Abstract
Oxazyme (OC4) is an orally administered formulation that has as an active component a recombinant mutant form of Bacillus subtilis oxalate decarboxylase (OxDC) enzyme C383S, designed to degrade dietary oxalate in the stomach. Fourteen-day repeat-dose studies were conducted in rats and dogs to evaluate toxicity and determine a no observed adverse effect level (NOAEL). Animals were administered OC4 by oral gavage twice daily for 14 consecutive days. Reversibility, progression, and delayed appearance of any observed changes were evaluated in a subset of animals that underwent a recovery of 7 days following 14 days of control or test-article. There were no test-article-related adverse effects or deaths in either species. Results indicate that the NOAEL under the conditions used in the studies was 720.8 mg/kg/d in rats and 187.2 mg/kg/d in dogs, the high dose tested in each species.
Oxalate is a metabolic end product in humans. It is endogenously produced by the liver and also derived by absorption from the diet. The majority of oxalate is eliminated from the body via the kidneys, with a small percentage eliminated through the gastrointestinal (GI) tract. Oxalate forms a calcium-oxalate salt, which is insoluble at physiological pH, and its accumulation can result in serious renal conditions. Consistent high levels of urinary oxalate can result in recurrent kidney stones and renal complications. 1-3 Six percent to 12% of the population in Western civilizations develop occasional or recurrent kidney stones during their lifetime. 4,5
Hyperabsorption of dietary oxalate unaccompanied by any underlying GI disease is seen in 20% to 30% of recurrent calcium-oxalate stone formers. 6 The underlying cause of this increased absorption is not known, but these individuals are known to respond to low-oxalate diets. As an alternative to dietary restriction, ingestion of oxalate decarboxylase (OxDC) may reduce urinary oxalate levels in individuals by enzymatically degrading dietary oxalate prior to absorption, starting in the stomach.
Oxalate decarboxylases are common enzymes found in nature, most often as extracellular proteins produced by fungi, and are thought to control extracellular oxalate levels. Two edible fungi consumed in the United States, Portabella and Enoki, produce OxDC for oxalic acid control. 7,8 Furthermore, natto, a common Japanese dish made from fermented soybeans by way of the bacterium Bacillus subtilis, contains OxDC and is frequently ingested by humans with no apparent adverse effects. 9
The active component of OC4 (Oxazyme) is a recombinant mutant B subtilis OxDC enzyme, C383S. OxDC from B subtilis has 1 C-terminal cysteine residue, which tends to form disulfide linkages, thus accounting for the majority of OxDC aggregates detected during purification and storage (unpublished results; Li, 2006). Therefore, this cysteine residue at position 383 was replaced with serine to prevent cysteine-induced aggregates while retaining all catalytic properties. Hereafter, this C383S mutant of OxDC from B subtilis is simply referred to as OxDC.
OxDC catalyzes the cleavage of the oxalate C-C bond, yielding CO2 and formate (Figure 1 ), and has been shown to be specific for oxalate as substrate. 10 This enzyme exhibits optimal activity at acidic pH (3.5-5.0) and needs only catalytic quantities of molecular oxygen. 11 In its native state, OxDC can degrade approximately 2 to 3 mg of oxalate per minute per 1 mg of enzyme. The daily oxalate intake of individuals consuming Western diets has been shown to lie within the range of 44 to 351 mg/d. 12 By degrading dietary oxalate in the stomach, prior to its absorption, OxDC may lower urinary oxalate levels and thereby reduce the risk for kidney stone formation.
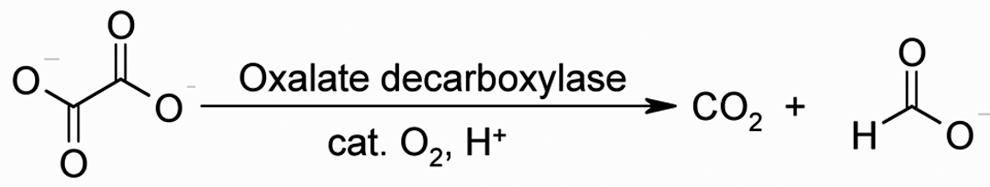
Reaction mechanism of oxalate decarboxylase (OxDC).
Toxicity studies were conducted to evaluate the safety of OC4 administered orally twice daily for 14 consecutive days in rats and dogs. Reversibility, progression, and delayed appearance of changes during a 7-day postdose recovery period were evaluated in recovery group animals from both studies.
Methods
Test and Control Materials
The test article in both the rat and dog study was powdered OC4. In OC4, OxDC is physically entrapped in microencapsulated particles consisting of Eudragit L100-55 (a copolymer used as an excipient in a number of approved drugs), trehalose (classified as generally recognized as safe [GRAS] in GRN-000045), and triethyl citrate (affirmed GRAS in 21 CFR 184.1911). The particles were spray-dried, resulting in a final cream-colored dry powder. Two lots (OC4-SB-081114-1-A1-C1-N1-D1 and OC4-SB-081114-1-A1-C1-N2-D1) manufactured for Good Laboratory Practice (GLP) preclinical studies were provided for the rat and dog toxicity studies described herein. The powdered test article was stored refrigerated at 5°C ± 3°C in foil pouches pending suspension in vehicle for preparation of OC4 dose suspensions.
The vehicle, 50 mM sodium citrate solution pH 4.0, was manufactured by Hyclone (Logan, UT) (lot RRJ082403) and maintained refrigerated at 5°C ± 3°C pending use for suspension of OC4 and subsequent dosing.
The high dose administered to either species was selected to be greater than 50 times the anticipated clinical dose. The test article was suspended in vehicle at 72 mg/mL and 90.1 mg/mL to provide high-dose suspensions for dogs and rats, respectively, whereas the control was vehicle alone. This suspension was diluted ~10-fold (to 7.2 mg/mL and 9.22 mg/mL with stirring) in citrate buffer to provide the low-dose suspensions. Dose suspensions were used within 1 week of preparation and were stored refrigerated at 5°C ± 3°C between doses. The dose volumes administered to rats and dogs were 4.0 mL/kg and 1.3 mL/kg, respectively. Individual dose volumes were calculated using the most recent body weights.
Retention samples were collected from dose suspensions to demonstrate stability and uniformity. Three separate 1-mL retention samples were obtained from the top, middle, and bottom of the dose suspensions while stirring. Separate samples were collected from high- and low-concentration test-article dose suspensions following the first dose administrations on days 1 and 14.
Study Organization and Regulatory Status
The 14-day rat repeat-dose study was conducted at MPI Research (Mattawan, MI) and the 14-day dog repeat-dose study at Comparative Biosciences (Sunnyvale, CA) in accordance with the GLP regulations of the US Food and Drug Administration (FDA) 13 and the Organization for Economic Cooperation and Development (OECD). 14 Except for having only 2 rather than 3 test-article dose groups, both studies comply with Toxicological Principles for the Safety Assessment of Food Ingredients 15 and with OECD Guidelines for the testing of chemicals for both rodents and nonrodents 16,17 except for the 14-day duration rather than 28 or 90 days.
Animals and Maintenance
Rats
A total of 63 male and 63 female CD [Crl:CD (SD)] rats approximately 6 weeks of age were obtained from Charles River Laboratories (Portage, Mich). During the 8-day acclimation period, the animals were observed daily with respect to general health and clinical signs of abnormality. Prior to the start of dosing, the animals were given a mock dose of tap water twice daily on 2 occasions in the same manner and same volume intended for use on study to acclimate them to the gavage procedure. Using a simple randomization procedure, 114 rats (57 males weighing 168-204 g and 57 females weighing 141-186 g at randomization) were chosen for the study. The control and both dosing groups included 30 animals, 15 of each sex: 5 animals of each sex were assigned to recovery groups. Additionally, 4 rats of each sex constituted the toxicokinetic study control group, and 8 rats of each sex received the high dose as the toxicokinetic test (TK) group. Each animal was assigned an animal number and was implanted with a microchip bearing a unique study identification number. Animals assigned to study had body weights within ±20% of the mean body weight for each sex.
The animals were individually housed in suspended, stainless steel, wire-mesh cages in an environmentally controlled room (temperature and relative humidity). The designated ranges were 64°F to 79°F and 30% to 70%, respectively. Fluorescent lighting was provided for approximately 12 hours per day.
Meal Lab Diet (Certified Rodent Diet 5002, PMI Nutrition International, Richmond, IN) was available ad libitum, except during designated periods. Tap water was available ad libitum via an automatic watering system. The water supply was monitored for specified contaminants at periodic intervals.
