Abstract
The Expert Panel for Cosmetic Ingredient Safety reviewed updated information that has become available since their original assessment from 2001, along with updated information regarding product types, and frequency and concentrations of use, and reaffirmed their original conclusion that Methyl Alcohol is safe as used to denature alcohol in the practices of use and concentration as described in this report.
Keywords
The Expert Panel for Cosmetic Ingredient Safety published the Final Report on the Safety Assessment of Methyl Alcohol in 2001. 1 The Expert Panel concluded that based on the data presented in the safety assessment, Methyl Alcohol is safe as used to denature alcohol used in cosmetic products.
Frequency (2022; 1998) and Concentration (2021; 1984) of Use According to Likely Duration and Exposure and by Product Category.
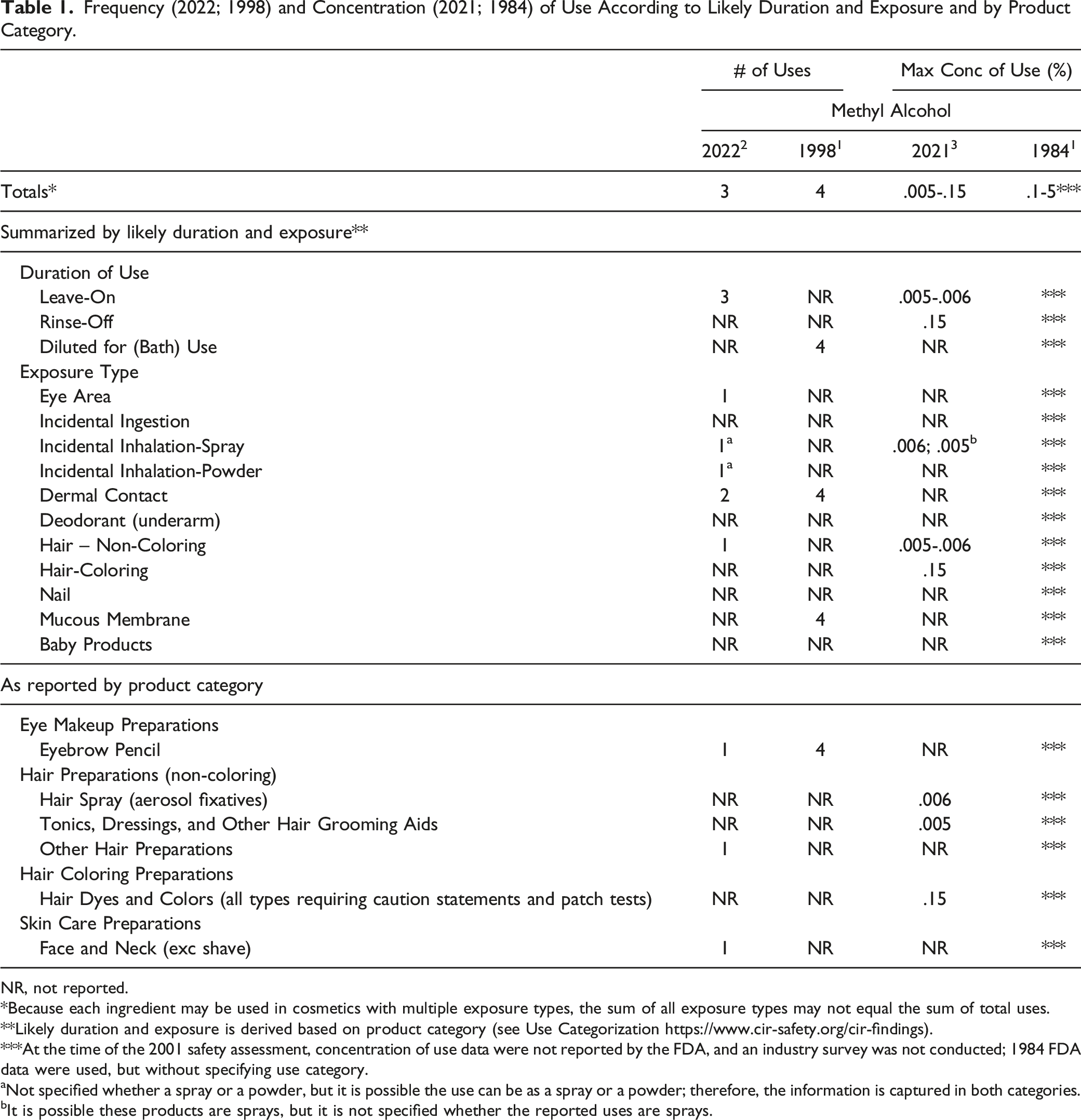
NR, not reported.
*Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
**Likely duration and exposure is derived based on product category (see Use Categorization https://www.cir-safety.org/cir-findings).
***At the time of the 2001 safety assessment, concentration of use data were not reported by the FDA, and an industry survey was not conducted; 1984 FDA data were used, but without specifying use category.
aNot specified whether a spray or a powder, but it is possible the use can be as a spray or a powder; therefore, the information is captured in both categories.
bIt is possible these products are sprays, but it is not specified whether the reported uses are sprays.
In October 2022, an extensive search of the world’s literature was performed for studies dated 1996 forward, and a substantial amount of new data was found from two sources, a European Chemicals Agency dossier and a screening assessment dataset initial assessment profile issued by the Organisation for Economic Co-operation and Development.4,5 A wide breadth of data was included; notably, a short-term and a subchronic oral toxicity study, several short-term and chronic inhalation toxicity studies, 2 Ames tests, carcinogenicity studies in mice and rats, and a guinea pig maximization test for sensitization were found, all of which were not present in the original report. Additionally, chemical properties, dermal, oral, and inhalation toxicokinetic and acute toxicity studies, multiple in vitro genotoxicity studies, an in vivo genotoxicity study (administered via the inhalation route), an in vitro developmental toxicity study in mice, a skin irritation study in rabbits, and an ocular irritation study in rabbits were also found that were supplemental to, and did not conflict with, results found in the original report. It was also noted that Methyl Alcohol is now approved as a food additive [21CFR173.250] as well as a component of adhesives, defoaming agents, and paper and paperboard products that come in contact with food [21CFR175.105; 21CFR176.200; 21CFR176.210; 21CFR176.80].
The Expert Panel stated that the newly available data was mostly additive to the original report, with studies reporting negative results, and that the number of reported uses remained relatively constant over time. Thus, the Expert Panel agreed that in spite of the volume of data that was available, the published literature did not reveal toxicity or other data considered significantly different to warrant re-evaluation of the safety of this ingredient in cosmetic products.
The Expert Panel did observe that Methyl Alcohol now has a reported use in aerosol hair sprays (.006%). However, negative inhalation toxicity data from the previous report, as well as 18-month and 24-month inhalation toxicity studies with negative results considered for this re-review, mitigated any concern. Furthermore, the Expert Panel reasoned that the maximum reported concentration of use for Methyl Alcohol in sprays is far below the permissible exposure limit issued by the Occupational Safety and Health Administration (200 ppm).
In summary, the Expert Panel reviewed 2022 frequency of use and 2021 concentration of use data, in addition to any new, available, relevant safety data. Considering this information, as well as the information provided in the original safety assessment, the Expert Panel reaffirmed the 2001 conclusion.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contributions
The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Declaration of Conflicting Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
