Abstract
Lacto-N-triose II (LNT II), an essential human milk oligosaccharide and precursor to lacto-N-tetraose (LNT) and lacto-N-neotetraose (LNnT), was evaluated for safety. Genotoxicity was assessed through in vitro tests including Bacterial Reverse Mutation Test and mammalian cell micronucleus test, and a subchronic oral gavage toxicity study was conducted on juvenile Sprague-Dawley rats. In this study, LNT II was administered at dose levels of 0, 1,500, 2,500, or 5,000 mg/kg body weight (bw)/day for 90 days, followed by a 4-week treatment-free recovery period. LNT II was non-genotoxic in the in vitro assays. No compound-related effects were observed across all dosage levels based on various measures, including clinical observations, body weight gain, feed consumption, clinical pathology, organ weights, and histopathology. Consequently, the highest dosage of 5,000 mg/kg bw/day was established as the no-observed-adverse-effect-level (NOAEL). These results suggest the safe use of LNT II in young children formula and as a food ingredient, within the limits found naturally in human breast milk.
Keywords
Introduction
Human milk oligosaccharides (HMOs) are the third most abundant endogenous component of human milk, following lactose and lipids. These non-digestible oligosaccharides are believed to foster the growth of infant gastrointestinal tract microbiota and maturation of the intestinal mucosal immune system. Structurally, HMOs consist of Glu, Gal, GlcNAc, fucose, and N-acetyl-neuraminic acid moieties. All HMOs contain lactose (Galβ1-4Glc) at the reducing end and elongated oligosaccharide chains, composed of lacto-N-biose (Galβ1-3GlcNAc) or N-acetyllactosamine (Galβ1-4GlcNAc) disaccharide units, linked by β1-3 or β1-6 glycosidic bonds at the non-reducing ends. To date, over 200 different HMOs have been identified, with the highest concentrations found in human colostrum (20 to 25 g/L), and ranging between 5 and 20 g/L in mature human milk.1–3
Three major types of HMOs exist: neutral N-containing HMOs, neutral (fucosylated) HMOs, and acidic (sialylated) HMOs. According to Zivkovic, the non-fucosylated, neutral N-containing HMOs constitute 42-55% of the total HMO fraction in breast milk. 4 Lacto-N-triose II (LNT II) falls within the “neutral core” sub-fraction of HMOs, which contains oligosaccharides with the aminosugar GlcNAc. These “core” HMOs can undergo further modifications by the addition of fucose or sialic acid, leading to the formation of fucosylated and sialylated HMO fractions, respectively. While the presence of “core” HMOs is consistent across all milk types and maternal phenotypes, genetic factors can induce variations in fucosylation patterns.
LNT II, naturally present in human breast milk, has been consumed by infants for millennia. It also can be produced via the catabolism of major neutral N-containing HMOs including lacto-N-tetraose (LNT) and lacto-N-neotetraose (LNnT) through gut microbiota fermentation or acid hydrolysis, suggesting that LNT II formation likely takes place in the gastrointestinal (GI) tract during the ingestion of LNT and LNnT.5–8 Several studies have sought to quantify the concentration of LNT II in human breast milk.9–11 In colostrum, the concentration of LNT II is observed to be between 129 mg/L and 136 mg/L. This concentration diminishes to approximately 30-40 mg/L by the 42nd day of lactation and remains relatively consistent up to the 168th day of lactation.
The positive impact of HMOs on infants’ digestive system, intestinal health, and immune system improvement has spurred extensive research. 1 The limited accessibility of human milk poses a significant challenge in acquiring adequate amounts of LNT II for broader applications, including in infant or toddler formulas and functional foods. 12 This issue is further compounded by ethical and logistical hurdles surrounding the extraction of LNT II from human milk, highlighting the need for alternative production methodologies. 13 The burgeoning global demand for HMOs exacerbates challenge as producing more HMOs in a sustainable fashion. 14 To overcome this challenge, the biosynthetic production of LNT II using engineered microorganisms has surfaced as a promising approach for scalable and sustainable production. 4 Synthetic biology techniques are employed to produce bioactive oligosaccharides or “nature-identical” compounds using genetically modified microbial hosts. Several studies have explored the safety and efficacy of biosynthesized LNT II to meet the growing demand for these crucial oligosaccharides. 15
Despite their chemical similarity to their naturally occurring counterparts, synthetically produced HMOs are derived using novel technologies, necessitating regulatory approvals in global markets. The first commercialized HMOs, 2′-fucosyllactose (2′-FL) and LNnT, synthesized chemically and through fermentation, have been granted Generally Recognized as Safe (GRAS) status in the United States (for use in infant formula up to 2.4 or .6 g/L for 2′-FL and LNnT, respectively) in 2015.16,17 They are also approved as novel infant food ingredients in the European Union.18,19 LNT II, produced via novel technology, is included in the Novel Food list of 2 approved HMOs, LNnT and LNT by EFSA in 2015 and 2019, respectively,20,21 and have received GRAS acknowledgement from the US FDA.17,22
The LNT II examined in this study is a synthetic biological product, derived from the fermentation of a bioengineered microorganism. The safety assessment of LNT II involved in vitro genotoxicity tests and subchronic toxicity studies in juvenile rats. The genotoxicity tests were conducted to evaluate potential mutagenicity, clastogenicity, and aneugenicity of LNT II in vitro. The subchronic tests preceded a 90-day oral gavage study in juvenile rats dosed starting from postnatal day (PND) 19, simulating human toddler exposure. This approach allowed for the assessment of potential systemic toxicity following exposure from post-weaning into adulthood, to corroborate the safety and establish the No-observed-adverse-effect-level (NOAEL) of LNT II. This study design aligns with guidelines accepted by regulatory authorities for evaluating the safety of novel infant formula ingredients such as HMOs.16,17,22
Materials and Methods
Good Laboratory Practice and Regulatory Guidelines
All studies were conducted at Pharmaron (Beijing) TSP Services Limited in adherence to U.S. FDA Good Laboratory Practice (GLP) Regulations for Nonclinical Laboratory Studies (21 CFR Part 58), and the Organisation for Economic Co-operation and Development (OECD) Principles of Good Laboratory Practice. 23 Pharmaron TSP is accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International (AAALAC International). All animals were provided with environmental enrichments upon arrival. Additionally, all portions of the study performed at Pharmaron TSP adhered to the study protocol, protocol amendments, and applicable standard operating procedures.
The 90-day oral gavage toxicity study adhered to OECD Guideline Test No. 408, 24 “Repeated dose 90-day oral toxicity study in rodents,” and FDA Guidance for Industry: Nonclinical safety evaluation of pediatric drug products (February, 2006).
The bacterial reverse mutation test followed OECD Guideline Test No. 471, 25 “Bacterial reverse mutation test.” The in vitro mammalian cell micronucleus test was carried out in accordance with OECD Guideline Test No. 487 (OECD, 2016), 26 “In vitro mammalian cell micronucleus test.”
Test Material
Composition of LNT II Batches Used for the Safety Studies (Batch A220701M and A220901M).
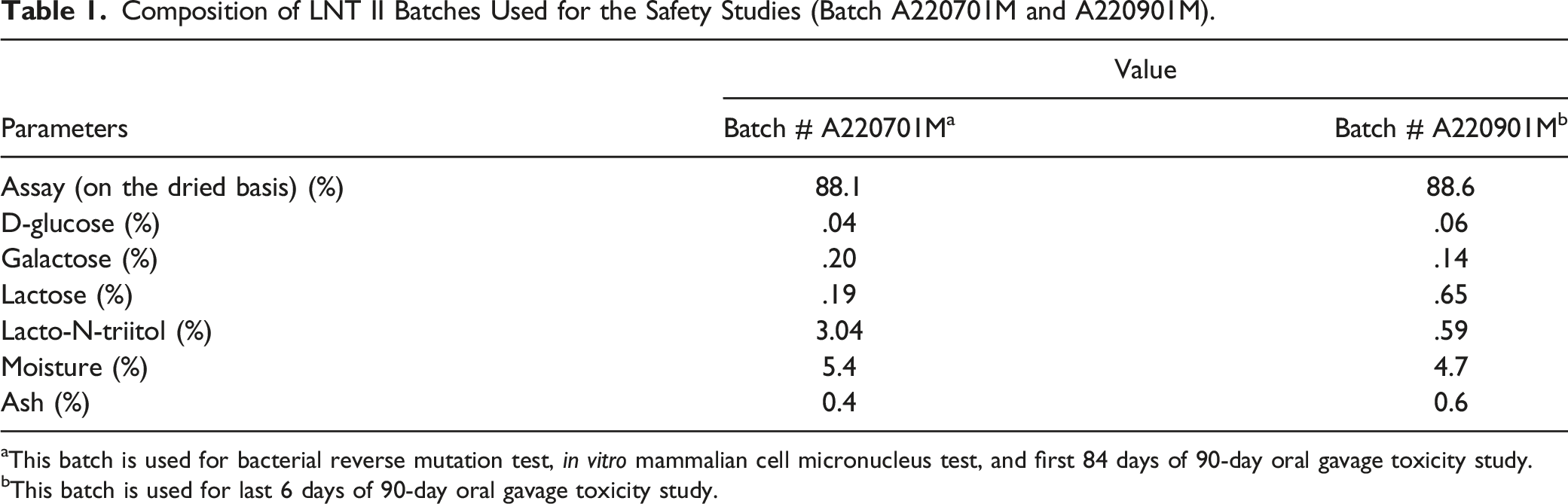
aThis batch is used for bacterial reverse mutation test, in vitro mammalian cell micronucleus test, and first 84 days of 90-day oral gavage toxicity study.
bThis batch is used for last 6 days of 90-day oral gavage toxicity study.