Dogs
A total of 40 Beagle dogs (Canis lupus familiaris; 20 males, 20 females), age 8 to 12 months and weighing 5 to 9 kg, were obtained from Marshall BioResources (North Rose, NY). Animal arrival was followed by a 5-day acclimation period with daily observations. Following acclimation and prior to the first dose administration, animals received pretreatment veterinary, ophthalmologic, and cardiologic/respiratory examinations.
Upon receipt at the testing facility, dogs were assigned sequential temporary animal identification numbers, which in addition to the ear tattoo number (breeder assigned) and study number served as identification until random assignment to study groups. Upon assignment to a study group, each animal was weighed, assigned a permanent identification number, and ear-marked with a unique permanent identification number. The animal groups (Table 1 ) included 6 dogs of each sex, 2 of which were randomly assigned to the recovery groups.
Dose Levels and Group Composition
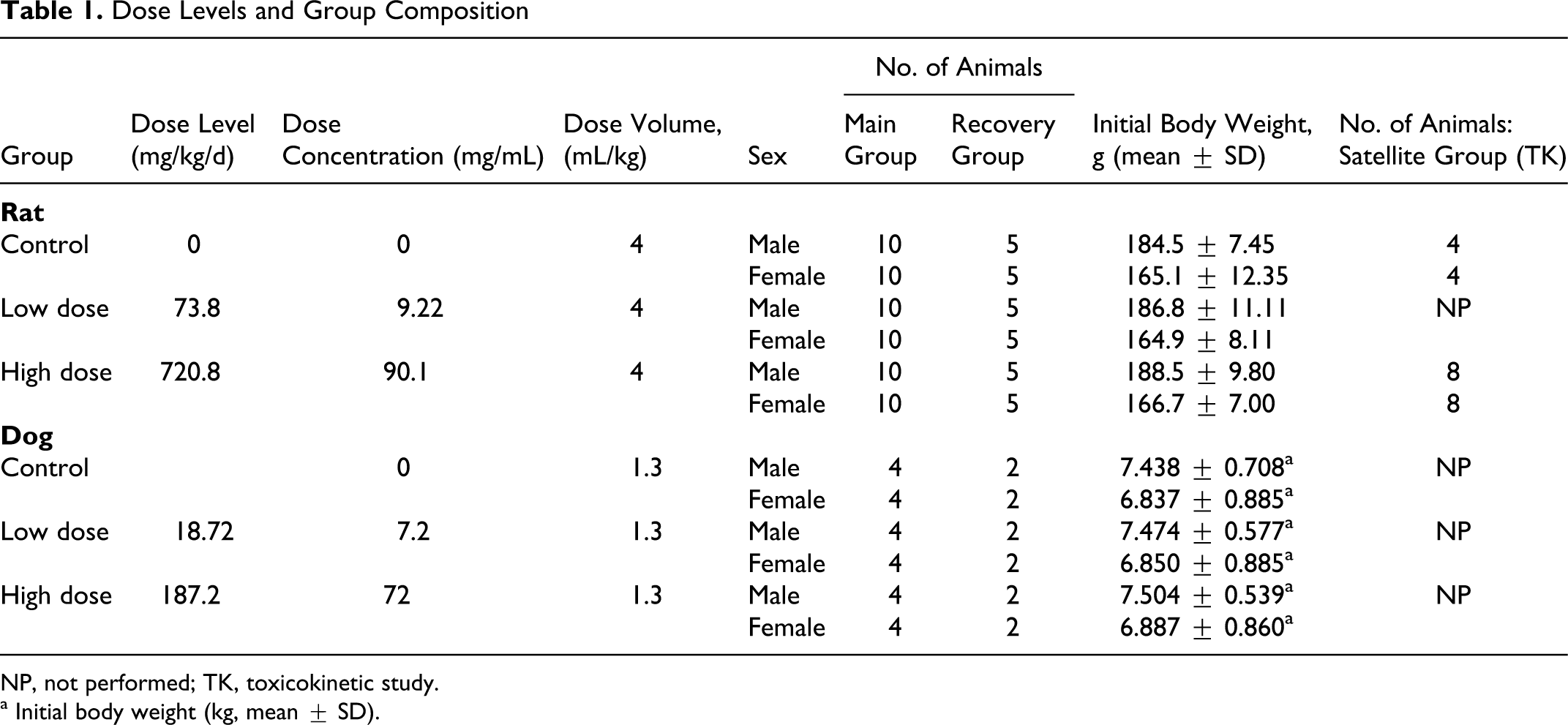
NP, not performed; TK, toxicokinetic study.
a Initial body weight (kg, mean ± SD).
Animals were housed individually in stainless steel cages with elevated grid floors within a room dedicated to dogs. General procedures for animal housing and husbandry met all regulations concerning use of animals in research, including the US Department of Agriculture regulations 18 implementing the Animal Welfare Act 19 and the recommendations of the National Research Council’s Guide for Care and Use of Laboratory Animals. 20
The temperature of the animal rooms was set at 64°F to 84°F. The relative humidity (RH) was not regulated but was measured and recorded daily. The RH generally fell between 30% and 70%, the range specified by the National Institutes of Health. Environmental parameters were monitored and recorded daily. Fluorescent lighting was provided for approximately 12 hours per day.
LabDiet 5007 Certified Canine Diet (Purina Mills, St Louis, MO) was fed throughout the acclimation, treatment, and recovery periods. Animals were fed ~300 g once a day prior to the first of 2 daily doses. Prior to necropsy, dogs were fasted overnight. Fresh tap water was provided ad libitum to the animals via water pans. The water supply was periodically monitored by the testing facility for chlorine content and bacterial contamination.
All animals appeared normal and were approved for use in the study after exam and assessment of an electrocardiogram. On day –1, prior to the start of dose administration, body weights of the 36 study animals (18 males, 18 females) were recorded.
Administration of Test Article
Rats and dogs
Homogeneous vehicle and test-article suspensions were administered in 2 equal daily doses via oral gavage, at least 6 hours apart, for 14 consecutive days. The total daily dose levels for the rat study were 0, 73.8, and 720.8 mg/kg/d, at a dose volume of 4 mL/kg/dose (Table 1). Dogs were administered 0, 18.72, or 187.2 mg/kg/d at a dose volume of 1.3 mL/kg/dose (Table 1). The TK study animals (rats only) received the vehicle or test article in the same manner as the main study animals at respective total daily dose levels of 0 and 720.8 mg/kg/d. Individual dose volumes were based on the most recent body weights.
Clinical Observations, Body Weight, Electrocardiography, and Ophthalmoscopy
Rats
All animals were observed twice daily for morbidity, mortality, injury, and the availability of food and water throughout the duration of the study. A detailed clinical examination of each animal was performed pretest and on all study animals twice weekly during the study. The observations included evaluation of the skin, fur, eyes, ears, nose, oral cavity, thorax, abdomen, external genitalia, limbs and feet, respiratory and circulatory effects, autonomic effects such as salivation, and nervous system effects including tremors, convulsions, reactivity to handling, and unusual behavior.
The Functional Observational Battery (FOB) is a widely used test to evaluate potential test-article effects on neurological and neurobehavioral parameters. FOB evaluations, including those conducted in the home cage during handling, in the open field, and at other times, were conducted on all main study animals on days 1, 7, and 14 prior to the first daily dose and at 1 (±0.5) and 4 (±1) hours after the first daily dose. During open-field evaluations, each animal was observed for a minimum of 3 minutes in a black Plexiglas observation box measuring 20 × 20 × 8 in. The parameters evaluated in the FOB were based on those proposed by Moser et al. 21,22 The observations included evaluation of activity and arousal, posture, rearing, unusual behavior, clonic and tonic seizures, gait, mobility, stereotypy, righting reflex, response to stimulus (approach, click, tail pinch, and touch), palpebral closure, pupillary response, piloerection, exophthalmus, lacrimation, salivation, and respiration. The amounts, qualitative and/or quantitative measures, of defecation and urination were also recorded. Forelimb and hindlimb grip strength was measured, and hindlimb splay was quantitatively measured as described by Edwards and Parker. 23,24 Pain perception was assessed by measuring the latency of response to a nociceptive (thermal) stimulus when each animal was placed on a hot plate apparatus set to 52°C (±1°C). 25 Body weight and temperature were also measured.
Body weights for all animals were measured and recorded at receipt, 2 days prior to the initial dose, and twice weekly during the study.
Indirect ophthalmoscopic examinations were conducted on all animals predose and on study animals prior to the terminal or recovery necropsy. The anterior portion of the eyes, intermediate optic media, and fundus were all examined. Mydriasis was induced by instillation of 1 drop of 0.5% aqueous tropicamide solution into each eye 5 to 10 minutes prior to examination. The ophthalmoscopic examinations were also conducted prior to the start of dosing for TK animals.
