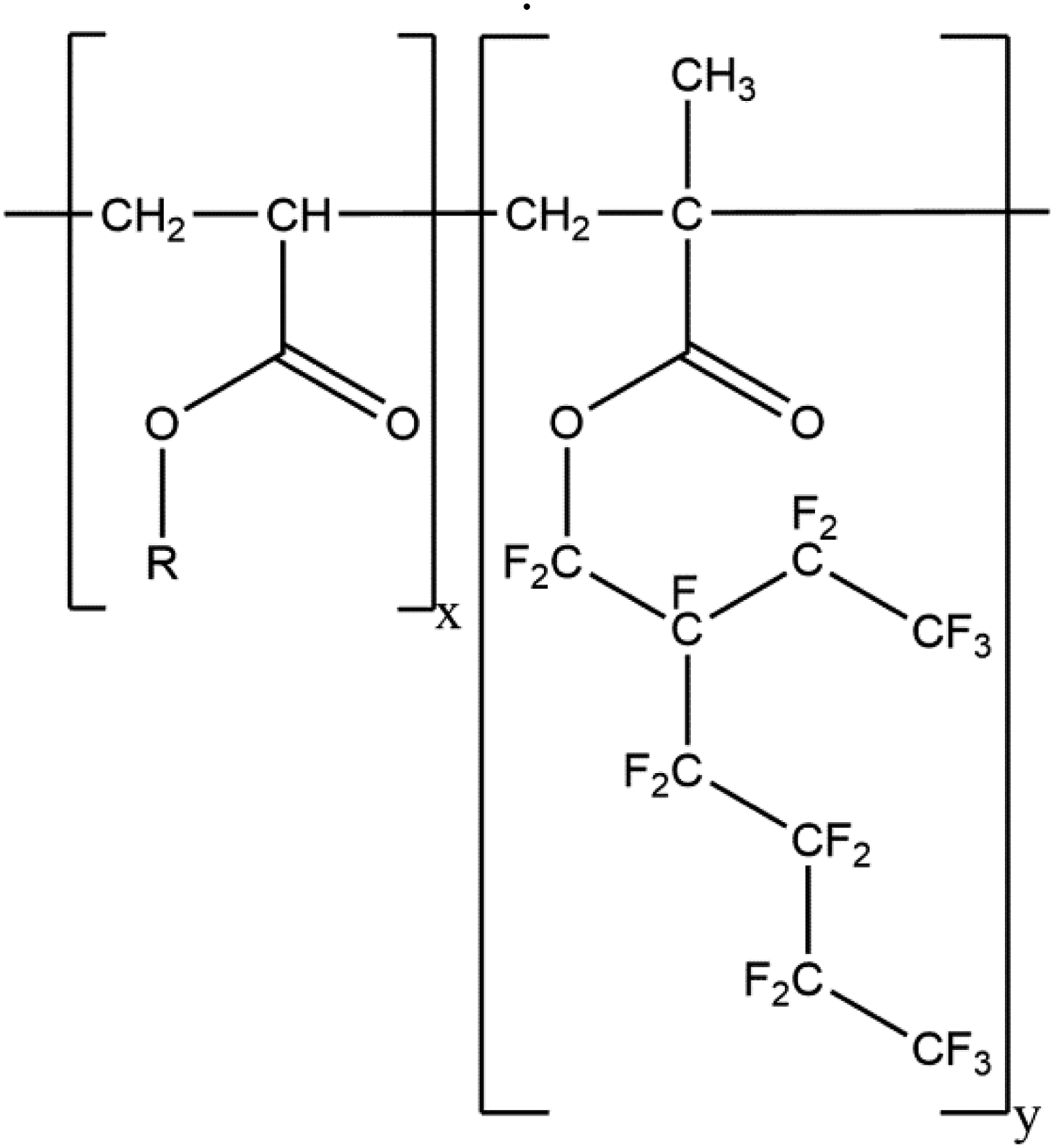
The remainder of the polyfluorinated polymers in this report comprise a fluorinated polyethers sub-group. This sub-group of ingredients comprises copolymers of polyethers and fluorinated monomers. In some cases, Polyperfluoroethoxymethoxy Difluorohydroxyethyl Ether for example, these fluorinated polyethers also comprise end-capping units (Figure 3). Polyperfluoroethoxymethoxy Difluorohydroxyethyl Ether – comprising 2 perfluorinated monomers and end-capping units.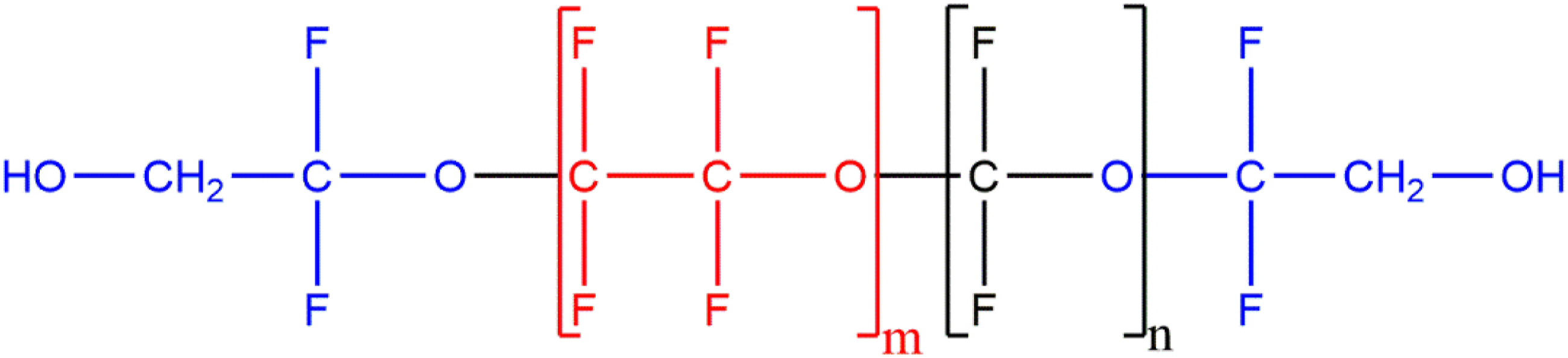
As with any polymeric ingredient, the number of monomeric repeat units (and thus polymeric size) and degree of linearity (i.e., branched or straight) have the potential to greatly impact the physical characteristics (e.g., matter phase, hardness, etc.) imbued on the substance. Accordingly, size, distribution, and connectivity variations, as used in cosmetic ingredients, are important considerations for understanding the nature of these materials.
According to one source, PTFE is composed of at least 20,000 tetrafluoroethylene monomer residues linked into very long, unbranched chains. 2 According to some of the toxicity data in this report, some of the test articles are PTFE fluorotelomers. The International Union of Pure and Applied Chemistry (IUPAC) Gold Book suggests that such telomers comprise very small polymers with between 2 and 10 repeat units. 3 For PTFE, these numbers of monomer residues would result in polymers (telomers) with molecular weights between 238 and 1038 Da.
Chemical and Physical Properties
Chemical and Physical Properties of Polyfluorinated Polymers.

