Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) assessed the safety of soy proteins and peptides, which function in cosmetics primarily as hair conditioning agents and skin-conditioning agents-miscellaneous. The Panel considered relevant data related to these ingredients. The Panel concluded that soy proteins and peptides are safe in cosmetics in the present practices of use and concentration described in this safety assessment.
Introduction
Soy protein and peptide ingredients function mainly as skin and hair conditioning agents in personal care products. 1 This report assesses the safety of the following 6 soy ingredients:
Glycine Max (Soybean) Polypeptide, Glycine Soja (Soybean) Peptide, Glycine Soja (Soybean) Protein, Hydrolyzed Soy Protein, Hydrolyzed Soy Protein Extract, Hydrolyzed Soymilk Protein
The safety of several hydrolyzed proteins as used in cosmetics has previously been reviewed by the Panel. The Panel concluded that hydrolyzed collagen, hydrolyzed corn protein, hydrolyzed rice protein, and hydrolyzed silk are safe for use in cosmetics.2–6 Additionally, the Panel concluded that hydrolyzed wheat gluten and hydrolyzed wheat protein are safe for use in cosmetics when formulated to restrict peptides to a weight-average molecular weight (MW) of 3 500 Da or less. 7
Soy proteins and peptides are used as food. The US Food and Drug Administration (FDA) determined that the use of peptones (ie, water-soluble protein hydrolysates) as direct food substances is generally recognized as safe (GRAS), and that soybean protein is GRAS as an indirect food additive for substances migrating to food from paper and paperboard products. Because daily exposure from food use would result in much larger systemic exposures than from use in cosmetic products, the systemic toxicity potential of soy peptide ingredients via oral exposure is not addressed further in this report. The primary focus of the safety assessment is the review of safety based on topical exposure.
Chemistry
Definition
Definitions and Functions of the Ingredients in This Safety Assessment. 1

The soy protein and peptide derivatives form a broad category of materials which are prepared by extraction from soy and partial hydrolysis to yield cosmetic ingredients. Soy proteins and peptides can also be separated on the basis of molecular size. By removing oil at lower temperatures, soy protein isolate is obtained, and is widely used in the food industry.9–11 Whole aqueous extractable soybean proteins can be separated into storage globulin and whey fractions by acidification.
The FDA defines the term “protein” to mean any α-amino acid polymer with a specific defined sequence that is greater than 40 amino acids in size. 12 The FDA considers a “peptide” to be any polymer composed of 40 or fewer amino acids; however, these definitions of protein and peptide are not necessarily adhered to in the naming of cosmetic ingredients.
The acid-precipitable fraction of whole aqueous extractable soybean proteins includes the major soybean storage proteins. The remaining part consists of the minor globulin, γ-conglycinin, and contaminating proteins, including whey proteins. Whey proteins are composed of lipoxygenase (102 kDa), bamylase (61.7 kDa), lectin (33 kDa), and Kunitz trypsin inhibitors (20 kDa). The proportion represented by these whey proteins in the acid-precipitated globulins is unknown. Soy protein isolate is a mixture of various proteins, and the main ingredients are classified into 4 protein categories according to their sedimentation coefficients 2S, 7S, 11S, and 15S which sediment at different gravitational forces when the solution is subjected to a centrifugal field. Among these proteins, 7S (β-conglycinin) and 11S (glycinin) represent 80-90% of all soybean protein, and the ratio 7S/11S has been reported to be about .5-1.3 depending on varieties. The 7S globulin consists of 3 subunits α (ca 67 kDa), α’ (ca 71 kDa), and β (ca 50 kDa). The 11S globulin is a hexamer and is made up of 5 different subunits, each of which consists of an acidic subunit A (ca 35 kDa) and a basic subunit B (ca 20 kDa), linked by a disulfide bond. The 11S globulin was found to dissociate into 2S, 3S, or 7S forms in solutions of various pH values and ionic strengths. Amino acid compositions of β-conglycinin and glycinin have been analyzed, but the 3-dimensional structures are not well established in spite of much effort.
Chemical and Physical Properties
Chemical and Physical Properties.