Dogs
The animals were weighed once prior to dose initiation, twice weekly during the treatment and recovery periods, and at kill. Clinical observations were recorded at least once daily during acclimation and twice daily during the dosing and recovery periods. All signs of clinical abnormality were recorded. Dogs also were observed on day 1 at ~2 to 4 hours after each of the first 2 twice-daily dose administrations.
Cardiovascular/respiratory, ophthalmologic, and physical examinations, including auscultation, were performed prior to start of dosing and prior to kill for both treatment and recovery phases. Recovery animals therefore, underwent 1 additional cardiovascular, ophthalmic, and physical exam compared with main study (treatment) animals. Cardiovascular/respiratory examinations consisted of electrocardiography and determinations of blood pressure, respiratory rate, capillary refill time, and hemoglobin saturation.
Ophthalmologic examinations consisted of gross ocular observation, slit lamp biomicroscopy, and indirect funduscopy. Gross observations included inspection for ocular irritation and ocular discomfort. Slit lamp and indirect ophthalmic examinations used topically applied 1% Tropicamide to dilate the eyes 15 to 20 minutes prior to examination. The slit lamp examination included inspections of the conjunctiva, retina and retinal vessels, optic disc, and lens.
Food Consumption (Rats and Dogs)
In the rat study, food consumption was measured and recorded twice weekly for all study animals except the TK animals. In the dog study, food consumption was measured in all animals once daily throughout the treatment and recovery phases.
Hematology, Coagulation, Clinical Chemistry, and Urinalysis
Rats
Clinical pathology evaluations were conducted on all surviving study animals at terminal and recovery necropsies. The animals had access to drinking water but were fasted overnight prior to sample collection. Terminal blood samples (approximately 4 mL) were collected via the vena cava after carbon dioxide inhalation. Samples were collected into tubes containing K2EDTA for evaluation of hematology parameters and sodium citrate for evaluation of coagulation parameters. No anticoagulant was used for the clinical chemistry samples. The order of bleeding was conducted by alternating 1 animal from each dose group and then repeating to reduce handling and time biases. The animals were housed for 16 hours in stainless steel metabolism cages to collect urine for urinalysis evaluation.
Dogs
Blood specimens (approximately 2 mL) for clinical pathology assessment were collected from all animals 1 day prior to start of dosing (day 0) and 1 day after the completion of dosing (day 15). Blood collection was repeated for recovery animals 1 week after the last dose was administered prior to necropsy (day 22). Blood was collected as described previously. Urine specimens for urinalysis were collected from each animal at necropsy (day 15 or 22). Urinalysis was performed using a lateral flow assay (Urispec 11-Way; Henry Schein, Melville, NY) to determine urinalysis parameters.
Organ Weights (Rats and Dogs)
Body and organ weights were recorded at each scheduled necropsy, and appropriate organ weight ratios were calculated (relative to body and brain weights). Paired organs were weighed together. A combined weight of the thyroid gland with bilateral parathyroid glands was obtained along with a combined weight of the glandular and nonglandular stomach. In the rat study, approximately 2 cm of duodenum was left attached to the stomach, whereas in the dog study no duodenum was left attached to the stomach. The right mandibular/sublingual salivary glands were weighed together in the rat study. Only 1 mandibular salivary gland was excised and processed for examination, but it was not weighed in the dog study. The thyroid/parathyroid gland and pituitary gland were excised and weighed at necropsy in the dog study but were weighed following fixation in the rat study.
Toxicokinetics
Rats
Blood samples (approximately 0.5 mL) were collected via the orbital sinus from 3 TK animals per sex in both control and test-article-treated groups at alternating time points. TK blood samples were collected on the first and last day of dosing (days 1 and 14) in glass tubes containing approximately 0.5 mL of Alsever’s solution as the anticoagulant. Blood was drawn from control group rats predose and at 1 hour postdose and from test-article-treated animals predose and again at 0.5, 1, 2, 4, and 6 hours postdose. The samples were mixed well, stored on ice, and centrifuged under refrigeration. The animals were not fasted overnight prior to blood collection. After final blood collection, TK animals were killed via carbon dioxide inhalation followed by cervical dislocation.
A qualitative Western blot based assay was used to detect OxDC in plasma at concentrations 125 ng/mL or greater. This assay was validated based on the applicable requirements of ICH Q2R1, 26 namely precision, specificity, limit of detection, and robustness, including sample stability. The blood was first centrifuged to isolate blood plasma. Samples and standards were reduced with β-mercaptoethanol, diluted with SDS-PAGE Laemmli sample buffer, boiled, and separated by SDS-PAGE to separate proteins present in the rat plasma. The separated proteins were then transferred and fixed onto a nonreactive membrane. A rabbit anti-OxDC IgG antibody was added to the membranes to label any OxDC present in the blood plasma. Subsequent treatment with an anti-rabbit IgG antibody labeled with horseradish peroxidase (HRP) was used to visualize the presence of OxDC.
Dogs
A single predose TK blood sample was collected from each animal 1 day prior to start of dosing and from all animals predose (time 0) and again 0.5, 1, 2, 4, and 6 hours after the first dose on day 14, the final day of dosing. Each TK sample was split into 2 aliquots, which were processed separately to provide plasma or serum. The resulting plasma and serum specimens were stored at 5°C ± 3°C pending analysis. The same method for detecting the presence of OxDC described in the rat section was used for assaying dog plasma samples.
Immunogenicity (Rats and Dogs)
Blood samples were collected from all study animals at the 14-day (end of dosing) and recovery necropsies, processed to serum, and used to determine whether antibodies to OxDC were present. Serum samples were stored frozen at approximately –70°C until assessed for antibody formation.
A validated qualitative indirect enzyme-linked immunosorbent assay (ELISA) was used to test for the presence of anti-OxDC antibodies in the serum at concentrations 10 ng/mL or greater in rat sera or 400 ng/mL or greater in dog sera. The validation was performed in accordance with ICH Q2R1 26 based on precision, limit of detection, robustness, and specificity. The blood was first centrifuged to isolate blood serum. OxDC was affixed to nonreactive media, and the serum test samples were exposed to the captured OxDC and visualized.
Histopathology
Rats
Necropsies were performed on 1 animal found dead during the dosing phase and on all surviving study animals at the scheduled end of dosing and recovery necropsies. The animals were killed by carbon dioxide inhalation and examined for external abnormalities including masses. The skin was reflected from a ventral midline incision, and any abnormalities were identified and correlated with antemortem findings. The abdominal, thoracic, and cranial cavities were examined for abnormalities, and the organs were removed, examined, and, where required, placed in fixative. The pituitary was fixed in situ. All tissues were fixed in neutral buffered formalin, except for the eyes (including the optic nerve) and testes, which were fixed in modified Davidson’s fixative. 27 Formalin was infused into the lung via the trachea and into the urinary bladder. Additionally, portions of the bone marrow, spleen, thymus, and lymph node (mesenteric and mandibular) were collected from all main study animals. The tissues were frozen in optimal cutting temperature (OCT) and stored at approximately –70°C.
Microscopic examination of fixed hematoxylin and eosin–stained, paraffin-embedded tissue sections was performed. A 4-step grading system was used to define gradable lesions for comparison between dose groups.
Dogs. Animals were killed and necropsied on day 15 (treatment-phase animals) or day 22 (recovery-phase animals). Necropsies included inspection for gross lesions as well as collection and weighing of tissues/organs. The collected tissues/organs were fixed in 10% neutral buffered formalin (NBF), except for the eyes and testes, which were fixed in modified Davidson’s solution for 12 to 24 hours before being transferred to NBF. Bone marrow smears also were prepared. Additional specimens of bone marrow, thymus, spleen, and mesenteric lymph node were frozen in OCT and stored at –70°C.
Following fixation, tissues were processed, including trimming, dehydration, embedding in paraffin, sectioning at 3- to 5-μm thickness, and staining with hematoxylin and eosin. Tissues from control and high-dose OC4-treated animals were examined histopathologically via light microscopy.
Statistical Analysis
Data for each sex within a set were analyzed separately. The raw data were tabulated within each time interval, and the mean and standard deviation were calculated for each endpoint by sex and group. For each endpoint, treatment groups were compared with the control group. Data for some endpoints, as indicated, were transformed by either a log or rank transformation prior to conducting the specified analysis.