Method of Manufacture
PTFE
PTFE is prepared by the polymerization of tetrafluoroethylene. 2 Because PTFE is poorly soluble in practically all solvents, the polymerization occurs as an emulsion in water. 8 Alternatively, polymerization may be carried out using a surfactant, such as perfluorooctanoic acid (PFOA). According to another source, “the ammonium salts (in some cases also the sodium salts) of long chain perfluoroalkyl carboxylic acids (PFCAs) such as PFOA and perfluorononanoic acid (PFNA) have been applied as processing aids (emulsifiers) at low concentrations (around .5 wt%) in the polymerization of certain polyfluorinated polymers (i.e., PTFE, fluorinated ethylene propylene (FEP), perfluoroalkoxy (PFA), and polyvinylidene difluoride (PVDF)) and fluoroelastomers. After 2006, many fluoropolymer manufacturers in China, Japan, Western Europe and the United States (US) started to replace the salts of long-chain PFCAs with the salts of short-chain PCFAs (such as perfluoro-n-hexanoic acid (PFHxA)) or other nonperfluoroalkyl alternatives (such as polyfluoroalkyl ether carboxylic acids) for fluoropolymer manufacturing.” 9
The method of manufacture of PTFE (from one supplier) is summarized as follows: virgin-grade high molecular weight PTFE is the starting material, and the molecular weight needs to be reduced in order for PTFE to be micronized into a fine powder. 10 Electron beam irradiation is used to lower the molecular weight, typically into the 15,000 to 50,000 Da range. This process is controlled via melting point reduction (starting around 341°C and ending around 330°C). Irradiated PTFE is post-baked to remove volatiles (including any trace PFOA to below 25 ppb). The resulting material is jet mill micronized to the particle size specification (mean particle size = 5.58 μm).
Impurities
According to a chemical supplier, all commercial grades of PTFE contain some trace level of PFOA and perfluorooctyl sulfonate (PFOS); levels of PFOA are fractionally lower than PFOS. 11 The incidental content of PFOA and PFOS is detectable in the ppb range. The supplier also noted that, in 2017, the European Union (EU) published measures to regulate PFOA and its salts and related substances under Annex XVII of Registration, Evaluation, Authorization, and Restriction of Chemicals (REACH). The new law (EU 2017/1000) will be implemented in phases, starting July 4, 2020. Under this new law, trace content of PFOA will be regulated to <25 ppb, and, trace content of PFOA-related substances, regulated to <1000 ppb. The content of tetrafluoroethylene monomer in PTFE has been described as undetectable (detection limit: 75 ppb). 10
Use
Cosmetic
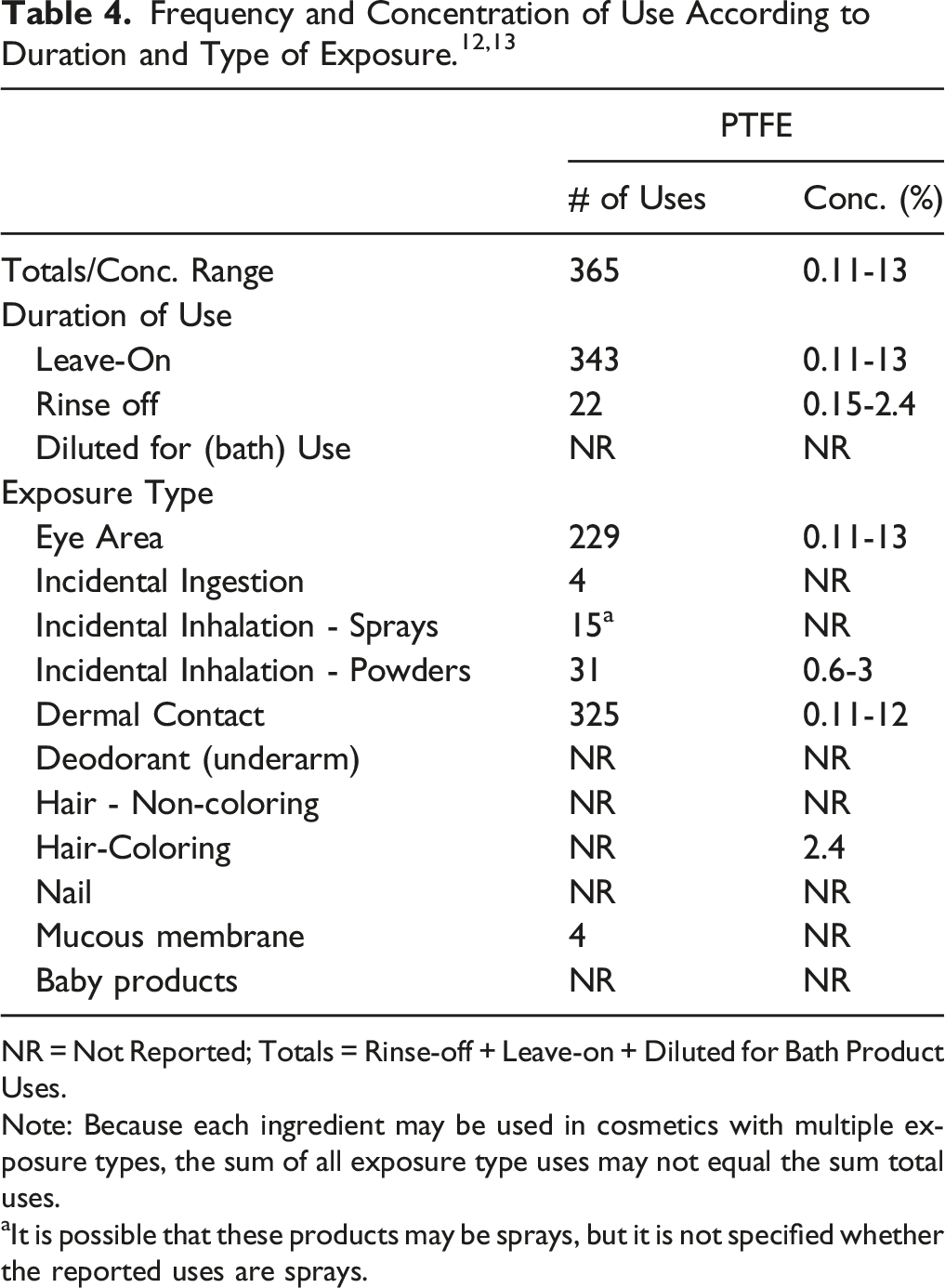
The safety of the polyfluorinated polymers is evaluated based on data received from the US Food and Drug Administration (FDA) and the cosmetics industry on the expected use of these ingredients in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in FDA’s Voluntary Cosmetic Registration Program (VCRP) database. 12 Use concentration data are submitted by the cosmetics industry in response to surveys, conducted by the Personal Care Products Council (Council), of maximum reported use concentrations by product category. 13
NR = Not Reported; Totals = Rinse-off + Leave-on + Diluted for Bath Product Uses.
Note: Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure type uses may not equal the sum total uses.
aIt is possible that these products may be sprays, but it is not specified whether the reported uses are sprays.
Cosmetic products containing PTFE may be applied to the skin and hair or, incidentally, may come in contact with the eyes (at maximum use concentrations up to 13% for PTFE in mascara) and mucous membranes (frequency of use, but not use concentrations, was reported for lipsticks formulations). Ingredient use in lipsticks may result in incidental ingestion. Products containing PTFE may be applied as frequently as several times per day and may come in contact with the skin or hair for variable periods following application. Daily or occasional use may extend over many years.
