Abstract
Antioxidant 1 copper chaperone (Atox1) may contribute to preventing DDP cochlear damage by regulating copper transport family and cell cycle proteins. A rat model of cochlear damage was developed by placing gelatin sponges treated with DDP in the cochlea. HEI-OC1 cells were treated with 133 μM DDP as a cell model. DDP-induced ototoxicity in rats was confirmed by immunofluorescence (IF) imaging. The damage of DDP to HEI-OC1 cells was assessed by using CCK-8, TUNEL, and flow cytometry. The relationship between Atox1, a member of the copper transport protein family, and the damage to in vivo/vitro models was explored by qRT-PCR, western blot, CCK-8, TUNEL, and flow cytometry. DDP had toxic and other side effects causing cochlear damage and promoted HEI-OC1 cell apoptosis and cell cycle arrest. The over-expression of Atox1 (oe-Atox1) was accomplished by transfecting lentiviral vectors into in vitro/vivo models. We found that oe-Atox1 increased the levels of Atox1, copper transporter 1 (CTR1), and SOD3 in HEI-OC1 cells and decreased the expression levels of ATPase copper transporting α (ATP7A) and ATPase copper transporting β (ATP7B). In addition, the transfection of oe-Atox1 decreased cell apoptosis rate and the number of G2/M stage cells. Similarly, the expression of myosin VI and phalloidin of cochlea cells in vivo decreased. Atox1 ameliorated DDP-induced damage to HEI-OC1 cells or rats' cochlea by regulating the levels of members of the copper transport family.
Introduction
Cisplatin (DDP), cis-diamminedichloroplatinum (II), is a metal coordination compound and one of the most effective and commonly used alkylation chemotherapeutic agents for the treatment of various solid tumors. 1 In 1978, DDP was approved by the U.S. Food and Drug Administration (FDA) and has been widely used to treat many tumors. 1 When radiotherapy is not an option, platinum derivatives are often used as first-line treatment for solid cancers, in combination with other chemotherapeutic agents for malignancies. 2 The anticarcinogen properties of DDP stem from the substitution of chlorine ligands by nucleophilic moieties (such as DNA strands), thus hindering the replication of cancer cells. 1 Despite its high toxicity and frequent recurrence of drug resistance, it remains the only treatment option for multiple tumors.3,4 Therefore, lowering DDP toxicity remains the main objective of current clinical and basic research.
Ototoxicity is a common side effect of cancer treatment using DDP, limiting its use and dose. 5 Ototoxicity refers to the damage caused by drugs that affect the structure of the inner ear and related neural tissues, resulting in cochlear or vestibular dysfunction.6-11 DDP-induced human ototoxicity was more dominant in the cochlea than in the vestibular apparatus. 12 The mechanism of DDP-induced cochlear dysfunction has been associated with cell damage and the destruction of cochlear homeostasis. Ototoxicity stems from the overproduction of reactive oxygen species in the cochlea, leading to apoptosis of outer hair cells, spiral ganglion cells, and vascular lines. 13 Treatment with DDP was found to impair the synapses between Corti outer organ hair cells, vascular fringe cells, spiral ganglion cells, inner hair cells, and spiral ganglion neurons.14,15 Unlike other side effects, DDP-induced ototoxicity results in irreversible damage to the inner ear as the hair cells cannot usually regenerate. 16
Copper transport family proteins are common transporters of copper (Cu) and DDP and were found to reduce the adverse effects of DDP accumulation on the body.17,18 Copper is an indispensable trace element necessary for biological functions 19 but can be fatal in toxic concentrations. Copper hydroxyl radicals cause oxidative damage to macromolecules, react with mercaptan groups in proteins, and replace essential metals. 20 Therefore, maintaining copper homeostasis is an essential process in living systems. Copper transporters were found to regulate complex intracellular Cu import, intracellular flux, and Cu efflux. 21 High levels of DDP were shown to inhibit Cu transport families in regulating the retention amount of DDP in cells. 22 As a Cu transport companion factor, Atox1 was demonstrated to be involved in the cell transport of various Cu proteins and could regulate the cell cycle.23-25
Antioxidant 1 copper chaperone (Atox1) transfers cytosolic Cu to Cu pumps in trans Golgi network, thereby facilitating the function of various enzymes. 26 Atox1 facilitates the intracellular transport of DDP. 27 A lack of Atox1 was demonstrated to induce the arrest of many cells in the G2/M stage, slowing the proliferation rate. 28 Protein kinase D inhibitors were demonstrated to sensitize cancer cells to DDP by regulating ATPase copper transporting α/β (ATP7A/B). 29
However, the role of Atox1 in DDP cochlear damage is not fully understood. Our previous study showed that Atox1 and members of the Cu transport family were highly expressed in the inner ear of rats, and that the Cu transport family was regulated by DDP and copper sulfate in the cochlea. Therefore, we aimed to investigate the role of Atox1, members of the Cu transport family (CTR1, ATP7A, and ATP7B), cyclin D1, and superoxide dismutase 3 (SOD3) in DDP-induced cell damage. The knowledge gained in this study will facilitate the development of novel strategies and therapeutic targets for the prevention and treatment of DDP cochlear damage.
Materials and Methods
Cell Counting Kit-8
The mouse cochlear hair cells HEI-OC1 (M8-0401, OriCell, China) were incubated with a conventional Dulbecco’s Modified Eagle Medium (DMEM) at 33°C. First, HEI-OC1 cells were treated with 0 μM, 10 μM, 25 μM, and 50 μM DDP (P4394-25MG, Sigma-Aldrich, USA) for 24 h. After the HEI-OC1 cells reached the logarithmic phase, these cells were inoculated into 96-well plates at 5 × 103 cells/well. Next, the cells were treated with different concentrations of DDP for 24 h. Subsequently, 10 μL Cell Counting Kit-8 (CCK-8) working solution (NU679, DOJINDO) was added to each well. After incubation for 4 h, the absorbance was measured using a microplate reader. The dose causing 50% cell growth inhibition (IC50) was calculated from measurement of cell viability in different concentrations of DDP.30,31
HEI-OC1 Cell Transfection and Cisplatin Treatment
The HEI-OC1 cells were grouped as follows: control, DDP group, DDP + over-expression of Atox1 (DDP + oe-Atox1) group, and the DDP + over-expression of negative control (DDP + oe-NC) group. Cells were treated with 133 μM DDP. The oe-Atox1 and oe-NC were purchased from GenePharma Co., Ltd. (China Shanghai). HEI-OC1 cells were grown on plates and then transfected as per the manufacturer's instructions.