Test Animals
The 90-day toxicity study utilized Sprague-Dawley rats [Crl:CD (SD) SPF/VAF], obtained from Beijing Vital River Laboratory Animal Technology Co. Ltd. At the commencement of dosing, the rats were at postnatal day (PND) 19-22, with weight ranges of 31.4-72.3 g for females and 34.9-72.8 g for males. Prior to the study, rats were provided with softened food every day throughout their 6-day quarantine and acclimatization period at Pharmaron TSP. During this acclimation period, each rat was identified with a unique cage card and tail mark.
Bacterial Reverse Mutation (Ames) Assay
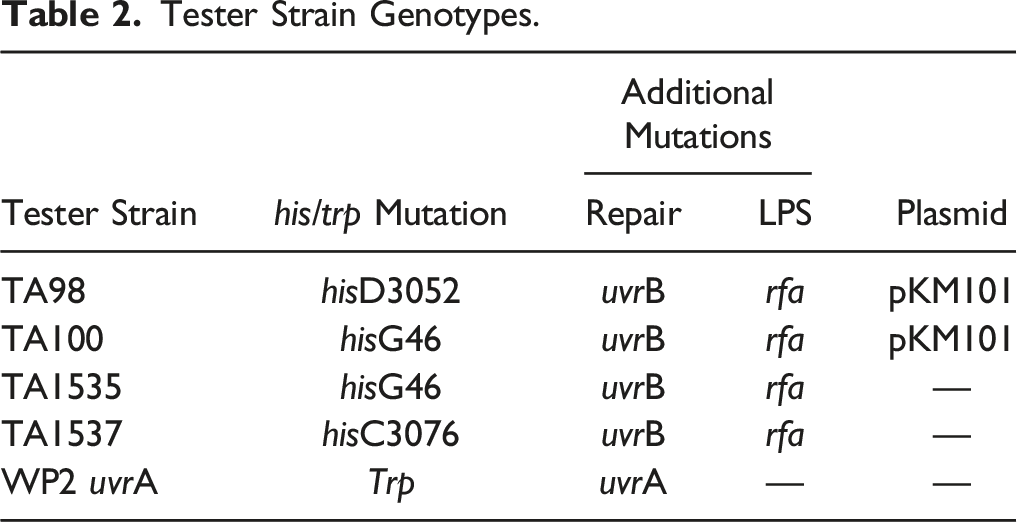
Tester Strain Genotypes.
Positive Controls.

2-AA, 2-aminoanthracene; 2-NF, 2-nitrofluorene; MNNG, N-Methyl-N-nitro-N-nitrosoguanidine.
In Vitro Micronucleus Assay
The clastogenic/aneugenic effects of LNT II were assessed via a micronucleus assay using Chinese hamster ovary (CHO-WBL) cells, both with and without an induced rat liver S9 activation system using β-naphthoflavone and phenobarbital. This assay determines whether the test article or its metabolites can inhibit cell cycle or induce micronuclei, particularly in cells cultivated in the presence of cytochalasin B (CYB), which inhibits cytokinesis, allowing only nuclear mitosis. 29 The choice of the CHO-WBL cell line was based on the OECD guideline 487, due to its demonstrated sensitivity for detecting clastogens/aneugens. 30 These cells were obtained from Litton Bionetics, Inc. (Maryland, USA), and underwent multiple rounds of cloning before storage in liquid nitrogen at the testing facility. They were routinely screened for chromosomal stability and mycoplasma contamination, and were not used after the 15th passage.
Sterile water for injection served as the solvent due to its compatibility with both the test article and target cells, CHO-WBL cells. The cells underwent LNT II treatment for 3 and 24 hours without S9 activation, and for 3 hours with S9 activation. Harvesting was performed 24 hours post LNT II treatment initiation.
The in vitro micronucleus assay utilized test article in solution with nominal LNT II concentrations of 100, 250, and 500 μg/mL, selected based on preliminary tests that revealed no precipitation or cytotoxicity. The exposure duration was 3 hours and 24 hours without S9, and 3 hours with S9 activation. The highest concentration of the test article did not exceed .5 mg/mL and was selected to ensure cytotoxicity did not significantly surpass 50% of concurrent solvent controls. The cytokinesis block proliferative index (CBPI) was calculated by adding the number of mononucleated cells to twice the number of binucleated cells and 3 times the number of multinucleated cells, divided by total cell count. Two thousand binucleated cells (1,000 per culture) were scored for each test and control concentration for micronucleated cell (MN-BN) counting.
Subchronic (90-Day) Oral Repeat Dose Toxicity Study
LNT II was administered orally to SD rats (via gavage) once daily, starting from PND 19-21. This treatment was segmented into 3 subsets: one lasting 13 weeks, another for 13 weeks followed by a 4-week recovery period to evaluate reversibility, progression, and other delayed effects, and the third subset lasting 43 days to examine development and reproductive function, a commonly used assessment for young animals. The study was conducted in compliance of with the OECD principles of GLP, followed OECD guidelines for the testing of chemicals (408)—Repeated dose 90-day oral toxicity study in rodents and FDA guidelines: Nonclinical safety evaluation of pediatric drug products, OECD guidelines for the testing of chemicals (414)—One-generation reproduction toxicity study.
The Animal Grouping Information.

Note: M, male; F, female; DIW, deionized water.
aDosing for consecutive 91 days, and were necropsied on day 92.
bDosing for consecutive 91 days, and were necropsied as recovery animals on day 120 after 4 weeks of recovery.
cDosing for 43 consecutive days, female rats were performed C-section examination on gestation day 15, and the male rats were performed sperm analysis and necropsy after cohabitation completion to assess development and reproductive function.
Dose levels of 1,500, 2,500, and 5,000 mg/kg body weight/day were established based on a 14-day dose range-finding toxicity study, which yielded no significant findings. These dose levels were selected for correlation with the doses used in the 90-day study. The testing formulation of LNT II was prepared by suspending the designated amount of LNT II in deionized water (DIW), which made up 60% of the final volume. This suspension was then homogenized using a combination of stirring and ultrasonication. After this, DIW was added incrementally to reach the desired final concentration. Dose formulations were prepared on a weekly basis. In accordance with stability data, aliquots were created for daily use and stored at a temperature of 5 ± 3°C, shielded from light. The dose volume was 10 mL/kg body weight once daily.
During the study period, various observations and assessments were conducted on animals. These included cageside observation, detailed clinical observations, body weight measurement, food consumption monitoring, ophthalmology examination, as well as developmental and behavioral assessments.
Observation
Cageside observations were performed twice daily beginning on study day 1 (first day of dosing) and thereafter. Detailed clinical observations were conducted once before dosing, twice weekly thereafter, and one day before and on the days of sacrifices of subset 1 and subset 2 animals. For mating females from subset 3, observations were conducted on gestation day 0 and every third day thereafter until gestation day 15. Detailed clinical observations included monitoring changes in skin, fur, eyes, mucous membranes, respiratory and circulatory systems, autonomic and central nervous systems, as well as somatomotor activity and behavioral patterns.
Body Weight and Food Consumption
Body weight evaluations were conducted once before dosing, twice weekly thereafter, and one day before and on the days of necropsies of subset 1 and subset 2 animals. For mating females from subset 3, these testing were conducted on gestation day 0 and every third day thereafter until gestation day 15. Food consumption was measured once on day 1, twice weekly thereafter, and once before sacrifices of subset 1 and subset 2 animals. For subset 3 animals, food consumption was measured once on day 1, twice weekly thereafter, and once on the day of cohabitation. Mating females in gestation were also monitored for food consumption once on gestation day 0 and every third day thereafter until gestation day 15.
Ophthalmology Examinations
Ophthalmology examinations using a pen light and indirect ophthalmoscope were conducted prior to randomization of animals, and then again with the addition of slit lamp were conducted once prior to the scheduled necropsies for subset 1 and subset 2 animals.
Neurobehavioral Evaluations
Functional observational battery examinations were conducted for subset 1 animals once prior to the scheduled terminal necropsy and for subset 2 animals once prior to the scheduled recovery necropsy.
