Abstract
The Expert Panel for Cosmetic Ingredient Safety reviewed updated information that has become available since their original assessment from 1991, along with updated information regarding product types, and frequency and concentrations of use, and reaffirmed their original conclusion that Polyacrylamide is safe as a cosmetic ingredient in the practices of use and concentration as described in this report.
Keywords
The Expert Panel for Cosmetic Ingredient Safety first published a Final Report of the Safety Assessment of Polyacrylamide in 1991, with the conclusion that Polyacrylamide, with less than .01% acrylamide monomer content, is safe as a cosmetic ingredient as currently used. 1 Subsequently, because a large number of relevant safety studies on acrylamide became available, in 2005 the Expert Panel published an Amended Final Report on the Safety Assessment of Polyacrylamide and Acrylamide Residues in Cosmetics. 2 The Expert Panel concluded that Polyacrylamide is safe as a cosmetic ingredient in the present practices of use and concentration described in the safety assessment, if the level of acrylamide monomer in formulation is not greater than 5 ppm; it was acknowledged that acrylamide is a demonstrated neurotoxicant in humans (at high exposure levels that could not be attained with cosmetic use), a known carcinogen in animal tests, and is probably carcinogenic to humans. 3
Current and Historical Frequency and Concentration of Use According to Duration and Exposure.
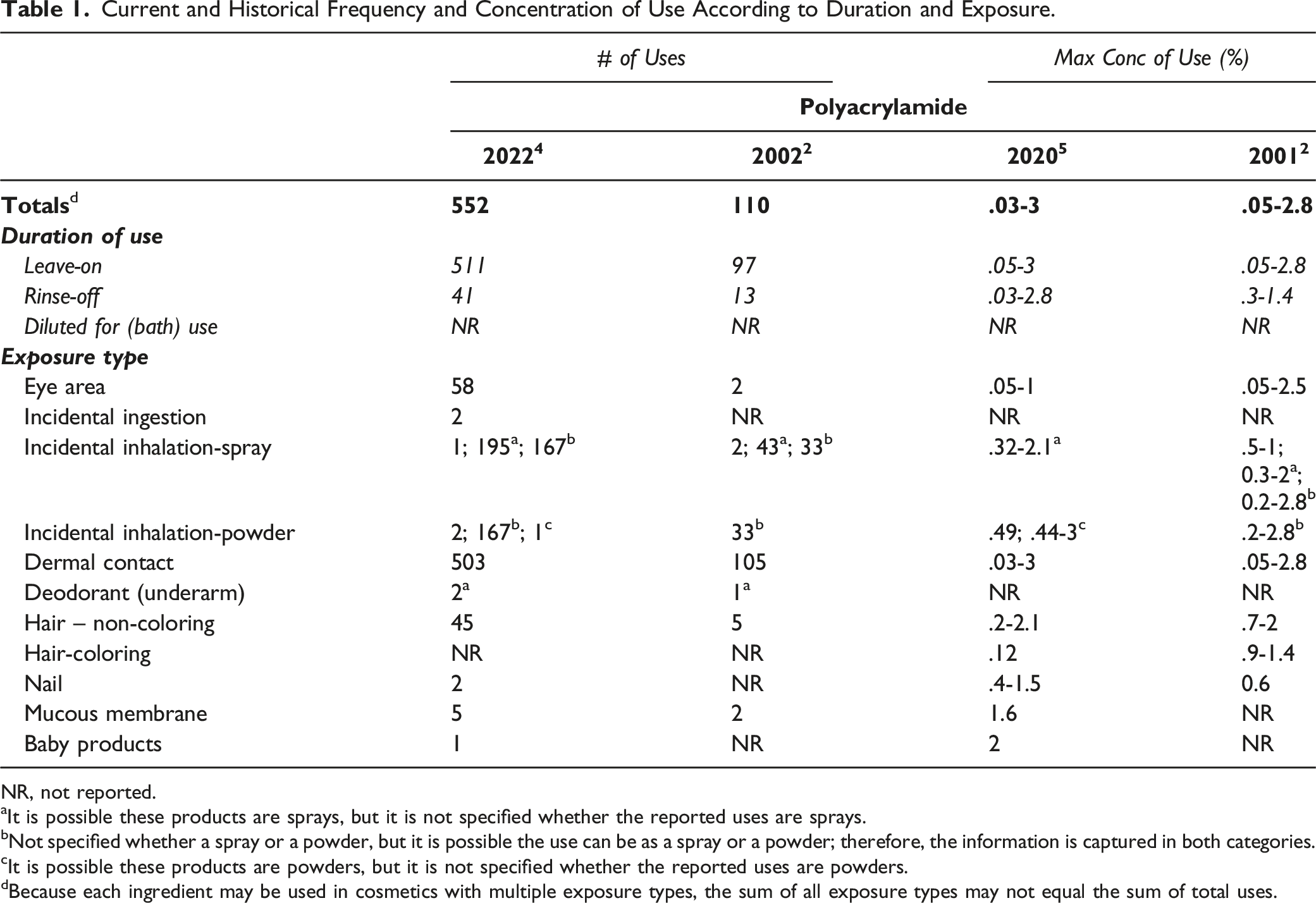
NR, not reported.
aIt is possible these products are sprays, but it is not specified whether the reported uses are sprays.
bNot specified whether a spray or a powder, but it is possible the use can be as a spray or a powder; therefore, the information is captured in both categories.
cIt is possible these products are powders, but it is not specified whether the reported uses are powders.
dBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
An extensive search of the world’s literature was performed for studies dated 2000 forward. No new published data were found. The Expert Panel considered the reported use in a baby lotion (concentration of use not reported) and discussed the typical average daily consumption of Polyacrylamide in foods, which mitigated systemic toxicity concerns. The Expert Panel noted that the mean dietary intake of Polyacrylamide has been reported to average .5 μg/kg/bw/d in adults, whereas intake is higher among children. 6
After reviewing updated frequency and concentration of use data, and considering the lack of new toxicological data, the Expert Panel determined to not reopen this safety assessment on Polyacrylamide and reaffirmed the conclusion published in 2005.
Footnotes
Author Contributions
The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Declaration of Conflicting Interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