Soy Peptide
Dipeptide- or tripeptide-rich forms of soy peptide were described to have a mean MW of around 500 Da. 13
Hydrolyzed Soy Protein
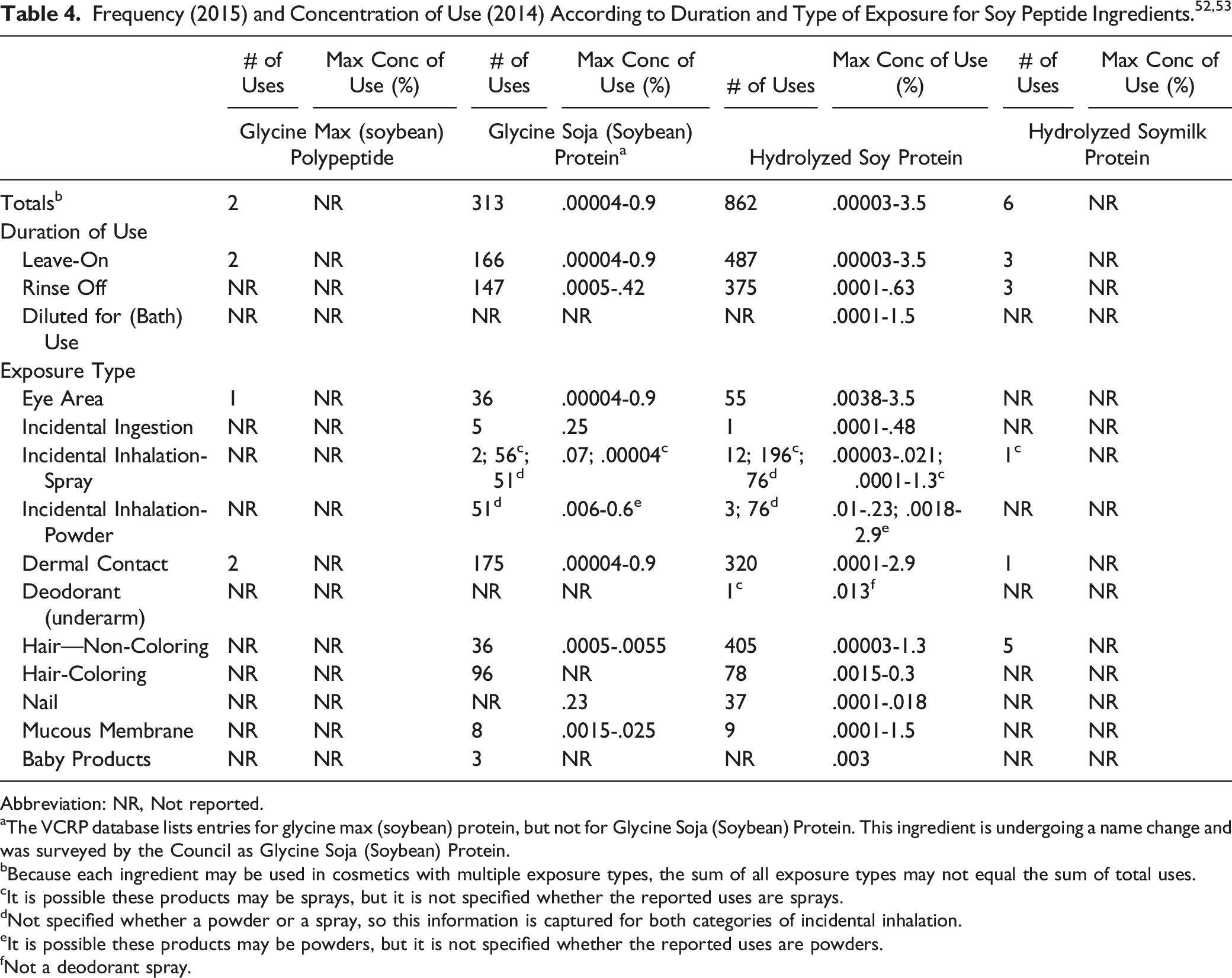
A histogram showing the approximate distribution of MWs for a Hydrolyzed Soy Protein product from one supplier is shown in Figure 1.
14
The figure shows that approximately 35% of the MW distribution falls between 490 and 1 030 Da. One source has indicated the average MW is 300 Da
15
; however, other sources have reported the MW of their Hydrolyzed Soy Protein product to be approximately 1 000-2 000 Da.16–18 Molecular weight distribution of Hydrolyzed Soy Protein from a supplier.
14
Hydrolyzed Soymilk Protein
A supplier has reported the MW of their Hydrolyzed Soymilk Protein product to be approximately 1 000-2 000 Da. 18
Method of Manufacturing
Glycine Soja (Soybean) Protein
Glycine soja (soybean) protein can be prepared from defatted low-heat soybean meal. 10 A dispersion of soy flour is prepared by adding distilled water (1: l5, w/v) with final protein concentration of 3.1% (w/w). Then, 2 mol/l sodium hydroxide is used to adjust the dispersion to pH 8.5. The dispersion is stirred for 1 h at room temperature and then centrifuged (10 000 × g, 20 min). The supernatant is adjusted to pH 4.5 with 2 mol/l hydrochloric acid and centrifuged (10 000 × g, 20 min). The obtained sediment is resuspended with distilled water (1: 5, v/v) and adjusted to pH 7.0 with 2 mol/l sodium hydroxide. Then it is dialyzed against deionized water and freeze-dried.
A supplier has indicated that a Glycine Soja (Soybean) Protein product is manufactured by adding water to the seed of Glycine max Merrill (Leguminosae), extracting, and then filtering. 19 The solvent 1,3-butylene glycol is then added to the filtrate. More than 80% of the resulting protein product has a MW less than 5 000 Da.