Terminal Deoxynucleotidyltransferase-Mediated dUTP-Biotin Nick End Labeling (TUNEL) Assay
HEI-OC1 cells were evaluated using the TUNEL Apoptosis Assay Kit (40306ES50, Yeasen Biotechnology, China) to detect HEI-OC1 cell apoptosis. HEI-OC1 cells grown on glass slides at a density of 2 × 104/cm2 were fixed with paraformaldehyde for .5 h. Proteinase K working solution was used to treat slides at 37°C for 20 min. Subsequently, the slides were incubated for .5 h in equilibration buffer. Terminal deoxynucleotidyl transferase incubation buffer and 2-(4-amidinophenyl)-6-indolecarbamidine dihydrochloride (DAPI) were then added. Finally, slides were sealed with glycerin and examined under a fluorescence microscope. The number of positively stained cells was counted in the same-sized field of view. The ratio of TUNEL-positive cells to total cells was calculated as the positivity rate, which represented the cell apoptosis rate.
Flow Cytometry
After the HEI-OC1 cells were inoculated with DDP in 6-well plates for 24 h, the cells were digested by pancreatic enzymes. Then the cells were rinsed and resuspended with PBS at low temperature, and precooled ethanol was added to mix the cells. After removing the ethanol with PBS, propidium iodide was used to culture the cells for .5 h. Finally, the cell cycle was measured using a flow cytometer (A00-1-1102, Beckman).
Establishment of Cisplatin-Induced Cochlear Damage Rat Model
Six-month-old Sprague–Dawley (SD) rats of equal genders were purchased from the Shanghai Laboratory Animal Research Center (Shanghai, China). Rats were intraperitoneally injected with 1% pentobarbital (35-40 mg/kg). The corneal reflex was checked with soft cotton. 32 After the corneal response was absent, the hair behind the bilateral ears was cut off and the skin was disinfected. A 1 cm incision was made 2 mm behind the ear to expose the auditory vesicle. Next, a 3-4 mm hole was drilled above the auditory bubble to expose the round window niche. Gelatin sponges (1 mm3) were then soaked with 1.0 μL DDP (.5, 1.0, and 1.5 mg/mL) 33 and placed in the round window niche and referred to as the DDP treatment groups (n = 3 rats per group). The control group was received gelatin sponges soaked with 1.0 μL normal saline at the same position. The wound surface was closed with dental glue. After 30 min of regaining consciousness, all rats were fed separately and freely. Follow-up tests were performed three days later. For rats received the same treatment on both cochleae; bilateral cochleae were used as a single sample. 34
Overexpression of Antioxidant 1 Copper Chaperone in Rats In Vivo and Cisplatin Treatment
The lentivirus transfection complex (Atox1-oeRNA LV and control-oeRNA LV) was diluted according to the manufacturer’s instructions. The scala media at the basal turn of the antennal cochlea was punctured by a Bonn micro-probe, and .6 μL of lentiviral vectors were injected. 35 The concentration of oe-NC/oe-Atox1 lentivirus was determined using flow cytometry and subsequently adjusted to 5 × 106 Vp/μL. 36 The rats were injected with the same volume of lentivirus for three consecutive days. All lentivirus were synthesized by GenePharma Co., Ltd. (China Shanghai). The rats were administered DDP/normal saline through the round window after being transfected for three days. The rats were euthanized three days later with a 4% pentobarbital sodium overdose. The cochleae were fixed in 4% paraformaldehyde and preserved for immunofluorescence.
Immunofluorescence Detection
The rat cochlear cells were stained for myosin VI and phalloidin as previously described. 37 After being fixed with 4% paraformaldehyde, the cochleae were treated with xylene. Then, cochleae were embedded in paraffin wax and sliced into 2 μm sections. After heating at 60°C for 12 h, the cochleae sections were progressively treated with xylene and graded concentrations of ethanol. After antigen retrieval with urea for 30 min, cochleae sections were washed with PBS. The cochleae sections were further treated with sodium borohydride, 75% ethanol, and Sudan black dye. Subsequently, the sections were blocked with bovine serum albumin solution for 1 h. The samples were then successively treated with primary antibody (myosin VI, 1:50, 26778-1-AP, Proteintech, USA) and secondary antibody (CoraLite488-conjugated Affinipure Goat Anti-Rabbit IgG, 1:200, SA00013-2, Proteintech, USA). Sections were then incubated with phalloidin (5 µg/mL, SIGMA, USA) for 30 min in the dark. After staining with DAPI, the samples were coverslipped and fluorescence images were collected.
Western Blot
Cells and cochlear tissues were homogenized in Radioimmunoprecipitation Assay (RIPA) buffer (AWB0136, Abiowell, China) and centrifuged at 4 degrees C. The bicinchoninic acid protein kit (AWB0104, Abiowell, China) was used to measure protein levels in the resulting supernatant. The supernatants (20 μg protein per well) were electrophoresed on 10% PAGE gels at 75 V for 130 min. Proteins were transferred to nitrocellulose membranes at a constant current of 300 mA. The transfection time required for detection of protein Atox1, Copper transporter 1 (CTR1), ATP7A, ATP7B, cyclin D1, SOD3, cyclin-dependent kinase 2 (CDK2), CDK1, cyclin A, cyclin B, B-cell lymphoma-2 (Bcl-2), BCL-2-associated X protein (Bax), and Caspase-9 were 20, 45, 130, 180, 50, 50, 53, 54, 72, 80, 46, 41, and 66 min, respectively. The membrane was treated with 5% skimmed milk powder for 1.5 h. The primary antibody Atox1 (ab154179, 1:500, Abcam), CTR1 (#13086, 1:1000, CST), ATP7A (MA5-27720, 1:1000, ThermoFisher), ATP7B (ab124973, 1:1000, Abcam), cyclin D1 (60186-1-Ig, 1:12000, Proteintech), SOD3 (ab21974, 1:500, Abcam), CDK2 (10122-1-AP, 1:1000, Proteintech), CDK1 (67575-1-Ig, 1:5000, Proteintech), cyclin A (18202-1-AP, 1:4000, Proteintech), cyclin B (55004-1-AP, 1:2000, Proteintech), Bcl-2 (12789-1-AP, 1:2000, Proteintech), Bax (50599-2-Ig, 1:6000, Proteintech), and Caspase-9 (10380-1-AP, 1:3000, Proteintech) were added to the membrane. The secondary antibody HRP goat anti-mouse IgG (SA00001-1, 1:5000, Proteintech) or HRP goat anti-rabbit IgG (SA00001-2, 1:6000, Proteintech) was added to the membrane. The membrane was incubated with chemiluminescence solution source for 1 min. Then, the membrane was wrapped and imaged with a Gel imaging system (ChemiScope6100, Clinx, China), with an exposure time set for 1 min. β-actin was used as an internal reference.
