Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) reviewed the safety of M piperita (peppermint)–derived ingredients. The Panel reviewed data relevant to the safety of these ingredients. Because final product formulations may contain multiple botanicals, each containing the same constituent(s) of concern, formulators are advised to be aware of these constituents and avoid reaching levels that may be hazardous to consumers. Industry should continue to use good manufacturing practices to limit impurities that could be present in botanical ingredients. The Panel concluded that M piperita (Peppermint) Oil, Extract, Leaf, and leaf-derived ingredients are safe in cosmetics in the present practices of use and concentration when formulated to be non-sensitizing, and that the available data are insufficient for determining that M piperita (Peppermint) Flower/Leaf/Stem Extract, M piperita (Peppermint) Flower/Leaf/Stem Water, and M piperita (Peppermint) Meristem Cell Culture are safe under the intended conditions of use in cosmetic formulations.
Keywords
Introduction
The safety of four of the cosmetic ingredients named in this safety assessment has been previously reviewed by the Panel; in 1998, the Panel issued a final report (published in 2001) with a conclusion stating that Mentha Piperita (Peppermint) Oil, Mentha Piperita (Peppermint) Leaf Extract, Mentha Piperita (Peppermint) Leaf, and Mentha Piperita (Peppermint) Leaf Water are safe as used in cosmetic formulations. 1 The conclusion also stated that the concentration of pulegone, a constituent of these botanical ingredients, should not exceed 1%. In accordance with the Cosmetic Ingredient Review (CIR) Procedures, the Panel evaluates the conclusions of previously issued reports every 15 yr, and therefore a re-review was initiated. The new conclusion reached in this re-review supersedes the original conclusion. According to the web-based International Cosmetic Ingredient Dictionary and Handbook (wINCI; Dictionary), most of these ingredients are reported to function as fragrance ingredients and/or skin conditioning agents in cosmetic products. 2
In addition to the four M piperita (peppermint)–derived ingredients that are mentioned above, this re-review included 6 related, previously unreviewed ingredients. The complete list of ingredients included in this assessment is: Mentha Piperita (Peppermint) Extract Mentha Piperita (Peppermint) Flower/Leaf/Stem Extract Mentha Piperita (Peppermint) Flower/Leaf/Stem Water
Mentha Piperita (Peppermint) Leaf
Mentha Piperita (Peppermint) Leaf Cell Extract
Mentha Piperita (Peppermint) Leaf Extract
Mentha Piperita (Peppermint) Leaf Juice
Mentha Piperita (Peppermint) Leaf Water
Mentha Piperita (Peppermint) Meristem Cell Culture
Mentha Piperita (Peppermint) Oil
*Previously reviewed ingredients are indicated in italics.
The cosmetic ingredient names, according to the Dictionary, are written as above, that is, capitalized, without italics, and unabbreviated. When referring to the plant from which these ingredients are derived, the traditional taxonomic nomenclature practice of using italics to identify genus and species will be followed (e.g., M piperita).
This safety assessment includes relevant published and unpublished data for each endpoint that is evaluated. Published data are identified by conducting an exhaustive search of the world’s literature. A list of the typical search engines and websites used, sources explored, and endpoints that Panel evaluates is available on the CIR website (https://www.cir-safety.org/supplementaldoc/preliminary-search-engines-and-websites; https://www.cir-safety.org/supplementaldoc/cir-report-format-outline). Unpublished data are provided by the cosmetics industry, as well as by other interested parties.
Toxicity Data on Components of M piperita (Peppermint)–Derived Ingredients.

Excerpts from the 2001 safety assessment on the previously reviewed ingredients are disseminated throughout the text of this re-review document, as appropriate, and are identified by italicized text. For complete and detailed information, please refer to the original report, which is available on the CIR website (https://www.cir-safety.org/ingredients).
Chemistry
Definition and General Characterization
Definitions and Functions of the Ingredients in This Safety Assessment. 2

Chemical and Physical Properties
Method of Manufacture
M piperita (Peppermint) Oil
European and American peppermint oil is distilled with steam from the fresh, above-ground parts of the flowering plant M piperita Linne, rectified by distillation and not dementholized. 1 It has been reported that the menthone content decreases while the menthol content increases in peppermint leaves upon storage for 1 to 2 months, at 22 to 24°C. However, the relative menthone to menthol proportion remained practically constant during the total storage time.
According to one source, Mentha Piperita (Peppermint) Oil has been extracted (distilled; water solvent) from the leaves of M piperita harvested (first in July and second harvest in September) in Washington state. 8
M piperita (Peppermint) Extract
According to one source, the main steps in the process of manufacturing a trade name mixture defined as an aqueous solution containing 7.5% Mentha Piperita (Peppermint) Extract are: solubilization of M piperita in water, separation of soluble and insoluble phases, and filtration and sterilizing filtration. 9
M Piperita (Peppermint) Leaf Extract
The following method relates to preparation of the butylene glycol/water extract of Mentha Piperita (Peppermint) Leaf Extract. 10 Dried raw material is extracted with 50 vol% 1,3-butylene glycolic solution. After extraction, the additional steps in the production process include: filtrate → sedimentation → filtrate → adjustment → packaging.
In another method, the preparation of a water/ethanol extract is described.
11
Dried raw material is extracted with 30 vol% ethanol solution. After extraction, the additional steps in the production process include: filtrate
The production method described herein relates to preparation of Mentha Piperita (Peppermint) Leaf Extract (powder form).
10
According to the method of production, dried raw material is extracted with 30 vol% ethanol solution. After extraction, the additional steps in the production process include: filtrate
In the supercritical fluid extraction with natural carbon dioxide production method for Mentha Piperita (Peppermint) Leaf Extract from the dried leaves of M piperita, neither additives nor other technical adjuncts are introduced during the production process. 12
M Piperita (Peppermint) Leaf Water
In the preparation of Mentha Piperita (Peppermint) Leaf Water, dried raw material is subjected to steam distillation.
10
After distillation, the remaining steps in the production process are: water soluble fraction obtained
Composition and Impurities
Pulegone is found in young peppermint leaves, and is metabolized to menthol as the leaves mature. It has also been reported that pulegone is found only in Mentha Piperita (Peppermint) Oil from young plants and in trace amounts in “inferior” oils; pulegone is absent from “good quality” Mentha Piperita (Peppermint) Oil. 1 However, a supplier of Mentha Piperita (Peppermint) Oil reported pulegone concentrations of 1 to 4%, depending on the origin of the oil. Published studies that have investigated the pulegone content of Mentha Piperita (Peppermint) Oil also reported a range of <1 to 4% for Mentha Piperita (Peppermint) Oils of a North American origin.
M Piperita (Peppermint) Oil
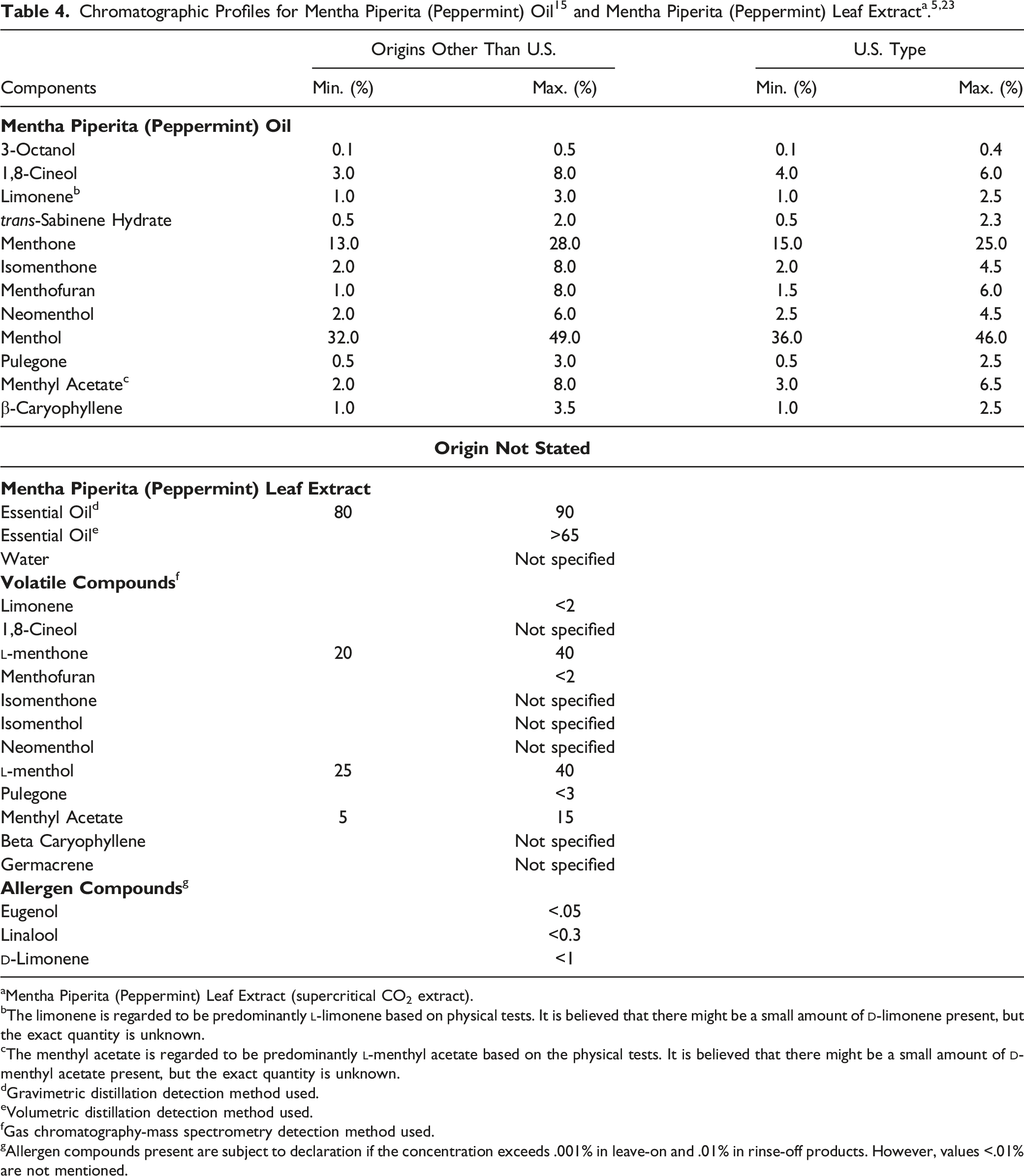
The major constituents of Mentha Piperita (Peppermint) Oil include the terpenes: (−)-menthol (30%–55%), (−)-menthone (14%–32%), (+)-isomenthone (1.5%–10%), (−)-menthyl acetate (2.8%–10%), (+)-menthofuran (1.0%–9.0%), and 1,8-cineol (3.5%–14%). 13
Certain trends were observed between oil extracted from first and second harvest leaves, and oil extracted from fresh leaves vs dried leaves.