PTFE is reported in the VCRP as used in [fragrance] powders (dusting and talcum, excluding aftershave talcum) and in face powders, which may result in incidental inhalation exposure. Also, data from the Council’s survey indicate that PTFE is used in face powders at maximum use concentrations ranging from .5 to 3%. According to one supplier, micronized PTFE (fine powder; mean diameter = 5.58 μm) is being used in cosmetic products. 10 However, the particle size of PTFE in the finished cosmetic product was not provided. This mean PTFE particle size diameter (of the raw material ingredient) is within the respirable size range of particles. Respirable fraction is defined as the mass fraction of particles that can reach the alveoli, and the median value of the distribution of particle sizes in this category is 4.25 μm (geometric standard deviation (GSD) = 1.5 μm). 14 It has been shown that 50% of the particles with an aerodynamic diameter of 4 μm will be in the respirable fraction.
The ingredients reviewed in this safety assessment are not restricted from use in any way under the rules governing cosmetic products in the European Union. 15
Non-Cosmetic
Polyfluorinated polymers (e.g., PTFE and Tetrafluoroethylene/Hexafluoropropylene Copolymer) are used in a wide variety of thermal and electrical applications. This is due in part to associated low heats of combustion, low rates of flame spread, high resistance to ignition, and inherent chemical resistance. 16
PTFE
PTFE is ubiquitous in materials that are commonly used in cooking (e.g., coatings for cookware), due to its thermal stability and non-stick properties. 17 PTFE membrane filters have been used in the collection of particulate matter (i.e., nano or ultrafine particulate matter fraction). 18 Diffusion cells that are used in some in vitro percutaneous absorption experiments are made of PTFE. 19 PTFE skin graft chambers have been used to isolate wounds and prevent epidermal healing from the skin edge. 20 Flexible PTFE feeding tubes have been used in oral carcinogenicity studies. 21 Other non-cosmetic uses of PTFE include: hookup and hookup-type wire in electronic equipment; computer wire, electrical tape, electrical components and spaghetti tubing; seals and piston rings, basic shapes, bearings, mechanical tapes, and coated glass fabrics; tubing and sheets for chemical laboratory and process work; lining vessels; in gaskets and pump packings, sometimes mixed with graphite or glass filters; electrical insulator, especially in high-frequency applications; filtration fabrics; protective clothing; and as a prosthetic aid. 2
PTFE is included on the list of resinous and polymeric coatings that the US FDA has determined may be safely used as the food-contact surface of articles intended for use in producing, manufacturing, packing, processing, preparing, treating, packaging, transporting, or holding food (21CFR175.300). PTFE is also included on the list of polymers categorized as exempt from the requirement of a tolerance (i.e., after meeting the criteria specified for defining a low-risk polymer), whereby this categorization relates to use as an inert ingredient in a pesticide chemical formulation (40CFR180.960).
Hexafluoropropylene/Tetrafluoroethylene Copolymer
Hexafluoropropylene/Tetrafluoroethylene Copolymer is included on the list of perfluorocarbon resins that the US FDA has determined may be safely used as articles or components of articles intended to contact food, subject to the provisions that are stated in the CFR (21CFR177.1550).
Toxicokinetic Studies
Dermal Penetration
Dermal penetration data on the polyfluorinated polymers reviewed in this safety assessment were not found in a search of the published literature, and unpublished data were not submitted.
Absorption, Distribution, Metabolism, and Excretion
Absorption, distribution, metabolism, and excretion data on the polyfluorinated polymers reviewed in this safety assessment were neither found in the published literature, nor were these data provided.
Toxicological Studies
Acute Toxicity Studies
Dermal
PTFE
Results relating to the acute dermal toxicity of PTFE are presented in a study evaluating the skin irritation potential of this ingredient. 22 Skin irritation data from this study are summarized later in this report. The test substance (powder, .5 g) was applied to abraded and intact skin of the trunk (area not stated) of 6 New Zealand White rabbits for 24 h. None of the animals died, and no clinical signs or behavioral alterations were observed during the study.
Oral
PTFE
The acute oral toxicity of 2 anti-cohesive coating materials containing PTFE was evaluated using 4 groups of Kunming mice (10 males and 10 females per group). 23 One of the materials contained 60% PTFE, and the other material contained 68 to 73% PTFE. Both materials were administered by gavage. The two 60% PTFE groups received doses of 12.5 and 25 g/kg, respectively. The two 68 to 73% PTFE groups received doses of 2.5 and 5 g/kg, respectively. Dosing was followed by a 1-wk observation period and LD0 (dose at which no animals are expected to die) values were determined. In the 60% PTFE groups, there were no deaths at 12.5 g/kg and 2 deaths at 25 g/kg. In the 68 to 73% PTFE groups, there were no deaths at 2.5 g/kg and 3 deaths at 5 g/kg. The LD0 values in mice were determined to be 12.5 g/kg for the 60% PTFE material and 2.5 g/kg for the 68% to 73% PTFE material.
The acute oral toxicity of 2 anti-cohesive coating materials containing PTFE was evaluated according to the same procedure (stated above) using 4 groups of Wistar rats (10 males and 10 females per group). 23 The two 60% PTFE groups received doses of 6.25 and 18.8 g/kg, respectively. The two 68 to 73% PTFE groups received doses of 1.25 and 3.75 g/kg, respectively. In the 60% PTFE groups, there were no deaths at 6.25 g/kg and 5 deaths at 18.8 g/kg. In the 68 to 73% PTFE groups, there were no deaths at 1.25 g/kg and 1 death at 3.75 g/kg. The LD0 values in rats were determined to be 6.25 g/kg for the 60% PTFE material and 1.25 g/kg for the 68 to 73% PTFE material. 23
A low-molecular-weight PTFE resin (fluorotelomer, chemical characterization data not included) was administered orally to rats (strain and dosing method not stated) at doses as high as 17 g/kg. 24 None of the animals died, and there were no clinical effects or organ changes that were related to test substance administration.
Short-Term Toxicity Study
Dermal
Polyperfluoroethoxymethoxy Difluorohydroxyethyl Ether
Results relating to the short-term dermal toxicity of Polyperfluoroethoxymethoxy Difluorohydroxyethyl Ether are presented in a guinea pig maximization test in which the test substance was injected (0.1 mL)/applied (undiluted) topically to 10 guinea pigs during a 7-d period. 25 Skin sensitization data from this study are summarized later in this report. No mortalities occurred and there were no signs of general toxicity in any of the animals tested.
Inhalation
PTFE
Spray inhalation experiments on a low-molecular-weight PTFE resin (fluorotelomer, molecular weight range and other chemical characterization data not included) were performed using 4 rats (strain not stated). 24 The rats were exposed for 9 d (3 times per day) to a 20% dispersion of the fluorotelomer in dichloro(fluoro)methyl (CCl2F)–chlorodifluoromethyl radical (CClF2) from a pressurized container. After spraying, the jars were sealed and exposure to the dispersion continued for 15 min. A total of 26 exposures were performed. During exposure, incoordination, labored breathing, and irritation of the nose were observed. It was noted that these signs were primarily due to propellants and the dispersing agent (not stated). Recovery occurred immediately after exposure, and it was noted that there was no evidence of pathology that could have been attributed to exposure.
Subchronic Toxicity Studies
Oral
PTFE
Three types of PTFE resin (chemical characterization data not included, 25% in the diet) were fed to male and female rats (strain and number per group not stated) for 90 d. 24 After feeding with each type of PTFE resin, there were no adverse effects on growth rate or behavior and there was no microscopic evidence of tissue changes. However, a slight shift in the distribution and number of white blood cells was observed. Also, feeding with one of the 3 types of resin (unsintered PTFE resin) caused an increase in the size of the liver (relative to body weight). This finding was not accompanied by any histological abnormality. Whether or not the increase in liver size was statistically significant was not stated.