Quantitative Real-Time Polymerase Chain Reaction
Primer Sequences.

Statistical Analysis
GraphPad Prism 9 (GraphPad Software, La Jolla, CA) was used for statistical analysis.
39
The data were represented as the mean ± standard deviation (
Results
Cisplatin Promoted HEI-OC1 Cell Apoptosis and Cell Cycle Arrest
First, we measured the effect of DDP on HEI-OC1 cell proliferation rate. The HEI-OC1 cells were incubated with 0, 10, 25, 50, 100, and 200 μM DDP for 24 h. The CCK-8 results (Figure 1(A)) showed that a higher DDP concentration had a stronger inhibitory effect on the proliferation effect of HEI-OC1 cells (P < .05). The IC50 value of DDP was 133 μM. Therefore, HEI-OC1 cells treated with 133 μM DDP were used throughout this study and referred to as the DDP group. In addition, the TUNEL results (Figure 1(B)) also showed that higher DDP had a stronger pro-apoptotic effect on HEI-OC1 cells (P < .05). In the DDP group (Figure 1(C)), the number of HEI-OC1 cells remaining in the G2/M phase was higher than that in the control group (P < .05). The level of cyclin in HEI-OC1 cells also fluctuated (Figure 1(D)). Compared with the control group, the expression levels of CDK1, cyclin A, and cyclin B increased, while the expression levels of CDK2 and cyclin D1 decreased (both P < .05). Compared with the control group, the levels of Bax and Caspase-9 increased, and the levels of Bcl-2 decreased in the DDP group (both P < .05). In conclusion, DDP had pro-apoptotic and cell cycle arrest effects on HEI-OC1 cells. DDP promoted HEI-OC1 cell apoptosis and cell cycle arrest. (A) The cell proliferation was measured by CCK-8 at increasing DDP concentrations; (B) after treatment with DDP, HEI-OC1 cells were detected by using the TUNEL assay. Scale bar = 25 μm; (C) cell cycle was measured by flow cytometry; (D) levels of CDK-1, cyclin A, cyclin B, DCK2, cyclin D1, Bax, Caspase-9, and Bcl-2 were measured using western blots. *P < .05 vs 0 μM or control. #P < .05 vs 10 μM. & P < .05 vs 100 μM.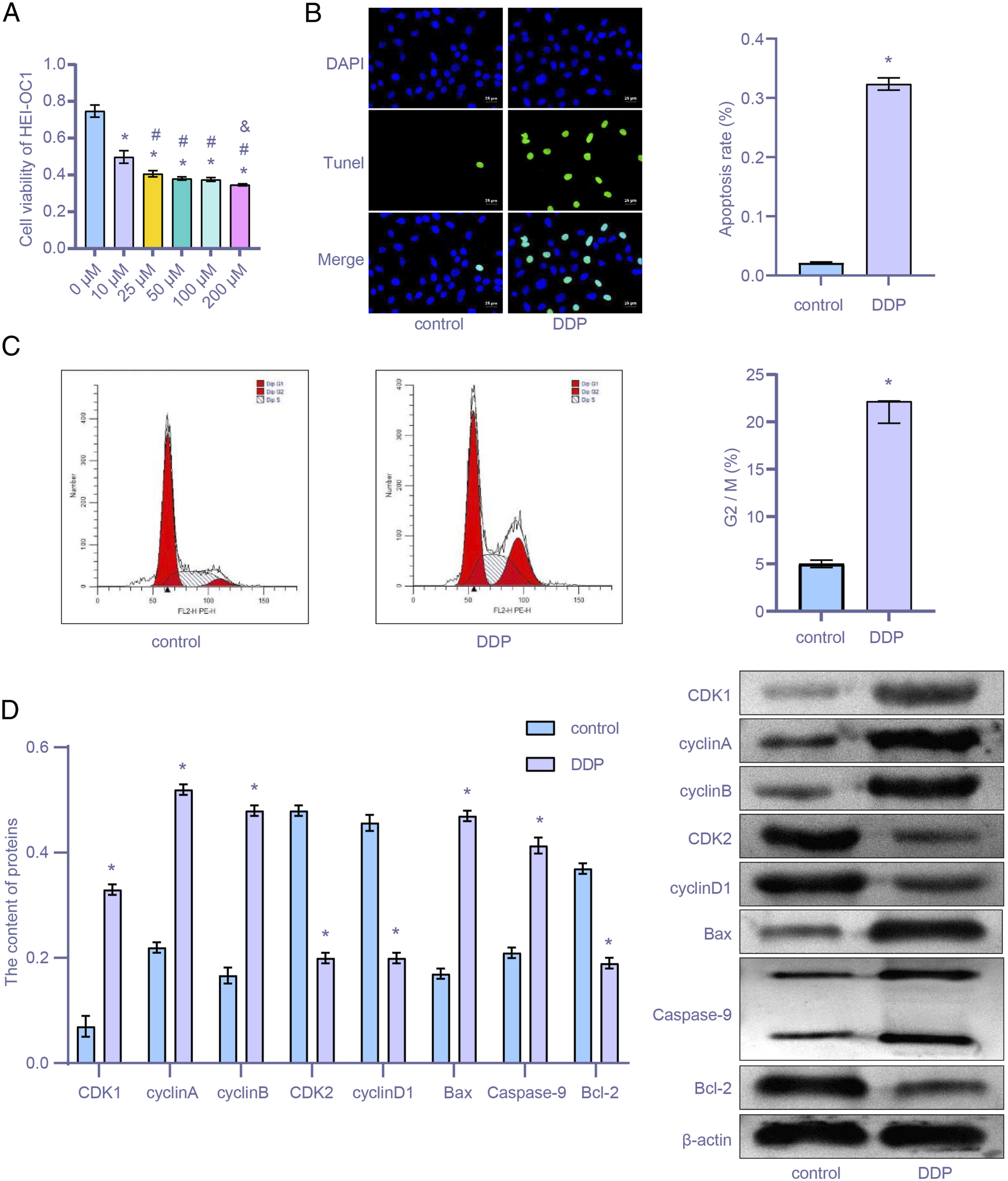
Antioxidant 1 Copper Chaperone Regulated the Cu Transport Family to Suppress HEI-OC1 Cell Damage