8
When compared to the second harvest, oils from the first harvest were generally higher in (Z)-3-hexenol, 1,8-cineol, α-pinene, β-pinene, sabinene hydrate, isomenthone, menthofuran, pulegone, β-caryophyllene, and germacrene
Major components of the essential oil of M piperita adult plants from Poland include menthone, menthol, menthyl acetate, carvone, piperitone, 1,8-cineol, and pulegone. 14
According to the United States Pharmacopeial Convention’s (USP) Food Ingredients Expert Committee, the acceptance criteria for Mentha Piperita (Peppermint) Oil include not less than 5% total esters (calculated as menthyl acetate) and not less than 50% menthol. 7
aMentha Piperita (Peppermint) Leaf Extract (supercritical CO2 extract).
bThe limonene is regarded to be predominantly
cThe menthyl acetate is regarded to be predominantly
dGravimetric distillation detection method used.
eVolumetric distillation detection method used.
fGas chromatography-mass spectrometry detection method used.
gAllergen compounds present are subject to declaration if the concentration exceeds .001% in leave-on and .01% in rinse-off products. However, values <.01% are not mentioned.
M Piperita (Peppermint) Extract
Mentha Piperita (Peppermint) Extract (combined in a trade name mixture) is an aqueous solution composed of 7.5% (maximum percentage) Mentha Piperita (Peppermint) Extract, with <40 ppm pulegone and <50 ppm menthol. 9 The following statement relating to composition was also provided: “Our active can be divided in sugars (47%), mineral ashes (38%), proteins (13%), and polyphenols (2%).” Composition data on this trade name mixture also include the following impurities: alkaloids (<.05 g/L; assay of alkaloids performed with Dragendorff reagent), copper (.23 ppm), iron (3.76 ppm), manganese (21 ppm), nickel (.19 ppm), and zinc (3.14 ppm). An assay of allergens was performed to characterize and quantify 26 allergen compounds in this trade name mixture in order to comply with the requirements of European Regulation 12234/2009. Allergens were not detected. Thus, the concentrations of the 26 allergens were less than the sensitivity of the method (<1 ppm).
The following information relating to impurities is included in a certificate of analysis for a trade name mixture containing 2.5% Mentha Piperita (Peppermint) Extract: lead (<10 ppm), arsenic (<3 ppm), mercury (<1 ppm), and pesticide residues (meets USP specification). 6
M Piperita (Peppermint) Leaf
The major monoterpene constituents of Mentha Piperita (Peppermint) Leaf are: (−)-limonene; 1,8-cineol; (+)-pulegone; (−)-menthone; (+)-isomenthone; (+)-menthofuran; (−)-menthol; and (+)-neomenthol.
17
Mentha Piperita (Peppermint) Leaf also contains caffeic acid, rosmarinic acid, and the following flavonoids: apigenin-, diosmetin-, and luteolin-glycosides, and free lipophile methoxylized flavones such as xanthomicrol and gardenine
The following elemental contaminants have been detected in M piperita herbal tea (tea leaves) samples (n = 3) from Serbia: manganese (111.97 mg/kg dry weight), iron (443.90 mg/kg), copper (17.15 mg/kg), and zinc (26.86 mg/kg), molybdenum (2.695 mg/kg), cobalt (.161 mg/kg), nickel (1.882 mg/kg), selenium (.107 mg/kg), aluminum (554 mg/kg), and tin (3.66 mg/kg). 19
It is possible that pesticide residues may be present as impurities in the leaves of M piperita. In a study in which Mentha Piperita (Peppermint) Leaves were soaked in pesticides, the dissipation rate of pesticide residues during the drying process was said to have been satisfactory, except for the pirimiphos-ethyl pesticide, because of its high octanol-water partition coefficient and low vapor pressure. 20
M piperita (Peppermint) Leaf Extract
An analysis of Mentha Piperita (Peppermint) Leaf Extract indicated that the leaves principally contained cinnamic acid, caffeic acid, rosmarinic acid, and various flavonoids (flavones and flavanones). 21 The following solvents were used to extract the peppermint leaves: light petroleum, dichloromethane, acetonitrile, ethyl acetate, methanol, n-butanol, and water. Eriocitrin (383.3 ± 2.2 mg/g extract) and rosmarinic acid (381.2 ± 1.9 mg/g extract) were the most abundant components identified within the leaves, while naringenin-7-O-glucoside (.8 ± .01 mg/g extract) was the least abundant component identified. Kynurenic acid (3.82 ± .46 μg/g) has also been detected in Mentha Piperita (Peppermint) Leaf Extract. 22 It should be noted that kynurenic acid is a constituent of human synovial fluid.
Composition data provided by the Council indicate that Mentha Piperita (Peppermint) Leaf Extract (butylene glycol/water extract) contains tannin and terpenoid (which contains 2.8 ppm pulegone) and that Mentha Piperita (Peppermint) Leaf Extract (water/ethanol extract) contains essential oil, tannin, and terpenoid. 10
Data on impurities provided by the Council indicate that Mentha Piperita (Peppermint) Leaf Extract (butylene glycol/water extract) contains not more than 10 ppm heavy metals and not more than 2 ppm arsenic. 10 Mentha Piperita (Peppermint) Leaf Extract (water/ethanol extract) contains not more than 10 ppm heavy metals and not more than 1 ppm arsenic.
The supercritical fluid extraction with natural carbon dioxide production method for M piperita (Peppermint) Leaf Extract from the dried leaves of M piperita results in no solvent residues, no inorganic salts, no heavy metals, and no reproducible microorganisms. 12
According to data provided by the Council, M piperita (peppermint) leaf extract powder contains not more than 10 ppm heavy metals and not more than 2 ppm arsenic. 10
A chromatographic profile for Mentha Piperita (Peppermint) Leaf Extract (supercritical CO2 extract) is presented in Table 4.5,23
M Piperita (Peppermint) Leaf Water
Composition data provided by the Council indicate that Mentha Piperita (Peppermint) Leaf Water contains essential oil (menthol). 10 The data also included specifications that indicate that Mentha Piperita (Peppermint) Leaf Water contains not more than 10 ppm heavy metals and not more than 1 ppm arsenic. 10
Use
Cosmetic
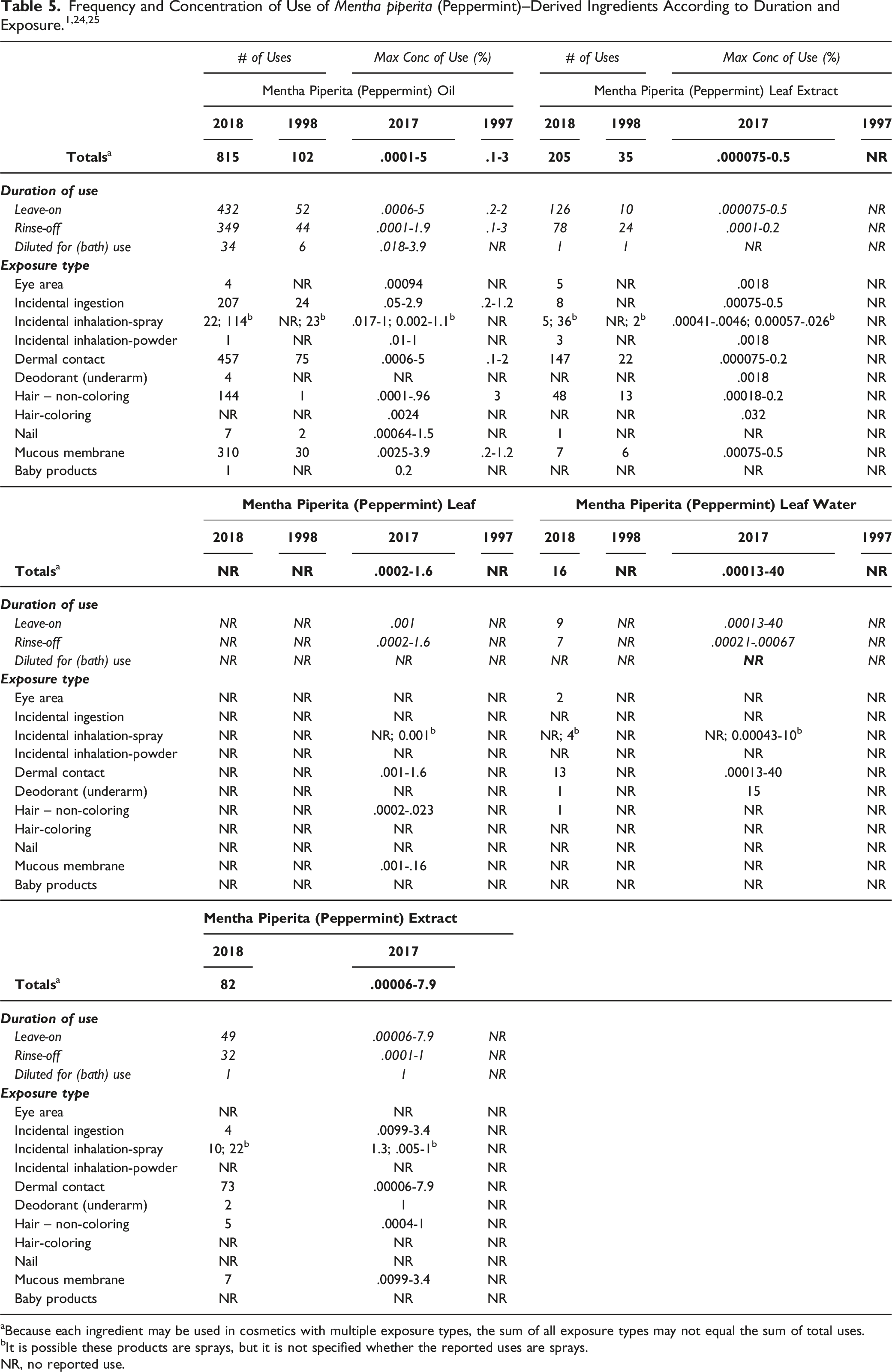
The safety of M piperita (peppermint)–derived ingredients is evaluated based on data received from the US Food and Drug Administration (FDA) and the cosmetics industry on the expected use of these ingredients in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in FDA’s Voluntary Cosmetic Registration Program (VCRP) database. 24 Use concentration data are submitted by the cosmetics industry in response to surveys, conducted by the Council, of maximum reported use concentrations by product category. 25
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
bIt is possible these products are sprays, but it is not specified whether the reported uses are sprays.