Hydrolyzed Soy Protein
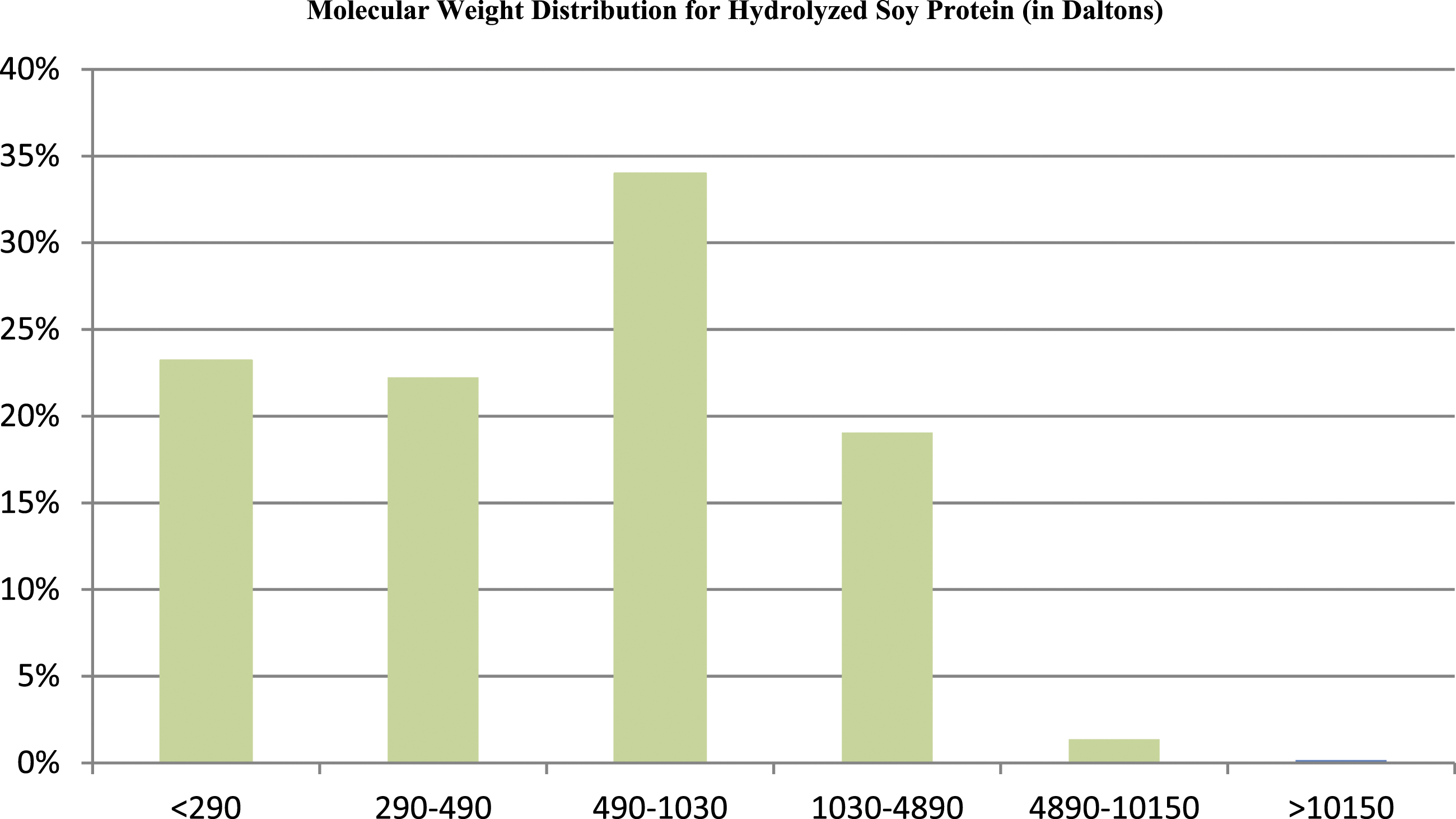
A manufacturing flow chart from a Hydrolyzed Soy Protein supplier is shown in Figure 2. Manufacturing process of a Hydrolyzed Soy Protein product from a supplier.
45
The preparation of hydrolysates can be afforded via acid and enzyme. 10 The above Glycine Soja (Soybean) Protein dispersion (4% w/v) is adjusted to pH 2.0 with 1 mol/l hydrochloric acid, and incubated at 37°C for 30 min. Then, an enzyme (such as pepsin) is added to each part at an enzyme to substrate ratio of .3% (w/w) to start the enzymatic hydrolysis reaction. Each fraction is incubated at 37°C (10-900 min) and the enzyme is deactivated by adjusting the pH to 7.0 with 2 mol/l sodium hydroxide.
A supplier has reported that Hydrolyzed Soy Protein is produced from isolated soy proteins that are hydrolyzed with a protease enzyme for 2 h. 20 The enzyme is inactivated by heat once the target MW is achieved. The resultant solution may then be concentrated.
Another supplier reported that Hydrolyzed Soy Protein (MW = 300 Da) may be prepared by both alkaline and enzyme hydrolysis. 15 These processes occur for several hours until the desired MW is reached. The final product is a 25% water solution of Hydrolyzed Soy Protein.
In a Hydrolyzed Soy Protein product of 8.5%, the supplier states that enzymatic hydrolysis yields a material with 87% of the proteins with a MW less than 2000 Da, 11% of the proteins with a MW between 2 000 and 5 000 Da, and 2% of the proteins with a MW greater than 5 000 Da. 21
Hydrolyzed Soymilk Protein
While method of manufacturing for Hydrolyzed Soymilk Protein was not discovered in the published literature nor was information provided by industry, soymilk is reported to be made through water extraction of whole soybeans. 22
Composition and Impurities
A supplier has stated that Hydrolyzed Soy Protein and Hydrolyzed Soymilk Protein do not contain fragrance ingredients designated as sensitizing by the European Union’s cosmetics regulations.23–26
Glycine Soja (Soybean) Protein
A supplier states that Glycine Soja (Soybean) Protein product (>80% has MW less than 5 000 Da) is comprised of .4% protein, 30% butylene glycol, and 69.6% water. 19
Soy Peptide
Amino Acid Composition of a Soy Peptides Sample. 13
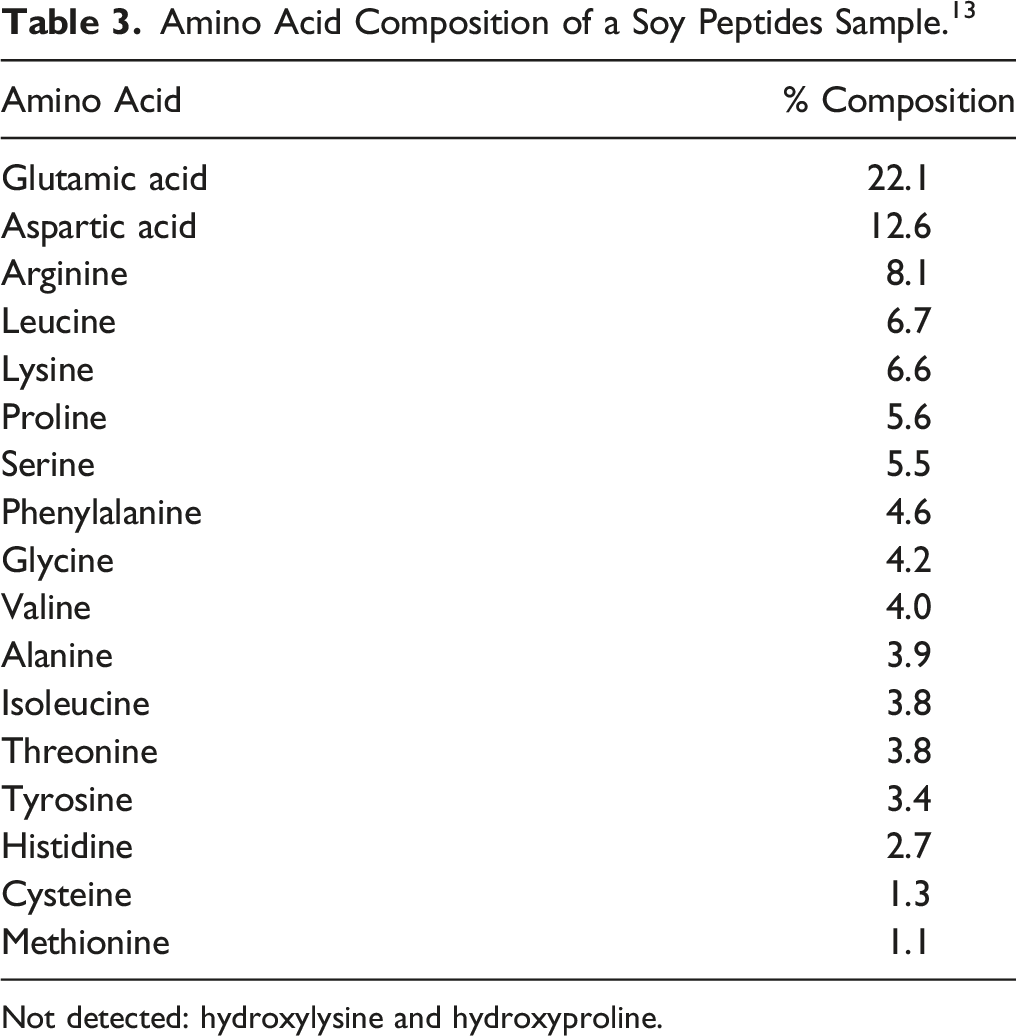