After overexpression of Atox1, the relationship between Atox1, Cu transport family, and damage of HEI-OC1 cells was investigated. As shown in Figure 2(A), the expression level of Atox1 in the DDP + oe-Atox1 group was higher than that in the DDP + oe-NC group (P < .05), suggesting successful transfection of oe-Atox1. As shown in Figure 2(B), the proliferation rate of HEI-OC1 cells in the DDP + oe-Atox1 group was higher than that in the DDP + oe-NC group (P < .05). TUNEL staining results showed (Figure 2(C)) that the apoptosis rate of HEI-OC1 cells in the DDP + DDP + oe-Atox1 group was lower than in the DDP + oe-NC group (P < .05). After observing the apoptosis rate of HEI-OC1 cells, the levels of Bax and Caspase-9 in the DDP + oe-Atox1 group were found to be lower than in the DDP + oe-NC group (both P < .05). However, the change in Bcl-2 levels was the opposite. In Figure 2(D), the expression levels of CTR1 and SOD3 in the DDP + oe-Atox1 group were higher than in the DDP + Atox1-NC group, and the expression levels of ATP7A and ATP7B were lower than in the DDP + Atox1-NC group (both P < .05). These results suggest that Atox1 regulates members of the Cu transport family and ameliorates HEI-OC1 cell damage. Atox1 regulated members of the Cu transport family to suppress HEI-OC1 cell damage. With changes in the expression of Atox1, corresponding changes to members of the Cu transport family were observed. These changes included (A) the expression level of Atox1 under qRT-PCR or western blot; (B) the cell proliferation ability under CCK-8 assay; (C) the HEI-OC1 cells apoptotic rate measured using a TUNEL assay; (D) and the expression level of Bax, Caspase-9, Bcl-2, Atox1, CTR1, ATP7A, ATP7B, and SOD3 as measured by using qRT-PCR or a western blot assay (D). *P < .05 vs control. #P < .05 vs DDP. & P < .05 vs DDP + oe-Atox1.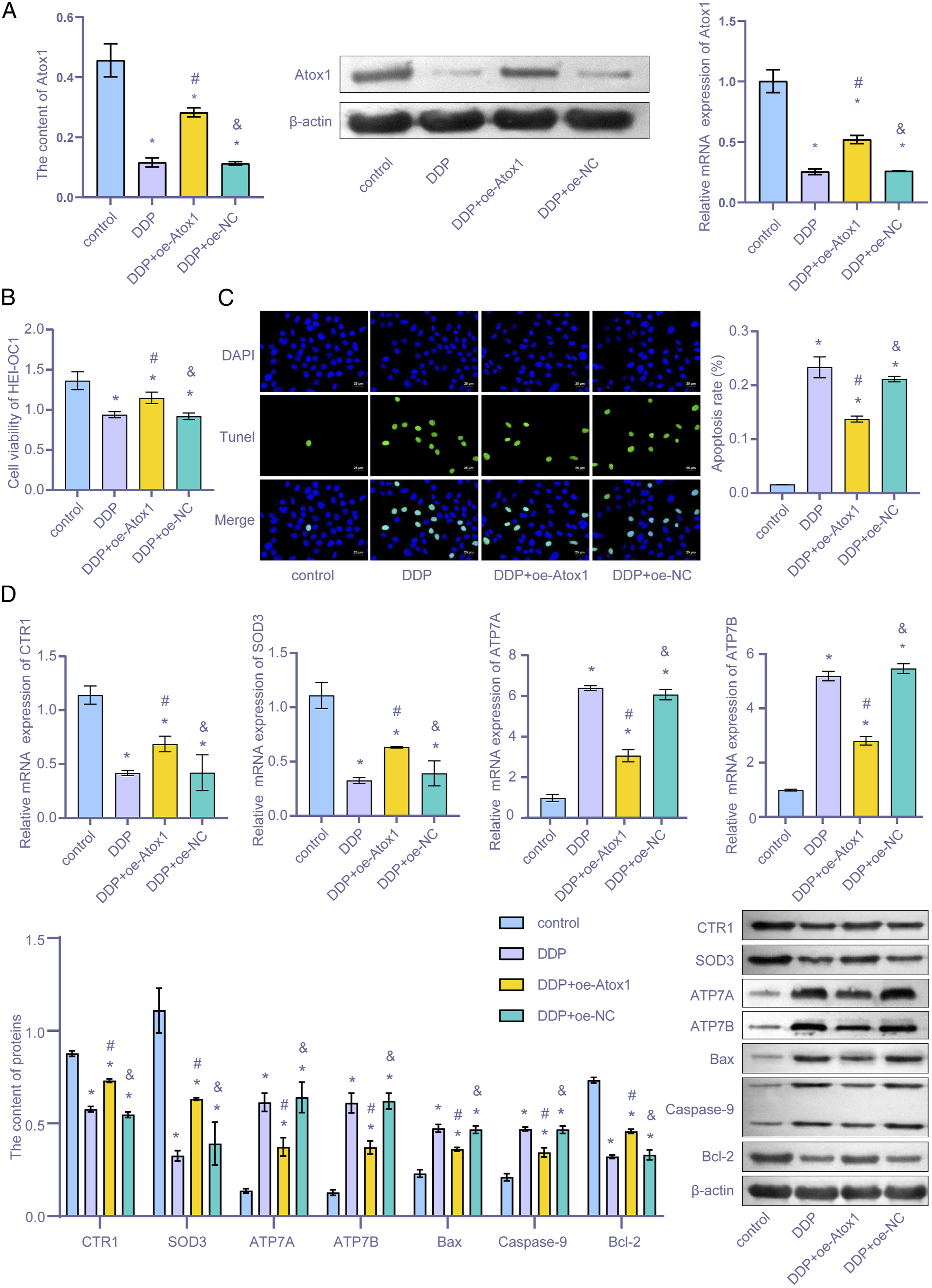
Regulating Antioxidant 1 Copper Chaperone Relieved HEI-OC1 Cell Cycle Arrest
Flow cytometry and western blot assays were performed to study the effect of Atox1 on the HEI-OC1 cell cycle. As shown in Figure 3(A), the number of HEI-OC1 cells in the G2/M stage of the DDP + oe-Atox1 group was lower than in the DDP + oe-NC group (P < .05). After cyclin level changes were detected (Figure 3(B)), expressions of CDK1, cyclin A, and cyclin B in HEI-OC1 