NR, no reported use.
According to VCRP and Council survey data, the following M piperita (peppermint)–derived ingredients are not being used in cosmetic products: M piperita (Peppermint) Flower/Leaf/Stem Water, M piperita (Peppermint) Flower/Leaf/Stem Extract, M piperita (Peppermint) Leaf Cell Extract, M piperita (Peppermint) Leaf Juice, and M piperita (Peppermint) Meristem Cell Culture.
Cosmetic products containing M piperita (peppermint)–derived ingredients may be applied to the skin and hair or, incidentally, may come in contact with the eyes (at maximum use concentrations up to .0018% M piperita (Peppermint) Leaf Extract in eye lotions) and mucous membranes (at maximum use concentrations up to 3.9% M piperita (Peppermint) Oil in bath oils, tablets, and salts). Additionally, use in lipstick products (at maximum use concentrations up to 2.9% M piperita (Peppermint) Oil) is being reported, the application of which may result in incidental ingestion. Products containing M piperita (peppermint)–derived ingredients may be applied as frequently as several times per day and may come in contact with the skin or hair for variable periods following application. Daily or occasional use may extend over many years.
M piperita (Peppermint) Oil is being used in both pump hair sprays (maximum use concentrations up to .02%) and aerosol hair sprays (maximum use concentrations up to .017%) which may result in incidental inhalation exposure. Additionally, use of this ingredient in foot sprays at maximum use concentrations up to .5% is being reported. M piperita (Peppermint) Leaf Extract is also being used in pump and aerosol hair sprays, but at lower maximum use concentrations, and in face and neck/body and hand spray products at maximum use concentrations up to .001%. M piperita (Peppermint) Extract is being used in face and neck sprays at a highest maximum use concentration of 1.3%. In practice, most droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 μm, with propellant sprays yielding a greater fraction of droplets/particles below 10 μm, compared with pump sprays.26-29 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (i.e., they would not enter the lungs) to any appreciable amount.26,27
M piperita (Peppermint) Oil is being used in foot powders at maximum use concentrations up to 1%, and M piperita (Peppermint) Leaf Extract is being used in face powders at maximum use concentrations up to .0018%. Conservative estimates of inhalation exposures to respirable particles during the use of loose powder cosmetic products are 400-fold to 1000-fold less than protective regulatory and guidance limits for inert airborne respirable particles in the workplace.30-32
Noncosmetic
Mentha Piperita (Peppermint) Oil is a generally recognized as safe (GRAS) ingredient according to the FDA for use in dietary supplements. 1 It is described as a naturally occurring carminative that relaxes gastrointestinal smooth muscle. A final ruling by the FDA labeled Mentha Piperita (Peppermint) Oil as safe and effective as an antitussive (topical/inhalant). Final rulings cautioned that Mentha Piperita (Peppermint) Oil is not safe and effective for use as an expectorant in either topical/inhalant or lozenge form, or for use as a nasal decongestant, mouthwash, or digestive aid.
M piperita (Peppermint) Oil, M piperita (Peppermint) Extract, M piperita (Peppermint) Flower/Leaf/Stem Extract, and Mentha Piperita (Peppermint) Leaf Juice are generally recognized as safe (GRAS) by the US FDA for use in food for human consumption (21CFR182.20). M piperita (Peppermint) Oil is an inactive ingredient in drug products that have been approved by the U.S. FDA. 33 A number of active ingredients, M piperita (Peppermint) Oil included, have been present in over-the-counter (OTC) drug products for various uses. 34 However, the FDA has determined that, based on evidence currently available, there are inadequate data to establish general recognition of the safety and effectiveness of Mentha Piperita (Peppermint) Oil as an active ingredient in the following drug products: nasal decongestant drug products, digestive aid drug products, insect bite and sting drug products, and astringent drug products.
M piperita (Peppermint) Oil is on the U.S. Environmental Protection Agency (EPA) list of active ingredients eligible for minimum risk pesticide products. 35
According to the European Medicines Agency Committee on Herbal Medicinal Products (HMPC) community herbal monograph on Mentha x piperita L., aetheroleum (i.e., peppermint oil; aetheroleum is a term used to describe a preparation made from the above-ground parts of a plant), this herbal medicine is administered orally for the symptomatic relief of minor spasms of the gastrointestinal tract, flatulence, and abdominal pain, especially in patients with irritable bowel syndrome. 36 It is also an herbal medicine that is administered cutaneously for the symptomatic relief of mild tension-type headache. These uses have been identified as well-established uses by the HMPC. The highest recommended daily dose in the European Union is 1.2 mL peppermint oil (i.e., 1,080 mg peppermint oil, which contains maximum 140 mg pulegone + menthofuran). For a 60 kg person, this would correspond to a daily intake of 2.3 mg/kg body weight.
Toxicokinetic Studies
Dermal Penetration
M piperita (Peppermint) Oil
Eserine in a Mentha Piperita (Peppermint) Oil vehicle was applied to a 2.2 cm2 shaved area on the abdomen of mice. The absorption rate for Mentha Piperita (Peppermint) Oil was measured as the latent period between application and appearance of eserine-induced signs of cholinergic excess. 1 Mentha Piperita (Peppermint) Oil had a latent period of 58 min.
Penetration Enhancement
M piperita (Peppermint) Leaf Extract
The skin penetration enhancement potential of Mentha Piperita (Peppermint) Leaf Extract (aqueous ethanol extract) was evaluated using dorsal porcine skin (dermatomed to thickness of 500 μm). 37 A square section of skin was cut to provide a dose area of 1 cm2 and placed in a flow-through diffusion cell. [14C]Caffeine (hydrophilic) or [14C]salicylic acid (hydrophobic) was applied topically with 10% Mentha Piperita (Peppermint) Leaf Extract to porcine skin. The receptor fluid for the diffusion cell was “a Krebs-Ringer bicarbonate buffer spiked with dextrose and bovine serum albumin (BSA).” Ethanol alone served as the control. When compared to [14C]caffeine in the presence of ethanol (control), the dermal absorption of [14C]caffeine was significantly greater (P > .05; flux and permeability of caffeine increased by over 3-fold) in the presence of Mentha Piperita (Peppermint) Leaf Extract. However, this was not true for [14C]salicylic acid.
Penetration Inhibition
M piperita (Peppermint) Oil
Mentha Piperita (Peppermint) Oil and ring-UL-[14C]benzoic acid were applied to full-thickness human skin (breast or abdominal) samples in a static diffusion cell. 38 The receptor fluid for the diffusion cell was “0.9% sodium chloride and 1% Tween in water.” As the concentration of M piperita (Peppermint) Oil increased from zero to 5% in the donor phase, the maximal flux of benzoic acid decreased. The differences were significant at 1.0% and 5.0% M piperita (Peppermint) Oil, where the maximal fluxes were reduced to 81% and 52% of the control, respectively.
Absorption, Distribution, Metabolism, and Excretion
Oral
M piperita (Peppermint) Oil
The rate of Mentha Piperita (Peppermint) Oil absorption and excretion following oral administration was determined by measuring urinary menthol glucuronide.
1
Four male volunteers ingested 180 mg of an enteric-coated Mentha Piperita (Peppermint) Oil-coated capsule following a 16-h fast. Menthol was liberated from its glucuronide metabolite by treating the urine with β-
Mentha Piperita (Peppermint) Oil is relatively rapidly absorbed after oral administration and eliminated mainly via the bile.