Not detected: hydroxylysine and hydroxyproline.
Hydrolyzed Soy Protein
A supplier states that Hydrolyzed Soy Protein (MW = 300 Da) has heavy metals, arsenic, and iron at levels <10 ppm, 1 ppm, and 10 ppm, respectively. 15 Another supplier states that Hydrolyzed Soy Protein (87% has MW <2000 Da) did not have detectable levels of alkaloids, pesticides, or aflatoxins. 21 No traces of arsenic or mercury were detected. Other metals were reported as cadmium (2 ppb), chromium (57 ppb), cobalt (15 ppb), nickel (.7 ppm), lead (60 ppb), and iron (<10 ppm).
The compositional breakdown of products containing 25% and 35% Hydrolyzed Soy Protein included 74.6% or 64.6% water, respectively, and .2% methylparaben, and .2% quaternium-15, each.23,24
Hydrolyzed Soymilk Protein
The compositional breakdown of a product containing 21.5% Hydrolyzed Soymilk Protein included 76.6% water, 1.2% phenoxyethanol, and .7% DMDM hydantoin. 25
Use
Cosmetic
The safety of the cosmetic ingredients included in this safety assessment is evaluated on the basis of the expected use in cosmetics. The Panel utilizes data received from the FDA and the cosmetics industry. The data received from the FDA are those it collects from manufacturers on the use of individual ingredients in cosmetics by cosmetic product category in its Voluntary Cosmetic Registration Program (VCRP), and those from the cosmetic industry are submitted in response to a survey of the maximum reported use concentrations by category conducted by the Council.
Abbreviation: NR, Not reported.
aThe VCRP database lists entries for glycine max (soybean) protein, but not for Glycine Soja (Soybean) Protein. This ingredient is undergoing a name change and was surveyed by the Council as Glycine Soja (Soybean) Protein.
bBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
cIt is possible these products may be sprays, but it is not specified whether the reported uses are sprays.
dNot specified whether a powder or a spray, so this information is captured for both categories of incidental inhalation.
eIt is possible these products may be powders, but it is not specified whether the reported uses are powders.
fNot a deodorant spray.
Based on the VCRP data and the results of the Council’s concentration of use survey, Glycine Soja (Soybean) Peptide and Hydrolyzed Soy Protein Extract are not in use.