Chronic Toxicity Studies
Oral
PTFE
The chronic oral toxicity of PTFE was evaluated using 6 Swiss mice. 26 The mice were fed a standard diet supplemented with PTFE (concentration not stated) for 6 months. The animals developed spotty loss of fur, skin lesions, and a 50% loss of weight. A control group (fed standard diet only) was included in the study, but results for this group were not reported.
Developmental and Reproductive Toxicity Studies
Oral
PTFE
The teratogenicity of 2 anti-cohesive coating materials containing PTFE (one containing 60% PTFE, and the other containing 68 to 73% PTFE) was evaluated using groups of 10 Wistar rats (5 males and 5 females per group). 23 The positive control, N,N'-methylene bis-(2-amino-1,3,4-thiadiazole), was administered to a group of 12 rats (6 males and 6 females), and the negative control (soybean oil) was administered (by gavage) to a group of 10 rats (5 males and 5 females). The 60% PTFE material was administered at dose of 6.25 g/kg, and the 68 to 73% PTFE material was administered at a dose of 1.25 g/kg. The positive and negative controls were administered at doses of .0005 and .005 g/kg, respectively. All materials were administered once daily on gestation days 7–16. The results for both PTFE materials were classified as negative.
Genotoxicity Studies
In Vitro
PTFE
Two anti-cohesive coating materials containing PTFE (one containing 60% PTFE, and the other containing 68 to 73% PTFE) were negative in the Ames test at doses up to 10,000 μg/plate in Salmonella typhimurium TA98, TA100, and TA1535, with and without metabolic activation. 23
In Vivo
PTFE
The genotoxicity of 2 anti-cohesive coating materials containing PTFE (one containing 60% PTFE, and the other containing 68 to 73% PTFE) was evaluated in the micronucleus test using groups of 10 Kunming mice (5 males and 5 females per group). 23 Cyclophosphamide served as the positive control. Doses were administered by gavage. The 60% PTFE material was administered at a dose of 12.5 g/kg, and the 68 to 73% PTFE material was administered at a dose of 2.5 g/kg. The positive control was administered at a dose of .06 g/kg. Each dose was administered twice, separated by a 24-h interval. Results for both PTFE materials were classified as negative.
Carcinogenicity Studies
Inhalation
Tetrafluoroethylene (PTFE Monomer)
Because tetrafluoroethylene is used primarily in the synthesis of PTFE, it is important to note that the National Toxicology Program (NTP) has evaluated the safety of tetrafluoroethylene in inhalation carcinogenicity studies involving F344/N rats and B6C3F1 mice.
5
Groups of 60 male F344 rats were exposed (inhalation) to 156, 312, or 625 ppm tetrafluoroethylene, and groups of 60 female F344 rats were exposed (in inhalation chamber) to 312, 625, or 1250 ppm tetrafluoroethylene, 5 d/wk (6 h/d) for 104 wk. Groups of 58 male and 58 female B6C3F1 mice were exposed (in an inhalation chamber) to 312, 625, or 1250 ppm tetrafluoroethylene 5 d/wk (6 h/d) for 95 to 96 wk. NTP’s conclusion is stated as follows: Under the conditions of these 2-yr inhalation studies, there was clear evidence of carcinogenic activity of tetrafluoroethylene in male F344/N rats based on increased incidences of renal tubule neoplasms (mainly adenomas) and hepatocellular neoplasms. There was clear evidence of carcinogenic activity of tetrafluoroethylene in female F344/N rats based on increased incidences of renal tubule neoplasms, liver hemangiosarcomas, hepatocellular neoplasms, and mononuclear cell leukemia. There was clear evidence of carcinogenic activity of tetrafluoroethylene in male and female B6C3F1 mice based on increased incidences of liver hemangiomas and hemangiosarcomas, hepatocellular neoplasms, and histiocytic sarcomas.
Carcinogenicity of Implanted PTFE.

Subcutaneous
PTFE
The following results were reported in carcinogenicity studies in which various forms of PTFE (see Table 5) were implanted s.c. in mice of the following strains: 89 random-bred female Swiss mice (fibrosarcomas: 11 of 89 mice),27,28 groups of random-bred male and female Swiss mice (fibrosarcomas: 8 of 89 mice; 1 of 61 mice; 23 of 103 mice; 10 of 53 mice; 7 of 54 mice; and 4 of 40 mice),28,29 19 male and 27 female inbred C57BL mice (sarcomas: 4 of 20 females and 4 of 15 females that retained implant),28,30 40 male and 40 female random-bred, CTM albino mice (sarcomas: 18 of 40 females; 9 of 40 males),28,31 38 BALB/c female mice (fibrosarcomas: 17 of 38 mice),28,32 38 C3Hf/Dp female mice (fibrosarcomas: 36 of 38 mice),28,32 and 39 C57BL/He female mice (fibrosarcomas: 12 of 39 mice).28,32
When PTFE was implanted s.c. in carcinogenicity studies involving rats, the following results were reported: 15 rats of unknown strain (malignant sarcomas: 4 of 15 rats),28,33 65 male and female weanling Wistar rats (sarcomas: 2 of 65 rats),28,34 2 groups of Wistar rats, number per group unknown (sarcomas: 8 of 34 rats and 6 of 32 rats that survived minimum latent period),28,35 39 male Evans rats (no tumors),28,36 40 male Evans rats (no tumors).28,36
Intraperitoneal
PTFE
When PTFE (rod or powder form) was implanted i.p. in rats, the results were as follows: 16 weanling Wistar rats (no sarcomas; fibroadenoma: 1 of 16 rats tested with rods) and 17 weanling Wistar rats (sarcomas: 2 of 17 rats; fibroadenoma: 1 of 17 rats tested with powder; fibrosarcomas: 2 of 17 rats tested with powder).28,37 Rats subjected to the same i.p. implantation surgical procedure (but no material was implanted) served as controls.
Other Relevant Studies
Muscle Necrosis
PTFE
A PTFE patch was implanted (size of implant not stated) in the muscle of rabbits. 38 Medical grade vinyl tubing served as the negative control. At specified time intervals, ranging from 24 h to 12 wk following implantation, the rabbits were killed. The paravertebral muscles were isolated and dissected to recover the implanted material and adjacent tissue. Each site was examined grossly for signs of tissue reaction and the appropriate score was recorded. The implant and adjacent tissue were removed and prepared for microscopic examination. The initial type of necrosis exhibited by rabbit skeletal muscle in response to the physical injury of implant insertion, and the chemical injury sustained by the toxic qualities of the implant, was coagulative necrosis. Coagulative necrosis was soon followed by liquefactive necrosis. The necrotic debris was removed, partly by phagocytic macrophages and giant cells. Fibrosis immediately adjacent to and completely surrounding the implant was observed. In addition to regenerating and encapsulating, fatty infiltration was associated with the repair process. PTFE caused an occasional to mild eosinophilic infiltrate at each time interval investigated.
Inflammatory Response
PTFE
Four animal groups, each consisting of 2 mongrel dogs, 5 New Zealand White rabbits, and 10 BALB/c mice, were injected with PTFE particulate, defined by the following particle size distribution, in a glycerin carrier: 4% of total particulate = 79.1 ± 38 nm; 24% of total particulate = 6100 ± 1000 nm; 30% of total particulate = 7000 to 25,000 nm; and 42% of total particulate = 485 ± 200 nm. 39 The groups were followed for periods of 1 wk, 3 mo, 6 mo, and 1 yr. Mice received one s.c. dorsal injection each, rabbits received 2 subareolar injections each, and dogs received 3 subareolar injections each in addition to 2 periurethral injections. There was no indication that controls were used in the study. Histologic examination of the biopsy sites revealed a persistent chronic inflammatory reaction with progressive growth of the involved tissue volume. In addition to giant cells and macrophages, lymphocytes became apparent at 3 mo and comprised up to 40% of the cellular infiltrate by 1 yr. Plasma cells were also noted at the 1-yr period in the rabbit model.