13
The major biliary metabolite is menthol glucuronide, which undergoes enterohepatic circulation. The urinary metabolites result from hydroxylation at the C-7 methyl group at C-8 and C-9 of the isopropyl moiety, forming a series of mono- and dihydroxymenthols and carboxylic acids, some of which are excreted, in part, as glucuronic acid conjugates. Studies with tritiated
Toxicological studies
Acute Toxicity Studies
Oral
M piperita (Peppermint) Oil
Mentha Piperita (Peppermint) Oil had a 24-h oral LD50 of 4441 mg/kg in fasted Wistar rats; the 48-h LD50 was 2426 mg/kg. 1 In a study involving fasted mice, an LD50 of 2410 mg/kg was reported for Mentha Piperita (Peppermint) Oil diluted in olive oil.
Short-Term Toxicity Studies
Oral
M piperita (Peppermint) Oil
In 3 of 4 short-term oral toxicity studies (28-d or 5-wk studies) involving 20 to 28 rats per group, brain lesions (specifically, cyst-like spaces in the cerebellum) were observed at Mentha Piperita (Peppermint) Oil doses up to 100 mg/kg/d. 1 In the remaining study (12 rats per group), these lesions were not observed in rats dosed with M piperita (Peppermint) Oil at doses of 20, 150, or 500 mg/kg/d for 5 wk.
Subchronic Toxicity Studies
Oral
M piperita (Peppermint) Oil
Groups of 28 Wistar rats were given oral doses of 10, 40, and 100 mg/kg M piperita (Peppermint) Oil (diluted with soybean oil) daily for 90 d. 1 All hematological and biochemical parameters were within normal range, and there were no significant differences in absolute and relative organ weights. Brain lesions (specifically, cyst-like spaces in the cerebellum) were observed in all dose groups, but these results were classified as significant only for animals of the 100 mg/kg/d dose group. No other lesions of encephalopathy were observed. Nephropathy (hyaline droplet formation) was observed only in male rats of the 100 mg/kg/d dose group, and there was no evidence of epithelial degeneration. The no-observed-adverse-effect level (NOAEL) for Mentha Piperita (Peppermint) Oil was 40 mg/kg/d in this study.
Genotoxicity Studies
In Vitro
M piperita (Peppermint) Oil
The mutagenic potential of Mentha Piperita (Peppermint) Oil was investigated using the Salmonella/mammalian microsome test. 1 The following Salmonella typhimurium strains were used: TA1535, TA100, TA1537, and TA98. The sample tested contained 38.1% menthol, 33.7% menthone, and 1.7% pulegone; the remaining components were not identified. Mentha Piperita (Peppermint) Oil, tested at doses of 6.4, 32, and 160 μg/plate, produced the same number of revertants as the negative control. Toxicity was noted at the next (and maximum) dose of 800 μg/plate. Metabolic activation appeared to have made the oil less toxic to the bacteria. Mentha Piperita (Peppermint) Oil was not genotoxic.
In an in vitro chromosomal aberration test using a Chinese hamster fibroblast cell line, Mentha Piperita (Peppermint) Oil, at a maximum concentration of .25 mg/mL (in ethanol), produced polyploidism in 3% of the cells and structural aberrations in 7% of the cells at 48 h after treatment. The results were considered equivocal, as scores of either ≥10% or ≤4.9% were necessary for classification as either positive or negative, respectively. The results for Mentha Piperita (Peppermint) Oil (150 μg/ml) were negative in a mouse lymphoma L5178Y TK +/− cell mutagenesis assay. Results were also negative for this ingredient (at 155 μg/ml) in an unscheduled DNA synthesis assay using rat hepatocytes. 1
The genotoxicity of Mentha Piperita (Peppermint) Oil was evaluated in a chromosome aberration test using human peripheral blood lymphocytes. 39 Lymphocyte cultures were incubated for 24 h with test substance concentrations up to .30 μl/ml. When chromosome aberrations (chromatid breaks, chromatid exchanges, chromosome breaks, and chromosome exchanges) were scored, not less than 100 metaphases per culture were analyzed. Mentha Piperita (Peppermint) Oil was the most clastogenic at a concentration of .20 μl/ml (8-fold increase over acetone solvent control); the number of aberrant cells decreased at higher concentrations. The authors noted that the dose-response curve for M Piperita (Peppermint) Oil was complicated, with a clear peak response at a concentration of .20 μl/ml.
M piperita (Peppermint) Oil was tested at concentrations up to .30 μl/ml in the sister chromatid exchange (SCE) test involving human lymphocytes. 39 The test conditions were essentially the same as those in the preceding chromosome aberration test, with the exception that 5-bromo-2'-deoxyuridine was added (10 μg/ml) to cultures initially. To determine the replicative index, 200 cells were scored. M piperita (Peppermint) Oil induced SCEs in a dose-independent manner. The authors noted that, seemingly, the saturation of SCE-inducing capacity occurred at high concentrations of Mentha Piperita (Peppermint) Oil. Results also indicated that Mentha Piperita (Peppermint) Oil inhibited cell replicative kinetics, some signs of which were observed at a concentration of .15 μl/ml. At concentrations ≥.20 μl/ml, statistically significant inhibition of cell replicative kinetics was evident.
M piperita (Peppermint) Extract
The genotoxicity of a trade name mixture containing 2.5% Mentha Piperita (Peppermint) Extract was evaluated in the Ames test using the following Salmonella typhimurium strains, with and without metabolic activation: TA97a, TA98, TA100, TA102, and TA1535. 40 The test substance was diluted with sterile distilled water to a concentration of 10% (effective concentration of extract = .25%) prior to testing each strain. Sterile deionized water served as the solvent control and positive controls (not stated) were also used. The test substance was not cytotoxic to the test system and was not genotoxic to any of the strains tested, either with or without metabolic activation. The bacterial strains tested were sensitive to the positive control mutagens and had a spontaneous reversion rate that was well within the accepted values for each strain.
Antigenotoxicity Studies
M piperita (Peppermint) Leaf Extract
Oral pretreatment with Mentha Piperita (Peppermint) Leaf Extract (aqueous extract) (1 g/kg/d for 3 consecutive days) before exposure to gamma radiation was found to be effective in protecting against chromosomal damage in the bone marrow of Swiss albino mice (number tested not stated). 41 The exposure of mice to 8 gray (Gy) ionizing radiation (1 Gy is equivalent to the absorption of one joule of radiation energy per kilogram of matter) only resulted in chromosomal aberrations in the form of chromatid breaks, chromosome breaks, centric rings, dicentrics, exchanges, and acentric fragments. In mice pretreated with Mentha Piperita (Peppermint) Leaf Extract, there was a significant decrease in the frequency of aberrant cells when compared to the irradiated control. A significant increase in the percentage of chromatid breaks, chromosome breaks, centric rings, dicentrics, exchanges, acentric fragments, total aberrations, and aberrations/damaged cell was observed at 12 h post-irradiation necropsy time in control animals. However, a significant decrease in the percentage of aberrations of this type was observed in mice pretreated with M piperita (Peppermint) Leaf Extract.
The modulatory effects of M piperita (Peppermint) Leaf Extract (aqueous extract) on genotoxicity and lung tumor incidence were evaluated using 4 groups of 30 to 76 Swiss albino mice. 42 Beginning at 3 wk of age (weaning), the mice received a single subcutaneous injection of benzo[a]pyrene and were then dosed orally (by gavage) with either water (group of 53 mice) or M piperita (Peppermint) Leaf Extract (1 g/kg; group of 76 mice). The remaining 2 groups of mice in the study were identified as no benzo[a]pyrene or M piperita (Peppermint) Leaf Extract dosing (30 mice) and M piperita (Peppermint) Leaf Extract alone (30 mice). When compared to mice in the benzo[a]pyrene only group, M piperita (Peppermint) Leaf Extract reduced the frequency of chromosomal aberrations and micronuclei in bone marrow cells. M piperita (Peppermint) Leaf Extract had an antigenotoxic effect in this study. Results relating to the modulatory effect of M piperita (Peppermint) Leaf Extract on lung tumor formation are included in the Anticarcinogenicity section of the report text.
Carcinogenicity Studies
Oral
M piperita (Peppermint) Oil
In a carcinogenicity study of toothpaste and its components, groups of 52 male pathogen-free CFLP (ICI-redefined) mice were dosed by gavage with 4 or 16 mg M piperita (Peppermint) Oil/kg/d, 6 d/wk for 80 wk. Treatment was followed by a 16- to 24-wk observation period. An untreated group of 52 male mice and a vehicle control group of 260 male mice that received the toothpaste base (which did not contain chloroform, eucalyptol, or M piperita (Peppermint) Oil) were maintained as controls. At least one neoplasm at any site was observed in 73, 69, 65, and 71% of mice of the low-dose, high-dose, untreated control, and vehicle control groups, respectively. The incidence of neoplasms of the lungs and kidneys was comparable among mice of the treated and nontreated groups. Hepatic cell tumor incidence for M piperita (Peppermint) Oil-dosed mice (25%) was comparable to the incidence for mice of the vehicle control group (27%); the incidence for the untreated group was 19%. Malignant lymphoma was found in 25, 21, 10, and 14% of mice of the low-dose, high-dose, untreated, and vehicle control groups, respectively. The researchers did not discuss whether the differences in tumor incidence were significant. 1
Anticarcinogenicity Studies
M piperita (Peppermint) Leaf Extract
The modulatory effects of M piperita (Peppermint) Leaf Extract (aqueous extract) on genotoxicity and lung tumor incidence were evaluated using 4 groups of 30 to 76 Swiss albino mice. 42 The dosing procedure (including groups tested) was identical to that stated for Swiss mice in the second study of the Antigenotoxicity section in this report. The mice were killed at 9 wk of age and evaluated for lung tumor incidence. Dosing with M piperita (Peppermint) Leaf Extract caused a significant reduction in the number of lung adenomas from an incidence of 67.92% in mice given benzo[a]pyrene only to 26.31%, which amounted to 61.26% inhibition. Tumor multiplicity was 1.22 in the benzo[a]pyrene only group and 1.15 in the benzo[a]pyrene + M piperita (Peppermint) Leaf Extract group. M piperita (Peppermint) Leaf Extract had an inhibitory effect on lung tumor formation in this study. Results relating to the modulation of genotoxicity are included in the Antigenotoxicity section of the report text.