As mentioned previously, some of these ingredients are used in products that are used near the eye. Additionally, some of these ingredients may be used in products that can be incidentally ingested or come into contact with mucous membranes. For example, Hydrolyzed Soy Protein is used in bath soaps and detergents at up to 1.5%. Additionally, some of these ingredients were reported to be used in hair sprays and body and hand sprays and could possibly be inhaled. For example, Glycine Soja (Soybean) Protein was reported to be used in body and hand sprays at a maximum concentration of .07%. In practice, most droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 μm, with propellant sprays yielding a greater fraction of droplets/particles below 10 μm compared with pump sprays.27–30 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount.28,29
The soy peptide ingredients in this report are not restricted from use in any way under the rules governing cosmetic products in the European Union. 26
Non-Cosmetic
The FDA determined that the use of peptones as direct food substances is GRAS. These GRAS peptones are defined as “the variable mixture of polypeptides, oligopeptides, and amino acids that are produced by partial hydrolysis of …soy protein isolate…” (21 CFR §184.1553). Additionally, soybean protein (described as Glycine Soja (Soybean) Protein) is GRAS for substances migrating to food from paper and paperboard products (21CFR §182.90). The FDA requires allergen labeling when major allergens are included in food; these allergens include soybeans. 31
The FDA has also reviewed soybean protein for use as an active ingredient in over-the-counter drugs. Based on evidence currently available, there are inadequate data to establish general recognition of the safety and effectiveness of this ingredient in weight control drug products (21CFR §310.545). Soy proteins are used in adhesives and plastics industries. 32
Toxicokinetics
Hydrolyzed Soy Protein
While no experimental data were available for the dermal absorption of Hydrolyzed Soy Protein, gastrointestinal absorption would allow for significantly higher bioavailability than dermal absorption. 33 In worst-case scenarios, no signs of systemic toxicity were observed with human oral exposures to greater than 2 000 mg/kg Hydrolyzed Soy Protein; therefore, it was concluded that systemic toxicity from cutaneous exposure would be negligible.
Toxicological Studies
The soy proteins that serve as the sources for the ingredients that are addressed in this safety assessment are found in the foods we consume daily. The potential for systemic effects, other than sensitization, from the possible absorption of soy ingredients through topical exposure is much less than the potential for systemic effects from absorption through oral exposures. This is because the rates of absorption and metabolism of these ingredients in the skin are expected to be negligible compared to the corresponding rates in the digestive tract. Thus, the potential for systemic effects, other than sensitization, are not discussed in detail in this report.
Genotoxicity
Glycine Soja (Soybean) Protein
A trade name mixture containing .4% Glycine Soja (Soybean) Protein (>80% with a MW <5000 Da) produced no genotoxicity in a reverse mutation assay and in a chromosomal aberration study (with and without metabolic activation). 34 No further details were provided.
Hydrolyzed Soy Protein
Hydrolyzed soy protein product with 54% of the MW distribution below 5 000 Da was analyzed for mutagenic potential in an assay using Salmonella typhimurium TA1535/pSK1002 with and without S9 metabolic activation. 33 Concentrations tested were 625, 1 250, 2 500, or 5 000 μg/mL. No sign of mutagenicity was observed with or without S9. It was concluded that Hydrolyzed Soy Protein was not mutagenic.
Irritation and Sensitization
Irritation
Dermal
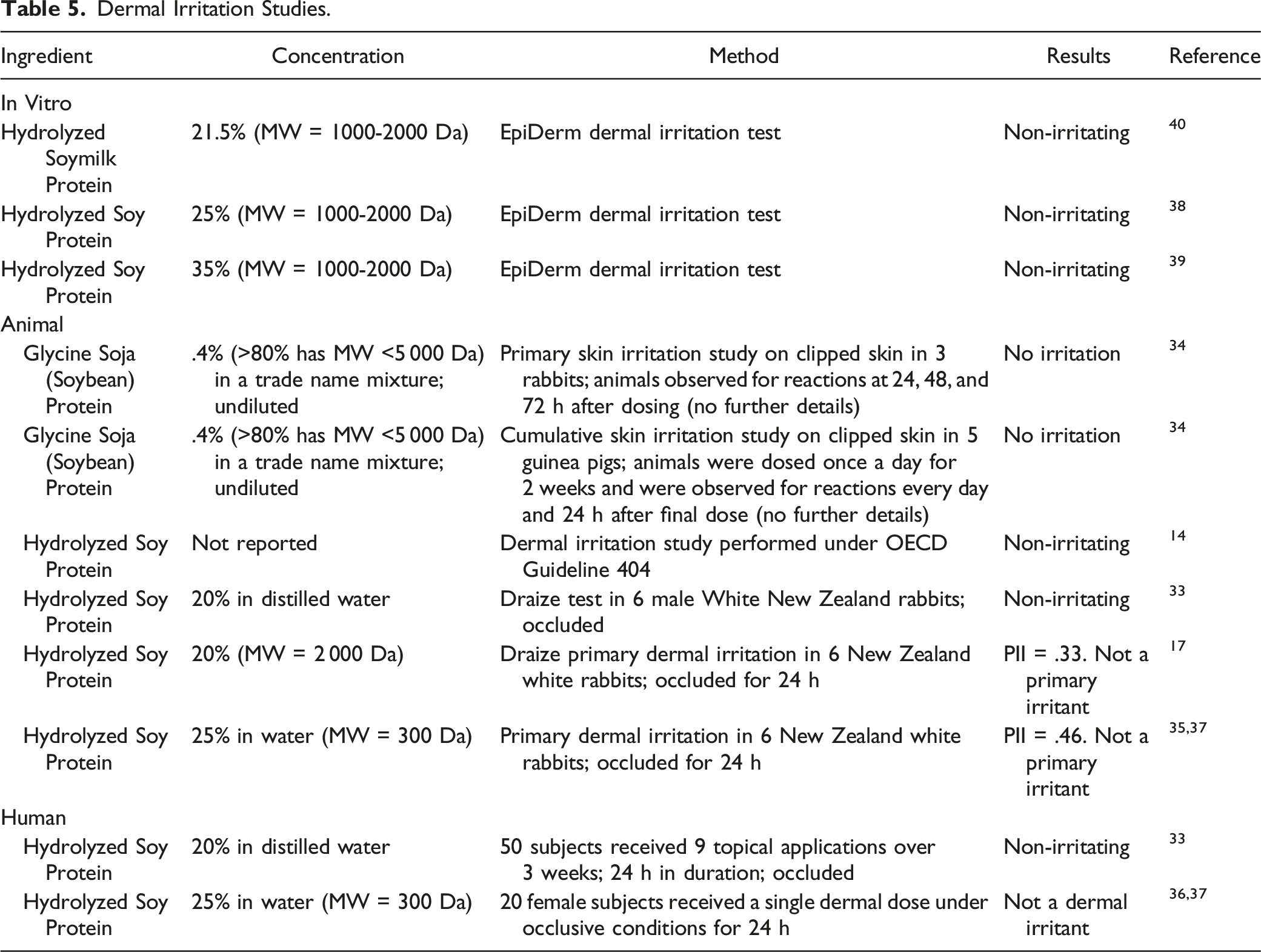
Dermal Irritation Studies.
