Abstract
The Expert Panel for Cosmetic Ingredient Safety reviewed newly available studies since their original assessment in 1982, along with updated information regarding product types and concentrations of use, and confirmed that Quaternium-18 and Quaternium-18 Bentonite are safe as cosmetic ingredients in the practices of use and concentration as described in this report.
The Expert Panel for Cosmetic Ingredient Safety (Panel) first published the safety assessment of Quaternium-18, Quaternium-18 Bentonite, and Quaternium-18 Hectorite in 1982 and concluded that these ingredients were safe as used. 1 In 2003, after considering new studies and updated use data on these ingredients, the Panel determined to not re-open the safety assessment. 2 In 2019, Quaternium-18 and Quaternium-18 Bentonite were again re-reviewed, and it was determined to re-open the safety assessment to evaluate the sufficiency of inhalation data on Quaternium-18 Bentonite. Additionally, an exhaustive search of the world’s literature was performed for studies dated 1995 forward. No relevant published data were found; however, unpublished data provided by the Personal Care Products Council regarding Quaternium-18 Bentonite were provided. 3 (Some of the data provided were already included in the original report.)
Current and Historical Frequency and Concentration of Use of Quaternium-18 and Quaternium-18 Bentonite.
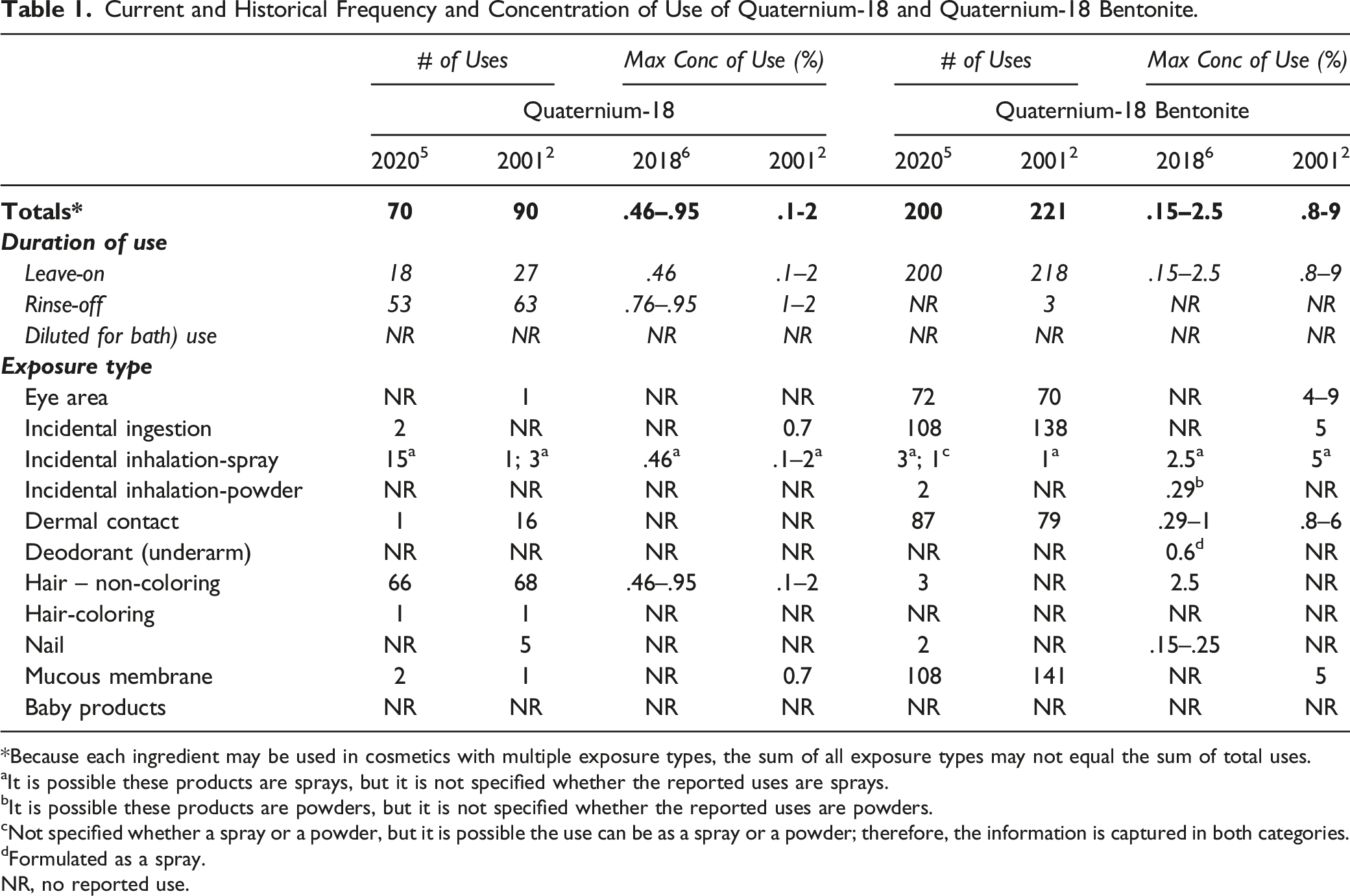
*Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
aIt is possible these products are sprays, but it is not specified whether the reported uses are sprays.
bIt is possible these products are powders, but it is not specified whether the reported uses are powders.
cNot specified whether a spray or a powder, but it is possible the use can be as a spray or a powder; therefore, the information is captured in both categories.
dFormulated as a spray.
NR, no reported use.
It should be noted that Quaternium-18 Hectorite was also included in the 1982 safety assessment and 2001 re-review. However, Quaternium-18 Hectorite is not included in the current assessment because it was recently (2013) part of a separate assessment (Safety Assessment of Ammonium Hectorites as Used in Cosmetics). 4 In that assessment, Quaternium-18 Hectorite was determined to be safe as used in cosmetics in the present practices of use and concentration.
Footnotes
Author Contributions
The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Declaration of Conflicting Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