The anticancer potential of M piperita (Peppermint) Leaf Extract (double-distilled; water extract) was studied using Swiss albino mice (number not stated). 43 Two stage mouse skin carcinogenesis was initiated by 7,12-dimethyl-benz[a]anthracene (DMBA). Two weeks later, croton oil (promoter) was applied 3 times per week for 14 wk. The mice were dosed orally with M piperita (Peppermint) Leaf Extract (800 mg/kg/d) for the same period. At the end of the dosing period, average latent period, tumor incidence, size, burden, weight and cumulative number of papillomas were assessed. Dosing with M piperita (Peppermint) Leaf Extract caused inhibition of skin papilloma formation induced by DMBA and the application of croton oil, in terms of a significant decrease in the cumulative number of papillomas, tumor burden, and tumor incidence. In the control group, the tumor incidence was 100%. However, after dosing with the test substance for 15 d, the tumor incidence was reduced to 64%. There was a significant increase in the latency period for the appearance of papillomas in test animals (11 wk in control group; 13 wk in test group).
The possible molecular mechanisms underlying the cytotoxicity and anticarcinogenic potential of M piperita (Peppermint) Leaf Extract (petroleum ether, benzene, chloroform, ethyl acetate, methanol, or water extract) on 6 human cancer (HeLa, MCF-7, Jurkat, T24, HT-29, and MIAPaCa-2) and normal (IMR-90 and HEK-293) cell lines were evaluated. 44 In the human cancer cell lines tested with doses of 1, 10, and 100 μg/ml for 6 h, the number of apoptotic cells was incremental with an increase in the dose of M piperita extracts. However, of all the extracts tested, the chloroform and ethyl acetate extracts resulted in a significantly higher apoptotic index after 6 h, and the results were dose-dependent. When compared to the cancer cell lines, no significant changes were observed in normal cells. Similarly, of all of the extracts tested, the chloroform and ethyl acetate extracts of M piperita had significant dose- and time-dependent anticarcinogenic activity, leading to G1 cell cycle arrest and mitochondrial-mediated apoptosis, perturbation of oxidative balance, upregulation of Bax gene, elevated expression of p53 and p21 in the treated cells, and acquisition of senescence phenotype, while inducing pro-inflammatory cytokines response.
A study was performed to evaluate the antitumor activity of M piperita (Peppermint) Leaf Extract (methanol extract), using SW-480 human colon adenocarcinoma cells in a relevant cell anti-proliferation assay. 45 Statistically significant (α = .05) growth inhibition was observed at a concentration of 31 µg/ml. An IC50 (concentration required for 50% inhibition, μg/ml) of 92.3 μg/ml was reported for M piperita (Peppermint) Leaf Extract.
Other Relevant Studies
Cytotoxicity
M piperita (Peppermint) Oil
The cytotoxicity of Mentha Piperita (Peppermint) Oil was evaluated in the (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay using 2 human cancer cell lines, MCF-7 and LNCaP. 46 Mentha Piperita (Peppermint) Oil from plants that were harvested during the summer and winter was tested. The following IC50 values (μg/ml) were reported: MCF-7 cell line (75.2 ± 2.9 [summer]; 80.8 ± 3.2 [winter]) and LNCaP cell line (90.4 ± 3.7 [summer]; 95.7 ± 4.5 [winter]). IC50 values in the 10 to 100 μg/ml range represented a potentially toxic chemical, and IC50 values <10 μg/ml represented a potentially very toxic chemical.
In another study, essential oil was extracted from the leaves of M piperita. 47 The oil essential oil was found to be cytotoxic in the following 4 human cancer cell lines: human lung carcinoma SPC-A1 cells (IC50 = 10.89 μg/ml), human leukemia K562 cells (IC50 = 16.16 μg/ml) and human gastric cancer SGC-7901 cells (IC50 = 38.76 μg/ml). The essential oil was inactive against human hepatocellular carcinoma BEL-7402 cells.
M piperita
The inhibitory effect of polysaccharides extracted from M piperita on A549 non-small cell lung adenocarcinoma cells was investigated using the MTT assay. 48 The results indicated that polysaccharides extracted from M piperita had a moderate toxic effect on the A549 cell line (IC50 = 879.52 ± 22.55 μg/mL). The growth of A549 cells was inhibited by M piperita in a dose-dependent manner. The inhibitory rate was 54.54% ± 1.38% at the highest concentration tested (1 mg/mL).
Hepatotoxicity
M piperita (Peppermint) Leaf Extract
M piperita (Peppermint) Leaf Extract (methanol extract) and other botanical extracts were tested on both human (HepG2/C3A) and rat (MH1C1) hepatoma cells, using a battery of toxicity endpoints. 49 The extract was dissolved in dimethyl sulfoxide (DMSO) and then diluted in culture medium to a final concentration of 1000 μg/ml. The following 8 endpoints covering a variety of biological activities relevant to hepatotoxicity were used for hepatotoxicity evaluation: oxidative stress, mitochondrial membrane permeability, cellular neutral and polar lipid accumulation, CYP1A, 2B, 3A activities, albumin excretion, and total DNA content. Cluster analysis was used to group the phenolics into 4 clusters for each cell type. Two of the clusters were cluster 1 (compounds clustering with the solvent control (DMSO)) and cluster 2 (compounds with reported in vivo liver toxicity). Overall and individual liver activity of the phenolics on both human and rat hepatoma cell lines were compared. For HepG2/C3A cells, 100% of the observations for M piperita (Peppermint) Leaf Extract and thyme extract, 92% for cinnamon extract, and 89% for juniper berry extract were assigned to cluster 1 (control group). For rat MH1C1 cells, 100% of the juniper berry extract and M piperita (Peppermint) Leaf Extract observations were assigned to cluster 1. The authors noted that because there are currently no reports of liver toxicity associated with peppermint, M piperita (Peppermint) Leaf Extract is useful as a negative control.
Nephrotoxicity
M piperita (Peppermint) Leaf Extract
The effects of M piperita (Peppermint) Leaf Extract (“M piperita tea” (i.e., aqueous extract)) on rat kidney tissue were evaluated. The tea (prepared daily) was made by pouring 250 mL of boiling water over one heaped teaspoon (5 g) of the dried leaves of M piperita L (grown in Turkey) and steeping for 5 to 10 min. Groups of 12 male Wistar albino rats were used. Test animals received M piperita tea (20 g/L) in drinking water for 30 d. Control rats were given commercial drinking water during the study. The following histopathological changes, described as slight, were reported for the group dosed with M piperita tea: hydropic degeneration of tubular epithelial cells, epithelial cells with pyknotic nuclei and eosinophilic cytoplasm, tubular dilatation and enlargements in Bowman capsules. In conclusion, the results indicate that M piperita is not nephrotoxic to rats. 50
Effect on Histamine Release
M piperita (Peppermint) Leaf Extract
The 50% ethanol extract of peppermint leaves and stems significantly inhibited histamine release from rat peritoneal mast cells that was induced by compound 48/80 (polymer produced by the condensation of N-methyl-p-methoxyphenethylamine with formaldehyde) in vitro. 51
M piperita
In a study involving human basophil cell suspensions, obtained from workers who were exposed to an additive containing penicillin, the cell suspensions were incubated with .1 to .001 mg/mL Peppermint (dry aroma). A dose-dependent increase in histamine release was noted, and it was concluded that this release was due to nonimmunological mechanisms. 1
Immune System Effects
M piperita (Peppermint) Oil
The results of a host-resistance assay involving groups of 20 mice that had been dosed orally with M piperita (Peppermint) Oil (up to 1250 mg/kg/d for 5 d) suggested immunosuppression and/or increased susceptibility to bacterial-induced mortality. The results of a plaque-forming assay involving groups of 10 mice that received the same oral doses were negative. 1
Effect on Hair Growth
In a study involving C57BL/6 mice, the data suggest that 3% M piperita (Peppermint) Oil (diluted in jojoba oil) facilitates hair growth by promoting the conservation of vascularization of hair dermal papilla, which may contribute to the induction of early anagen stage. 52
Dermal Irritation and Sensitization Studies
Dermal Irritation and Sensitization Data.

Irritation
In Vitro
M Piperita (Peppermint) Leaf Extract
The skin irritation potential of M Piperita (Peppermint) Leaf Extract (water/ethanol extract) at concentrations of 10% and 100% was evaluated using the in vitro reconstructed human epidermis test method, and results were negative.
Animal
M Piperita (Peppermint) Oil
Hairless sites on 5 white rabbits were injected intradermally with .05 mL M Piperita (Peppermint) Oil. Gross examinations were performed at 24 h and 48 h, at 1 and 2 wk, and, in some cases, at 1 mo after dosing. Dosing was repeated between 5 and 10 times. At microscopic examination of skin samples, moderate reactions characterized by polymorphonuclear leucocytes, lymphocytes, and plasma cells (without necrosis) were observed in 3 rabbits. Severe reactions, which were marked by the above as well as necrosis, were observed in the other 2 rabbits. 1
M Piperita (Peppermint) Extract
The skin irritation potential of a trade name mixture containing 7.5% Mentha Piperita (Peppermint) Extract was evaluated using 3 rabbits (strain not stated). 9 No cutaneous reactions were observed, and the authors concluded that the mixture was a non-irritant.