A material consisting of 72% PTFE and 28% zinc oxide by volume was implanted (size of implant not stated) in the mandibles of 14 guinea pigs. 40 Surgical sites that had no implanted material served as negative controls. Seven animals were killed at intervals of 4 and 12 wk after surgery (i.e., total of 14 animals killed), and tissue sections were prepared. Mild-to-moderate inflammation was observed at 4 wk, but the inflammation was predominantly moderate in intensity. In regions where the material appeared to be loosely dispersed or poorly condensed, a round cell infiltrate was present with active phagocytosis of the material by multinucleated giant cells. The inflammatory response at 12 wk was predominantly mild. The material was surrounded by a moderately thick fibrous capsule with very few inflammatory cells, except for tissue samples in which the material appeared to be poorly condensed. In areas where the material was loosely condensed, active phagocytosis and chronic inflammation persisted and were characterized by the presence of macrophages, plasma cells, and multinucleated giant cells.
Ocular Toxicity
To investigate the effects of focal implantation of expanded PTFE episcleral implants (i.e., explants or exoplants) on surrounding ocular tissues, an experimental and histopathological study was performed. 41 PTFE episcleral implants were inserted (for a period of 3–11 mo) into the eyes of 27 Fauve de Bourgogne rabbits. A newly formed capsule constantly encased the implants. Only 2 severe complications were observed, i.e., 2 eyes had an endocapsular acute inflammation and could not be included in the study. Finally, 25 eyes were studied histopathologically. Neither intrusion nor extrusion of episcleral implants was observed. The inner surface of the capsule was often covered with numerous giant cells, i.e., a foreign-body granuloma developed against the irregular outline of the episcleral implants. The sclera was both thinned and invaginated under the episcleral implants.
Dermal Irritation And Sensitzation Studies
Irritation
Animal
PTFE
The skin irritation potential of PTFE (powder) was evaluated using 6 New Zealand White rabbits (3 males, 3 females). 22 Two areas on the trunk (size of area not stated) were clipped free of hair and one area of skin was abraded. The test substance (.5 g) was applied to occlusive patches that were applied to both intact and abraded sites for 24 h. The test sites were examined for reactions at 24 and 72 h after patch application. Skin reactions were not observed at intact or abraded skin sites in any of the animals tested, and PTFE was classified as a non-irritant.
Human
PTFE
The skin irritation potential of a formula containing 7.6% PTFE was evaluated in a 48-h semi-occlusive patch test involving 26 subjects. 42 The dose per area was not stated. Skin irritation was not observed (primary irritation index (PII) = 0).
A single-insult semi-occlusive patch test (24-h) on an eye shadow containing 12% PTFE was performed using 15 subjects. 43 The location and size of the patch are not stated in this study. Skin irritation was not observed in any of the subjects tested (PII = 0).
Sensitization
Animal
PTFE
A 20% dispersion of the fluorotelomer in CCl2F–CClF2 (defined in Short-Term Inhalation toxicity section) was applied to the skin of 10 guinea pigs (strain not stated). 24 The method and duration of test substance application and dose per area were not stated. When the CCl2F–CClF2 evaporated, the material hardened and moderate mechanical irritation was observed. There was no evidence of sensitization in any of the animals tested.
Polyperfluoroethoxymethoxy Difluorohydroxyethyl Ether
The skin sensitization potential of Polyperfluoroethoxymethoxy Difluorohydroxyethyl Ether was evaluated in a maximization test using 10 test and 5 control male Dunkin Hartley albino guinea pigs. 25 On day 0, the 10 test animals received the following 3 pairs of intradermal injections: Freund’s complete adjuvant (FCA) emulsion (.1 mL), 1% Polyperfluoroethoxymethoxy Difluorohydroxyethyl Ether (.1 mL), and Polyperfluoroethoxymethoxy Difluorohydroxyethyl Ether in FCA (.1 mL). Similarly, the 5 control animals received the following 3 pairs of intradermal injections: FCA emulsion (.1 mL), petrolatum oil vehicle (.1 mL), and vehicle in FCA (.1 mL). On day 6, the animals were treated topically with 10% sodium lauryl sulfate in petrolatum oil (.5 mL). On day 7, the same area was treated with dermal applications of undiluted Polyperfluoroethoxymethoxy Difluorohydroxyethyl Ether or the vehicle for 48 h using an occlusive patch. The test sites were observed for signs of skin irritation 24 h after patch removal. At challenge on day 20, an occlusive patch containing 75% Polyperfluoroethoxymethoxy Difluorohydroxyethyl Ether or the vehicle was applied for 24 h to animals of the 2 groups. Sites were observed for any reactions at 24 h after patch removal and at 24 h later.
The injection of the test substance (in vehicle) caused slight irritation (number of animals not stated). Reactions were not observed after injection of the vehicle alone. At 24 h post-removal of the 48-h occlusive patch, signs of slight irritation (erythema) were observed at sites treated with the test substance. None of the animals had a positive reaction after treatment with the test substance during the challenge phase. Also, no skin reactivity was observed in the negative control group. The authors concluded that Polyperfluoroethoxymethoxy Difluorohydroxyethyl Ether did not appear to possess sensitizing capacity in this study. 25
Human
PTFE
The skin sensitization potential of a formula containing 2.89% PTFE was evaluated in a human repeated insult patch test (HRIPT, occlusive patches) involving 107 subjects. 42 The dose per area and duration of patch application were not stated. There was no evidence of dermal irritation or sensitization.
The skin sensitization potential of an eye shadow containing 6% PTFE was evaluated in 111 subjects. 44 Approximately .01 to .04 g of the product was applied to an occlusive patch that was placed on the back (left side; size of area not stated) of each subject. The patch remained in place for 24 h. This procedure was repeated for a total of 9 induction patch applications over an approximately 3-wk period. The challenge phase was initiated after a 2-wk non-treatment period. An occlusive patch containing .2 g of the product was applied to the right side of the back (new site) for 24 h. Reactions were scored at the time of patch removal and at 48 h and 96 h after patch application. Two subjects had a low-level (±; faint minimal erythema) reaction during the induction, and the same was true for 2 other subjects during the challenge phase. It was concluded that the eye shadow did not induce skin sensitization in any of the subjects tested.
An HRIPT on a cosmetic product containing 9% PTFE (undiluted) was performed using 206 subjects. 45 The subjects received 9 consecutive 24-h induction patches (4 cm2 semi-occlusive patch) of the product (.2 mL applied) on the infrascapular area of the back. Induction sites were evaluated at 48-h intervals. Patches applied on Friday were removed 24 h later, and reactions were evaluated on the following Monday, i.e., 72 h after patch application. The challenge phase was initiated (during week 6) after a 10- to 15-d non-treatment period. Challenge patches were applied for 24 h to new test sites, and reactions were scored at 48 h and 72 h post-application. There was no evidence of skin sensitization in any of the subjects tested.
