Abstract
The Expert Panel for Cosmetic Ingredient Safety reviewed information that has become available since their original assessment from 2001, along with updated information regarding product types, and frequency and concentrations of use, and reaffirmed their original conclusion that Lard, Hydrogenated Lard, Lard Glyceride, Hydrogenated Lard Glyceride, Lard Glycerides, and Hydrogenated Lard Glycerides are safe as cosmetic ingredients in the practices of use and concentration as described in this report.
Keywords
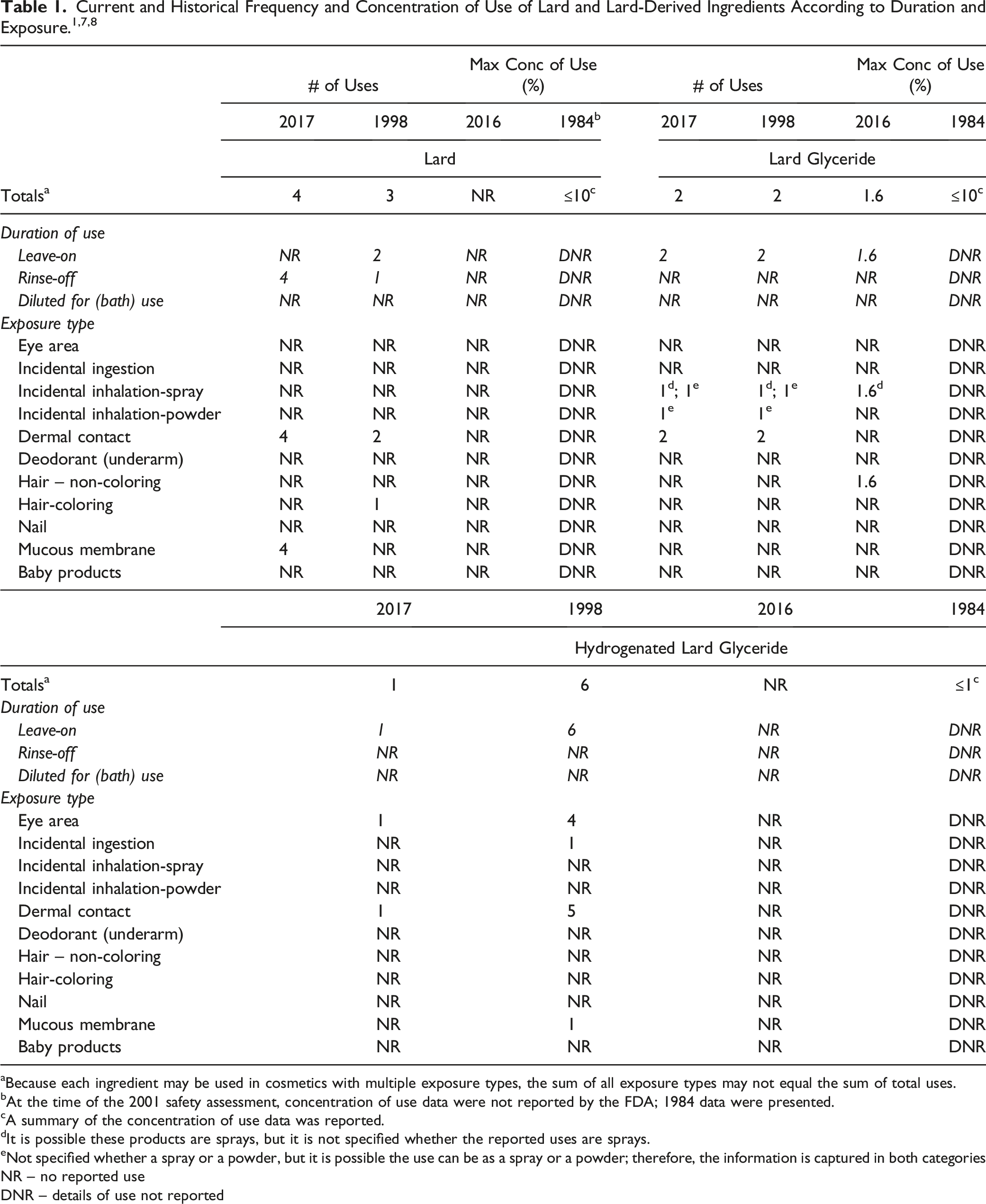
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
bAt the time of the 2001 safety assessment, concentration of use data were not reported by the FDA; 1984 data were presented.
cA summary of the concentration of use data was reported.
dIt is possible these products are sprays, but it is not specified whether the reported uses are sprays.
eNot specified whether a spray or a powder, but it is possible the use can be as a spray or a powder; therefore, the information is captured in both categories
NR – no reported use
DNR – details of use not reported
In 2017, Lard was reported to be used in 4 formulations (bath soaps and detergents); this is an increase from the 3 formulations reported in 1998. Lard Glyceride was reported to be used in 2 formulations in 2017 (a face and neck formulation and a moisturizing formulation) and 2 in 1998 (a body and hand care formulation and a moisturizing formulation). The reported use of Hydrogenated Lard Glyceride was less in 2017 (1 formulation [eyebrow pencil]) than in 1998 (6 formulations [4 eye formulations, a lipstick, and a makeup formulation]). Lard Glyceride was reported to be used at up to 1.6% in tonics, dressings and other hair grooming aids in 2016; in 1984, it was reported to be used at ≤ 10% in 1984 (types of formulations not specified). There were no reported concentrations of use in the 2016 Council survey for any of the other ingredients in this report. In 1984, Lard was reported to be used at concentrations ≤10% and Hydrogenated Lard Glyceride was reported at ≤ 1%. There were no current or historical uses reported for Hydrogenated Lard, Lard Glycerides, and Hydrogenated Lard Glycerides.
The Expert Panel expressed concern regarding pesticide and heavy metals that may be present in animal-derived ingredients. They acknowledged that the Lard-derived ingredients are formed from animal-derived constituents. The Expert Panel recognizes that these limits were developed for uses other than cosmetics but considers that such limits would assure that any cosmetic product with these ingredients can be used safely. They stressed that the cosmetics industry should continue to use the necessary procedures to limit these impurities in the ingredient before blending into cosmetic formulation.
Also of concern to the Expert Panel was the possible presence of dioxane impurities. They stressed that the cosmetics industry should continue to use the necessary procedures to limit these impurities from the Lard-derived ingredients before blending them into cosmetic formulations.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contributions
The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.