Human
M Piperita (Peppermint) Oil, M Piperita (Peppermint) Leaf Extract, and M Piperita (Peppermint) Leaf Water
The skin irritation potential of a lipstick containing .2961% M Piperita (Peppermint) Leaf Extract was evaluated in a 48-h occlusive patch test using the following group of 50 subjects: normal (25), with eczema (4 subjects), with allergy (4 subjects) and with sensitive skin (17 subjects). Results were classified as negative. 53 Slight erythema was observed in 1 of 50 subjects after repeated applications of a cleaning gel containing 50% M Piperita (Peppermint) Leaf Water in a product use study. 54 Mild and moderate erythema were observed in 12 and 6 subjects, respectively, patch tested with 50% M Piperita (Peppermint) Leaf Water (10% aqueous solution dilution; effective concentration = 5% M Piperita (Peppermint) Leaf Water). 55 In one of the skin sensitization studies on 20% M Piperita (Peppermint) Oil that is summarized in the following section, it was reported that there was no evidence of skin irritation in the 104 subjects tested. 56
Sensitization
Animal
M Piperita (Peppermint) Extract
The skin sensitization potential of a trade name material containing 7.5% Mentha Piperita (Peppermint) Extract was evaluated in the maximization test using 10 albino guinea pigs. 9 No macroscopic cutaneous reactions attributable to allergy were associated with application of the trade name material. There also were no cutaneous intolerance reactions in animals of the negative control group (further details not provided).
Human
Mentha Piperita (Peppermint) Oil
In the maximization test, 25 healthy male panelists received five 48-h occlusive induction patch (containing 8% M Piperita (Peppermint) Oil) applications. 1 Pre-treatment was for 24 h with an occlusive patch containing 5% sodium lauryl sulfate (SLS) prior to each exposure. After a 10-d non-treatment period, the subjects were challenged on the back with a 48-h patch (also preceded by SLS treatment). No evidence of sensitization was found.
M Piperita (Peppermint) Oil, M Piperita (Peppermint) Extract, M Piperita (Peppermint) Leaf Extract, and M Piperita (Peppermint) Leaf Water
In a human repeated insult patch test (HRIPT) on 20% M Piperita (Peppermint) Oil involving 104 subjects, results were negative for skin irritation and sensitization. 56 Skin sensitization also was not observed in another HRIPT on 20% M Piperita (Peppermint) Oil involving 101 subjects. 57 An HRIPT on a trade name mixture containing 2.5% M Piperita (Peppermint) Extract was performed using 52 male and female subjects, and results were negative for dermal irritation and allergic contact sensitization. 58 Results were also negative in an HRIPT evaluating the cumulative irritation and/or allergic contact sensitization potential of a cosmetic product containing .00554% M Piperita (Peppermint) Extract in 51 subjects. 59 In a maximization test on a cosmetic product containing .00554% M Piperita (Peppermint) Extract involving 26 subjects, there was no evidence of contact allergy. 60 HRIPT results for undiluted M Piperita (Peppermint) Leaf Extract (water/ethanol extract) in 52 subjects were also negative. 11 A face cream containing 20% M Piperita (Peppermint) Leaf Water did not induce cumulative skin irritation or sensitization in an HRIPT involving 107 subjects. 61
Photosensitization/Phototoxicity
Animal
M Piperita (Peppermint) Oil
Undiluted M Piperita (Peppermint) Oil was applied to the backs of 6 Skh: hairless mice. Thirty minutes later, the mice were irradiated for either 1 h with light from a fluorescent blacklight at an integrated UVA of 3 W/m2, or for 40 minutes with light from a Xenon lamp at a weighted erythema energy of .1667 W/m2. The mice were examined at 4, 24, 48, 72, and 96 h after radiation treatment. No effects were noted. In a second experiment, using 2 miniature swine and following the same protocol, no effect was produced by 100% M Piperita (Peppermint) Oil. 1
Ocular Irritation Studies
In Vitro
M Piperita (Peppermint) Extract
The ocular irritation potential of a trade name mixture containing 2.5% M Piperita (Peppermint) Extract was evaluated using an in vitro toxicity testing system consisting of normal, human-derived epidermal keratinocytes. 62 The cells had been cultured to form a stratified squamous epithelium that is similar to that found in the cornea. The procedure utilized a tetrazolium salt (3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyl-tetrazolium bromide (MTT)) that is reduced by succinate dehydrogenase (in viable mitochondria of viable cells) to a formazan derivative. The amount of MTT that is reduced by a culture is proportional to the number of viable cells. The trade name mixture, at a concentration of 10% in corn oil (effective concentration of extract = .25%) and a volume of 100 μl, was added to cell cultures; the incubation periods were 1, 4, and 24 h. Corn oil served as the negative control. An ET50 (time of exposure needed for a test material to reduce the viability of treated tissues to 50% of control tissues) was calculated. Values for % viability were: 108% (at 1 h), 100% (at 4 h), and 34% (at 24 h). Results indicated that the trade name mixture at a concentration of 10% (ET50 = 15.5 h (non-irritating, minimal)) had an ocular irritation potential that was somewhat less that sodium dodecyl sulfate at a concentration of .3% (ET50 = 740 min (12.3 h)).
Animal
M Piperita (Peppermint) Extract
A trade name mixture containing 7.5% M Piperita (Peppermint) Extract was instilled (.1 mL) into 1 eye of each of 3 New Zealand rabbits. 9 Slight conjunctival redness was observed in 2 animals and lacrimation was observed in 1 animal. The trade name mixture was classified as a slight ocular irritant.
Human
M Piperita (Peppermint) Leaf Water
The ocular irritation potential of a cleansing gel containing 50% Mentha Piperita (Peppermint) Leaf Water was studied using 50 subjects. 54 The subjects applied the product twice per day for 4 wk, and were instructed to record any signs felt or observed during product use. Product use did not cause any signs of ocular or palpebral irritation.
Clinical Studies
Multicenter Studies
M Piperita (Peppermint) Oil
Data from multicenter studies evaluating the skin irritation/sensitization potential of M Piperita (Peppermint) Oil in patients are summarized in Table 6.63,64
A multicenter study involving 13,398 patients was performed by the US/Canadian North American Contact Dermatitis Group (NACDG), whereby 71 patients were patch tested with M Piperita (Peppermint) Oil (2% in petrolatum). A positive reaction prevalence rate of .53% was reported for this ingredient. 64 In another multicenter study, neither irritant nor allergic reactions were observed in 73 patients patch tested with M Piperita (Peppermint) Oil according to International Contact Dermatitis Research Group (ICDRG) patch test procedures. 63
Case Reports
Case reports are summarized in Table 6.
Positive patch test reactions to M Piperita (Peppermint) Oil and Mentha Piperita were reported in 8 of 9 case reports on patients with diseased skin.65-71 Though positive patch test results were reported in one of the studies on M Piperita (Peppermint) Oil, prick test results in that study were negative. In patients without skin disease, but with peppermint sensitivity, positive prick test reactions to M Piperita (Peppermint) Leaf, M Piperita (Peppermint) Leaf Extract, and M Piperita were reported.72,73
Other Clinical Reports
M Piperita (Peppermint) Oil
Positive reactions were observed in 7 of 450 dermatitic patients who were patch tested with 2% M Piperita (Peppermint) Oil in yellow soft paraffin. 1 In another study, positive reactions to 2% M Piperita (Peppermint) Oil were observed in 6 of 86 dermatitic patients. A patch containing 1% M Piperita (Peppermint) Oil (vehicle unknown) was applied to the backs of 56 patients with chronic urticaria. No reactions were noted after a 1-h or 48-h exposure.
No reactions were observed in 25 spice factory workers who were patch tested with 2% M Piperita (Peppermint) Oil in petrolatum. It has been reported that the patch testing of individual components of M Piperita (Peppermint) Oil using 3 patients with allergic contact dermatitis established that the allergens were menthol and trace components such as piperitone or pulegone. 1
Dermal
A triple-blind clinical trial involved 96 randomly selected subjects (47 cases and 49 controls; all pregnant women) with a diagnosis of pruritus gravidarum. 74 The case and control subjects were instructed to apply .5% peppermint oil in sesame oil (from 60 mL bottle of the solution) and sesame oil (from 60 mL bottle), respectively, twice per day for 2 wk. Applications (volume per application not stated) were made to the area of the itch. The authors noted that M Piperita (Peppermint) Oil did not cause any special side effects in any of the subjects tested. Itch severity in the group treated with M Piperita (Peppermint) Oil in comparison with the group treated with sesame oil was statistically significant (P = .003). The authors stated that, based on the results of this study, M Piperita (Peppermint) Oil can be used for symptomatic treatment of skin itching in pregnant women.
Oral
Each of 6 pediatric patients with irritable bowel syndrome received a single oral dose of M Piperita (Peppermint) Oil (187 mg). 75 Each capsule contained 83 mg of menthol as a constituent of M Piperita (Peppermint) Oil. Each patient drank 125 mL of water after ingestion of the capsule. No adverse events were reported. The delayed appearance of menthol in the plasma was reported; a substantial lag time (range 1 to 4 h) was observed in all subjects. Thus, an apparent prolonged absorption time was demonstrated. The authors noted that reasons for the delayed time of peak (Tmax) likely related to formulation-specific factors (i.e., delayed release) and, potentially, enterohepatic recirculation.
Exposure Assessment
Dermal
M Piperita (Peppermint) Oil
The FDA calculated an estimated human exposure from cosmetic use based on the concentration of use information supplied by industry. 1 Using a body splash product containing .2% M Piperita (Peppermint Oil) and assuming 100% absorption over a body surface of 17,000 cm2 and a daily application of 1 mg/cm2 (∼17 mL of the product), the FDA estimated an exposure of 34 mg/d. For a 60-kg person, this amounted to an estimated daily dose of .6 mg/kg/day.