Ocular
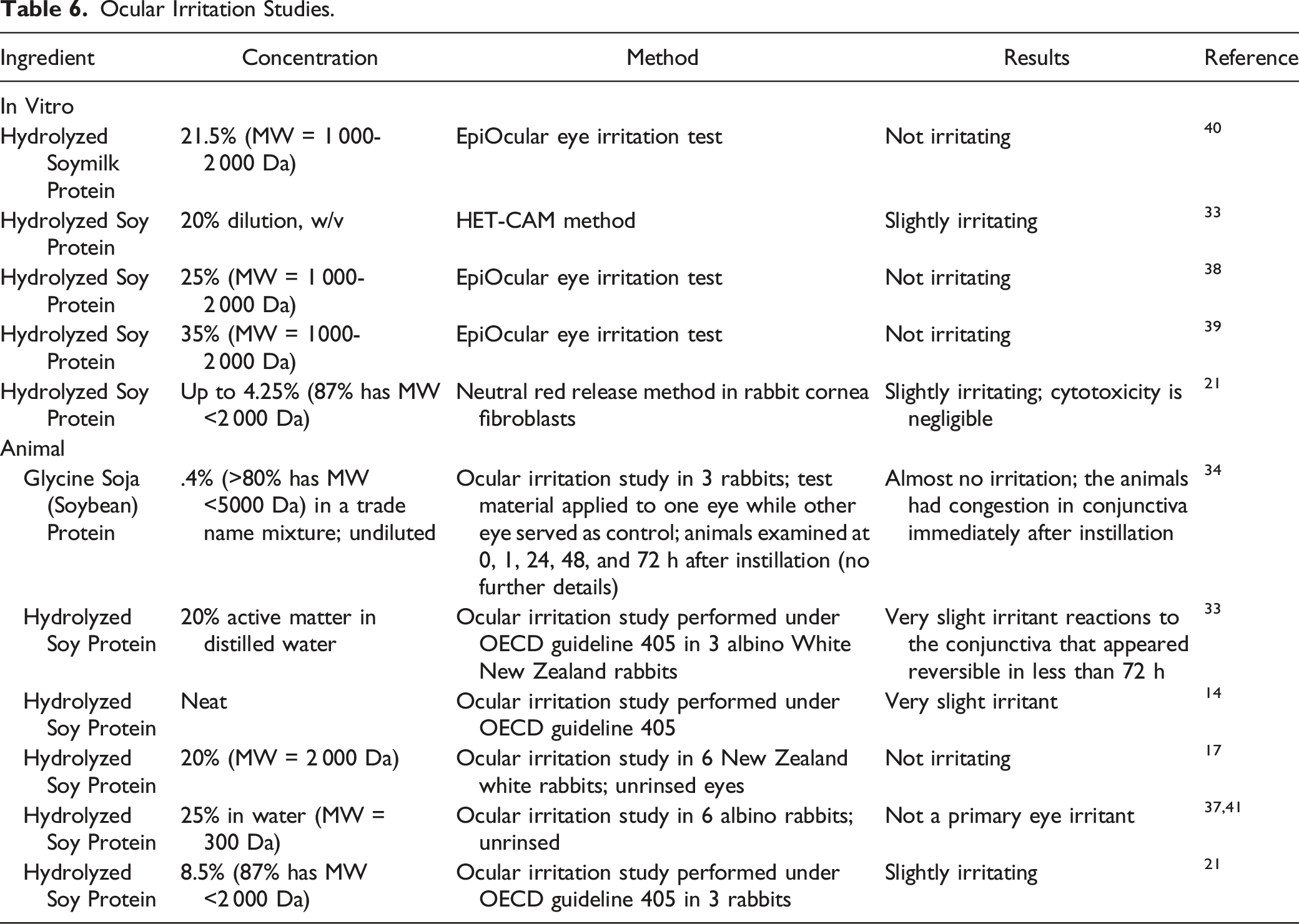
Ocular Irritation Studies.
