Abstract
The Expert Panel for Cosmetic Ingredient Safety reviewed newly available studies since their original assessment in 1998, along with updated information regarding product types and concentrations of use, and confirmed that Sodium Sulfite, Potassium Sulfite, Ammonium Sulfite, Sodium Bisulfite, Ammonium Bisulfite, Sodium Metabisulfite, and Potassium Metabisulfite are safe as cosmetic ingredients in the practices of use and concentration as described in this report.
Current and Historical Frequency and Concentration of Use of Sulfites According to Duration and Exposure.
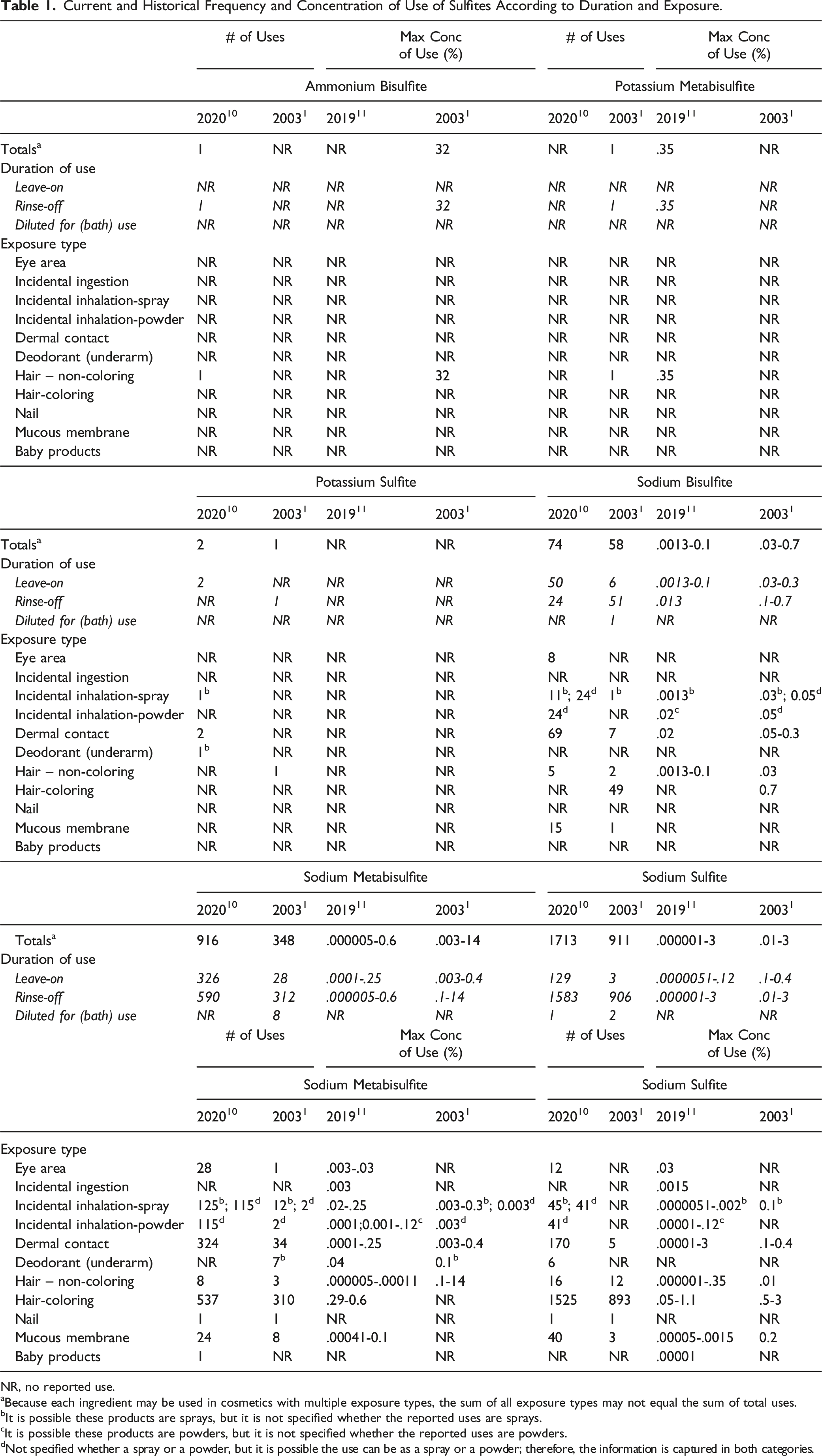
NR, no reported use.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
bIt is possible these products are sprays, but it is not specified whether the reported uses are sprays.
cIt is possible these products are powders, but it is not specified whether the reported uses are powders.
dNot specified whether a spray or a powder, but it is possible the use can be as a spray or a powder; therefore, the information is captured in both categories.
Frequency of use has increased substantially for Sodium Sulfite since the original review; in the 2003 report, 911 uses were reported, and in 2020, this ingredient was reported to have 1713 uses. 10 Sodium Metabisulfite also had a substantial increase in reported use frequency, from 348 uses in 2003 to 916 uses in 2020. The maximum concentration of use reported for this family of ingredient has decreased; in the 2003 report, Sodium Metabisulfite had the highest use concentration (14% in rinse-off products) 1 ; in 2019, this ingredient was reported being used at substantially lower concentrations of up to .6% in rinse-off products. 11 The sulfite with the highest reported use concentration in 2019 was Sodium Sulfite, at concentrations up to 3% in rinse-off products. (This is the same use concentration reported for this ingredient in the 2003 report. 1 )
The Expert Panel’s concern about sulfite-induced contact sensitization, following a review of patient studies, was allayed after considering negative results from two human repeated insult patch tests on Sodium Sulfite at concentrations greater than .25% (highest reported use concentration in leave-on products) in normal subjects. 1 The Expert Panel noted that results from a patient population are difficult to interpret in terms of their relevance to the general population, and, also, that few reactions to sulfites on standard panels used by dermatologists are being reported. 42 However, they acknowledged that sulfites can cause hypersensitivity, as evidenced by the enhancement of allergic sensitization in dust mite allergen-sensitized BALB/c mice. 36 Additionally, the Expert Panel noted that sulfites are associated with IgE-mediated allergic reactions in some individuals, and that individuals with sulfite allergies should exercise caution in using products containing sulfites that may be incidentally inhaled. 38 The lack of awareness of asthmatic responses to topical cosmetics was also acknowledged by the Expert Panel. 35
After considering that positive genotoxicity results (sister chromatid exchanges) were observed at the highest dose tested, the Expert Panel agreed that such a high dose would not be achieved in the current reported concentrations of use in cosmetics. Furthermore, the Expert Panel noted that the weight of evidence for sulfite-induced carcinogenicity in animal models is negative, and that the International Agency for Research on Cancer has concluded that there is inadequate evidence for the carcinogenicity of sulfites, bisulfites, and metabisulfites in experimental animals and humans. The minimal concern by the Expert Panel over the potential toxicity of sulfites from cosmetic exposure is further mitigated by the use of these ingredients at low concentrations and the low potential for absorption.
Footnotes
Author Contributions
The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