cells of the DDP + oe-Atox1 group were found to be lower than in the DDP + oe-NC group. However, expressions of CDK2 and cyclin D1 of the DDP + oe-Atox1 group were higher than in the DDP + oe-NC group (both P < .05). These results suggest that regulation of Atox1 ameliorated the stasis effect on the HEI-OC1 cell cycle. Regulating Atox1 reduced HEI-OC1 cell cycle arrest. With the Atox1 expression changes, corresponding changes were observed. These changes included (A) the HEI-OC1 cell cycle under flow cytometry; (B) expression levels of CDK2, cyclin D1, CDK1, cyclin A, and cyclin B using western blot. *P < .05 vs control. #P < .05 vs DDP. & P < .05 vs DDP + oe-Atox1.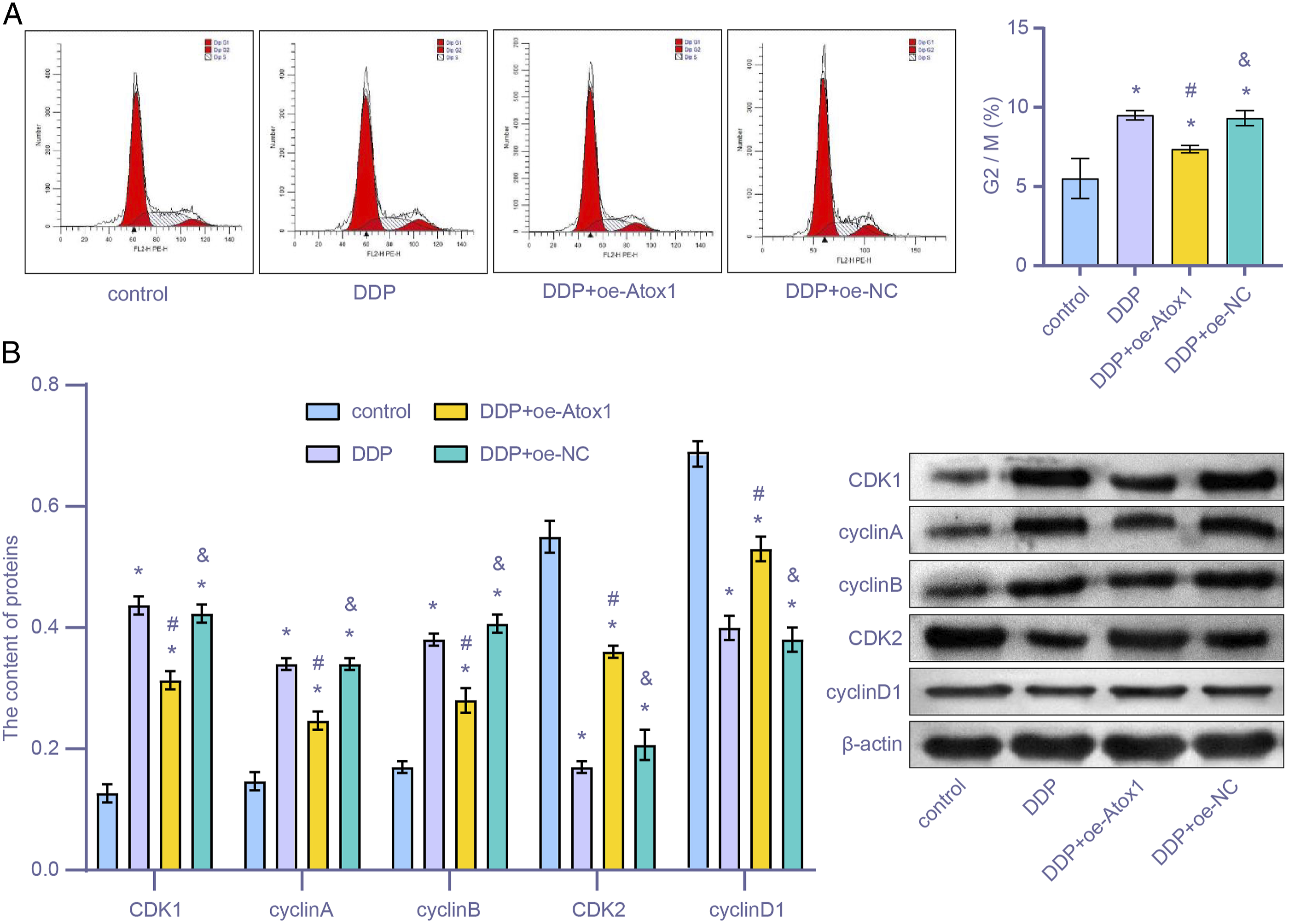
Cisplatin Toxicity Damaged the Cochlea of Rats
Based on the cell experiment results, we proceeded to confirm these results in SD rats. After the round window niche of cochlea was treated with .5, 1, and 1.5 mg/mL DDP for three days, immunofluorescence was performed to evaluate the extent of DDP damage to the cochlea. After three days, significant damage was observed in all hair cells treated with DDP (Figure 4(A)). The level of hair cells (myosin VI, red) and actin (phalloidin, green) gradually decreased (P < .05) with increasing DDP concentrations (Figure 4(A)). Similarly, increasing DDP concentrations (Figure 4(B)) reduced the expression levels of Atox1 and CTR1 and increased the expression levels of ATP7A and ATP7B in rats (both P < .05). As shown in Figure 4(C), the levels of cyclin D1 and SOD3 protein decreased in a dose-dependent manner (P < .05). In summary, DDP induced toxicity and altered protein expression, indicating cochlear damage. DDP toxicity damaged the cochlea of rats. (A) Immunofluorescence (IF) microscopy was used after treatment with .5, 1, and 1.5 mg/mL DDP. Scale bar = 50 μm; (B) The expression levels of Atox1, CTR1, ATP7A, and ATP7B were measured by qRT-PCR at increasing DDP concentrations; (C) levels of cyclin D1 and SOD3 were measured using a western blot assay. *P < .05 vs 0 mg/mL; #P < .05 vs .5 mg/mL; & P < .05 vs 1.0 mg/mL.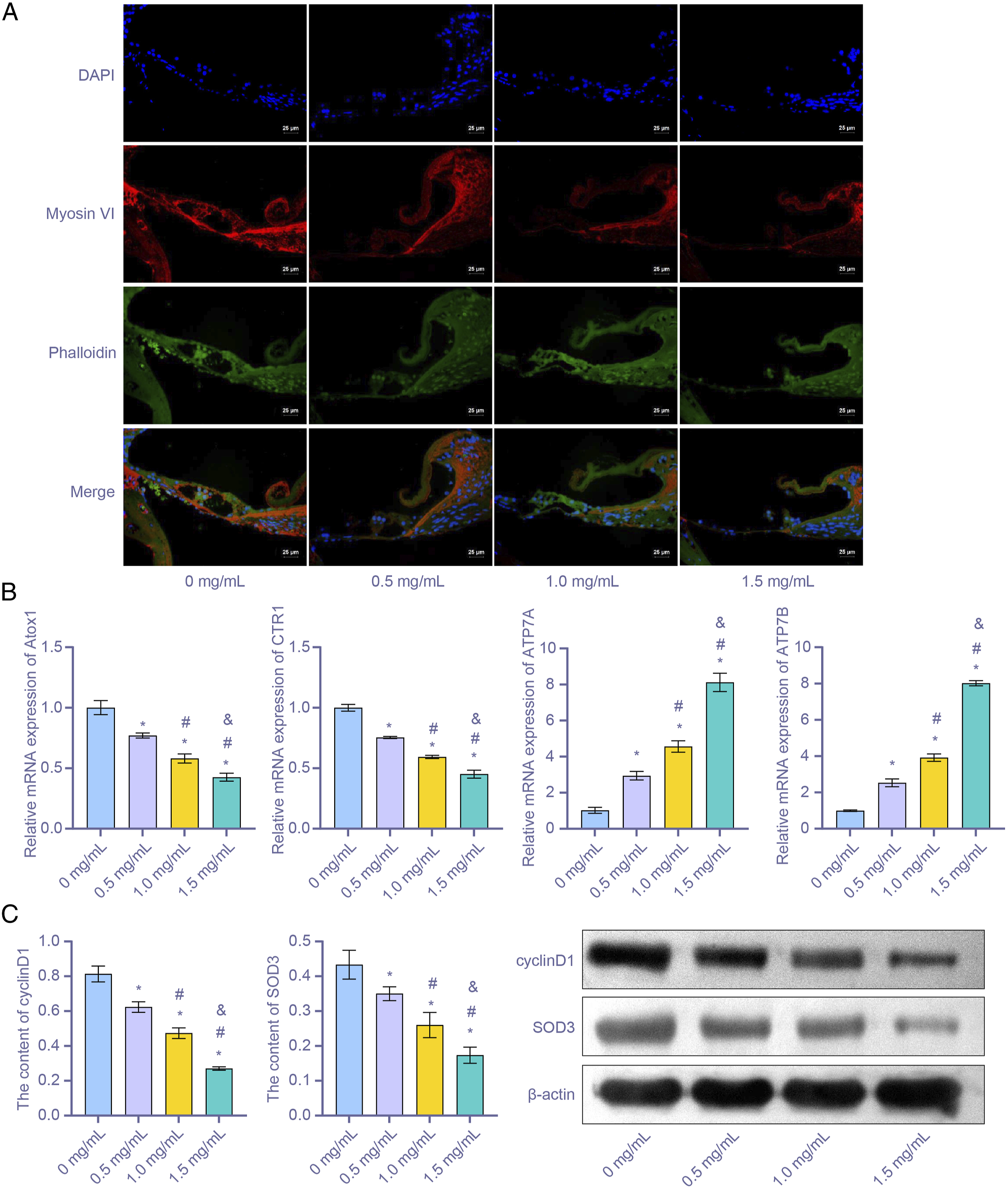
Transfection of Overexpression Antioxidant 1 Copper Chaperone Lentivirus Ameliorated Cochlea Injury Induced by Cisplatin