Sensitization
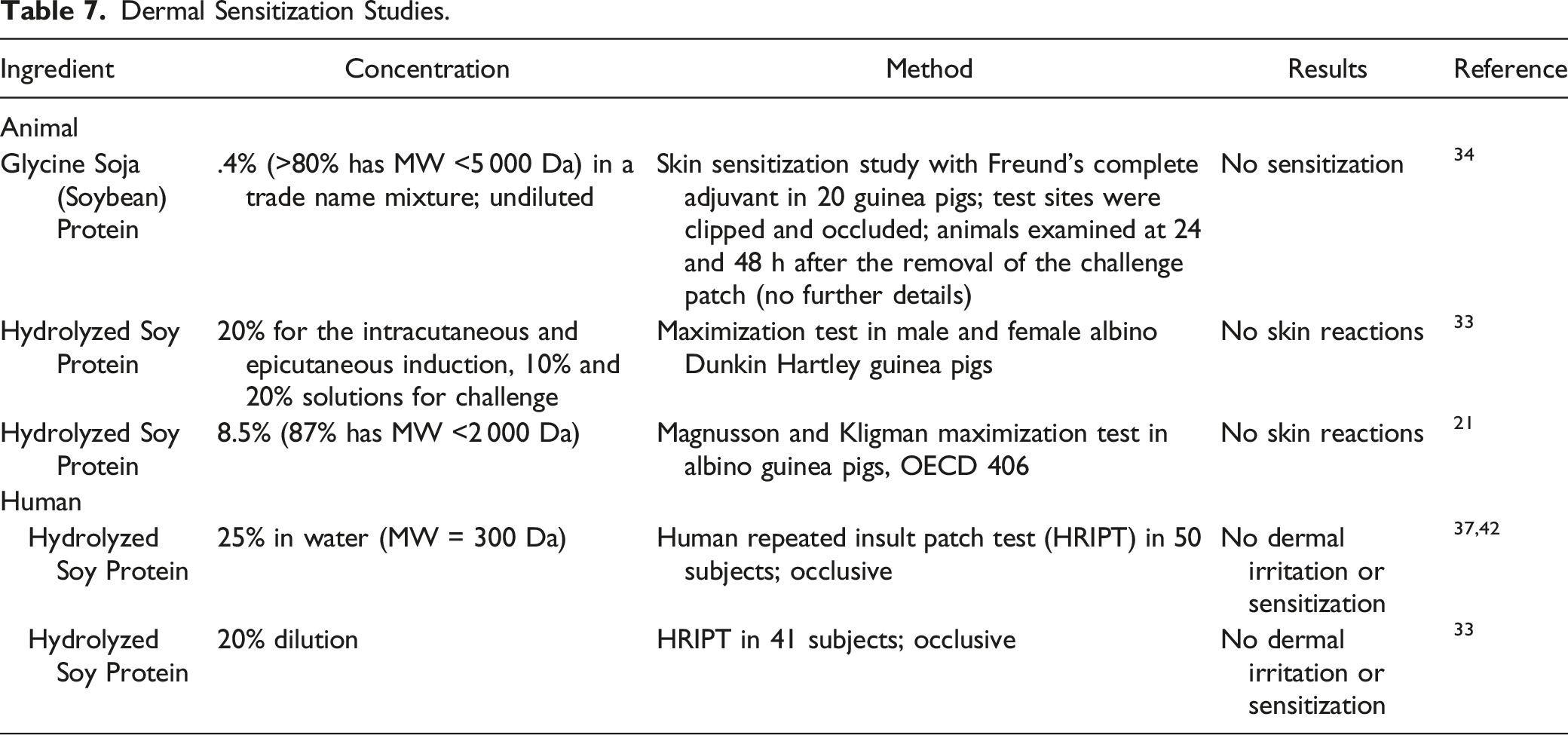
Dermal Sensitization Studies.
Type 1 Hypersensitivity
No occurrences of Type 1 (ie, immediate) hypersensitivity reactions to personal care products that contain soy peptide ingredients were reported in the public literature. An allergen must have at least 2 IgE-binding epitopes, and each epitope must be at least 15 amino acid residues long, to trigger a Type 1 hypersensitivity reaction. 43 Type 1 responses can be elicited in sensitized patients when pairs of IgE molecules against a specific allergen are bound to receptors on the surface of mast cells and other cells that mediate these types of immune reactions. The binding of an allergen molecule to 2 receptor-bound IgE molecules results in the cross-linking of the pair of IgE molecules. The cross-linking of sufficient numbers of IgE pairs bound to the receptors on the surface of a mast cell results in degranulation of the mast cell and the release of vasoactive amines, which are responsible for the Type 1 reaction.
Phototoxicity and Photosensitization
Glycine Soja (Soybean) Protein
An undiluted trade name mixture containing .4% Glycine Soja (Soybean) Protein (>80% with a MW <5 000 Da) produced no photo irritation in a phototoxicity study in 5 guinea pigs. 34 The test material was applied to 2 sites on clipped dorsal skin. One of the 2 sites was irradiated with ultraviolet light (UV) while the other site was covered and served as a control. Animals were examined for signs of erythema and edema on the first, second and third day after application. No further details were provided.
No photosensitization was observed in 20 guinea pigs exposed to an undiluted trade name mixture containing .4% Glycine Soja (Soybean) Protein (>80% with a MW <5 000 Da). 34 During the induction phase, the test sites were injected intradermally with Freund’s complete adjuvant in water and then stripped with cellophane tape prior to topical application of the test material and UV irradiation. For the photochallenge, a group of 10 guinea pigs received the test material on clipped skin while a second group of 10 animals served as a negative control group. One side of each animal was irradiated and the other side was protected with a cover. The animals were observed for signs of erythema and edema 24 and 48 h post-challenge. No further details were provided.