Developmental and Behavioral Assessments
For developmental and behavioral assessments, various tests were conducted for animals in subset 3. Sexual maturity was assessed daily starting from study day 5 and study day 15 for females and males, respectively. Auditory startle response was evaluated on study day 34. Locomotor activity tests, designed to evaluate behavioral function, were conducted on study day 32. Estrous cycle evaluations were performed daily beginning on study day 56 for females. Additionally, a shuttle box test was administered on study days 10 and 38 to assess learning and memory. 31 Reproductive function for both males and females were evaluated by mating them in a 1:1 ratio on study day 70.
Clinical Pathology
Parameters Evaluated in Clinical Chemistry, Hematology, and Coagulation.

Urinalysis
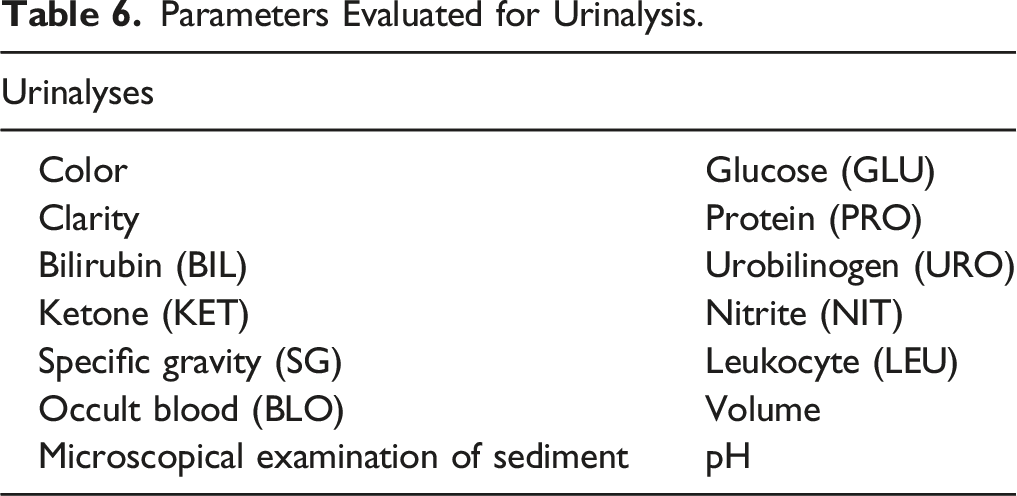
Parameters Evaluated for Urinalysis.
Necropsy
At the end of study, surviving animals were euthanized by exsanguination anesthesia via 70% CO2/30% O2 and subjected to detailed necropsy. This necropsy included a macroscopic examination of the external surface of the body, all orifices, the cranial cavity, the external surface of the brain, the thoracic, abdominal, and pelvic cavities along with their viscera, the cervical areas, the carcass, and the genitalia. Selected organs and tissues were weighed. Following the treatment period, selected organs/tissues were excised, tissues processed and staining, and microscopic evaluations were conducted from all animals in both the control and high-dose LNT II groups. Additionally, the length of the left tibia was measured in all animals in Subset 1 and 2 during the scheduled necropsy using a Vernier caliper. The organs or tissues weighed, collected, and placed in fixative and those examined histopathologically are listed in Supplemental Table 1.
Statistical Analysis
Quantitative results, including body weight, body weight gain, reproductive organ weight and relative to body weight (%), and food consumption, were analyzed using one-way analysis of variance (ANOVA) and Dunnett’s method, conducted in the Provantis™ 10.5 system which integrates the industry-standard SAS statistical tool.
Quantitative data comparisons between treatment and control groups were made, considering the homogeneity of the group variances. When homogeneous (P > .05), ANOVA was used, followed by Dunnett’s test for group mean comparisons if ANOVA revealed significant differences (P ≤ .05). For heterogeneous group variances (P ≤ .05), log transformation was performed. If the variance remained heterogeneous after transformation or data contained negative values, the non-parametric Kruskal-Wallis test was used, followed by Dunnett’s test on ranks for pairwise group comparisons if the Kruskal-Wallis test indicated significance (P ≤ .05).
Pairwise group comparisons of interest were undertaken via a two-sided test at the .05 significance level. Significant results were reported as P ≤ .01 or P ≤ .05. The data for males and females were analyzed separately with the significance threshold for all tests set at P < .05 (two-tailed).
Results
Genotoxicity Study
Bacterial Reverse Mutation (Ames) Assay
Results of Ames Assay Exposed to LNT II.
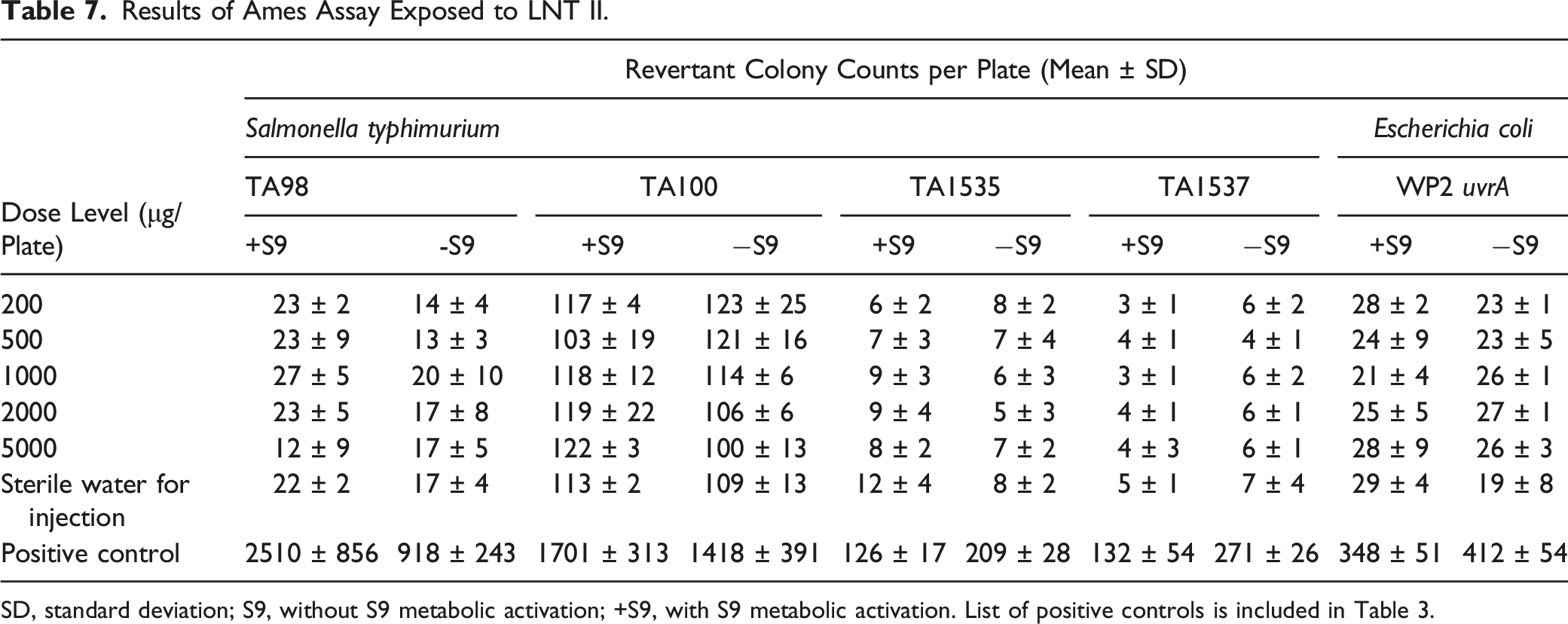
SD, standard deviation; S9, without S9 metabolic activation; +S9, with S9 metabolic activation. List of positive controls is included in Table 3.
In Vitro Micronucleus Assay
Result of in Vitro Micronucleus Assay Exposed to LNT II.