Ocular Irritation Studies
In Vitro
PTFE
The ocular irritation potential of a formula containing 2.89% PTFE was evaluated in the in vitro EpiOcularTM eye irritation test. An ET50 of >24 h (no eye irritation potential) was reported. 42
Animal
PTFE
In a study involving 6 New Zealand White rabbits (3 males, 3 females), PTFE (powder, .1 g) was instilled into the conjunctival sac of the right eye. 22 The lids were held together for ∼3 to 4 seconds in order to prevent loss of the test substance. The eyes were rinsed at 24 h post-instillation, and observations were made for up to 72 h post-instillation. No clinical signs or behavioral alterations were observed. Conjunctival redness was observed in 4 rabbits. After 24 h, the reactions had cleared in 3 animals. The reaction had cleared after 48 h in the fourth animal. PTFE was classified as non-irritating to the eye in this study.
The ocular irritation potential of a 20% dispersion of the fluorotelomer in CCl2F–CClF2 was evaluated using rabbits (number and strain of animals and test protocol not stated). 24 The test substance caused mild conjunctival irritation, which subsided in less than 72 h. Mild corneal injury was observed at 24 h, but not at 48 h. It was noted that the transient reactions observed in this study were no greater than those that were caused by CCl2F–CClF2 alone.
Clinical Studies
Other Clinical Reports
PTFE
The cellular tissue response to subcutaneously implanted PTFE (laminated to aluminum oxide; 5 × 10 mm2 implant blocks) was evaluated using 7 healthy volunteers. 46 PTFE was implanted s.c. in the iliac crest region. After 1, 2, 4, 12, and 26 wk, respectively, the implants with surrounding soft tissue were removed for histological and immunohistochemical examination using a panel of antibodies to various leukocyte markers. After 1 week, there were signs of edema, slight vessel proliferation, and fibroblast proliferation. At 2 wk, a foreign body reaction with giant cells and some decomposed microfragmented implant material dominated the peri-implant picture. At 4 wk, there were only some giant cells seen, the reaction having been mostly lymphohistiocytic. In one specimen, eosinophils were detected. At 12 wk, the vessel proliferation, fibroblast proliferation, and foreign body reactions were decreasing, but there was still a slight lymphohistiocytic reaction. Thus, PTFE implants primarily induced a slight foreign body reaction, leaving only a slight lymphohistiocytic reaction at 26 wk. The authors noted that the study provided no indication of a toxic, allergic, or traditional immunological pathogenesis of the tissue reaction being elicited by PTFE.
Occupational Exposure
PTFE
Pneumoconiosis was observed in a patient who had been exposed to PTFE during a PTFE spray-coating process over a period of 28 yr. 47 The spray solution contained 55 to 65% PTFE. Spraying was performed 2000 to 3000 times per day (50 cm away from the face), and the patient worked 6 d/wk (10 h/d) without a respirator. The presence of PTFE particles was confirmed, and the airborne concentration of PTFE was estimated to be .75 mg/m3. Particle size measurements in the personal air breathing zone ranged from 1 to 22 μm, and included a fraction less than 1 μm. Using computed tomography, lung biopsy, and electron microscopy, the patient was diagnosed with PTFE-induced granulomatous lung disease. The authors noted that this case demonstrates that long-term exposure to PTFE spraying can cause granulomatous lung lesions such as pneumoconiosis. Furthermore, it was noted that such lesions appear not to be caused by the degradation products of PTFE from high temperatures, but by spraying the particles of PTFE.
The percentage retention at 24 h of 4 µm PTFE particles (aerodynamic diameter of ∼6 μm) in the alveoli was studied using a total of 29 healthy male volunteers. 48 Students/workers at a university (11 total) inhaled 4.2 ± .7 and 4.3 ± .8 μm (mean ± SD) PTFE particles and the workers from a battery factory (18 workers) inhaled 3.9 ± .4 μm PTFE particles. Inhalation of the test particles resulted from 10 to 20 maximally deep inhalations. Radioactivity in the lungs was measured after inhalation. The 24-h retention correlated significantly with the first second of the forced expiratory volume (FEV1) and the forced vital capacity (FVC), and persisted when the subjects were divided into different categories according to profession and smoking habits. It was noted that the results suggest that exposure to particles larger than a few microns in workers with large FEV1 values may result in a greater risk for systemic toxic effects, when compared to workers with small FEV1 values.
Clinical phenomena in employees exposed to fumes from the processing of PTFE have been reported. 49 After exposure to the fumes, there is a latent period of a few hours and then a feeling of general malaise, aching muscles, a sense of oppression behind the mid-chest, a dry throat, and a cough followed, by shivering and profuse sweating. The symptoms abate after 24 h, with no after-effects. Seven cases were described, which included the 4 employees regularly working in the PTFE section of a fabrication works. Two cases were seen during the acute phase of the illness; x-ray examination of the chest revealed no abnormalities. One case had marked conjunctival congestion. Two employees working on a “dispersion process” (process similar to paint spraying, using PTFE dispersed in 10% chromic acid) complained of skin irritation.
An investigation concerning human exposure to PTFE took place at a fabricating plant that employed 130 persons. 50 Air levels of PTFE ranging from 0 to 5.48 mg/m3 were found. Urinary fluoride levels were investigated as an index of PTFE exposure, because carbonyl fluoride, a pyrolysis product of PTFE, is metabolized and excreted as inorganic fluoride ion. Spot urine samples and occupational histories relating to polymer fume fever were obtained from 77 workers. All urine values were below the level at which systemic effects are reported to occur. Analyses of the results (analysis of variance method) demonstrated that the mean urinary fluoride level among workers who had one or more years of exposure to PTFE (workers also had experienced one or more reported episodes of polymer fume fever) was significantly higher (P < .01) than that among employees with less than one year of exposure and no history of polymer fume fever. Additional exposure beyond one year and additional polymer fume fever episodes did not result in further elevation of urine fluoride levels.
Risk Assessment
The US Environmental Protection Agency (EPA) has not, as of December 2022, promulgated an enforceable drinking water standard for PFOA/PFOS under the Safe Drinking Water Act. Because of the persistence of PFOA and PFOS in the environment and their toxicity, mobility, and bioaccumulation potential, these pose potential adverse effects on human health and the environment. To provide Americans, including the most sensitive populations, with a margin of protection from a lifetime of exposure to PFOA and PFOS from drinking water, EPA’s Office of Water established health advisory levels at 70 ppt (equal to .07 ppb) (81 FR 33250; May 25, 2016). 51
Due to the adverse health effects of PFOA and PFOS, the Panel considered the risk from incidental ingestion of PTFE-containing cosmetic formulations. According to VCRP survey data received in 2018, PTFE is used in in 4 lipstick products. 12 However, the current use concentration data of PTFE in lipsticks has not been provided. 13 For the purpose of this risk assessment, considering a worst-case scenario, it is assumed that the use concentration of PTFE in lipsticks is 100%. Additionally, in an abundance of caution, this scenario includes the assumption that all of daily used lipstick is incidentally ingested. (However, it is extremely unlikely that an adult will ingest all of the lipsticks, and such ingestion certainly exceeds conditions of intended use).
In accordance with the Scientific Committee on Consumer Safety (SCCS) Notes of Guidance, the estimated daily exposure level for lipsticks (lip salve) is .057 g.