Data collected from rats were analyzed by Levene’s test, 1-way analysis of variance (ANOVA), and Welch’s t test with a Bonferroni correction to assess homogeneity of group variances for each specified endpoint and for all collection intervals. 28,29 All endpoints were analyzed using 2-tailed tests.
Raw data collected from dogs were analyzed by Bartlett’s test for equal variances to determine homogeneity of the data from multiple dosing groups. Where variance was homogeneous, 1-way ANOVA was used, followed by Tukey’s multiple comparisons post hoc test if the ANOVA was significant. 30 For nonhomogeneous variance, the Kruskal-Wallis (nonparametric) test was used, followed by Dunn’s posttest if the Kruskal-Wallis test was significant. 31
Histopathology lesion data were analyzed when relevant. Where appropriate, histopathology severity scores were analyzed statistically using nonparametric tests.
Results
Clinical Observations
Rats
No test-article-related mortality occurred during the study. On day 10, 1 high-dose female was found dead. The cause of death was determined to be a dosing error and was not due to a test-article-related effect. Microscopic examination revealed evidence of foreign material, and inflammation was noted within the fascia surrounding the larynx, suggesting perforation of the esophagus.
Clinical signs noted during the study were not considered test-article-related due to non-dose-responsive incidence and/or similar incidence in controls. No test-article-related clinical observations were noted at any dose level.
Clinical signs noted during FOB evaluations were not considered test-article related because there was a non–dose-responsive incidence and/or similar incidence in controls. Therefore, no test-article-related effects on FOB evaluations were noted at either dose.
Dogs
There were no unscheduled deaths during the in-life period (acclimation, treatment, or recovery phases). There were no clinical signs of abnormality during the acclimation periods. Slight swelling of the upper lip and single small raised spots or scabs in 1 high-dose female and 2 controls, respectively, were observed. No other clinical observations were noted. These findings were thought to be incidental and unrelated to treatment with the test article.
Body Weight and Food Consumption
Rats and Dogs
Body weight data (mean values) are illustrated in Figure 2 . The majority of the animals exhibited weight gain (up to 10% of predose value) during the 2 weeks of dosing. Differences at separate dose levels were not significant for the raw or normalized 2-week values (day 14) in males or females. No test-article-related effects on body weight or food consumption were noted at any dose level in either rats or dogs.
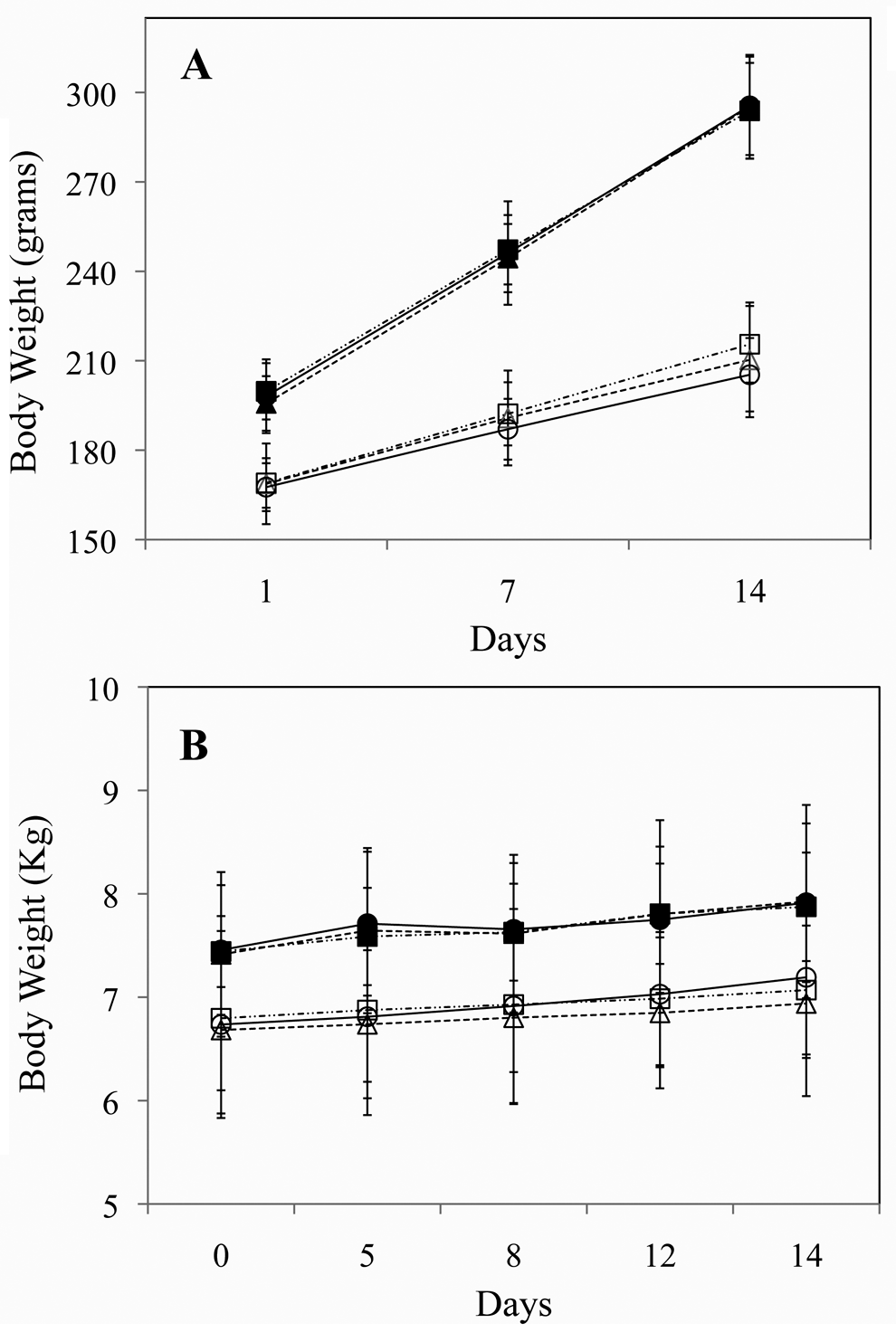
Mean body weights of rats (A) and dogs (B). Solid markers represent male animals and open markers females. High-dose groups are represented by squares with dashed-dotted lines, low-dose groups by circles with solid lines, and control groups by triangles with dotted lines. Error bars signify standard deviations.
Hematology
Rats
Hematology values are summarized in Table 2. Platelets were slightly but statistically significantly increased in females dosed with 720.8 mg/kg/d but were not considered adverse because the effect was seen in only 1 sex and the values were within a historical range for this strain and age of rat. There were no meaningful differences at recovery.
Significant Hematology Data (Mean ± SD)
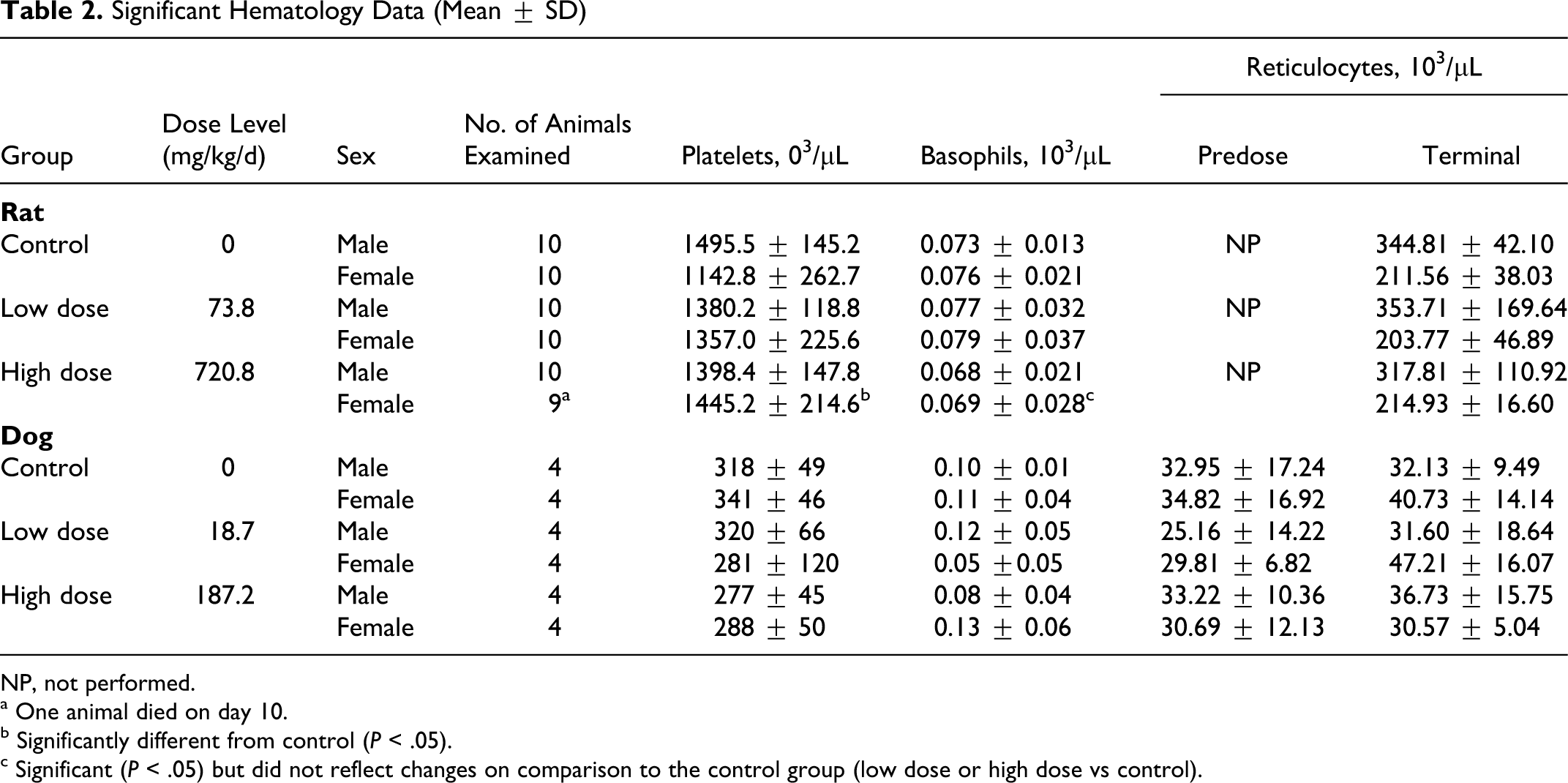
NP, not performed.
