Abstract
The Expert Panel for Cosmetic Ingredient Safety reviewed newly available studies since their original assessment in 1999, along with updated information regarding product types and concentrations of use, and confirmed that Erythorbic Acid and Sodium Erythorbate are safe as cosmetic ingredients in the practices of use and concentration as described in this report.
The Expert Panel for Cosmetic Ingredient Safety first published a Final Report on the Safety Assessment of Erythorbic Acid and Sodium Erythorbate, as part of a larger group of ingredients, in 1999. 1 The Expert Panel concluded that these ingredients are safe for use as cosmetic ingredients in the present practices of use. It should be noted that Ascorbyl Palmitate, Ascorbyl Dipalmitate, and Ascorbyl Stearate, which were a part of the original review, were added to a grouping of ethers and esters of ascorbic acid for which a safety assessment was published in 2017, and therefore, are not included as part of this rereview.
2022 and Historical Frequency and Concentration of Use of Erythorbic Acid and Sodium Erythorbate According to Duration and Exposure.
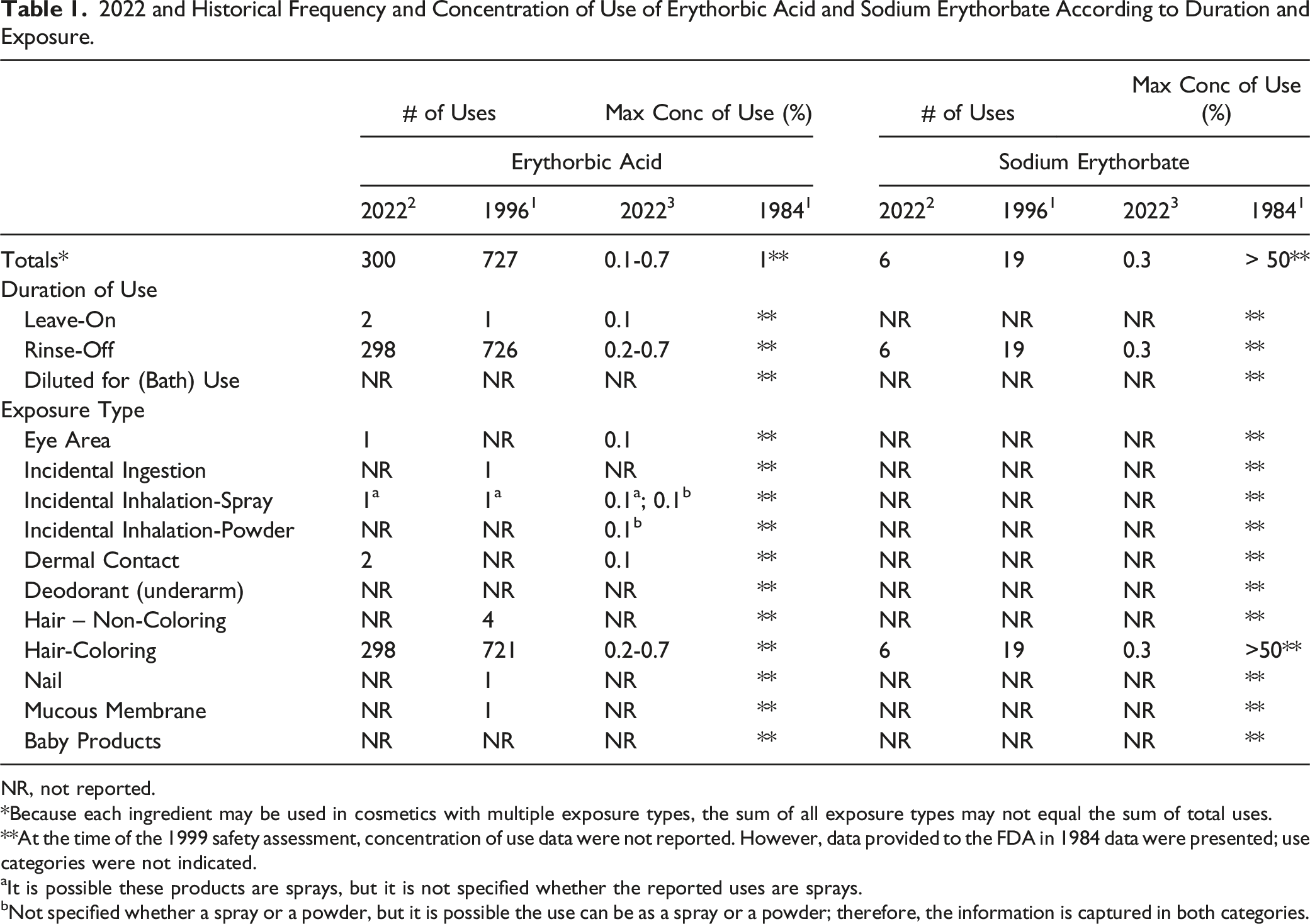
NR, not reported.
*Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
**At the time of the 1999 safety assessment, concentration of use data were not reported. However, data provided to the FDA in 1984 data were presented; use categories were not indicated.
aIt is possible these products are sprays, but it is not specified whether the reported uses are sprays.
bNot specified whether a spray or a powder, but it is possible the use can be as a spray or a powder; therefore, the information is captured in both categories.
An extensive search of the world’s literature was performed for studies dated 1994 forward, and minimal new published data were found.4-15 One noteworthy finding included a local lymph node assay performed in female mice in which Sodium Erythorbate was tested at 5, 10, or 25% in propylene glycol. 15 The stimulation index for the maximum concentration tested (25%) was 1.29; all stimulation index values were < 3, indicating that Sodium Erythorbate is not a sensitizer.
The Expert Panel reviewed 2022 frequency and concentration of use data, in addition to any new, available, relevant safety data, and reaffirmed the 1999 conclusion that these ingredients are safe for use as cosmetic ingredients in the present practices of use.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contributions
The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