Case Reports
A 43-year-old female presented with a 4-year history of dramatic erythematous eruption of the cheeks and nasal tip. 44 The patient had rosacea but did not respond to topical and systemic treatments. On examination, erythema was observed on the nasal tip and erythematous plaques with fine scale and pustules were observed on the cheeks. Also noted was partially eczematized seborrheic dermatitis of the scalp. The patient’s history included seborrheic dermatitis, lifelong atopic eczema and reactions to jewelry, perfumes, and certain cosmetics. The patient did not wear makeup but used topical products on her face, some of which contained soy ingredients. Previous patch testing yielded a +++ reaction to soy. The patient discontinued use of facial products containing soy and was treated with hydrocortisone ointment, oral erythromycin, and clobetasol foam. At 48 h, the cheek erythema and edema had resolved, and by 96 h, all pustules had cleared, and the seborrheic dermatitis was nearly cleared.
Summary
Soy protein and peptide ingredients function mainly as skin and hair conditioning agents in personal care products. Soy proteins are used as food, and daily exposure from food use would result in much larger systemic exposures than from use in cosmetic products. Additionally, the FDA determined that the use of peptones as direct food substances is GRAS, and that soybean protein is GRAS for substances migrating to food from paper and paperboard products.
According to the 2015 VCRP data, hydrolyzed soybean protein has the most reported uses of the ingredients listed in this safety assessment in cosmetic products with a total of 862; about half of the uses are in non-coloring hair products. Glycine soja (soybean) protein has the second greatest number of overall uses reported, with a total of 313; a third of those are used in leave-on skin care products and another third are used in hair dyes and colors. The results of the concentration of use survey conducted in 2014 by the Council indicate Hydrolyzed Soy Protein has the highest reported maximum concentration of use at up to 3.5% in mascara. Glycine soja (soybean) protein is used at up to .9% in eye lotion.
Soy proteins may also be used in adhesives and by the plastics industry.
While no experimental data were available for the dermal absorption of Hydrolyzed Soy Protein, it was noted that gastrointestinal absorption allows for significantly higher bioavailability than from dermal exposures.
A trade name mixture containing .4% Glycine Soja (Soybean) Protein produced no genotoxicity in a reverse mutation assay and in a chromosomal aberration study (with and without metabolic activation). Hydrolyzed soy protein was not mutagenic in an assay using S. typhimurium TA1535/pSK1002 with and without S9 metabolic activation at concentrations up to 5000 μg/mL.
Hydrolyzed soy protein (25 and 35%) and Hydrolyzed Soymilk Protein (21.5%) were predicted to be non-irritating in in vitro dermal studies. Glycine soja (soybean) protein (.4%) was not irritating in rabbits and guinea pigs. Hydrolyzed soy protein was not a dermal irritant in rabbits when tested up to 25% and in human studies when tested up to 25%.
In in vitro ocular assays, Hydrolyzed Soymilk Protein (21.5%) was predicted to be not irritating while Hydrolyzed Soy Protein (up to 35%) was predicted to be not irritating to slightly irritating. In rabbit ocular studies, Glycine Soja (Soybean) Protein (.4%) produced almost no irritation while Hydrolyzed Soy Protein (tested neat and at concentrations ranging from 8.5 to 25%) was not irritating to slightly irritating.
Glycine soja (soybean) protein was not a dermal sensitizer when tested at concentrations up to .4% in guinea pigs. Hydrolyzed soy protein was not a dermal sensitizer in non-human and human studies when tested at up to 25%. No occurrences of Type 1 (ie, immediate) hypersensitivity reactions to personal care products that contain soy peptide ingredients were reported in the public literature.
No phototoxicity or photosensitization was observed in guinea pig studies with an undiluted trade name mixture containing .4% Glycine Soja (Soybean) Protein.
A case study described aggravation of rosacea in a patient following use of facial products containing soy.
Discussion
The Panel noted that soy proteins are known food allergens that can elicit Type I immediate hypersensitivity reactions when ingested by sensitized individuals. However, the Panel was not concerned that such reactions would be induced by dermal exposure, because these ingredients are water soluble, would not penetrate the skin, and have MWs that are well below that which would cause IgE-cross-linking. The Panel reviewed studies showing no relevant ocular irritation in animals, no dermal irritation or sensitization in animals and human subjects, and no reported cases of Type I immediate hypersensitivity reactions from cosmetic use, which support their conclusion for these ingredients.
The Panel discussed the issue of incidental inhalation exposure from hair sprays and body and hand sprays. There were no inhalation toxicity data available. The Panel considered pertinent data indicating that incidental inhalation exposures to soy ingredients in such cosmetic products would not cause adverse health effects, including data characterizing the potential for soy ingredients to cause ocular or dermal irritation or sensitization. The Panel noted that 95%-99% of droplets/particles produced in cosmetic aerosols would not be respirable to any appreciable amount. The potential for inhalation toxicity is not limited to respirable droplets/particles deposited in the lungs. In principle, inhaled droplets/particles deposited in the nasopharyngeal and thoracic regions of the respiratory tract may cause toxic effects depending on their chemical and other properties. However, coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
The Panel also addressed concerns about pesticide residues and heavy metals that may be present in plant-derived ingredients. They emphasized that the cosmetics industry should continue to use the necessary procedures to limit these impurities in the ingredients before blending into cosmetic formulations.
Conclusion
The Expert Panel for Cosmetic Ingredient Safety concluded that the following soy-based ingredients are safe in cosmetics in the present practices of use and concentration described in this safety assessment:
Glycine Max (Soybean) Polypeptide
Glycine Soja (Soybean) Peptide*
Glycine Soja (Soybean) Protein
Hydrolyzed Soy Protein
Hydrolyzed Soy Protein Extract*
Hydrolyzed Soymilk Protein
*Not reported to be in current use. Were ingredients in this group not in current use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.