a One animal died on day 10.
b Significantly different from control (P < .05).
c Significant (P < .05) but did not reflect changes on comparison to the control group (low dose or high dose vs control).
Dogs
Differences in hematological parameters among the dosing groups were not significant predose or at day 15 for males or females (Table 2). The only exceptions were statistically significant decreases in basophil values (either as percentage of white blood cells or as cell counts) among high-dose females at day 15, but these values did not differ significantly from the control animals (Table 2). In the absence of apparent treatment-related differences at day 15, values following recovery (day 22) were not subjected to statistical analysis.
Blood Chemistry
Rats
There were no test-article-related effects on clinical chemistry parameters or coagulation values at the end of dosing or recovery. Sporadic statistical differences were observed that were considered not toxicologically meaningful due to the magnitudes of change.
Dogs
Differences in coagulation parameters among the dosing groups were not significant predose or at day 15 for males or females. In the absence of apparent treatment-related differences at day 15, values following recovery (day 22) were not subjected to statistical analysis.
Differences in clinical chemistry parameters among the dosing groups were not significant at day 1 or at day 15 for males or for females. The only exceptions were for phosphorus levels in low-dose males (P < .05 both predose and at day 14 vs controls) and triglycerides in high-dose females (P < .05 overall for predose females, although not supported by post hoc analysis). In the absence of apparent treatment-related differences at day 15, values following recovery (day 22) were not subjected to statistical analysis.
Ophthalmology, Urinalysis, and Electrocardiography (Dogs Only)
Rat and Dogs
No test-article-related effects were noted for the ophthalmological examinations at any dose level or for urinalysis parameters at termination or recovery. Furthermore, no treatment-related or dose-related cardiovascular findings were noted in any of the animals treated with OC4.
Gross Pathological Findings
Rats
There were no test-article-related macroscopic findings in any of the terminal or recovery groups in this study. All macroscopic findings in these rats were typical of rats of this strain, sex, and age or were considered incidental to administration of test article.
Dogs
Each animal was subjected to gross necropsy at kill. Virtually all animals, dosed with either vehicle or test article, displayed non–test-article-related enlargement of Peyer’s patches along the length of the intestines. A single low-dose female had accumulated gas within the intestines. The majority of the males (11/18) had testicular defects (1 testicle missing or of unequal size) prior to dosing. These testicular defects were thought to be congenital defects because they were found in approximately equal numbers in all male groups. Other observations were intermittent and limited to discoloration of 1 or both lungs (2 high-dose females, 1 high-dose male, and 1 low-dose male) and malformation of 1 kidney (1 low-dose female). Thus, no macroscopic treatment-related effects were noted.
Organ Weights
Rats
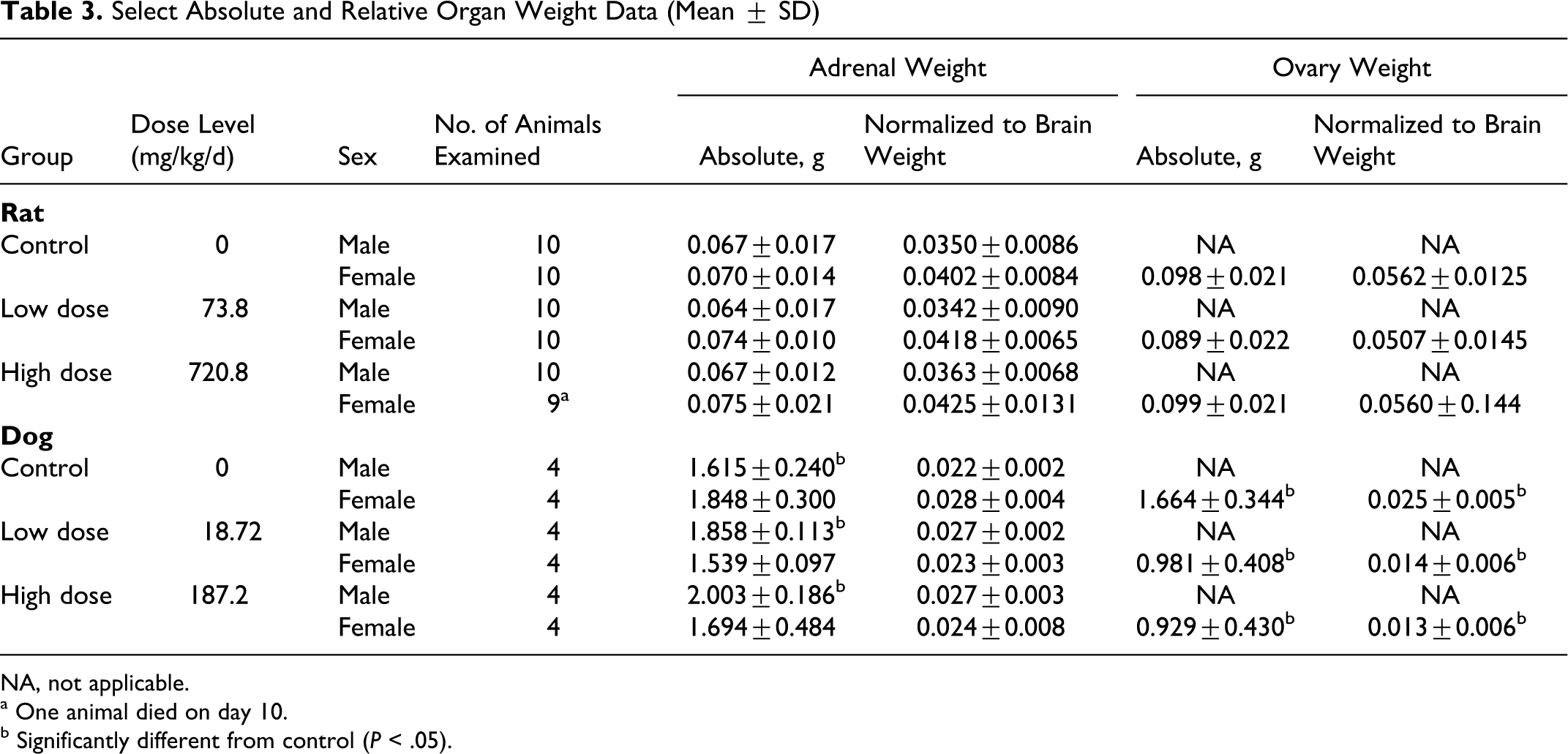
Weights of the organs were recorded at necropsy. Values are presented as raw weights and as normalized to brain weight (Table 3).
Select Absolute and Relative Organ Weight Data (Mean ± SD)
NA, not applicable.
a One animal died on day 10.
b Significantly different from control (P < .05).