Oral
M Piperita (Peppermint) Oil
In the European Union, the highest recommended daily dose of M Piperita (Peppermint) Oil is 1.2 mL, that is, 1080 mg M Piperita (Peppermint) Oil (contains a maximum of 140 mg pulegone + menthofuran). 16 For a 60 kg person, this would correspond to a daily intake of 2.3 mg/kg body weight. This recommended daily dose of M Piperita (Peppermint) Oil in medicinal products results in pulegone/menthofuran that exceeds the tolerated daily intake (TDI) (.1 mg/kg) that was established for food by the Committee of Experts on Flavoring Substances (CEFS).
Risk Assessment
Dermal
M Piperita (Peppermint) Oil
A maximum dermal use level of 5.4% has been recommended for M Piperita (Peppermint) Oil. 76 This dermal restriction is based on 8% menthofuran (pulegone metabolite) and 3% pulegone content, with limits of .5% for menthofuran and of 1.2% for pulegone. The authors also recommended that M Piperita (Peppermint) Oil, due to menthol content, should be avoided altogether in cases of cardiac fibrillation and in individuals with a glucose-6-phosphate dehydrogenase deficiency. No further information relating to these recommendations is provided.
Oral
M Piperita (Peppermint) Oil and Pulegone (a component of M Piperita (Peppermint) Oil)
Based on 8% menthofuran and 3% pulegone content, with limits of .2 mg/kg/d for menthofuran and .5 mg/kg/d for pulegone, the authors of one study recommended a maximum daily oral dose of 152 mg Mentha Piperita (Peppermint) Oil. 76
M piperita
A study was performed to characterize data on dietary botanical supplement (DBSs) associated with adverse event reports submitted to the FDA Center for Food Safety and Applied Nutrition’s Adverse Event Reporting System (CAERS). 77 FDA obtained CAERS data from 1999 to 2003 involving adverse effects associated with the 6 most frequently used DBSs, including peppermint. No adverse events were reported for single-ingredient peppermint supplements during the study period.
Summary
The safety of the following ingredients in cosmetics was previously reviewed by the Panel, and a final report with a conclusion stating that these ingredients are safe as used in cosmetic formulations was published in 2001: M Piperita (Peppermint) Oil, M Piperita (Peppermint) Leaf Extract, M Piperita (Peppermint) Leaf, and M Piperita (Peppermint) Leaf Water. The conclusion also stated that the concentration of pulegone, a constituent of these botanical ingredients, should not exceed 1% in the finished product. The current safety assessment is, in part, a re-review of the 4 M piperita (peppermint)–derived ingredients, and is inclusive of safety test data that have become available since the final report was issued.
The current safety assessment is also a first review of the following 6 M piperita (peppermint)–derived ingredients: M Piperita (Peppermint) Extract, M Piperita (Peppermint) Flower/Leaf/Stem Extract, M Piperita (Peppermint) Flower/Leaf/Stem Water, M Piperita (Peppermint) Leaf Cell Extract, M Piperita (Peppermint) Leaf Juice, and M Piperita (Peppermint) Meristem Cell Culture.
According to 2018 VCRP data, the greatest use frequency is being reported for M Piperita (Peppermint) Oil, which is being used in 815 cosmetic products, mostly leave-on products. The results of a concentration of use survey provided in 2016 indicate that M Piperita (Peppermint) Leaf Water is being used at a concentration up to 40% in leave-on products, which is the greatest use concentration that is being reported for M piperita (peppermint)–derived ingredients reviewed in this safety assessment.
In the US, M Piperita (Peppermint) Oil is GRAS for use in food for human consumption. It is also an inactive ingredient in drug products that have been approved by the FDA and is on the EPA list of active ingredients eligible for minimum risk pesticide products.
M Piperita (Peppermint) Leaf Extract (10% aqueous ethanol extract) caused a statistically significant increase in the penetration of caffeine, but not salicylic acid, through porcine skin. M Piperita (Peppermint) Oil inhibited the penetration of benzoic acid through human skin.
Following oral administration, M Piperita (Peppermint) Oil is relatively rapidly absorbed and eliminated mainly via the bile. The major biliary metabolite is menthol glucuronide. Additional metabolites are mono- or di-hydroxylated menthol derivatives.
M Piperita (Peppermint) Leaf Extract (aqueous extract
No adverse events were reported for single-ingredient peppermint supplements in a study that was performed to characterize data on dietary botanical supplement associated with adverse event reports submitted to the FDA Center for Food Safety and Applied Nutrition’s Adverse Event Reporting System (CaliforniaERS).
M Piperita (Peppermint) Oil was clastogenic in a chromosome aberration test involving peripheral blood lymphocytes. The authors noted that the dose-response curve for M Piperita (Peppermint) Oil was complicated, with a clear peak response at a concentration of .20 μl/ml; the number of aberrant cells decreased at higher concentrations. In a genotoxicity assay involving human lymphocytes, M Piperita (Peppermint) Oil induced sister chromatid exchanges in a dose-dependent manner. A trade name mixture containing 2.5% M Piperita (Peppermint) Extract (diluted to a concentration of 10% (effective concentration of extract = .25%)) was not cytotoxic or genotoxic in the Ames test, with or without metabolic activation. Numerous genotoxicity studies were evaluated, and the preponderance of the data was negative.
Oral pretreatment with M Piperita (Peppermint) Leaf Extract (aqueous extract) before exposure to gamma radiation was found to be effective in protecting against chromosomal damage in the bone marrow of Swiss albino mice. In another study, the oral administration of M Piperita (Peppermint) Leaf Extract had an antigenotoxic (i.e., reduced the frequency of chromosomal aberrations and micronuclei in bone marrow cells) in Swiss albino mice intraperitoneally injected with benzo[a]pyrene.
Oral dosing with M Piperita (Peppermint) Leaf Extract caused a significant reduction in the number of lung adenomas from an incidence of 67.92% in Swiss albino mice intraperitoneally injected with benzo[a]pyrene to 26.31%. Oral dosing with M Piperita (Peppermint) Leaf Extract also caused inhibition of skin papilloma formation induced by DMBA and the application of croton oil, in terms of a significant decrease in the cumulative number of papillomas, tumor burden, and tumor incidence.
In the human cancer cell lines tested with M Piperita (Peppermint) Leaf Extract (various extractants used), the number of apoptotic cells was incremental with an increase in the dose of M piperita extracts. M Piperita (Peppermint) Leaf Extract (only from chloroform and ethyl acetate extractants) had significant dose- and time-dependent anticarcinogenic activity, leading to G1 cell cycle arrest and mitochondrial-mediated apoptosis, perturbation of oxidative balance, upregulation of Bax gene, and elevated expression of p53 and p21 in the treated cells. In a study that was performed to evaluate the antitumor activity of M Piperita (Peppermint) Leaf Extract (methanol extract) in an anti-proliferation assay involving SW-480 human colon adenocarcinoma cells, statistically significant growth inhibition was observed.
Results were positive for M Piperita (Peppermint) Oil (from plants harvested during seasons of the year) in cytotoxicity assays involving human cancer cell lines: The following IC50 values (μg/ml) were reported: MCF-7 cell line (75.2 ± 2.9 [summer]; 80.8 ± 3.2 [winter]) and LNCaP cell line (90.4 ± 3.7 [summer]; 95.7 ± 4.5 [winter]). In another study, M Piperita (Peppermint) Oil was cytotoxic to the following human cancer cell lines: human lung carcinoma SPC-A1 cells (IC50 = 10.89 μg/ml), human leukemia K562 cells (IC50 = 16.16 μg/ml) and human gastric cancer SGC-7901 cells (IC50 = 38.76 μg/ml). The essential oil was inactive against human hepatocellular carcinoma BEL-7402 cells.
M Piperita (Peppermint) Leaf Extract (1000 μg/ml, methanol extract) did not induce hepatotoxicity in vitro assays involving human (HepG2/C3A) and rat (MH1C1) hepatoma cells.
The 50% ethanol extract of peppermint leaves and stems significantly inhibited compound 48/80-induced histamine release from rat peritoneal mast cells in vitro.
In a study involving C57BL/6 mice, it was concluded that 3% M Piperita (Peppermint) Oil (diluted in jojoba oil) facilitated hair growth by promoting the conservation of vascularization of hair dermal papilla.
A trade name mixture containing 7.5% M Piperita (Peppermint) Extract was non-irritating to the skin of 3 rabbits. No macroscopic cutaneous reactions attributable to allergy were observed in a maximization test in which 10 albino guinea pigs were patch tested with a trade name material containing 7.5% M Piperita (Peppermint) Extract during induction and challenged with the undiluted material and the material at a concentration of 50% (effective concentration of extract = 3.75%)
In a 48-h occlusive patch test, a lipstick product containing .2961% M Piperita (Peppermint) Leaf Extract did not cause skin irritation in any of the 50 subjects tested. In an in vitro skin irritation study on M Piperita (Peppermint) Leaf Extract (water/ethanol extract) involving reconstructed human epidermis, results were negative (MTT >50%).
There was no evidence of dermal irritation or allergic contact sensitization in an HRIPT in which 52 male and female subjects were patch tested with a 10% dilution of a trade name mixture containing 2.5% M Piperita (Peppermint) Extract (effective concentration of extract = .25%). A cosmetic product containing .00554% M Piperita (Peppermint) Extract did not cause dermal irritation or allergic contact dermatitis in 51 male and female subjects patch tested with a cosmetic product (an off-white cream) containing .00554% M Piperita (Peppermint) Extract. In the maximization test, a cosmetic product (off-white cream) containing .00554% M Piperita (Peppermint) did not induce contact sensitization in the 26 male and female subjects tested.