52
Thus, a total dose of PFOA exposure can be estimated: • Exposure dose of PFOA = 25 ppb highest trace content
10
x 100% (assumed maximum use concentration of PTFE in lipsticks) = 25 ppb = 25 ng/g. • Therefore, the total dose of PFOA exposure = 25 ng/g × .057 g = 1.425 ng =
Comparatively, the maximum dose suggested by EPA’s drinking water limitation can be calculated as follows: • EPA established a health advisory level of 70 ppt (.07 ppb) for PFOA and PFOS (combined) in drinking water. This level was based on a lifetime of exposure to total perfluorinated compounds.
53
• In setting this advisory level, EPA assumed a drinking water ingestion rate of 2 L/d for adults.
54
• Therefore, based on EPA’s health advisory, the maximum dose of PFOA exposure via drinking water should not exceed = .07 μg/l × 2 L =
Accordingly, a very conservative, worst-case scenario of cosmetic usage of a lipstick would result in a daily incidental ingestion dose of PFOA equal to .001425 μg/d, which is essentially 100-fold lower than the EPA’s advisory level for drinking water (.14 μg/d).
Though there is no reported maximum use concentration of PTFE for lipsticks, the maximum concentration of use reported across all categories is 13%.
13
Utilizing a concentration of 13% still results in an overestimation of ingredient exposure from lipstick use (as that particular use was intended for application to skin/hair (i.e., mascara), instead of mucous membrane (i.e., lips)). • When maximum use concentration of 13% PTFE in lipstick is assumed, the total dose of PFOA exposure = 25 ng/g × 13% × .057 g = .18525 ng = .00018525 μg/d.
This overly conservative estimate results in a total dose of PFOA exposure that is 755-fold lower than the EPA’s advisory level for drinking water.
Summary
The Panel assessed the safety of 12 polyfluorinated polymers as used in cosmetics. According to the Dictionary, these polyfluorinated polymers are reported to have the following functions in cosmetics: bulking agents, slip modifiers, film formers, viscosity increasing agents, dispersing agents, skin conditioning agents, skin protectants, hair conditioning agents, and solvents. Most of the ingredients have the film former function in common. However, PTFE, the only ingredient that is reported as being used in cosmetics, functions as a bulking agent and slip modifier, but not as a film former.
According to 2018 VCRP data, PTFE is reported to be used in 365 cosmetic products (343 leave-on and 22 rinse-off products). The results of a concentration of use survey conducted by the Council in 2017 indicate that PTFE is being used at concentrations up to 13% in leave-on products (mascara), which is the greatest use concentration that is being reported for PTFE, and at concentrations up to 2.4% in rinse-off products (hair bleaches). No use of the remaining 11 polyfluorinated polymers in cosmetics has been reported in VCRP or Council survey data.
A PTFE production method was provided by a supplier of this material. Using electron beam irradiation, the molecular weight of virgin grade, high molecular weight PTFE (as supplied) is reduced to a range of 15,000 to 50,000 Da. The irradiated PTFE is then post-baked to remove volatiles (including any trace PFOA to <25 ppb) and micronized into a fine powder (mean diameter = 5.58 µ). Trace tetrafluoroethylene monomer in PTFE is not detected (75 ppb detection limit). According to another supplier, all commercial grades of PTFE contain some trace level of PFOA (and fractionally lower levels of PFOS); this incidental content is detectable (if present) in the ppb range.
PTFE (powder, .5 g) was applied to abraded and intact skin of the trunk of 6 New Zealand White rabbits for 24 h. None of the animals died, and no clinical signs or behavioral alterations were observed.
A low molecular weight PTFE resin (fluorotelomer, chemical characterization data not included) was administered orally to rats at doses as high as 17 g/kg in an acute oral toxicity study. None of the animals died, and there were no test substance-related clinical effects or organ changes. In other acute toxicity tests, oral LD0 values were determined to be 12.5 g/kg for an anti-cohesive coating material containing 60% PTFE and 2.5 g/kg for an anti-cohesive coating material containing 68 to 73% PTFE) in Kunming mice. Oral LD0 values were determined to be 6.25 g/kg (for 60% PTFE material) and 1.25 g/kg (for 68 to 73% PTFE material) in Wistar rats.
In a guinea pig maximization test, Polyperfluoroethoxymethoxy Difluorohydroxyethyl Ether was injected (.1 mL) on day 0 and applied (undiluted) topically on day 7 to 10 guinea pigs. No mortalities occurred, and there were no signs of general toxicity.
In spray inhalation experiments, 4 rats were exposed for 9 d (3 times per day) to a 20% dispersion of a low molecular weight PTFE resin in CCl2F–CClF2. Incoordination, labored breathing, and irritation of the nose were observed, but there was no evidence of exposure-related pathology. It was noted that these signs were primarily due to propellants and the dispersing agent (not stated).
Three types of PTFE resin were fed to rats (25% in the diet) for 90 d. There were no adverse effects on growth rate or behavior, and there was no microscopic evidence of tissue changes. Feeding with one of the 3 types of resin (unsintered PTFE resin) caused an increase in the relative size of the liver.
When 3 types of PTFE resin were fed (at 25% in diet) to rats for 90 d, there was no microscopic evidence of tissue changes. However, an increase in liver size was observed after feeding with one of the 3 types of resin (unsintered PTFE resin). In a chronic oral toxicity study involving 6 Swiss mice fed a standard diet supplemented with PTFE (concentration not stated) for 6 mo, growth was normal, but the animals developed spotty loss of fur, skin lesions, and a 50% loss of weight.
Two anti-cohesive coating materials containing PTFE (60% PTFE and 68 to 73% PTFE) were not teratogenic in Wistar rats. The materials were administered once daily on gestation days 7–16.
Results for the two anti-cohesive coating materials were negative in the Ames test at doses up to 10,000 μg/plate in the S. typhimurium TA98, TA100, and TA1535, with and without metabolic activation. The two materials were also negative for genotoxicity in the micronucleus test.
When PTFE was implanted s.c. or i.p. in rats of different strains, tumor formation around the implantation site was observed. The same was true for PTFE implanted s.c. in mice of different strains. The tetrafluoroethylene monomer, used in the synthesis of PTFE, was found to be carcinogenic in mice and rats in an NTP inhalation carcinogenicity study.
In a study in which PTFE particulate in a glycerin carrier was injected into 2 mongrel dogs, 5 New Zealand White rabbits, and 10 BALB/c mice, histologic examination of the biopsy sites revealed a persistent chronic inflammatory reaction. Mild to moderate inflammation was observed in a group of 13 guinea pigs after mandibular implantation of a material consisting of 72% PTFE and 28% zinc oxide.
After an occlusive patch containing PTFE powder (.5 g) was applied to abraded and intact skin of 6 rabbits for 24 h, skin irritation was not observed. A 20% dispersion of a low molecular weight PTFE resin in CCl2F–CClF2 was applied to the skin of 10 guinea pigs. There was evidence of what was described as mechanical irritation, but no evidence of sensitization. The skin sensitization potential of Polyperfluoroethoxymethoxy Difluorohydroxyethyl Ether in 10 guinea pigs was evaluated using the maximization test. Induction and challenge concentrations applied were 100 and 75%, respectively. The test substance (1%) was also injected intradermally during induction. Slight skin irritation, but no sensitization reaction, was observed.
A formula containing 7.6% PTFE was classified as non-irritating to the skin in a 48-h patch test involving 26 subjects. An eye shadow containing 12% PTFE was not irritating to the skin of 15 subjects in a 24-h patch test. In an HRIPT involving 107 subjects, a formula containing 2.89% PTFE did not cause dermal irritation or sensitization. An eye shadow containing 6% PTFE did not induce skin sensitization in an HRIPT involving 111 subjects. There also was no evidence of skin sensitization in the 206 subjects patch tested with 9% PTFE.