There were no test-article-related absolute or normalized organ weight findings in any of the groups. Overall, all variations in organ weights were within the range of normal biological variation typically seen in rats of this strain, sex, and age.
Dogs
Among males, statistically significant (P < .05) increases at day 15 were seen for the adrenals (as absolute as well as body weight–relative and brain weight–relative values) in both low- and high-dose groups (Table 3). A dose-dependent increase in both absolute and normalized to both animal and body weight adrenal size was confirmed via post hoc analyses (P < .05) (Table 3). A correlation with adrenal size did not persist at the recovery kill, although the small sample sizes (n = 2) at day 22 precluded statistical analysis.
Among females, a statistically significant (P < .05) decrease was seen at day 15 in the ovary weight (as absolute and brain weight–normalized values) for both low- and high-dose animals. A dose-dependent decrease in ovary size was confirmed via post hoc analysis (P < .05) only as a fraction of brain weight (control vs high-dose group) (Table 3). A correlation with ovary size was still seen at the recovery kill, although the small sample sizes (n = 2) at day 22 precluded statistical analysis. However, the effect might also reflect estrous cycle or developmental differences among these young female dogs.
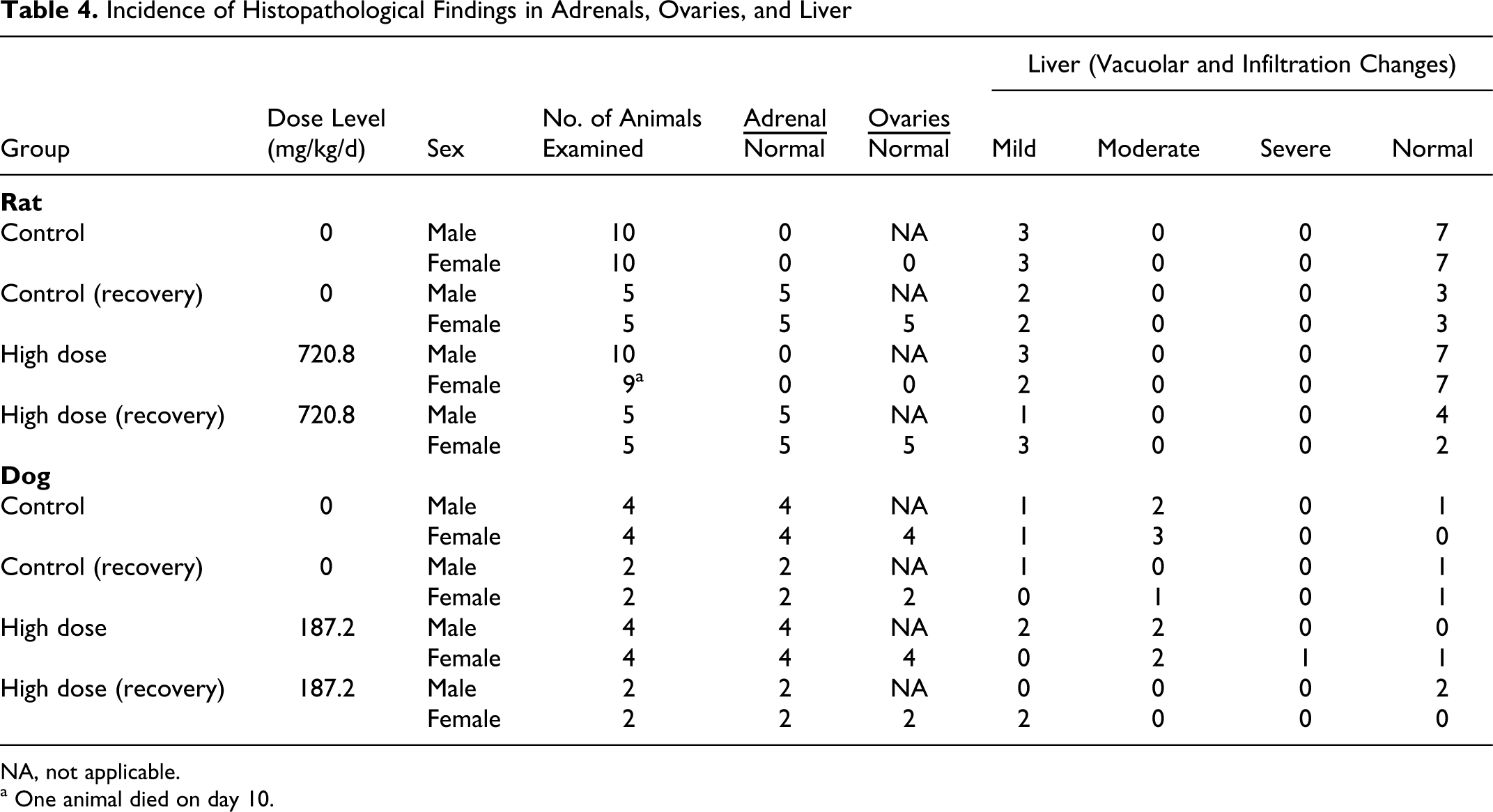
Histopathological Findings
Rats
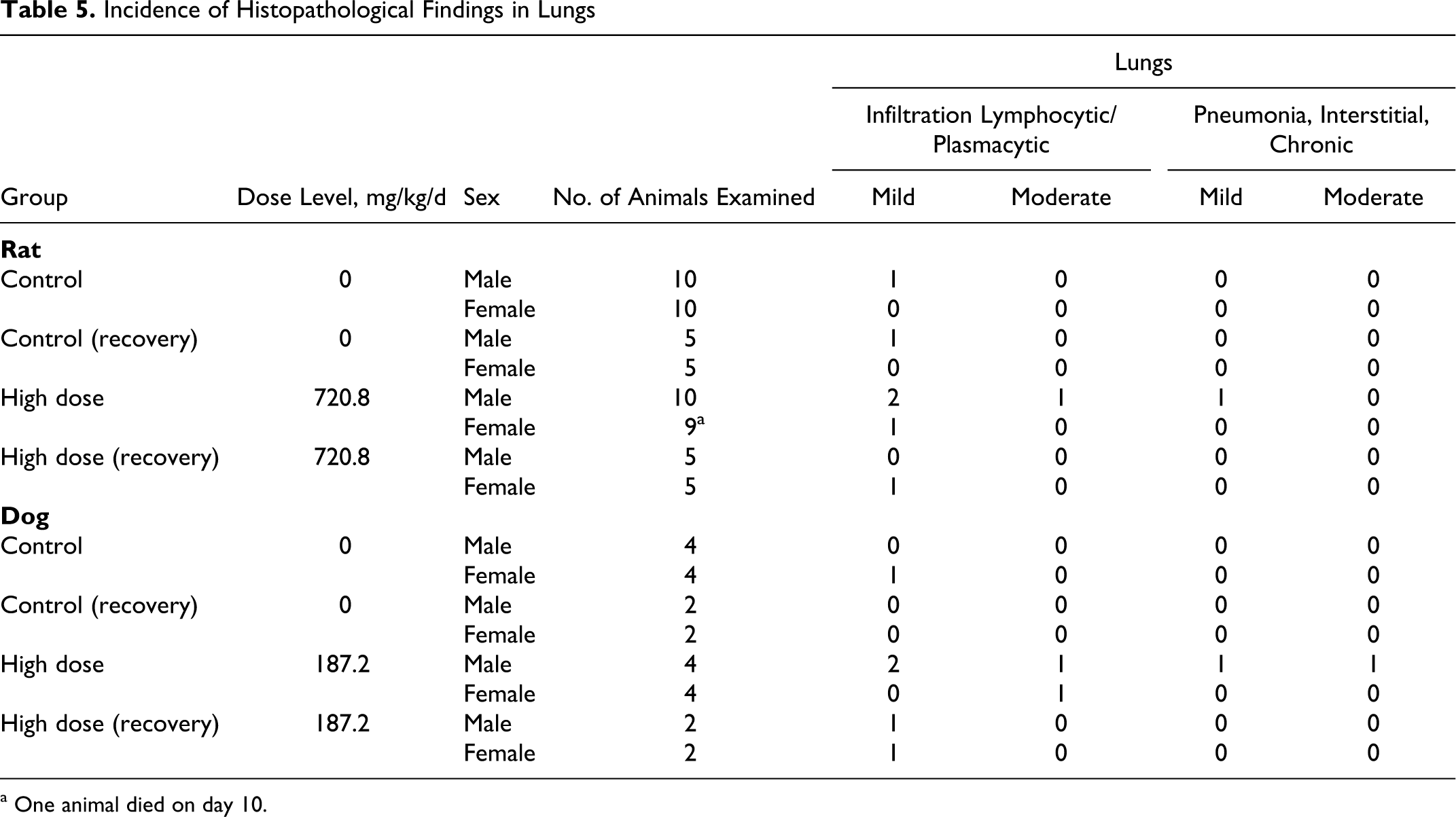
Significant microscopic observations are summarized in Tables 4 through 6. There were no test-article-related microscopic findings in any group. One or 2 animals in the male and female high-dose groups had mild to moderate perivascular, lymphocytic, and plasmacytic infiltrations compared with controls (Table 5 ). These changes were not considered related to OC4 administration given the low incidence of the finding and the presence in 1 male in the control group. All other microscopic findings were typical of rats of this strain, sex, and age or were considered incidental to administration of test-article.