A face cream containing 20% M Piperita (Peppermint) Leaf Water did not induce cumulative skin irritation or sensitization in an HRIPT involving 107 subjects. Negative results were also reported for a lipstick product containing .2961% M Piperita (Peppermint) Leaf Extract in a 48-h occlusive patch test (skin irritation test) involving 50 subjects. Slight erythema was observed in 1 of 50 subjects who applied a cleansing gel containing 50% M Piperita (Peppermint) Leaf Water twice per day for 4 wk; there were no signs of ocular or palpebral irritation in any of the subjects. In a 48-h, single-application patch test, the skin irritation potential of a cleansing gel containing 50% M Piperita (Peppermint) Leaf Water (10% aqueous dilution [effective concentration = 5%]) was evaluated using 52 subjects. A score of 1 (mild erythema) was reported for 12 subjects on day 2 and for 18 subjects on day 3. A score of 2 (moderate erythema) was reported for 2 subjects on day 2 and for 3 subjects on day 3. On day 4, 46 subjects had a score of zero and 6 had a score of 1. The authors concluded that the skin compatibility of the diluted product was considered good.
The skin irritation and sensitization potential of 20% M Piperita (Peppermint) Oil was evaluated in an HRIPT involving 104 subjects and results were negative. Similarly, there was no evidence of sensitization to 20% M Piperita (Peppermint) Oil in an HRIPT involving 101 subjects. HRIPT results for undiluted M Piperita (Peppermint) Leaf Extract (water/ethanol extract) in 52 subjects were also negative.
A trade name mixture containing 2.5% M Piperita (Peppermint) Extract was classified as non-irritating in an in vitro toxicity testing system, consisting of normal, human-derived epidermal keratinocytes, for evaluating ocular irritation potential. Slight ocular irritation was observed in a study in which 3 rabbits were tested with a trade name mixture containing 7.5% M Piperita (Peppermint) Extract.
In a multicenter study, neither irritant nor allergic reactions were observed in 73 patients patch tested with M Piperita (Peppermint) Oil according to International Contact Dermatitis Research Group (ICDRG) patch test procedures. Another multicenter study involving 13,398 patients was performed by the US/Canadian North American Contact Dermatitis Group (NACDG), whereby 71 patients were tested with Piperita (Peppermint) Oil (2% in petrolatum). The prevalence rate for this ingredient was .53%.
Mostly positive patch test reactions to M Piperita (Peppermint) Oil (2% in petrolatum) and M Piperita were reported in case reports on patients with diseased skin. In patients without skin disease, but with peppermint sensitivity, positive prick test reactions to M Piperita (Peppermint) Leaf, M Piperita (Peppermint) Leaf Extract, and M Piperita were reported. In other clinical reports, dermal application of M Piperita (Peppermint) Oil (.5% in sesame oil) did not cause any side effects in 47 pregnant female patients, and a single oral dose of M Piperita (Peppermint) Oil did not cause any adverse events in 6 pediatric patients.
Discussion
The safety of the following cosmetic ingredients was reviewed previously by the Panel, and a final report with a conclusion stating that these ingredients are safe as used in cosmetic formulations was published in 2001: M Piperita (Peppermint) Oil, M Piperita (Peppermint) Leaf Extract, M Piperita (Peppermint) Leaf, and M Piperita (Peppermint) Leaf Water. The conclusion also states that the concentration of pulegone, a constituent of these botanical ingredients, should not exceed 1% in the finished product. The current safety assessment is a re-review of the safety of these 4 ingredients, and is also an initial safety evaluation of the following 6 related ingredients that were not listed as cosmetic ingredients prior to development of the published safety assessment: M Piperita (Peppermint) Extract, M Piperita (Peppermint) Flower/Leaf/Stem Extract, M Piperita (Peppermint) Flower/Leaf/Stem Water, M Piperita (Peppermint) Leaf Cell Extract, M Piperita (Peppermint) Leaf Juice, and M Piperita (Peppermint) Meristem Cell Culture.
In the 2001 published final safety assessment on M piperita (peppermint)–derived ingredients, the Panel expressed concern that rat oral-dosing studies on M Piperita (Peppermint) Oil and pulegone reported cyst-like spaces in the cerebellum that were attributed to pulegone. Therefore, the Panel established a 1% concentration limit on pulegone in M piperita (peppermint)–derived ingredients due to toxicity concerns. In these studies, brain sections were fixed by immersion using 4% neutral buffered formaldehyde. Because immersion fixation of nervous tissue might cause artifacts observed as vacuolar retraction spaces around neurons, a study involving rats was performed, using both immersion and perfusion tissue fixation methods, to determine whether the cerebellar lesions observed in earlier studies were caused by dosing with pulegone. Study results for rats dosed with pulegone did not reveal the occurrence of test substance-related, cyst-like spaces in the white matter of the cerebellum using either perfusion or immersion tissue fixation techniques. A possible explanation for the observed dose-dependent cyst-like spaces seen in previous studies could have been due to an interaction between impurities in the test substance and the fixation agent used therein. Given this possibility, the Panel agreed that the brain lesions may have been an artifact of the fixation method, and that the 1% limitation on pulegone is no longer warranted.
The panel also considered the carcinogenic effects of pulegone in female rats, and in male and female mice, in the 2011 National Toxicology Program (NTP) oral carcinogenicity study. However, the Panel did not feel that the cytotoxic dose-response relationship (renal and liver toxicity) associated with cancer development would be relevant to pulegone exposure from a cosmetic product containing M piperita (Peppermint) Oil at current use concentrations.
The Panel expressed concern about pesticide residues, heavy metals, and other plant species that may be present in botanical ingredients. They stressed that the cosmetics industry should continue to use current good manufacturing practices (cGMPs) to limit impurities.
Additionally, the Panel recognized that M Piperita (Peppermint) Leaf Extract can enhance the penetration of other ingredients through the skin. The Panel cautioned that care should be taken in formulating cosmetic products that may contain M Piperita (Peppermint) Leaf Extract in combination with any ingredients whose safety was based on their lack of dermal absorption data, or when dermal absorption was a concern.
The issue of incidental inhalation exposure was discussed by the Panel, as M piperita (peppermint)–derived ingredients are being used in products that could possibly be inhaled. For example, M Piperita (Peppermint) Leaf Extract is used in face and neck sprays at maximum use concentrations up to 1.3%. Another example relating to inhalation exposure from products that are sprayed is the use of M Piperita (Peppermint) Oil in both pump hair sprays (maximum use concentrations up to .02%) and aerosol hair sprays (maximum use concentrations up to .017%) which may result in incidental inhalation exposure. The use of M Piperita (Peppermint) Leaf Extract at maximum use concentrations up to .0018% in face powders may also result in incidental inhalation exposure. Additionally, the Panel noted that droplets/particles from spray cosmetic products would not be respirable to any appreciable amount. Furthermore, droplets/particles deposited in the nasopharyngeal or bronchial regions of the respiratory tract present no toxicological concerns based on the chemical and biological properties of these ingredients. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at https://www.cir-safety.org/cir-findings.
Finally, the Panel agreed that the data relating to composition of M Piperita (Peppermint) Flower/Leaf/Stem Extract, M Piperita (Peppermint) Flower/Leaf/Stem/Water, and M Piperita (Peppermint) Meristem Cell Culture are insufficient. Also, after considering the available skin irritation and sensitization data, the Panel determined that skin sensitization data on these three ingredients are also insufficient. Thus, the Panel determined that the following additional data are needed in order to evaluate the safety of these three ingredients: • Composition data on M Piperita (Peppermint) Flower/Leaf/Stem Extract, M Piperita (Peppermint) Flower/Leaf/Stem/Water and M Piperita (Peppermint) Meristem Cell Culture. ○ Depending on the composition data that are received, other toxicological endpoints may be needed. • Skin irritation and sensitization data on M Piperita (Peppermint) Flower/Leaf/Stem Extract, M Piperita (Peppermint) Flower/Leaf/Stem/Water and M Piperita (Peppermint) Meristem Cell Culture.
The Panel also agreed that the available data are sufficient for determining that M Piperita (Peppermint) Oil, Leaf, and leaf-derived ingredients are safe in cosmetics in the present practices of use and concentration, when formulated to be non-sensitizing. Because final product formulations may contain multiple botanicals, each possibly containing the same constituent(s) of concern, formulators are advised to be aware of these constituents and to avoid reaching levels that may be hazardous to consumers. For M piperita (peppermint)–derived ingredients, the Panel was concerned about the presence of terpenes (e.g., limonene) and terpenoids (e.g., menthol) in cosmetics, which could result in sensitization. Thus, this non-sensitizing caveat relates to the avoidance of a cumulative effect of multiple botanicals, in a single formulation, that share one or more constituents in common.
Conclusion
The Expert Panel for Cosmetic Ingredient Safety concluded that the following 7 M piperita (peppermint)–derived ingredients are safe in cosmetics in the present practices of use and concentration described in the safety assessment, when formulated to be non-sensitizing: • M Piperita (Peppermint) Oil • M Piperita (Peppermint) Extract • M Piperita (Peppermint) Leaf • M Piperita (Peppermint) Leaf Cell Extract* • M Piperita (Peppermint) Leaf Extract • M Piperita (Peppermint) Leaf Juice* • M Piperita (Peppermint) Leaf Water
*Not reported to be in use. Were ingredients in this group not in current use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
The Panel also concluded that the available data are insufficient to make a determination that the following 3 M piperita (peppermint)–derived ingredients are safe under the intended conditions of use in cosmetic formulations: • M Piperita (Peppermint) Flower/Leaf/Stem Extract** • M Piperita (Peppermint) Flower/Leaf/Stem Water** • M Piperita (Peppermint) Meristem Cell Culture**
**Not reported to be in use. These ingredients are thus categorized as Insufficient Data—No Reported Use.
Footnotes
Author Contributions
The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
Declaration of Conflicting Interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review