A foreign-body reaction (slight lymphohistiocytic reaction) was observed in 7 healthy volunteers implanted s.c. with PTFE. Pneumoconiosis was observed in a patient who had been exposed to PTFE during a PTFE spray-coating process over a period of 28 yr. It was noted that such lesions appear not to be caused by the degradation products of PTFE from high temperatures, but by spraying the particles of PTFE.
A formula containing 2.89% PTFE was classified as having no ocular irritation potential in the in vitro EpiOcularTM eye irritation test. PTFE powder (.1 g) was classified as non-irritating to the eyes of 6 rabbits. The eyes were rinsed after instillation. Also, in rabbits, a 20% dispersion of a low molecular weight PTFE resin in CCl2F–CClF2 caused transient mild conjunctival irritation and corneal injury. The reactions observed were no greater than those that were caused by CCl2F–CClF2 alone.
Due to the adverse health effects of PFOA and PFOS, the Panel considered the risk from incidental ingestion of PTFE-containing cosmetic formulations. The highest maximum use concentration of PTFE (13%) in cosmetic products was utilized. Thus, an overly conservative estimation of ingredient exposure from lipstick use results in a total dose of PFOA exposure that is 755-fold lower than the EPA’s advisory level for drinking water.
Discussion
According to a cosmetic ingredient supplier, all commercial grades of PTFE contain some trace level of PFOA (detectable in ppb range) and fractionally lower levels of PFOS. More specific data on PTFE composition that were received indicate that the tetrafluoroethylene monomer is undetectable (75 ppb detection limit) and that perfluorooctanoic acid (PFOA) is present at concentrations of <25 ppb. The Panel noted that the EPA has established a health advisory level of 70 ppt (.07 ppb) for PFOA and PFOS (combined) in drinking water to provide Americans, including the most sensitive populations, with a margin of protection from a lifetime of exposure to PFOA and PFOS. Due to the adverse health effects (developmental toxicity/carcinogenicity) that are associated with these two impurities, the Panel considered the risk from incidental ingestion of PTFE-containing cosmetic formulations. The Panel previously determined that a value for the greatest possible amount of incidentally ingested PFOA and PFOS that would result from the use of oral hygiene products at the maximum use concentration of PTFE should be calculated and included in this safety assessment. Although the available use information now indicates that PTFE is not being used in cosmetic oral hygiene products, 2018 FDA VCRP data indicate that it is being used in lipsticks (use concentration data unavailable). Thus, the use of PTFE in products that are applied to the lips and the highest maximum use concentration of PTFE (13%) in cosmetic products (including those for which incidental ingestion is extremely unlikely) were used in this calculation. It was determined that an overly conservative estimation of ingredient exposure from lipstick use results in a total dose of PFOA exposure that is 755-fold lower than the EPA’s advisory level for drinking water (in addition to the 100-fold safety factor inherent in EPA’s limit).
The Panel discussed the issue of incidental inhalation exposure from powders. PTFE is reported in the VCRP data as used in [fragrance] powders (dusting and talcum, excluding aftershave talcum) and in face powders, which may result in incidental inhalation exposure. The Council’s survey results indicate that PTFE is used in face powders at maximum use concentrations ranging from .5 to 3%. The Panel noted that, according to one supplier, a raw material micronized PTFE ingredient (fine powder; mean diameter = 5.58 μm) is being used in final cosmetic formulations, and that this mean PTFE particle size diameter, before formulation, appears to be within the range of respirable particles. Respirable fraction is defined as the mass fraction of particles that can reach the alveoli, and the median value of the distribution of particle sizes in this category is 4.25 μm (GSD = 1.5 μm). However, the Panel’s concern over potential PTFE-induced inhalation toxicity was mitigated after results from a short-term inhalation toxicity study (4 rats; twenty-six 15-min exposures total) on a low molecular weight PTFE resin were reviewed. There was no evidence of pathology that could have been attributed to PTFE resin exposure in this study. Furthermore, the Panel noted that conservative estimates of inhalation exposures to respirable particles during the use of loose powder cosmetic products are 400-fold to 1000-fold less than protective regulatory and guidance limits for inert airborne respirable particles in the workplace. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at https://www.cir-safety.org/cir-findings.
The Panel noted that this safety assessment contains data from several subcutaneous implantation carcinogenicity studies on PTFE in various forms (e.g., disks, films, surgical mesh), the results of which indicated the formation of fibrosarcomas, fibroadenomas, and sarcomas (some of which were malignant) in mice/rats. However, taking into consideration the various types of cosmetic products in which PTFE is used, the Panel determined that these studies are not relevant to determining safety for cosmetic use. The Panel also reviewed data from an NTP inhalation carcinogenicity study indicating that the tetrafluoroethylene monomer was carcinogenic in rats and mice. However, the Panel noted that, according to the production method that was received from one supplier of PTFE, residual tetrafluoroethylene monomer in PTFE is not detected (detection limit: 75 ppb).
Finally, the Panel determined that additional data are needed for completion of the safety assessment of only the fluorinated side-chain polymers and fluorinated polyethers that are being reviewed. These 10 ingredients are not reported to be in use. The complete list of data needs includes: • Method of manufacture • Impurities • Skin sensitization data at maximum use concentration
It should be noted that method of manufacture, impurities, and skin sensitization data were received and considered sufficient for determining that Hexafluoropropylene/Tetrafluoroethylene Copolymer and PTFE would be safe at cosmetic use concentrations up to 13%. The need for skin sensitization at the highest maximum use concentration of PTFE (13%) was previously requested by the Panel. In response to that request, a negative HRIPT on a lower concentration, 9%, was received. The Panel agreed that these data at a lower concentration of PTFE, combined with the weight of the evidence, are sufficient for determining that PTFE is not a skin sensitizer at use concentrations in cosmetics; and, the same would be true if Hexafluoropropylene/Tetrafluoroethylene Copolymer were to be used in cosmetic products.
Conclusion
The Expert Panel for Cosmetic Ingredient Safety concluded that the following two fluoropolymers are safe in cosmetics in the present practices of use and concentration described in this safety assessment.
Fluoropolymers
PTFE
Hexafluoropropylene/Tetrafluoroethylene Copolymer*
*Not reported to be in current use. If this ingredient not in current use to be used in the future, the expectation is that it would be used in product categories and at concentrations comparable to PTFE.
The Panel also concluded that the data are insufficient to determine the safety of the following 10 ingredients, which are not reported as currently used in cosmetic products:
Fluorinated-Side-Chain Polymers
Acrylates/Perfluorohexylethyl Methacrylate Copolymer
Behenyl Methacrylate/Perfluorooctylethyl Methacrylate Copolymer
C6-14 Perfluoroalkylethyl Acrylate/HEMA Copolymer
Stearyl Methacrylate/Perfluorooctylethyl Methacrylate Copolymer
Fluorinated Polyethers
Acrylates/Methoxy PEG-23 Methacrylate/Perfluorooctyl Ethyl Acrylate Copolymer
PEG-10 Acrylate/Perfluorohexylethyl Acrylate Copolymer
Polyperfluoroethoxymethoxy Difluoroethyl PEG Diisostearate
Polyperfluoroethoxymethoxy Difluoroethyl PEG Ether
Polyperfluoroethoxymethoxy Difluorohydroxyethyl Ether
Polyperfluoroethoxymethoxy Difluoromethyl Ether
Footnotes
Author Contributions
The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.