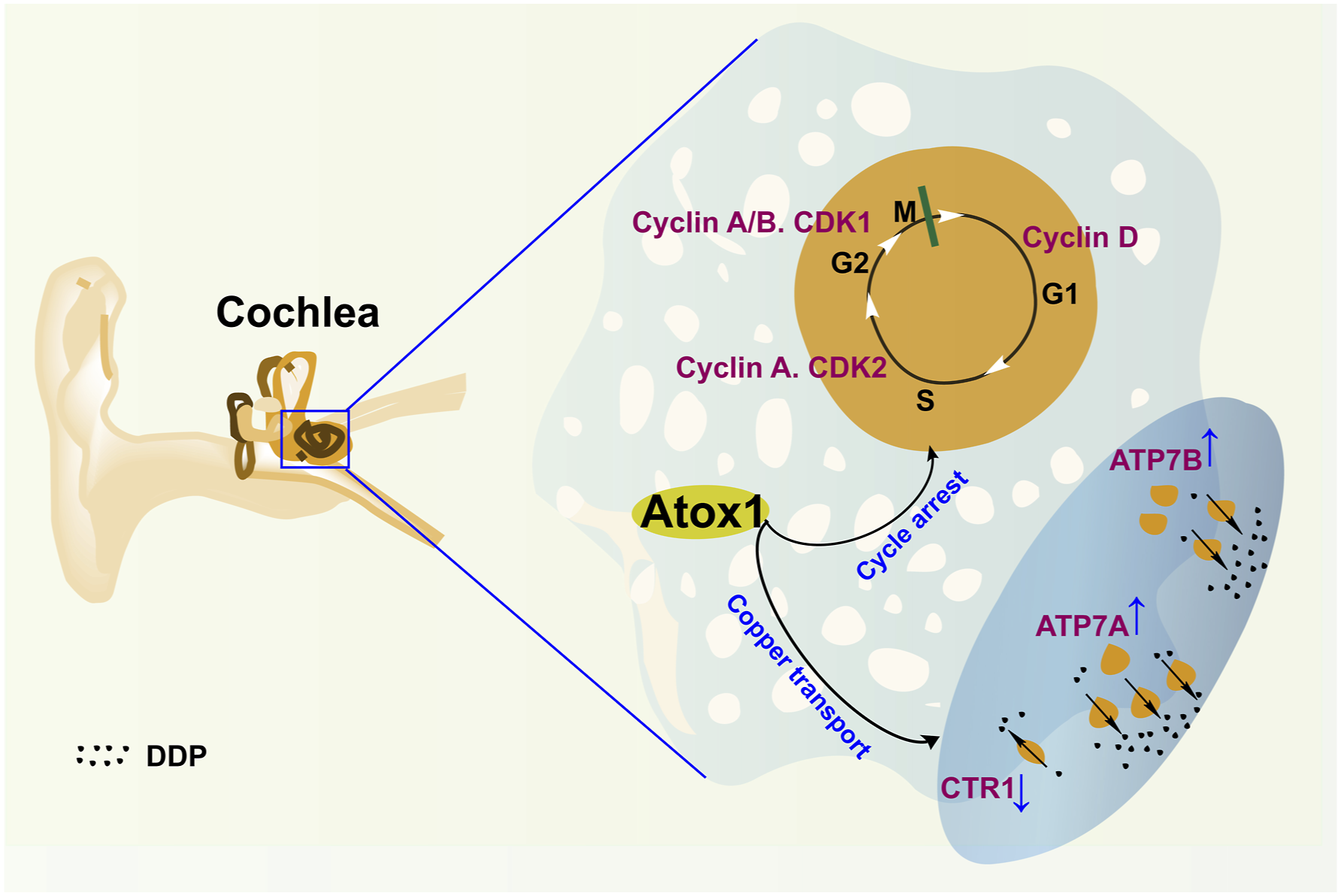
Finally, we investigated the role of overexpressed Atox1 in suppressing DDP injury. The rats' cochlea was treated with Atox1-overexpressed lentivirus preparation and treated with DDP to observe the damage to the cochlea. As shown in Figure 5(A), the extent of DDP damage to the cochlea in the DDP + oe-Atox1 group was higher than that in the DDP + oe-NC group (P < .05). When measuring gene and protein expression in the cochlea (Figure 5(B) and 5(C)), the levels of Atox1, CTR1, and SOD3 in the DDP + oe-Atox1 group were found to be higher than in the DDP + oe-NC group. The expression levels of ATP7A and ATP7B were lower than in the DDP + oe-NC group (P < .05). After observing the apoptosis indexes of cochlear cells, the levels of Bax and Caspase-9 in the DDP + oe-Atox1 group were lower than in the DDP + oe-NC group (both P < .05). The opposite effect was observed in the Bcl-2 levels. The western blot assay (Figure 5(D)) showed that the expression levels of CDK1, cyclin A, and cyclin B in the DDP + oe-Atox1 group were lower than in the DDP + oe-NC group (both P < .05). The opposite effect was observed in CDK2 and cyclin D1 levels. These results suggest that upregulating Atox1 could reduce the cochlea injury induced by DDP. The mechanism diagram is shown in Figure 6. Transfection of overexpression Atox1 lentivirus ameliorated cochlea injury induced by DDP. (A) IF images following DDP treatment or transfection of Atox1 associated lentivirus (scale bar = 50 μm); (B) Levels of Atox1, ATP7A, and ATP7B, SOD3, CTR1, Bax, Caspase-9, and Bcl-2 were detected by qRT-PCR; (C) Levels of Atox1, ATP7A, ATP7B, and SOD3 detected by western blot; (D) Levels of CDK2, cyclin D1, CDK1, cyclin A, and cyclin B detected by western blot. *P < .05 vs control. #P < .05 vs DDP. & P < .05 vs DDP + oe-Atox1. Atox1 protected the cochlea from DDP by regulating the copper transport family and cell cycle.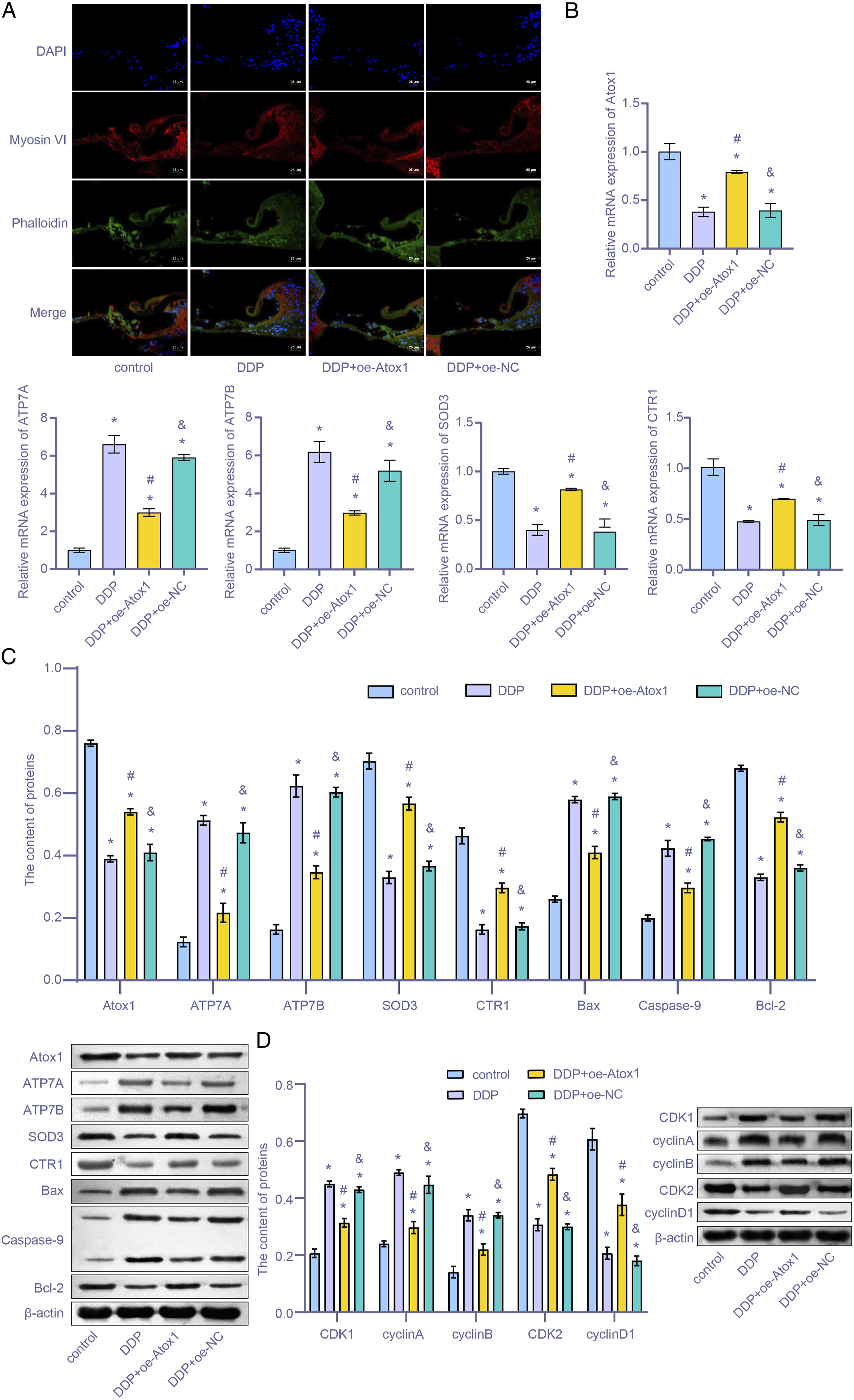
Discussion