Incidence of Histopathological Findings in Adrenals, Ovaries, and Liver
NA, not applicable.
a One animal died on day 10.
Incidence of Histopathological Findings in Lungs
a One animal died on day 10.
Dogs
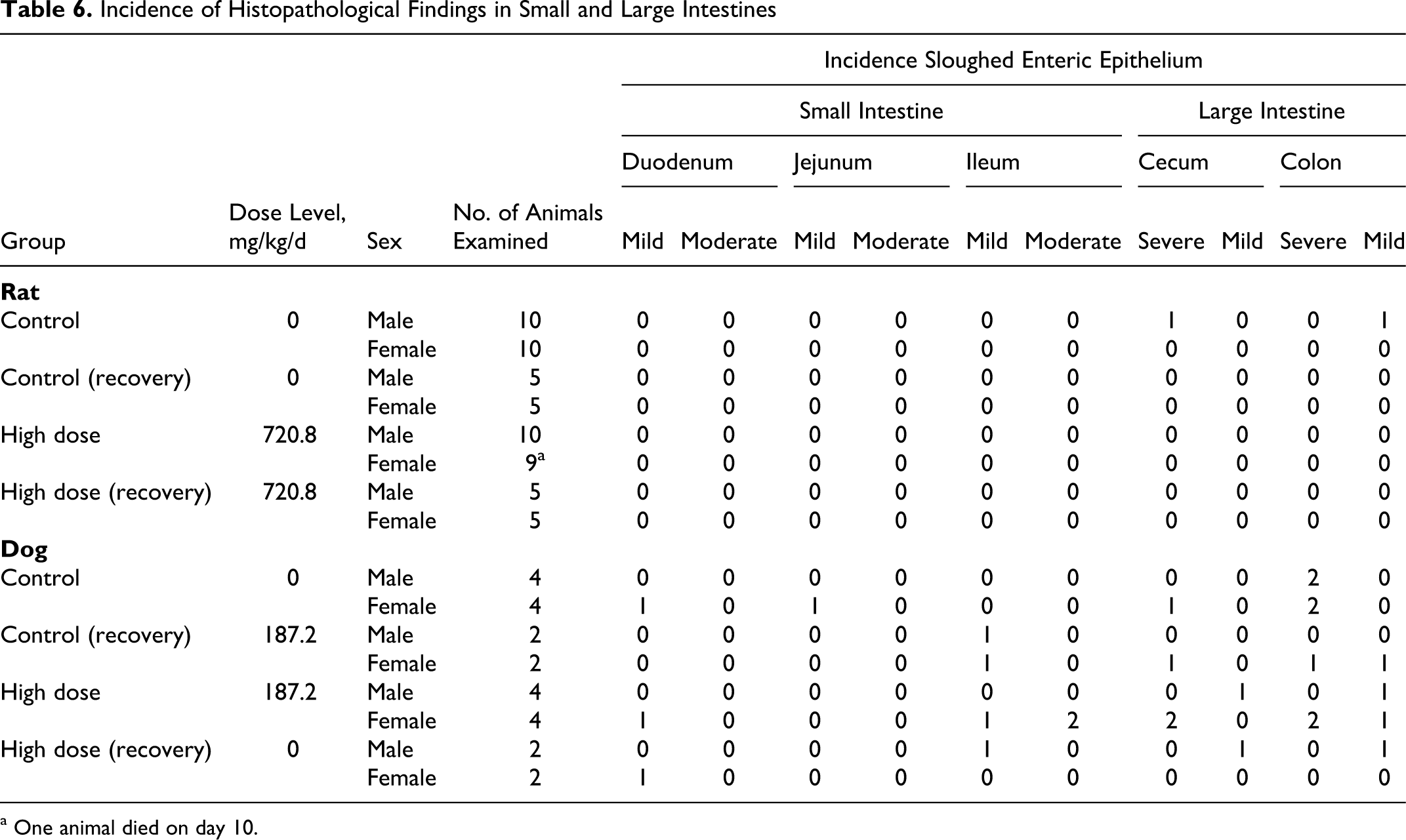
Histopathological examination revealed most tissues were within the normal limits with a few mild spurious, incidental, or idiosyncratic findings in a few tissues. Microscopic examinations of tissues from the high-dose and vehicle group animals revealed changes in several organs of both groups. Mild to moderate vacuolization (increased glycogen deposition) was noted in the liver with the same degree and frequency in both control and high-dose groups (Table 4). Mild to severe sloughing of enteric epithelium, especially in the colon and cecum, was identified with and without mucosal bacterial attachment (Table 6 ) in a few control and high-dose animals. This severity was higher in 2 high-dose animals, but the remaining high-dose animals were generally similar to controls. Similarly the lungs showed signs of mild to moderate perivascular, lymphocytic, and plasmacytic infiltrations (Table 5). One high-dose male had macrophage proliferation, and 2 high-dose males had mild to moderate pneumonia. The GI findings and the findings in the lungs were observed in only 1 or 2 animals, so they were not thought to be test-article related (note: only the high-dose and vehicle groups were examined histologically). There were no histological findings in the treated male adrenals or female ovaries corresponding with decreased organ weights, so the weight changes were not found toxicologically significant. The few incidental observations noted in the high-dose dogs were confined to 1 or 2 animals and therefore were not considered test-article related. Thus, no test-article-related histopathological findings were noted in the high-dose animals.
Incidence of Histopathological Findings in Small and Large Intestines
a One animal died on day 10.
Toxicokinetics and Immunogenicity
Rats and Dogs
Data collected from TK animal specimens demonstrated that OxDC was absorbed into the bloodstream and that anti-OxDC antibodies were generated. Measurable OxDC was detected in only 1 of 5 high-dose male rats in samples taken 4 hours after dosing, 1 of 5 high-dose females sampled 6 hours after dosing, 2 of 6 high-dose dog samples taken at 0.5 and 1 hour after administration, and 1 high-dose dog sample taken from the 2-hour and 6-hour time points (Table 7 ).
Toxicokinetic Analysis
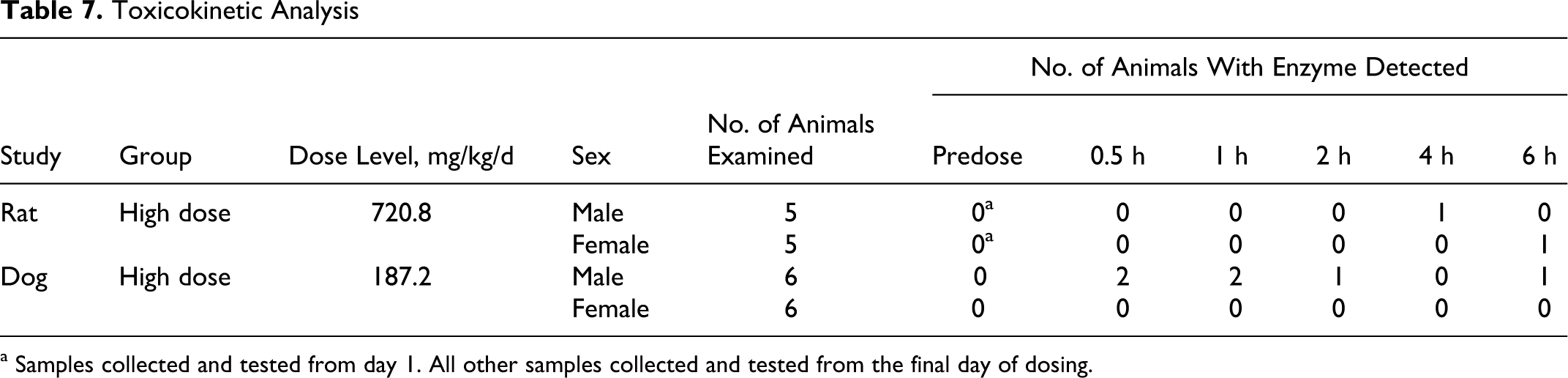
a Samples collected and tested from day 1. All other samples collected and tested from the final day of dosing.