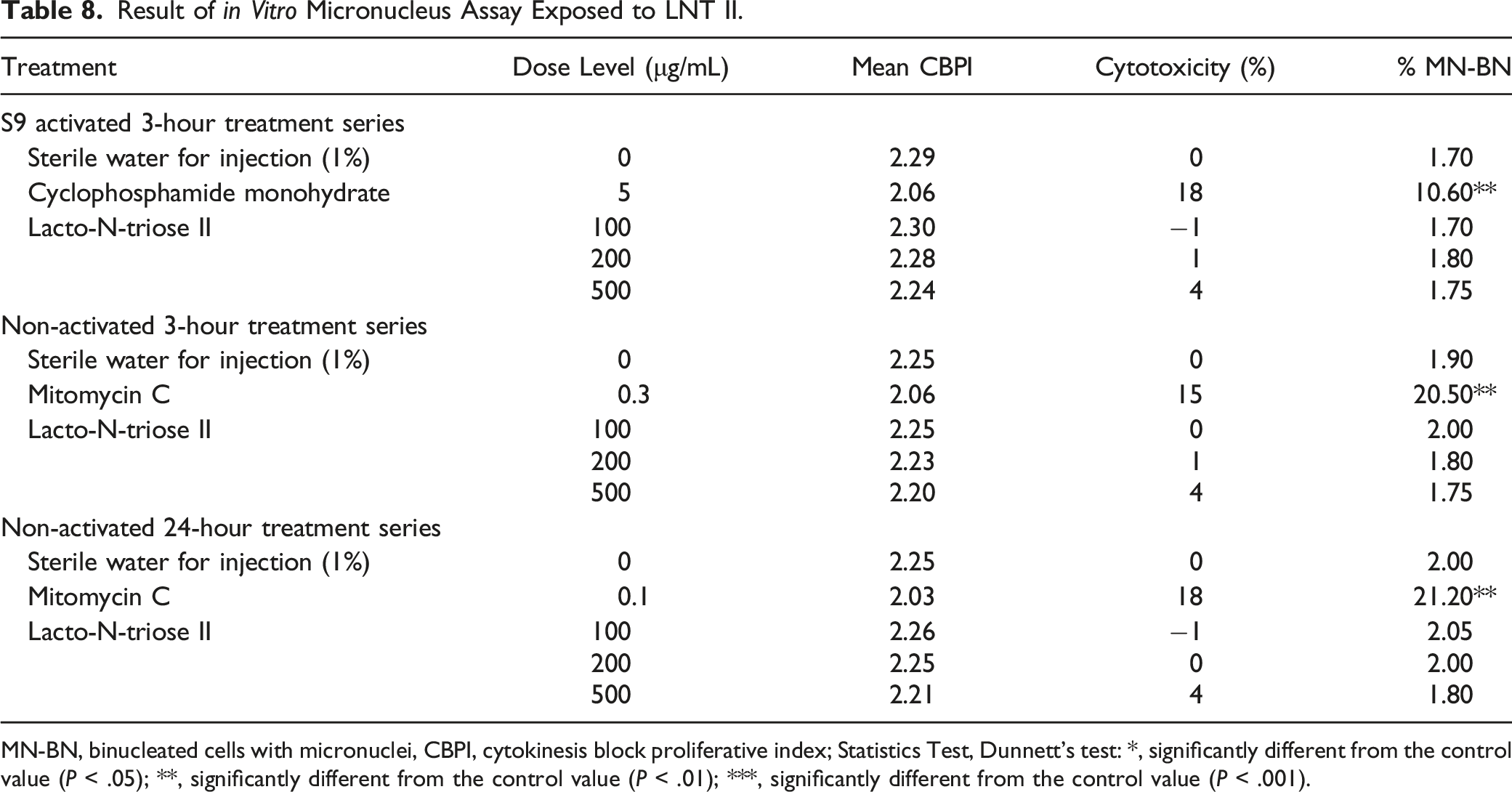
MN-BN, binucleated cells with micronuclei, CBPI, cytokinesis block proliferative index; Statistics Test, Dunnett's test: *, significantly different from the control value (P < .05); **, significantly different from the control value (P < .01); ***, significantly different from the control value (P < .001).
90-Day Repeat Dose Study
Mortality
All animals in the study survived until their scheduled necropsy.
Clinical Observations and Ophthalmoscopy
Summary of Clinical Observations.
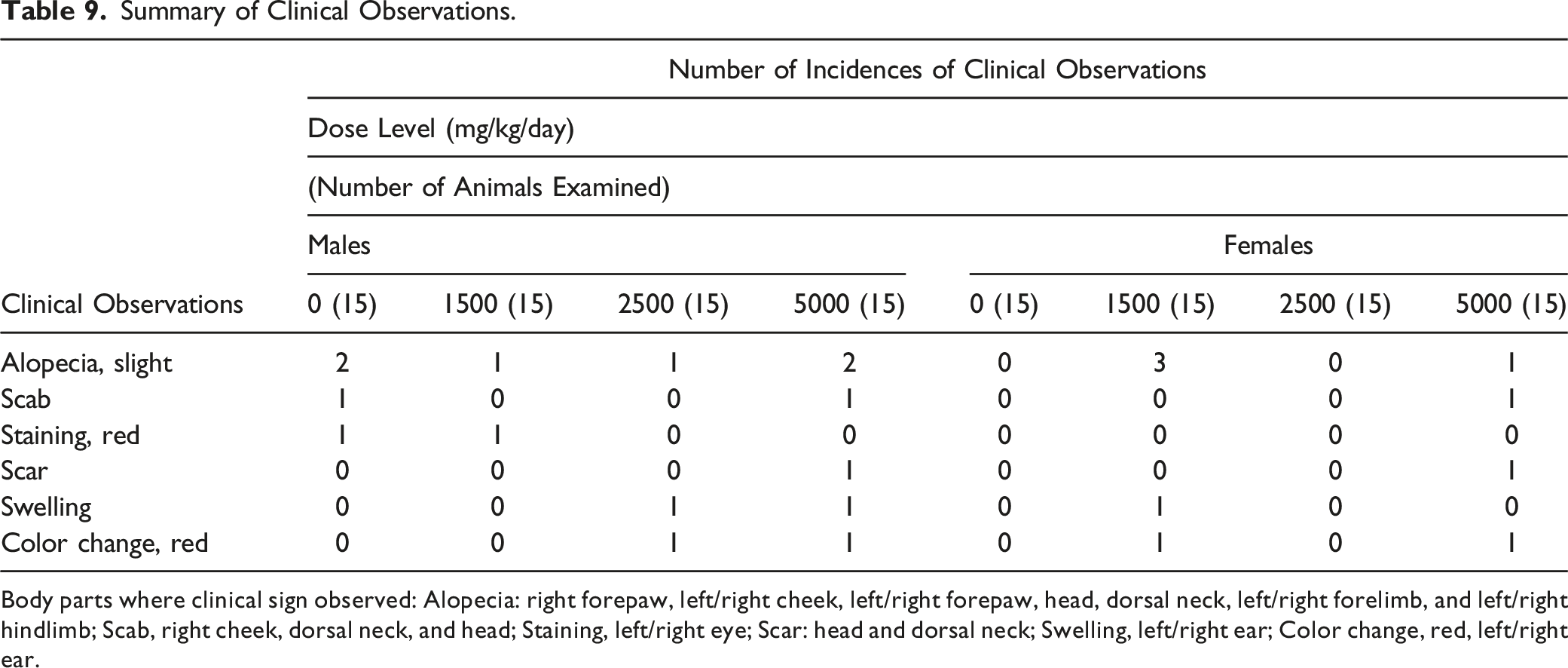
Body parts where clinical sign observed: Alopecia: right forepaw, left/right cheek, left/right forepaw, head, dorsal neck, left/right forelimb, and left/right hindlimb; Scab, right cheek, dorsal neck, and head; Staining, left/right eye; Scar: head and dorsal neck; Swelling, left/right ear; Color change, red, left/right ear.
Body Weight and Food Consumption
Animals gradually gained weight throughout the treatment and recovery periods without body weight decrease observed as presented in Figure 1A and B. Mean body weight for male and female rats during the 90-day treatment period. (A) Mean body weight of male rats during the 90-day treatment period. (B) Mean body weight of female rats during the 90-day treatment period.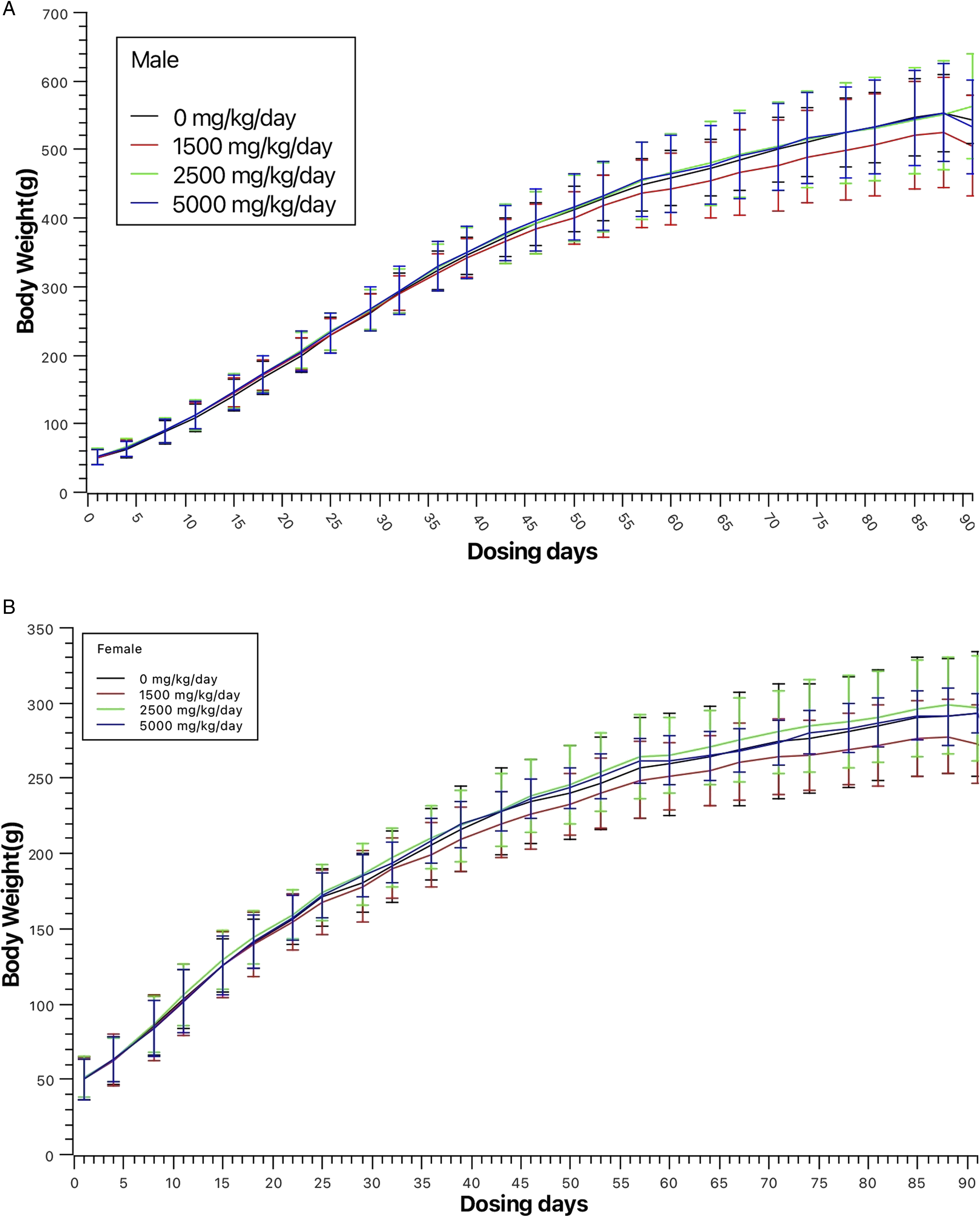
Statistically significant decreases in food consumption, by 6.8% to 12.3%, were observed in male rats from day 74 and in female rats from day 32 at doses ≥1,500 mg/kg/day of LNT II, which has a caloric content of 1598 kJ/100 g, in comparison to control groups. Despite these variations, body weight was not affected, as the rats continued to gain weight during both the treatment and recovery periods. The lowered food consumption did not significantly impact overall body weight. Such decreases in food consumption, without subsequent body weight impact, have been reported in scientific literature and are not considered biologically significant or adverse.