Ototoxicity, a common side effect of DDP treatment, can limit its use and dosage. This study aimed to investigate the damage caused by DDP to rat cochlear cells both in vitro and in vivo, as well as the role of DDP and Atox1 on cochlear damage. The results showed that the damage to rat cochlea hair cells caused by DDP is worsened in a dose-dependent manner. Additionally, DDP exerted pro-apoptotic and inhibitory effects on the cochlea cell cycle. By regulating members of the Cu transport family, Atox1 ameliorates cochlea cell damage and cycle arrest. Altogether, these findings highlight the potential utility of Atox1 as a protective agent for DDP-induced ototoxicity.
Previous studies have reported that DDP significantly affects tumor growth, but almost all treated patients experience varying degrees of ear injury. 42 Despite emerging clinical trial data, there are currently no effective protective or therapeutic strategies for DDP-induced ototoxicity. 43 In preventive studies of DDP ototoxicity, some researchers have investigated the effect of administration of vitamin E 44 and α-lipoic acid. 45 However, there are limited studies aiming at reducing ototoxicity by regulating members of the Cu transport family. Xie et al 46 indicated DDP homeostasis in the body could be maintained by Cu transporters. Among them, CTR1 proteins were demonstrated to control DDP uptake, while CTR2, ATP7A, and ATP7B proteins regulated DDP efflux.17,22,47-49 Sudhahar et al and Arnesano et al indicated that Atox1 is a Cu transport chaperone. Atox1 was involved in the cell transport of various Cu proteins and regulated the cell cycle.50,51 Ozumi K et al and Venzal et al indicated that Atox1 also upregulated the transcription of SOD3 gene, suppressed oxidative stress, and reduced the occurrence of apoptosis.52,53 Therefore, we tried to regulate Cu transport through Atox1 to influence DDP-induced rats cochlear damage. The rat cochlear model is damaged by DDP in a dose-dependent manner. The transfection of oe-Atox1 lentivirus in rats can ameliorate the ear damage caused by DDP through members of the Cu transport family. In addition, the expression levels of Atox1, CTR1, and SOD3 were higher after Atox1 was overexpressed. This is consistent with previous research reference. Thus, it is feasible to reduce ototoxicity through Atox1 regulation of Cu transporter family.