Results of analyses of rat and dog serum for antibodies to OxDC are shown in Table 8 . Antibodies to OxDC were detected in all rats and dogs tested from the high-dose group after 14 days of OC4 administration and a 7-day recovery. Although this assay is not quantitative, it is important to note that the optical density values observed in the least diluted (×50 dilution) of all positive samples indicate a significant response in all positives.
Immunoglobulin Detection
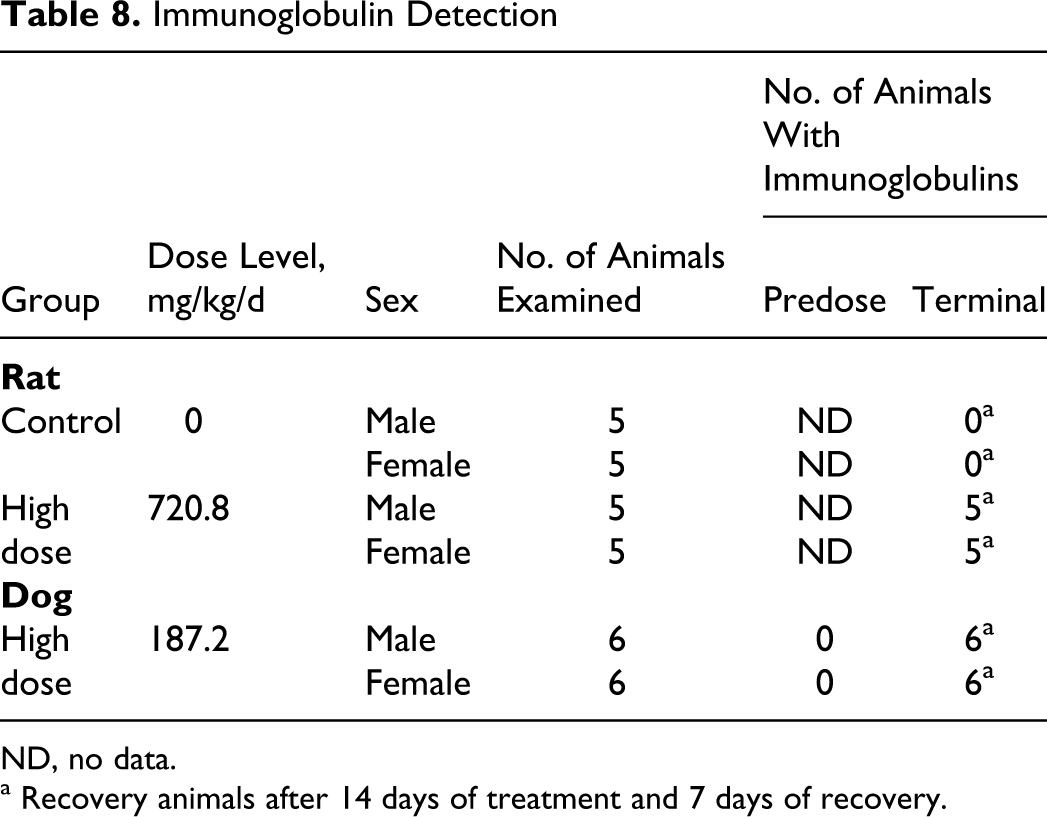
ND, no data.
a Recovery animals after 14 days of treatment and 7 days of recovery.
Dose Suspension Stability and Homogeneity
Analysis of retention samples revealed that all doses, independent of day collected or relative position within the mixing suspension, were homogeneous and fully potent.
Discussion
Administration of OC4 to both rats and dogs for 14 days revealed the absence of any toxicologically significant test-article-related clinical pathology effects. However, a few isolated effects were noted from the high-dose groups of both studies. Test-article-related effects were noted for platelet counts in the high-dose female rat group, but the increase was not considered adverse at the magnitude observed and was not observed at the end of the recovery period. The dog study revealed statistically significant dose-dependent changes in ovary and male adrenal gland weights, but no corresponding pathological changes in either the adrenals of males or the ovaries of females were found upon microscopic histological examination. Hence, these observations may be due to developmental changes or random variation and thus are probably unrelated to the test article and do not appear to be of toxicological significance. Furthermore, significant differences in basophil values among female dogs as well as phosphorous values among male dogs were noted but did not reflect changes on comparison to the control group. Finally, generally mild to moderate changes were noted from lungs, liver, and small and large intestines in both high-dose and control dog groups. Because these observed microscopic changes were present in both test-article and control animals and the incidence of moderate to severe findings was low, they were not considered test-article related.
Several TK animal specimens tested revealed the presence of OxDC in the bloodstream. The quantity of OxDC was not measured, but some samples were higher than the limit of detection. The absorption of macromolecules such as enzymes across the GI tract is not uncommon. 32 There may be a number of mechanisms by which orally ingested enzymes are efficiently absorbed.
Both animal species tested with OC4 showed an immunogenic response to the OxDC enzyme. In fact, all high-dose animals tested showed a significant immunoglobulin response after 14 days of administration and 7 days of recovery. This response indicates that absorption was readily occurring despite the lack of significant detection by the Western blot. The immunogenic response could be attributable to proteolyzed OxDC peptides, which would not be detected by Western blot analysis. Despite the quick response, there were no signs of toxicity associated with antibody generation. OxDC belongs to the cupin superfamily, a functionally highly diverse protein family found in a number of organisms. An immunogenic response to the cupin family of enzymes has been shown to occur infrequently as food allergens. 33 In fact, allergenicity within the cupin superfamily has been confined to the vicilin and legumin seed storage proteins. 33 However, an immunogenic response in laboratory animals is difficult to correlate to effects in humans. 34 Thus, despite the immunogenic response found in both rats and dogs and the available literature linking OxDC to food allergenic proteins, the potential for an allergenic response in humans is unlikely.
Formate and carbon dioxide are by-products of the enzymatic degradation of oxalate (substrate) in the stomach by OC4. Formate is a normal metabolic product in humans and many animals. The toxicokinetics and metabolism of formate have been extensively investigated in humans, nonhuman primates, rats, and pigs. Formate is rapidly absorbed from the GI tract. The major route of metabolism in rat and monkey is oxidation to CO2 and water by the folate biochemical pathway. 35 The half-life of formate in humans is short, less than 1 hour. 36
A 5-generation lifetime drinking water study with calcium formate was conducted in rats. 37 The average daily intake was between 150 and 200 mg/kg/d. 37 Results from these studies reveal no significant clinical or pathological changes (in particular ocular fundus). 37 By comparison, the amount arising from catabolism of dietary oxalate by OC4 in humans is minimal: degradation of 180 mg of dietary oxalate from a typical Western meal would yield ~90 mg each of formate and CO2.
A study to evaluate proof of principle of OxDC at degrading dietary oxalate was conducted in rats (unpublished results; Cowley, 2008). Six male rats were dosed twice daily with ~23 mg of OxDC for 10 days. Rats were given a special diet containing 2.3% oxalate. Urine was collected and tested for oxalate because a reduction of dietary oxalate by OxDC would be expected to reduce urinary oxalate concentrations. Results from this study demonstrated that OxDC administration resulted in a significant reduction in urinary oxalate in OC4-treated animals compared with untreated controls. Daily food intake, body weight, clinical chemistry, urinalysis, hematology, and limited histopathology (kidneys, large and small intestines, stomach, liver, heart, testes, lungs, spleen and thyroid glands) were evaluated. This limited safety assessment indicated no signs of adverse affects. Furthermore, this study provides additional evidence that the amount of formate formed from degradation of even greatly exaggerated levels of dietary oxalate is not harmful.
The no observed adverse effect level (NOAEL) of OC4 in the rat was determined to be 720.8 mg/kg/d, the highest dose tested, for twice-daily (360.4 mg/kg per dose) oral administration in males and females. Likewise, the NOAEL for OC4 in the dog is 187.2 mg/kg/d, again the highest dose tested.
Footnotes
Acknowledgments
We thank MPI Research and Comparative Biosciences for directing the conduct of these studies. We also acknowledge Matthew Glover, Dana Smith, Jianming Zeng, Julie Morris, Evan Chiswick, Gregory Barbe, and Matthew Battistini for manufacturing and testing OC4 and examining specimens associated with these 2 studies, as well as Anthony DeFusco and Carl-Gustaf Gölander for their diligent review of this manuscript.