32
Mean body weights for male and female rats during the dosing period are depicted in Figure 1A and B, while Figure 2B and B illustrate mean food consumption of during the same period. Food consumption for male and female rats during the 90-day treatment period. (A) Mean food consumption for male rats during the 90-day treatment period. (B) Mean food consumption for female rats during the 90-day treatment period.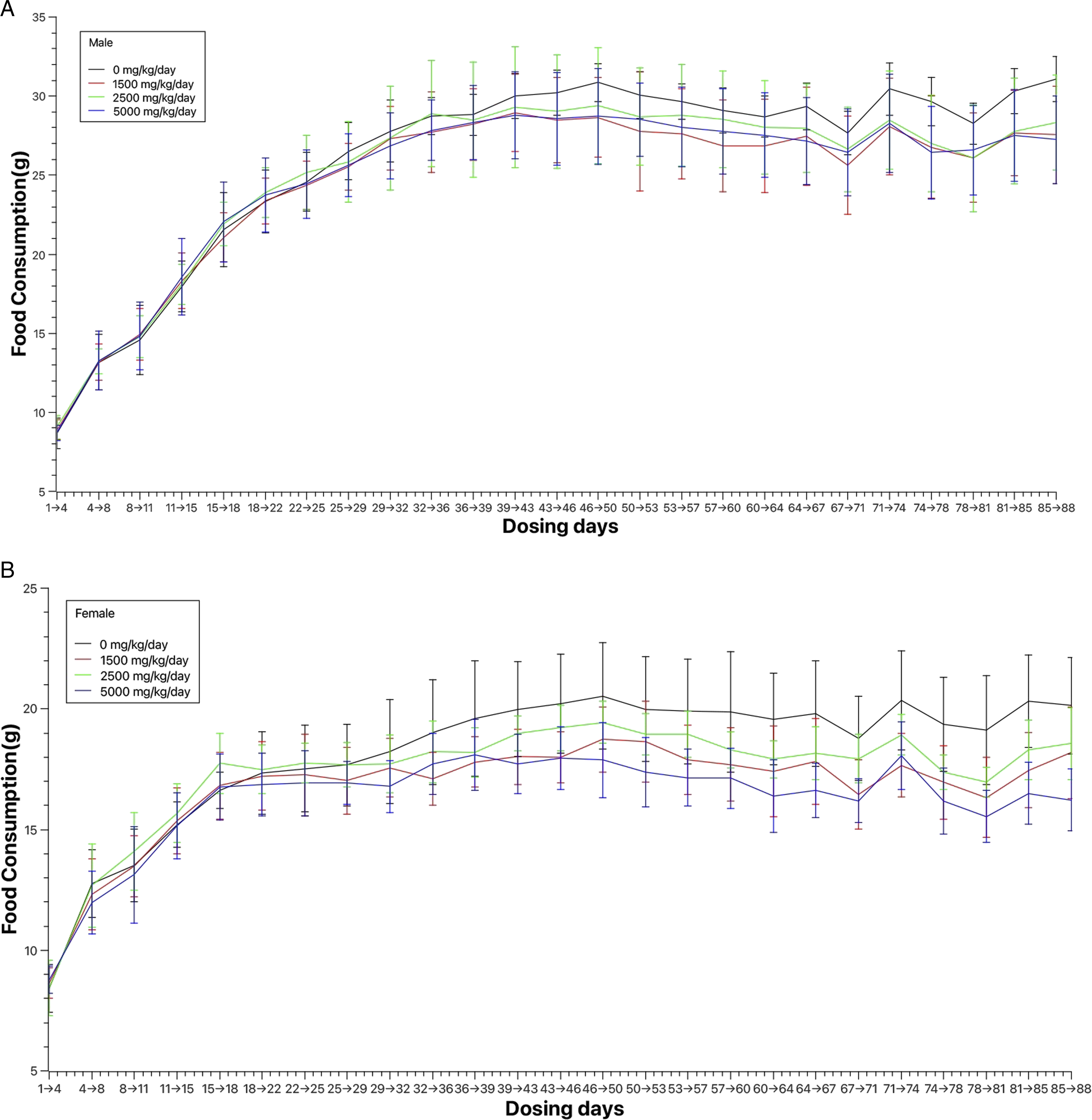
Developmental and Behavioral Assessments
There were no behavioral, learning and memory function parameters considered to be related to administration of LNT II.
Clinical Chemistry
Blood Chemistry Values for Male and Females Administered LNT II for At Least 90 Days.
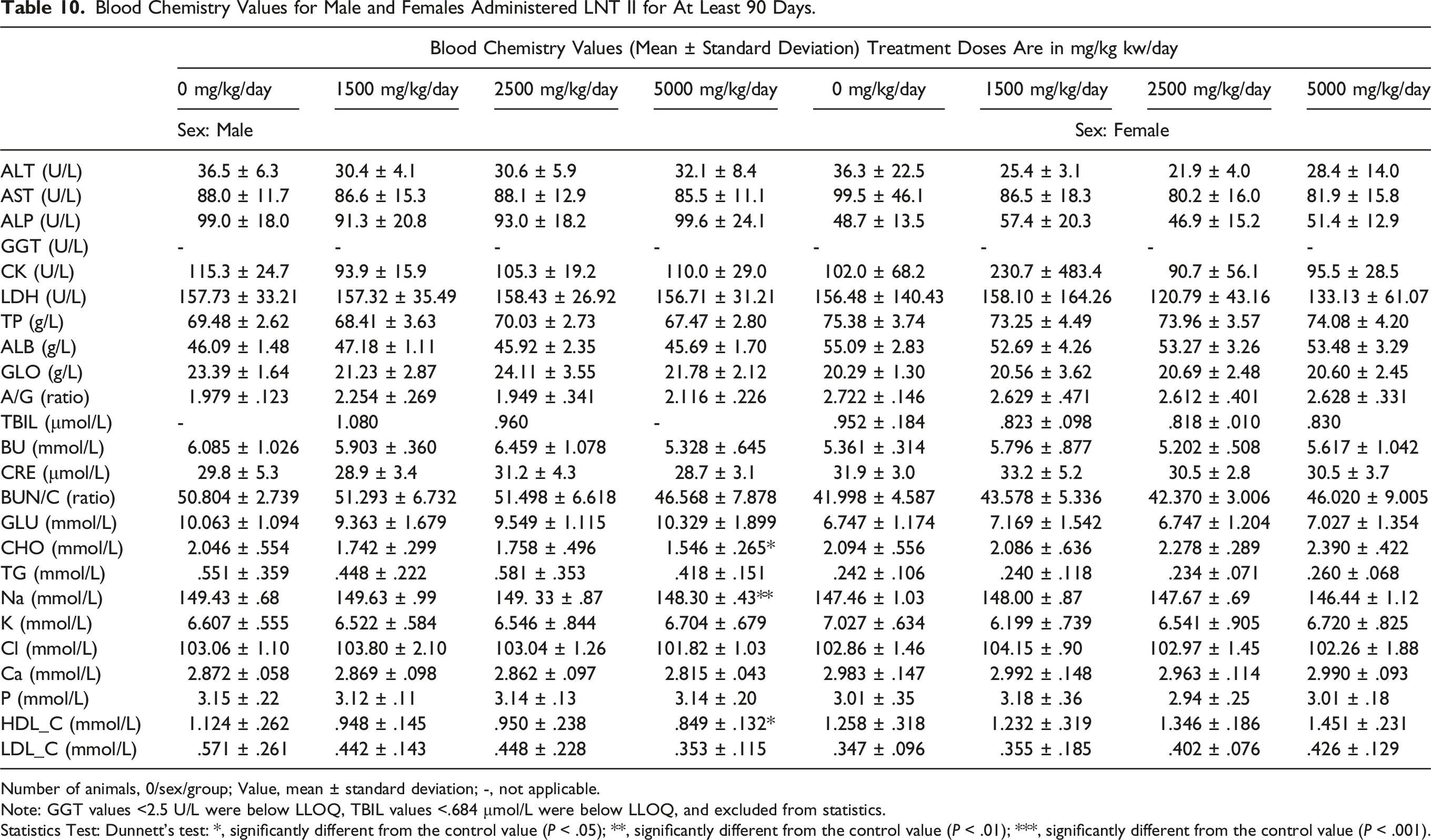
Number of animals, 0/sex/group; Value, mean ± standard deviation; -, not applicable.