Apart from reducing the toxicity of DDP via the Cu transport pathway, regulating the cell cycle could be another potential approach. Rao et al 54 indicated that DDP binds to DNA in the nucleus to regulate apoptosis or to proteins to induce cell dysregulation and death. Matson et al indicated that loss of Atox1 reduced DNA levels of the DDP-exposed nucleus and prolonged the duration of the G2/M phase, leading to G2/M enrichment. 28 We found HEI-OC1 cells were arrested in the G2/M stage after DDP treatment, with apoptosis rate increasing. The over-expression of Atox1 relieves DDP damage and shortens the duration of the G2/M phase. Importantly, the in vitro and in vivo results are consistent with each other. Thus, the regulation of Atox1 relieved cell cycle arrest caused by DDP.
The results of the current study suggest that Cu chaperone Atox1 protects the cochlea from DDP by regulating members of the Cu transport family and cell cycle. These findings could be used to identify novel molecular targets and set a theoretical basis for the prevention and treatment of DDP cochlear damage. However, the specific molecular mechanisms warrant further investigation.
Conclusion
In conclusion, DDP damaged the hair cells in a rat cochlea model in a dose-dependent manner. DDP exhibited pro-apoptotic effects and altered the cell cycle of HEI-OC1 cells. Atox1 ameliorated HEI-OC1 cell damage and cycle arrest by regulating members of the Cu transport family. In vivo, DDP damage was ameliorated by transfection of overexpressed Atox1 lentivirus and regulated the Cu transport family and cell cycle.
Footnotes
Author Contributions
Xubo Chen contributed to conception and design, contributed to interpretation, drafted the manuscript, and critically revised the manuscript; Weiren Xiang contributed to conception, contributed to acquisition, drafted the manuscript, and critically revised the manuscript; Lihua Li contributed to conception, contributed to analysis, drafted the manuscript, and critically revised the manuscript; Kai Xu contributed to conception, contributed to analysis, drafted the manuscript, and critically revised the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Jiangxi Province Key Research and Development Program (20171BBG70007), and the National Natural Science Foundation of China (81700919, 82260222).