Note: GGT values <2.5 U/L were below LLOQ, TBIL values <.684 μmol/L were below LLOQ, and excluded from statistics.
Statistics Test: Dunnett's test: *, significantly different from the control value (P < .05); **, significantly different from the control value (P < .01); ***, significantly different from the control value (P < .001).
Average phosphorus value for females administered 2,500 mg/kg·bw/day for 90 days with 28-day recovery was significantly higher (4.00 mmol/L, P < .05) compared to controls (3.24 mmol/L), females given 1,500 mg/kg bw/day (3.30 mmol/L) and 5000 mg/kg·bw/day (3.62 mmol/L). These differences were not observed in males, or in females treated with LNT II at different doses for 90 days or following the recovery period. No significant microscopic observations were found in animals treated with 2,500 mg/kg·bw/day that could account for these changes. Thus, the increase in phosphorus levels cannot be attributed to LNT II administration.
Hematology and Coagulation
Summary of Hematology Result of Males and Females Administered LNT II for At Least 90 Days.
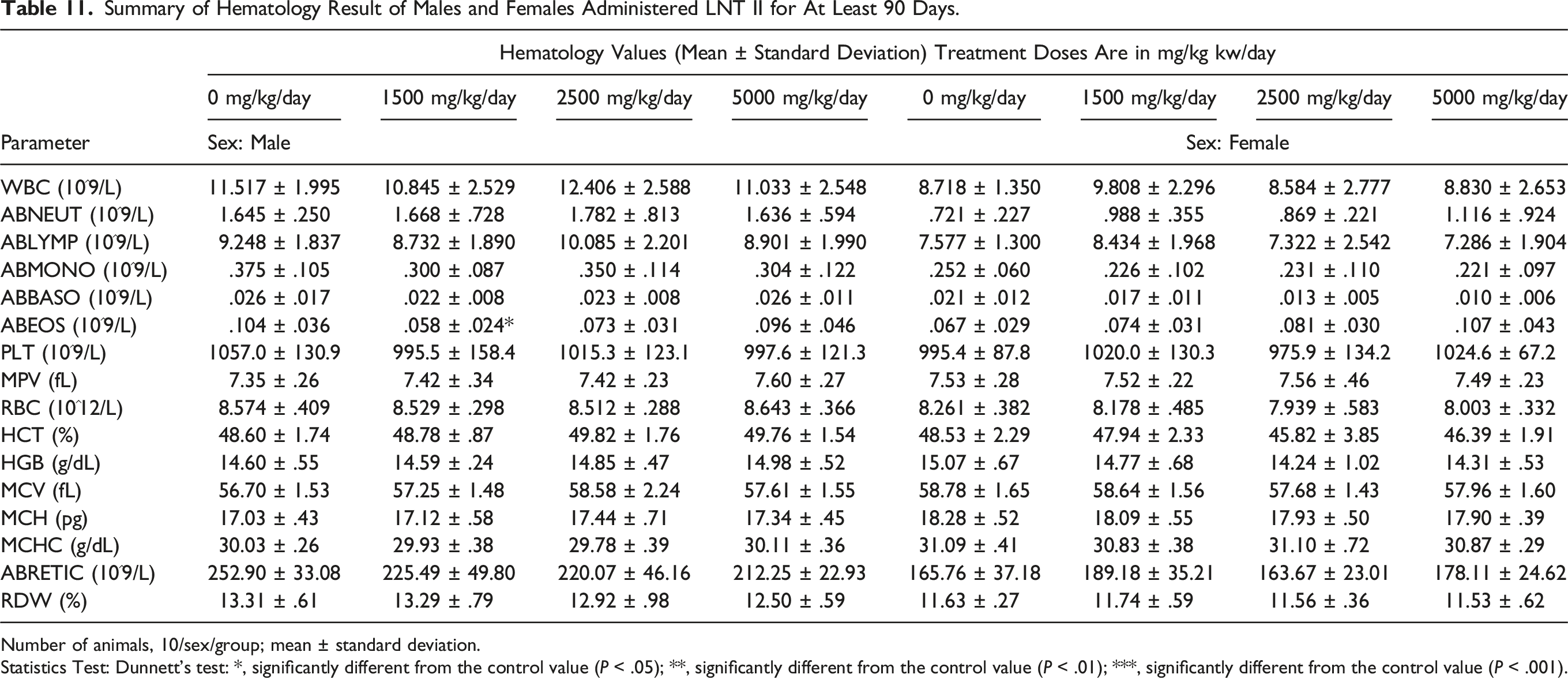
Number of animals, 10/sex/group; mean ± standard deviation.
Statistics Test: Dunnett's test: *, significantly different from the control value (P < .05); **, significantly different from the control value (P < .01); ***, significantly different from the control value (P < .001).
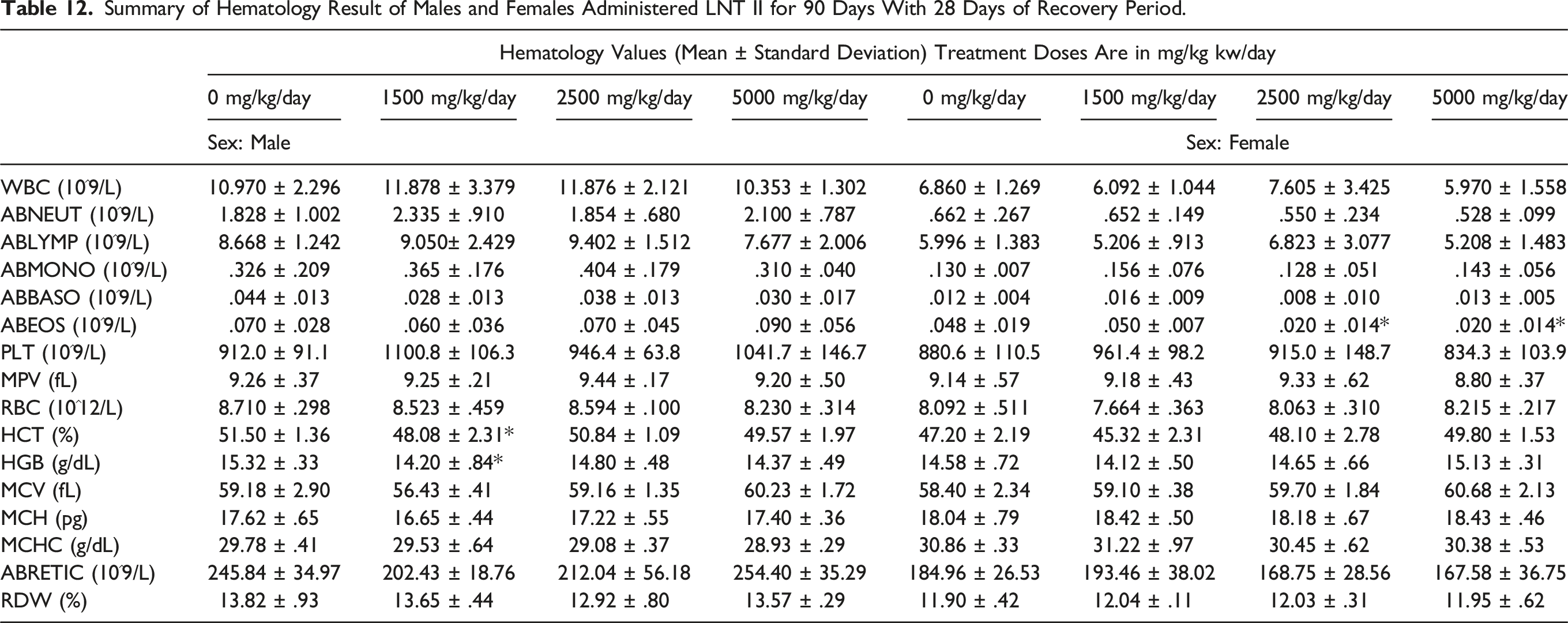
Summary of Hematology Result of Males and Females Administered LNT II for 90 Days With 28 Days of Recovery Period.
Summary of Coagulation Result.
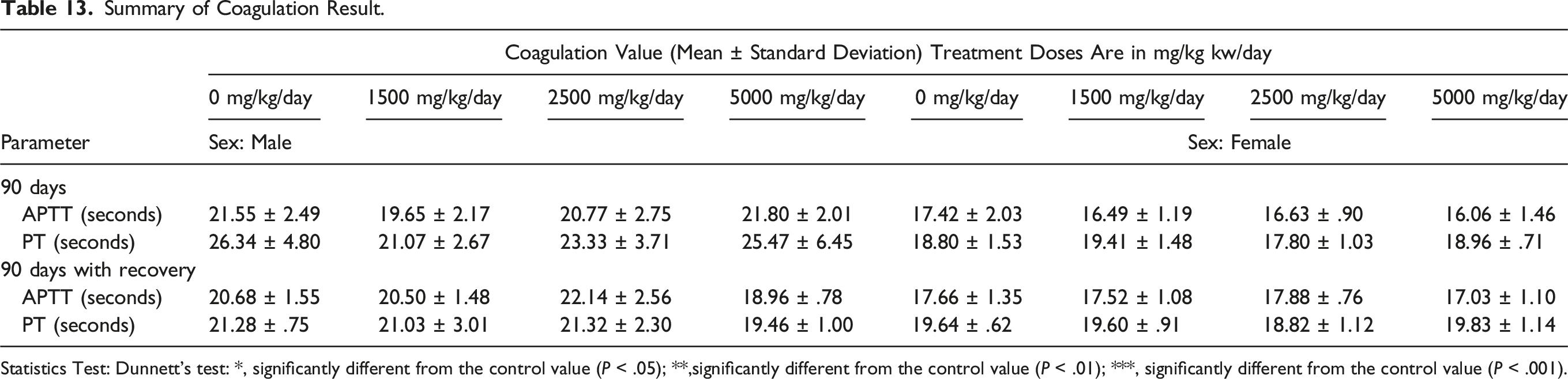
Statistics Test: Dunnett's test: *, significantly different from the control value (P < .05); **,significantly different from the control value (P < .01); ***, significantly different from the control value (P < .001).
Urinalyses
No LNT II related changes were observed in the urinalysis parameters evaluated during the treatment and recovery phase. However, positive Bilirubin (Bil) and Protein (Pro) were detected in both male and female animals, including the control group. Therefore, it is concluded that these changes are not related to the administration of LNT II.
Organ Weights
Absolute Organ Weights for Male and Female Rats Administered LNT by Oral Gavage for At Least 90 Days.
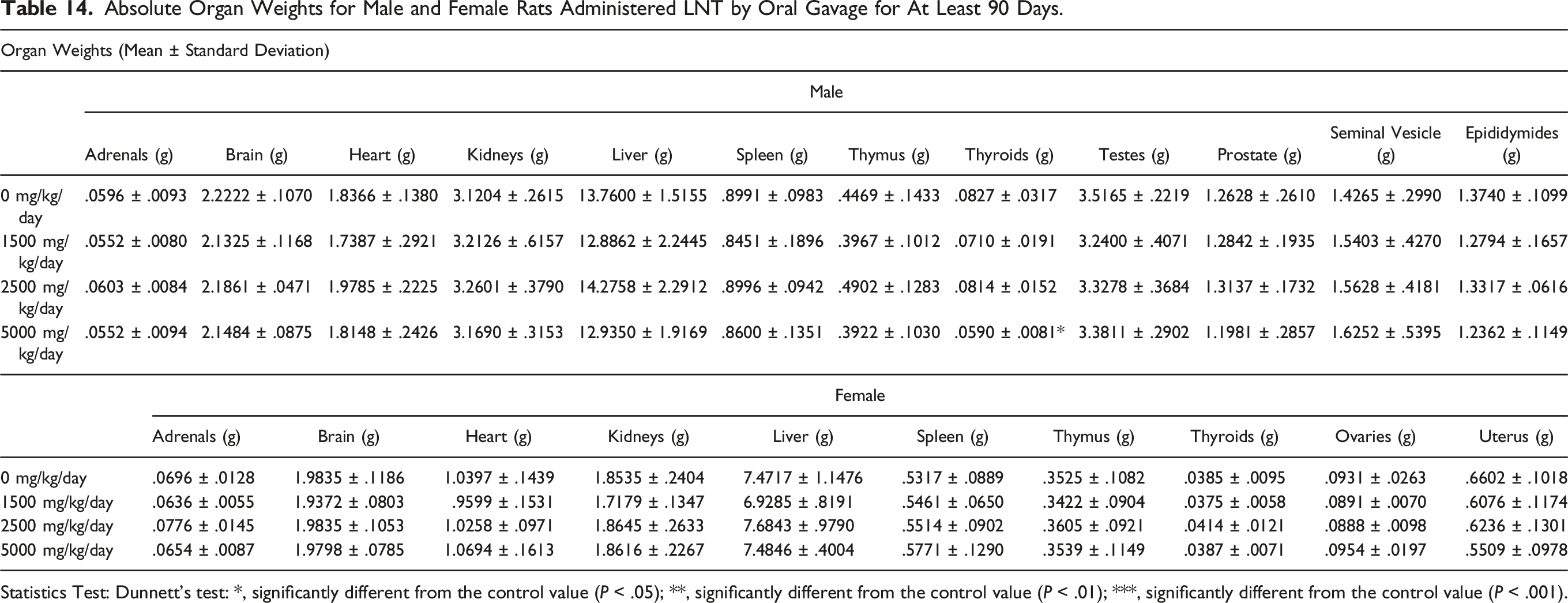
Statistics Test: Dunnett's test: *, significantly different from the control value (P < .05); **, significantly different from the control value (P < .01); ***, significantly different from the control value (P < .001).
Absolute Organ Weights for Male and Female Rats Administered LNT by Oral Gavage for At Least 90 Days With 28 Days of Recovery Period.
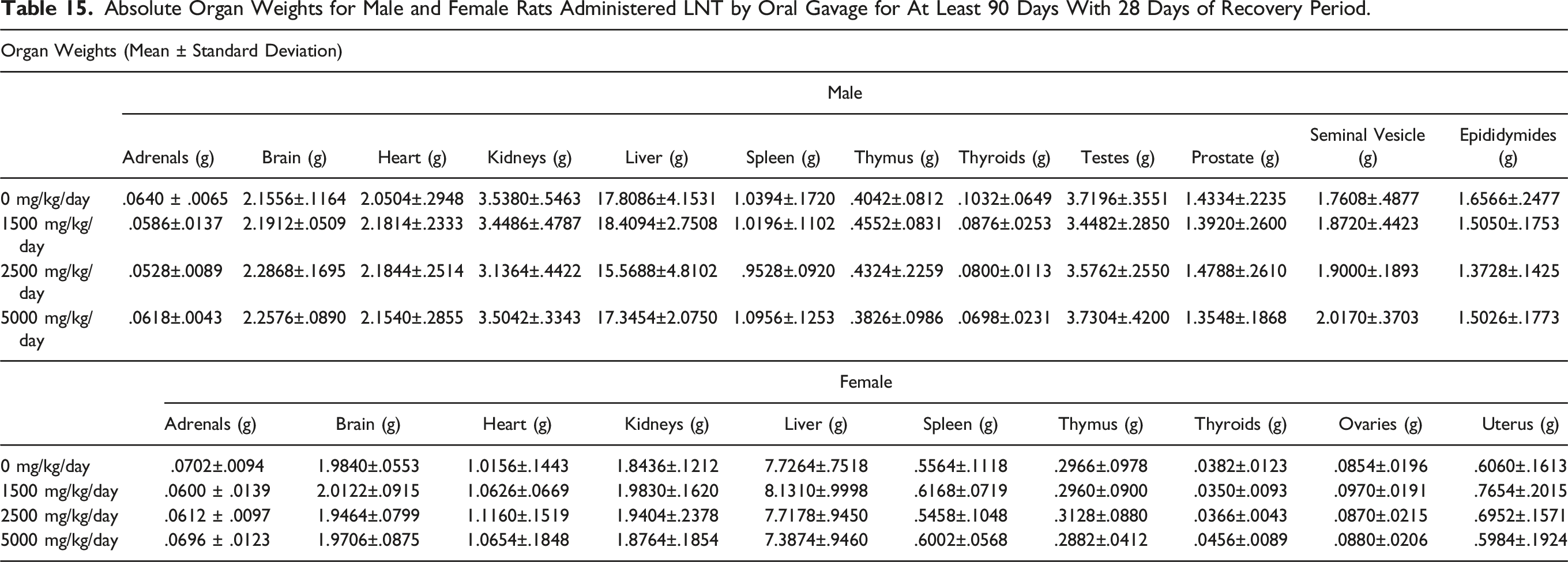
Macroscopic and Microscopic Findings
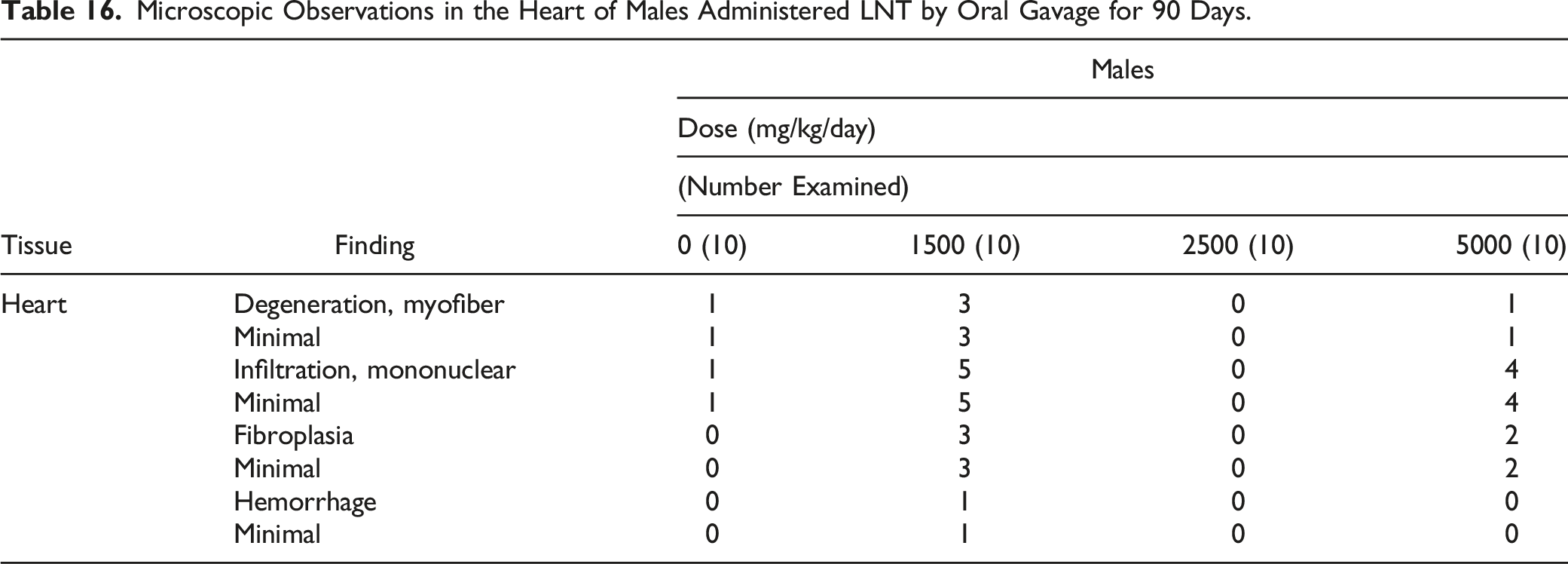
Microscopic Observations in the Heart of Males Administered LNT by Oral Gavage for 90 Days.
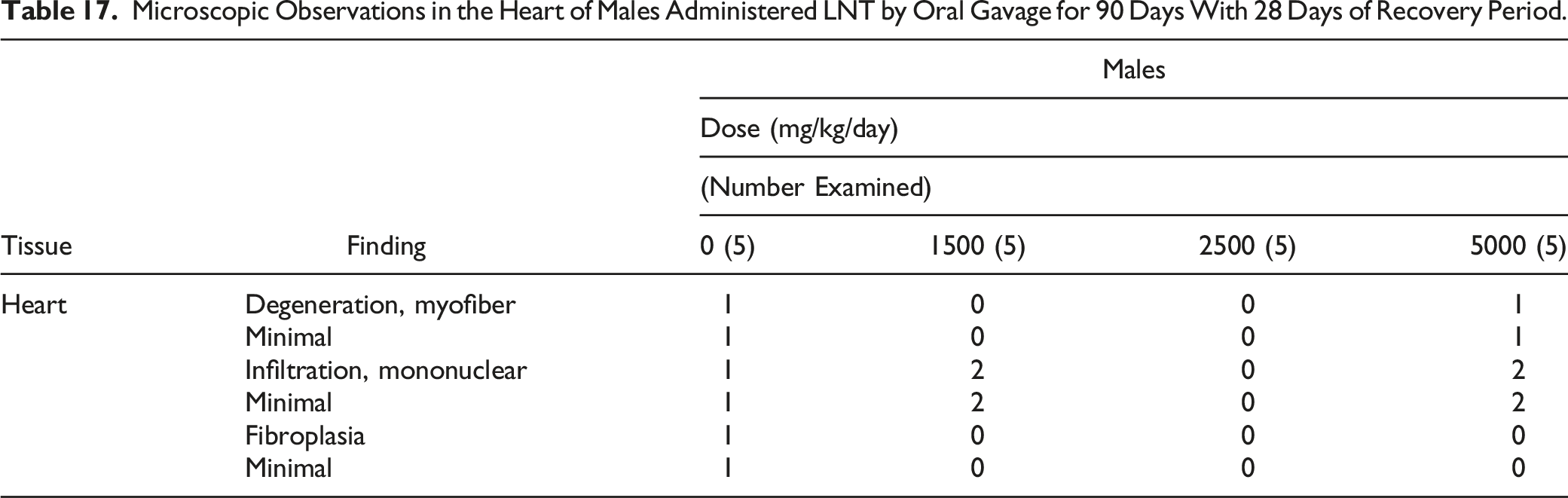
Microscopic Observations in the Heart of Males Administered LNT by Oral Gavage for 90 Days With 28 Days of Recovery Period.
Discussion
The potential advantages of HMOs for infant digestion, gut health, and immune system improvements have inspired a surge of research. However, the restricted supply of human milk creates a significant barrier to securing ample quantities of LNT II for broader use in infant or toddler formulas and functional foods. This predicament amplifies the need for efficient and safe alternative production methods. As a part of these efforts, LNT II, a tri-oligosaccharide found in human milk but absent in cow’s milk, has been evaluated for safety as a prospective new ingredient in young children 1 to 3 years of age foods. This safety examination employed standard protocols, which included in vitro genotoxicity tests and 90-day oral gavage studies on juvenile rats, mirroring previous studies on synthetic HMOs such as 2’-FL and LNnT.
The safety evaluation for human consumption of LNT II included assessments of potential genotoxicity via 2 in vitro genotoxicity assays: the bacterial reverse mutation assay and the in vitro mammalian cell micronucleus test. These represent the standard genotoxicity testing protocol covering major genetic endpoints with the minimum recommended tests. A 90-day study on juvenile rats from PND 19 was used to determine potential subchronic toxicity of orally administered LNT II, with a focus on the target population. A 14-day range-finding study was conducted to identify short-term effects, particularly gastrointestinal intolerance, at intended doses for the longer study.
No adverse effects were observed for LNT II up to 5,000 mg/kg bw/day based on various health indicators, suggesting it is safe for use in infant foods. The NOAEL was established at 5,000 mg/kg bw/day, in line with previous studies on HMOs.
This research provides a clear understanding of LNT II's upper tolerance level in rats, serving as a foundation for future human infant exposure estimations. Our findings strongly advocate for the safety of LN T II in infant foods.
In conclusion, the biosynthesized human milk oligosaccharide LNT II, evaluated across multiple studies, was non-genotoxic and well-tolerated in rats. Given the structural equivalence, safe exposure history, previous clinical data, and current preclinical investigations, LNT II’s safety for intended use is confirmed.
Supplemental Material
Supplemental Material - Preclinical Safety Evaluation of the Human-Identical Milk Oligosaccharide Lacto-N-Triose II
Supplemental Material for Preclinical Safety Evaluation of the Human-Identical Milk Oligosaccharide Lacto-N-Triose II by Xu Fang, Wei Zhu, Huiying Zhao, Wencheng Yin, Xiao Song, and Fengxin Liu in International Journal of Toxicology.
Footnotes
Acknowledgments
The authors wish to thank Dr Sanford Bigelow for reviewing the manuscript prior to journal submission.
Author Contributions
Fang, X. contributed to conception and design, contributed to interpretation, and critically revised manuscript; Zhu, W. contributed to design, contributed to analysis and interpretation, and drafted manuscript; Zhao, H. contributed to design, contributed to acquisition, analysis, and interpretation, and critically revised manuscript; Yin, W. contributed to conception, contributed to interpretation, and drafted manuscript; Song, X. contributed to design, contributed to interpretation, and critically revised manuscript; Liu, F. contributed to design and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was sponsored by Shandong Henglu Biotechnology Co. Ltd, who provided funding for the research.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
